Rheological Properties and Particle Size Distribution of Soy Protein Isolate as Affected by Drying Methods
Hima John1*, Shekh Mukhtar Mansuri2, Saroj Kumar Giri3 and Lalan Kumar Sinha3
1 PhD Scholar, ICAR-Central Institute of Agricultural Engineering, India
2 Scientist, ICAR-Central Institute of Agricultural Engineering, India
3Principal Scientist, ICAR-Central Institute of Agricultural Engineering, India
Submission: July 12, 2018; Published: October 11, 2018
*Corresponding author: Hima John PhD Scholar, ICAR-Central Institute of Agricultural Engineering, Bhopal, India.
Shekh Mukhtar Mansuri, Scientist, ICAR-Central Institute of Agricultural Engineering, Bhopal, India
How to cite this article: Hima J, Shekh M M, Saroj K G, Lalan K S. Rheological Properties and Particle Size Distribution of Soy Protein Isolate as Affected by Drying Methods. Nutri Food Sci Int J. 2018; 7(5): 555721. DOI: 10.19080/NFSIJ.2018.07.555721.
Abstract
Soy protein isolate (SPI) solution prepared by ultrafiltration was dried using spray, freeze and oven dryers. Rheological properties (storage and loss modulus, complex viscosity, loss tangent), protein solubility and particle size distribution (D4,3, D3,2 and span) of different isolate powders were compared with commercially available counterpart. Commercial SPI showed a shear thinning behavior while rest of the samples exhibited a shear thickening behavior. Commercial and oven dried SPI (60 °C for 48 hours) SPI shown almost constant loss tangent less than unity during oscillation test which indicated its solid or gel property. Commercial and oven dried SPI samples had comparable size distribution whereas freeze dried SPI shown higher peak and narrow distribution pattern. Commercial SPI had lesser uniformity compared to rest of the samples. Water solubility index of spray dried SPI samples were high compared to freeze and oven dried samples.
Keywords: Soy protein isolate; Spray dryer; Oven dryer; Particle size distribution; Storage and loss modulus; Protein solubility
Practical Application
Soybean is a rich and affordable source of plant protein and can be used as a protein supplement or ingredient in many food items. Soy protein isolate (SPI) is the most concentrated form of soy protein. Now a days, use of isolates in food industry is also increasing. Proteinsare used in food formulations to perform functional roles that are important for consumer food acceptance [1]. Lusas & Riaz [2] reported that soy proteins mainly used as ingredients in formulated foods and substitute for other ingredients. Soy protein can replace many ingredients in food formulations in meat or dairy products without changing the taste and quality of foods. It can be used as emulsifiers, texture enhancers and as ingredients to increase or replace protein content in food products like bread, pastry products, beverages and meat [3]. Numbers of investigations have been carried out to find out the variation in functional properties of SPIs under different processing conditions [4-6].
Rheological properties and particle size distribution of soy protein is equally important along with the functional properties it possesses. Rheological properties can be correlated with other flow behavior related functional properties. Similar studies have been conducted by [7,8] to incorporate different proteins in meat balls. Sabater [9] conducted an experimentto correlate flow characteristics of emulsions with sausage texture. Particle size distribution as well as the uniformity of particle is a major factor contributing the properties and behavior of food formulations and beverages. Particle size affects the solubility as well as the texture of final food preparations. Soluble proteins are easier to incorporate as food ingredients and other properties, like gelation and emulsification. Shen [10] reported that solubility of soy protein products are highly dependent on the physico-chemical states of protein molecules, which are either favorably or adversely affected by heating, drying and other processing treatments during their manufacture and storage. Kinsella [11] reported that heating of protein reduces its solubility, the extent of which depends on the intensity and duration of the heat treatment and therefore the solubility is an indicator of protein denaturation.
Over the year’s ultrafiltration has received considerable interest for concentration of soy proteins as proteins are retained by the membrane while the oligosaccharides and minerals are removed as permeate. The products with improved properties are obtained through ultrafiltration and there also with no excessive use of chemicals [12]. Different drying methods used to dry the protein solutions obtained through ultrafiltration influences the properties of protein isolate powder. Joshi et al. [13] reported that the drying method used for preparation of lupin protein isolate (LPI) can significantly affect the physicochemical properties, which in turn adversely affect the functionality of proteins. Variation of functional properties of SPI with different drying methods is reported by Hu et al., [14].
An attempt has been made to investigate the effect of drying methods on SPI properties. The objective of this study is to gain insights to how the rheological properties, particle size distribution and solubility characteristics of SPIs obtained through ultrafiltration are varying according to the drying methods.
Materials and Methods
Raw materials
Defatted soy flour used as the starting material for extraction of soy protein was purchased from local market. Commercially available SPI was procured from Sonic Biochem Extractions Ltd., Indore (M.P), India.
Preparation of soy protein isolate by ultrafiltration
Extraction of defatted soy flour was performed in purified water at pH 9 (adjusted with 0.2 M NaOH) and at 50 ºC with a solid/liquid ratio of 1/10, using a mechanical stirrer (Jyoti, model JSI-555, India) for one and half hours. Solid-liquid separation was performed in a centrifuge (Remi instruments-model K-70, India) at 10kg for 20 minutes at 15 °C temperature [15]. The supernatant obtained was used as a feed to the ultrafiltration after prefiltration through a micro filtration unit (Millipore). 10kDa hollow fibre type ultrafiltration cartridge (GE Healthcare, model UFP-10- C-4MA, USA) was used with maximum trans-membrane pressure 2kg/cm2 and a volume concentration ratio of 3.5 for getting a concentrate with total soluble solids 20 °Brix.
Drying of soy protein isolate
Concentrated solution obtained from ultrafiltration was dried using a freeze dryer (Bio Sync Technology, New Delhi), spray dryer (Yamato mini spray dryer, ADL 31) and hot air over dryer (Oric, reliable instruments, India).
For freeze drying protein solution was frozen for 12 hours at -18 °C. Frozen samples then freeze dried at 45 °C compressor temperature and 0.5mm vacuum pressure. Freeze dried samples were grounded using a mortar and pestle and sieved through a No. 100 mesh.
Laboratory scale spray dryer with double fluid nozzle arrangement and co-current flow pattern was used for study. Spray dryer was operated with a combination of three inlet temperatures (170, 180 and 190 °C) &three atomizing air pressure ranges (0.15-0.2, 0.2-0.25 and 0.25-0.3kg/cm2), while outlet temperature was fixed at 85 °C. Obtained SPI powders were stored under refrigerated condition for further analyses.
Laboratory scale hot air oven was used for drying SPI solution obtained by ultrafiltration. Combinations of drying time (24, 36 and 48 hours) and temperatures (40, 50 and 60 °C) were set for the study. Dried protein isolates were grounded using a mortar and pestle and sieved through a No. 100 mesh.
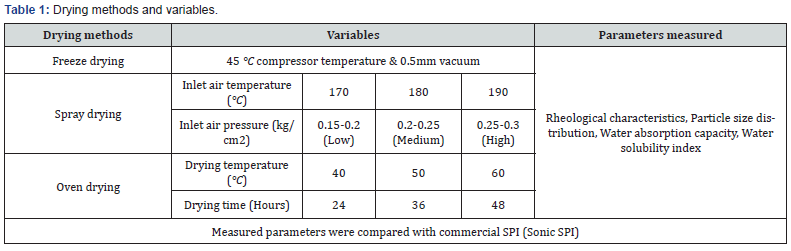
Experimental plan (Table 1)
Table 1 shows the drying methods adopted variables analyzed as different treatments and the parameters measured.
Rheological characteristics
Changes in rheological properties of SPI with different drying methods were measured with a rheometer (Anton Paar, MCR51, Austria (software: RHEOPLUS/32 V2.81)). Storage modulus(G’), loss modulus (G”) andviscosity (η) were measured for ultrafiltered spray dried, freeze dried and oven dried SPI. Obtained results were compared with commercially available counterpart. Parallel plate geometry (PP50-SN8586) with 50mm diameter was used for testing the samples. About 3ml of protein dispersion (25% w/v) was loaded onto the lower plate set at 25 °C. Gap between upper and lower plate was set as 1mm. Rheological characteristics of protein dispersion were measured using dynamic frequency sweep test by comparing the dependence of G’ and G’’ of protein isolate dispersion with angular frequency. Oscillation mode was set with 5% shear rate and dynamic angular frequency starting from 0.001 to 1000 1/s. 16 data points were measured with mean point duration 10 seconds. All measurements are made in triplicate.
Particle size distribution
Particle size distribution of ultra-filtered SPIs (spray dried, oven dried and freeze dried) and commercially available SPI were determined using a particle size analyser (Mastersizer, Malvern Inc., Worcestershire, United Kingdom) with wet feed attachment. Samples were dispersed in deionised water (1% w/v). Particle size analysis for average particle sizes (D[3,2] - Surface Area Moment Mean - Sauter Mean Diameter and D[4,3] - Volume or Mass Moment Mean - De BrouckereMean Diameter) and width of the distribution or Relative Span Factor (RSF) were analyzed.
The, Mean diameters D[3,2] and D[4,3] are defined by:
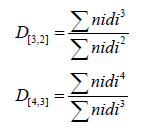
Where ni is the number of particles of diameter di.
RSF is defined as:
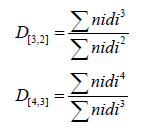
Where: D(v,0.5), D(v,0.9) & D(v,0.1) are the particle diameters where 50%, 90% and 10% volume distribution below this value respectively. The obscuration of all samples was maintained between 10 and 15%. Pump speed was set as 3000rpm and beginning of test, samples were sonicated. All measurements are made in triplicate.
Determination of Water absorption capacity (WAC)
WAC was determined according to the method of Beuchat [16] with some modifications. Approximately 0.5g of SPI was taken in 50ml centrifuge tube and 6ml distilled water was added to that. The tube was agitated on a vortex mixer for 1min at higher speed. The sample left for 30min and centrifuged at 5000g for 20min. WAC can be expressed as ml of water per gram of sample and was calculated using the following formula:
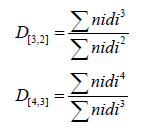
Determination of water solubility index (WSI)
WSI was determined according to the method of Morsy et al. [17] with some modifications. 0.2gram SPI was mixed with 5ml distilled water in a 50ml centrifuge tube and agitated in a vortex mixer for 2 minutes. The solution was centrifuged for 15 minutes at 3000rpm and the supernatant was separated. Water solubility index was calculated as:
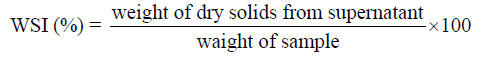
Statistical analysis
Triplicate runs were carried out for each experiment and the data were subjected to statistical analysis. Statistical analysis was done using SAS 9.3 software. One-way analysis of variance (ANOVA) was performed to evaluate the significance in differences (p<0.05) and LSD was used to determine whether the averages of two sets of measurements were significantly different at P<0.05.
Results and Discussion
Rheological characteristics
SPI samples variation in storage modulus (G’), loss modulus (G”) and complex viscosity (η) with angular frequency is shown in (Figure 1). G’ and G” showed similar kind of frequency dependence for all different treated samples except for commercial sample. Moduli of oven dried, and spray dried SPI samples showed more frequency dependence [higher slope of log (G’) and log (G”) vs. frequency] than that of the commercial SPI. Frequency sweep test G’ and G’’ values of for various treatment samples at the beginning were 30 to 3880 Pa and 11 to 187 Pa and at the end 33 to 6510 Pa and 165 to 407 Pa respectively.
Feed formulation
Normal control diet feed was prepared using soybean (27%), whole maize flour (Zea mays) (59%), vegetable oil (10%) and salt/vitamin mix (4%) was from Sigma-Aldrich Co. Ltd., Poole Dorset, UK. Same ingredients were used for test diet except Detarium senegalense seed oil was used wholly for 10% OSO feed and varying percentages (1.5%, 1.0%, and 0.5%) were supplemented with commercially consumed vegetable oil (VO) to make up the difference. Total oil composition was 10% for all formulated feeds used in this study. The different feed ingredients were thoroughly mixed, made into pellets for easy handling by animals and oven dried to prevent the growth of mold. Fully dried feeds were kept in previously labeled air tight bags and were stored at 4 °C to prevent microbial spoilage.
Commercial SPI showed almost constant G’ with increase in angular frequency whereas rest of the samples shown a gradual increase (Figure 1(a)). The increase in G’ was irrespective of the treatments and this shows the solid like characteristics of SPI samples with increased angular frequency. Initially highest G’ value was obtained for commercial sample followed by different spray dried and oven dried samples but at higher angular frequency all the treatments shows G’ values almost in the same range. G” of all samples increased with increase in angular frequency irrespective of the treatment (Figure 1(b)). Commercial SPI showed highest G” value but the rate of increase is less than spray dried and oven dried. Hence commercial SPI shows more liquid like properties with increase in angular frequency. Doublier [18] reported that the frequency independence in G’ and G” is an indication of a strong gel network, with no possibility of rupturing the junction zones within the time-scale of experiments. The strong frequency dependence of G’ and G” indicates that there is no specific interaction between molecules. Large slope of the G’ curve indicates low strength and a small slope indicate high strength [19].
Commercial SPI complex viscosity (η) decreased from 16.4 to 2.4 Pa.S with increase in angular frequency hence observed a shear thinning behavior but Spray dried, Oven dried and freeze dried samples exhibited a slight increase in η value with increasing angular frequency value (Figure 1(c)), hence exhibited a shear thickening behavior. The rate of increase is less for oven dried samples compared spray dried SPI. Koksel et al. [20] explained that the viscosity increases as a result of water uptake by the samples following hydrogen bond disruption due to heating. Granger et al. [21] reviewed that increase in viscosity may be due to an increase in droplet concentration and/or variation in the droplet size.
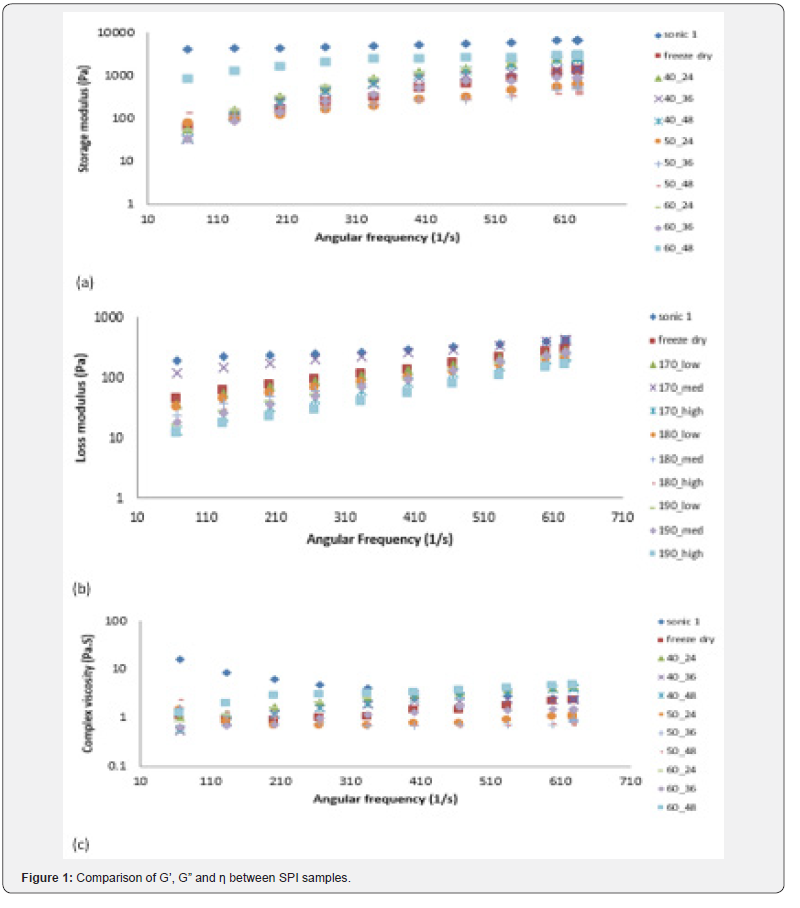
In commercial SPI, G’ and G” (G’ greater than G”) were followed almost parallel path with angular frequency (Figure 2(a)), shows the characteristics of a weak visco-elastic gel behavior. Low value of loss tangent (tan δ) also indicates the elastic like property of commercial SPI suspension. Where as in freeze dried, spray dried and oven dried sample exhibited a cross over point between G’ and G” with angular frequency below 100 1/s except for oven dried SPI which is dried under 60 °C for 48 hours (Figure 2(b,c,d,e,f)). Ross-Murphy [22] reported that the point at which G’ becomes just greater than G” (cross-over point), is taken as an empirical indication of gel formation and was accompanied by a rapid fall in tan δ. The rapid drop in Tan δ also indicates the transformation of solution into gel. For commercial SPI and oven dried SPI (60 °C for 48 hours) tan δ (less than unity) were almost constant (Figure 2(a,d)) during oscillation test indicated solid or gel property. Whereas spray dried SPI (190 °C inlet temperature and high air flow rate 0.25-0.3kg/cm2) tan δ decreased from 1.05 to 0.42 indicates transition from liquid or sol like characteristics to solid or gel like characteristics. For rest of the samples, tan δ values less than unity throughout the frequency sweep with a rapid drop after cross over point. Salunkhe & Kadam [23] also found that the spray-dried LPI produced a gel with more elastic properties than freeze-dried LPI.
Particle size distribution
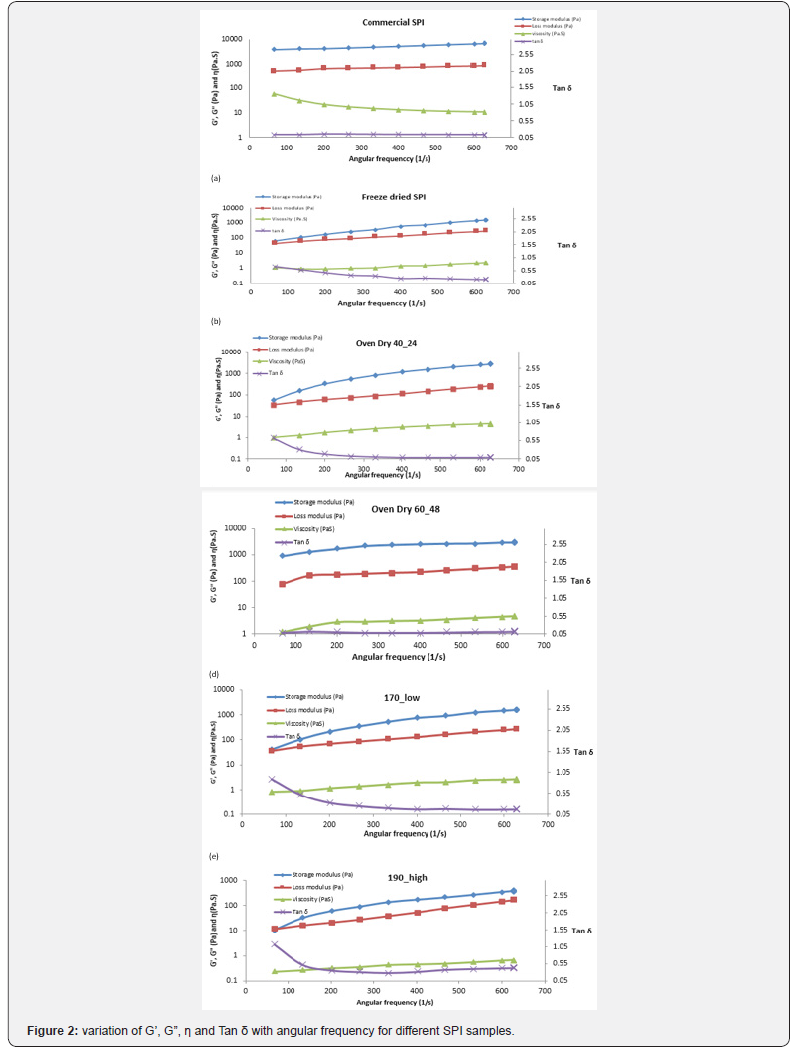
Figure 3 shows the particle size distribution for various SPI samples and Table 2 shows the corresponding D[4, 3], D[3,2] and span values. Single distinct peak was observed for all samples. Most of the samples had particle sizes above 100μm. Commercial SPI (Sonic Biochem) and oven dried SPI samples had comparable size distribution whereas there observed a higher peak and narrow distribution for freeze dried sample (Figure 3(a)). This is due to the fluffy nature of freeze-dried sample and this observation is on par with the findings of Liu et al. [24]. From Figure 3(b), significant higher peak for spray dried samples at higher temperatures and air flow rates were observed. The particle size of milk powder is studied by Laval & Pak [25] and he reported that the particle size is related to its appearance, reconstitution property and flow characteristic. He observed that particle size distribution is influenced by original milk characteristics, processing conditions and type of equipment used in drying process.
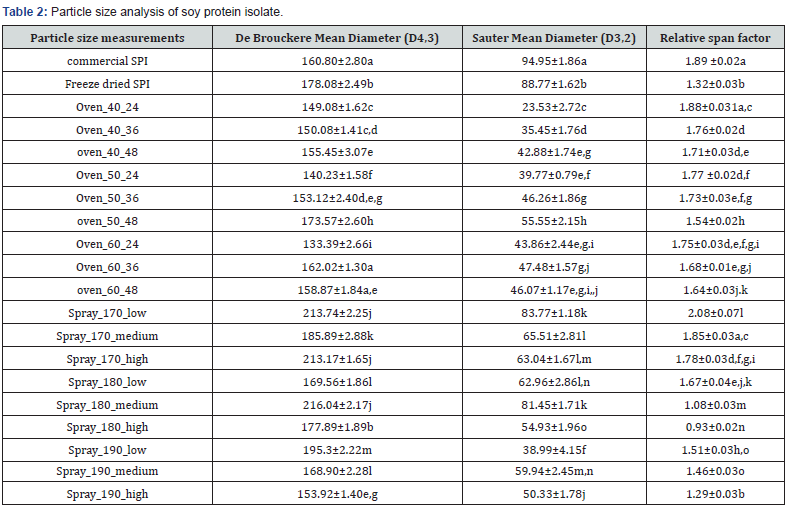
Oven_40_24: oven drying at 40 °C for 24 h; Oven_40_36: oven drying at 40 °C for 36 h; Oven_40_48: oven drying at 40 °C for 48 h; Spray_170_ low: spray drying at 170 °C inlet air temperature and 0.15-0.2 kg/cm2 air pressure; Spray_170_medium: spray drying at 170 °C inlet air temperature and 0.2-0.25 kg/cm2 air pressure; Spray_170_high: spray drying at 170 °C inlet air temperature and 0.25-0.3 kg/cm2 air pressure.
The De Brouckere Mean Diameter reflects the size of those particles which constitute the bulk of the sample volume and is sensitive to the presence of large particulates in the size distribution. D[4, 3] value indicates the size of the coarse particulates that make up the bulk of this sample. Oven dried samples had comparatively lesser coarse particles. Spray dried samples exhibited higher D[4, 3] value may be because of the use of double fluid type spray nozzle for atomizing the sample. This observation is in accordance with the findings by Elversson et al. [26] and he reported that there is a linear relationship between the droplet size and size of the powder particles.
The Sauter Mean Diameter D[3, 2] is most sensitive to the presence of fine particulates in size distribution and this can be used to monitor proportion of fine particles in samples. Similar to D[4, 3] oven dried samples had lesser D[3, 2] value and spray dried samples had higher value. Commercial SPI and freeze-dried SPI possessed good number of fine particles. Carić [27] conducted study on spray drying of milk powder and he observed that spray dried powder particles are usually spherical with diameters ranging from 10 to 250μm. He found that rapid dispersion requires a particle size of about 150 to 200μm and powder with large particle size has superior dispersibility. Singh [28] observed that dispersibility of powder decreases as the percentage of fine particles (<90μm) increases.
Relative span factor (RSF) is a dimensionless parameter indicative of the uniformity of the drop size distribution. It provides a practical means for comparing various size distributions, closer number to zero more uniform the sample will be. Commercial SPI had lesser uniformity compared to freeze dried, oven dried and spray dried samples. Also, there observed a slight increase in uniformity of particle distributions with the increase in drying time and air flow rate in the case of oven drying and spray drying respectively. Effect of inlet air temperature on particle size could not identify clearly from the observations. Similar conclusion was drawn by Seth et al. [29]. Agrahar-Murugkar et al. [30] observed that particle size of flour greatly influences the water absorption capacity, density and spread of biscuits.
Water absorption capacity (WAC) and water solubility index (WSI)
From Table 3, highest water absorption capacity was observed for commercial SPI (8.83±0.02ml/g) followed by freeze dried SPI (6.47±0.02ml/g). Oven dried, and spray dried samples show comparatively low WAC values, and this may be due to the denaturation of protein caused due to adopted processing methods. Jovanovich et al. [31] concluded that this variation is due to highly condensed and less porous particle morphology of the spray dried protein powders. Lili et al. [32] and Singh et al. [33] also observed that the proteins obtained from freeze-dried exhibited a much stronger capacity of absorbing water than that from spray-dried powders (p<0.05). Highest WAC of commercial SPI may due to the presence of additives.

Higher solubility of proteins helps to form stable dispersions when incorporated into beverages and other food systems. WSI of spray dried SPI samples were highly comparable to freeze dried and oven dried samples but for commercial SPI was lesser. Wang & Johnson [34] reviewed that functionalities such as gelling, emulsifying and foaming are closely associated with solubility. The high-water solubility of spray dried and freeze-dried protein powders can be attributed to less thermal stress encountered in drying process. Similar findings were reported by Pacheco et al., [35] and Yang et al. [36]. Another possible reason for the variation in WSI was explained by Wagner et al., [37] and Pace et al., [38], as the concentration of protein increases its solubility decreases caused by increased protein-protein interaction. Roesch & Corredig [39] concluded that when soy protein is at high concentration it will be aggregated; while at lower concentrations various types of soluble complexes may form. Corrigan et al. [40] also reported that high energy amorphous form caused by spray drying led to improve the functional properties of powder such as the enhanced solubility and faster dissolution rate. Lower WSI values for oven dried samples were due to the severity of heat treatment as well as the higher bulk density of oven dried samples compared with freeze dried and spray dried SPIs. This observation is on par with the findings of Mirhosseini & Amid [41].
Conclusion
The rheological properties, protein solubility and particle size distribution of isolate powder obtained through different drying methods were compared with commercial SPI. Storage and loss modulus of freeze dried, oven dried, and spray dried SPI samples have shown more frequency dependence than that of the commercial SPI. Commercial SPI showed almost constant G’ with variation in angular frequency whereas rest of the samples shown a gradual increase. G” of all samples increased with increase in angular frequency irrespective of the treatment. Commercial SPI showed a decrease in η with increase in angular frequency hence observed a shear thinning behavior. Spray dried, Oven dried, and freeze-dried samples exhibited a slight increase in η value with angular frequency value, hence exhibited a shear thickening behavior.
Commercial SPI and oven dried SPI samples had comparable size distribution whereas there observed a higher peak and narrow distribution for freeze dried sample. Oven dried samples had comparatively lesser coarse particles. Commercial SPI had lesser uniformity compared to freeze dried, oven dried and spray dried samples. Highest water absorption capacity was observed for commercial SPI and oven and spray dried samples showed comparatively low WAC values. WSI of spray dried SPI samples were high compared to freeze dried and oven dried samples. Commercial SPI also showed lesser WSI value than its other counter parts.
We conclude the drying methods and conditions have significant influence on rheological properties, solubility characteristics and particle size distribution of soy protein isolate obtained through ultrafiltration which in turn decides the functionality and end use of isolates in food applications.
References
- Lamsal BP, Jung S, Johnson LA (2007) Rheological properties of soy protein hydrolysates obtained from limited enzymatic hydrolysis. LWT-Food Science and Technology 40(7): 1215-1223.
- Lusas EW, Riaz MN (1995) Soy protein products: processing and use. J Nutr 125(3 Suppl): 573-580.
- Lai YP, Mondor M, Moresoli C, Drolet H, Gros-Louis M, et al. (2013) Production of soy protein isolates with low phytic acid content by membrane technologies: Impact of the extraction and ultrafiltration/ diafiltration conditions. Journal of food engineering 114(2): 221-227.
- Tavano OL (2013) Protein hydrolysis using proteases: an important tool for food biotechnology. Journal of Molecular Catalysis B: Enzymatic 90: 1-11.
- Kempka AP, Honaiser TC, Fagundes E, Prestes RC (2014) Functional properties of soy protein isolate of crude and enzymatically hydrolysed at different times. International Food Research Journal 21(6): 2229- 2236.
- Shih MC, Hwang TS, Chou HY (2016) Physicochemical and functional property changes in soy protein isolates stored under high relative humidity and temperature. J Food Sci Technol 53(1): 902-908.
- Hermansson AM, Akesson C (1975) Functional properties of added proteins correlated with properties of meat systems. Effect of concentration and temperature on water‐binding properties of model meat systems. Journal of Food Science 40(3): 595-602.
- Hermansson AM, Akesson C (1975) Functional propertles of added proteins correlated with properties of meat systems. Journal of Food Science 40(3): 603-610.
- Sabater de Sabates A (1976) Rheology of emulsions used in meat industry. Effect on sausages texture. In Anales de Bromatologia 28(1): 81-98.
- Shen JL (1976) Solubility profile, intrinsic viscosity, and optical rotation studies of acid precipitated soy protein and of commercial soy isolate. J Agric Food Chem 24(4): 784-788.
- Kinsella JE (1982) Relation between structure and functional properties of food proteins. In Food Proteins. Proceedings of the Kellogg Foundation International Symposium on Food Proteins held at University College, pp. 52-103.
- Alibhai Z, Mondor M, Moresoli C, Ippersiel D, Lamarche F (2006) Production of soy protein concentrates/isolates: traditional and membrane technologies. Desalination 191(1-3): 351-358.
- Joshi M, Adhikari B, Aldred P, Panozzo, JF, Kasapis S (2011) Physicochemical and functional properties of lentil protein isolates prepared by different drying methods. Food Chemistry 129(4): 1513- 1522.
- Hu XZ, Cheng YQ, Fan JF, Lu ZH, Yamaki K, Li, et al. (2010) Effects of drying method on physicochemical and functional properties of soy protein isolates. Journal of food processing and preservation 34(3): 520-540.
- Sinha LK, Chandra P (2011) Development of a process for production of soy protein concentrate and isolate by membrane separation technology. Final report (project no. 628). ICAR-CIAE, Bhopal, India.
- Beuchat LR (1977) Functional and electrophoretic characteristics of succinylated peanut flour protein. J Agric Food Chem 25(2): 258-261.
- Morsy NE, Rayan AM, Youssef KM (2015) Physico Chemical Properties, Antioxidant Activity, Phytochemicals and Sensory Evaluation of Rice- Based Extrudates Containing Dried Corchorusolitorius l. Leaves. Journal of Food Processing & Technology 6: 408.
- Doublier IL (1992) Viscoelastic properties of gels. Viscoelastic properties of foods, London, Elsevier Applied Science, New York, USA, pp. 371-434.
- Patel AR, Dumlu P, Vermeir L, Lewille B, Lesaffer A, et al. (2015) Rheological characterization of gel-in-oil-in-gel type structured emulsions. Food Hydrocolloids 46: 84-92.
- Koksel H, Masatcioglu T, Kahraman K, Ozturk S, Basman A (2008) Improving effect of lyophilization on functional properties of resistant starch preparations formed by acid hydrolysis and heat treatment. Journal of Cereal Science 47(2): 275-282.
- Granger C, Barey P, Combe N, Veschambre P, Cansell M (2003) Influence of the fat characteristics on the physicochemical behavior of oil-inwater emulsions based on milk proteins-glycerol esters mixtures. Colloids and Surfaces B: Biointerfaces 32(4): 353-363.
- Ross-Murphy SB (1991) Concentration dependence of gelation time. Cambridge, The Royal Society of Chemistry, UK, pp. 357-368.
- Salunkhe DK, Kadam SS (1989) Crc handbook of world food legumes: nutritional chemistry, processing technology, and utilization, CRC Press, Boca Raton, FL, USA, pp. 75-97.
- Laval A, Pak T (1995) Dairy processing handbook. Tetra Pak Processing Systems, Lund, Sweden.
- Liu J, Jiang W, Zeng JB, Yang Q, Wang YJ, et al. (2014) Effect of drying on particle size and sensitivities of nano hexahydro-1, 3, 5-trinitro-1, 3, 5-triazine. Defence Technology 10(1): 9-16.
- Elversson J, Millqvist, Fureby, A., Alderborn, G. & Elofsson, U. (2003) Droplet and particle size relationship and shell thickness of inhalable lactose particles during spray drying. J Pharm Sci 92(4): 900-910.
- Carić M (1994) Concentrated and dried dairy products. VCH Publishers Inc, New York, USA.
- Singh H (1992) Aspects of proteins in milk powder manufacture. Milk Proteins; Molecular, Physico-Chemical and Biological Aspects. Advanced dairy chemistry, Proteins (2nd edn), Elsevier Applied. Science Pub, New York, USA, pp. 735-765.
- Seth D, Mishra HN, Deka SC (2017) Functional and reconstitution properties of spray-dried sweetened yogurt powder as influenced by processing conditions. International Journal of Food Properties 20(7): 1603-1611.
- Agrahar-Murugkar D, Gulati P, Kotwaliwale N, Gupta C (2015) Evaluation of nutritional, textural and particle size characteristics of dough and biscuits made from composite flours containing sprouted and malted ingredients. J Food Sci Technol 52(8): 5129-5137.
- Jovanovich G, Puppo MC, Giner SA, Añón MC (2003) Water uptake by dehydrated soy protein isolates: comparison of equilibrium vapour sorption and water imbibing methods. Journal of food engineering 56(4): 331-338.
- Lili L, Huan W, Guangyue R, Xu D, Dan L, et al. (2015) Effects of freeze-drying and spray drying processes on functional properties of phosphorylation of egg white protein. International Journal of Agricultural and Biological Engineering 8(4): 116-124.
- Singh N, Kaur M, Sandhu KS (2005) Physicochemical and functional properties of freeze-dried and oven dried corn gluten meals. Drying technology 23(4): 975-988.
- Wang C, Johnson LA (2001) Functional properties of hydrothermally cooked soy protein products. Journal of the American Oil Chemists’ Society 78(2): 189-195.
- Pacheco-Aguilar R, Mazorra-Manzano MA, Ramírez-Suárez JC (2008) Functional properties of fish protein hydrolysates from Pacific whiting (Merlucciusproductus) muscle produced by a commercial protease. Food Chemistry 109(4): 782-789.
- Yang G, Yang B, Wu J (2009) Effect of drying on property of Zein. Food Science 30(24): 57-59.
- Wagner JR, Sorgentini DA, Añón MC (2000) Relation between solubility and surface hydrophobicity as an indicator of modifications during preparation processes of commercial and laboratory-prepared soy protein isolates. Journal of Agricultural and Food Chemistry 48(8): 3159-3165.
- Pace CN, Trevino S, Prabhakaran E, Scholtz JM (2004) Protein structure, stability and solubility in water and other solvents. Philos Trans R Soc Lond B Biol Sci 359(1448): 1225-1235.
- Roesch RR, Corredig M (2003) Texture and microstructure of emulsions prepared with soy protein concentrate by high-pressure homogenization. LWT-Food Science and Technology 36(1): 113-124.
- Corrigan DO, Healy AM, Corrigan OI (2003) The effect of spray drying solutions of bendroflumethiazide/polyethylene glycol on the physicochemical properties of the resultant materials. Int J Pharm 262(1-2): 125-137.
- Mirhosseini H, Amid BT (2013) Effect of different drying techniques on flowability characteristics and chemical properties of natural carbohydrate-protein Gum from durian fruit seed. Chem Cent J 7(1): 1.






























