Nutrition Support in Intensive Care: Which Practices?
Achwak Mehrez1*, Om kalthoum Sallem2, Imen Bannour1 and Ali majdoub2
1Intensive care department, University Hospital of Mahdia, Mahdia, Tunisia
2Nutrition-Gastrology department, University Hospital of Monastir, Monastir, Tunisia
Submission: December 27, 2023;Published: January 18, 2024
*Corresponding author: Achwak Mehrez, Intensive care Department, University Hospital of Mahdia, Mahdia, Tunisia
How to cite this article: Achwak M, Om kalthoum S, Imen B, Ali m. Nutrition Support in Intensive Care: Which Practices?. Nutri Food Sci Int J. 2024. 12(4): 555845.. DOI: 10.19080/NFSIJ.2023.12.555845.
Abstract
Background and Aims: To optimize feeding assistance in intensive care, multiple clinical practice guidelines have been published, providing evidence-based recommendations. Despite guidelines availability, considerable variations in practices are reported showing a lack of application at the bedside. We aimed to evaluate artificial feeding practices in multiple intensive care units and to determine the gap between these practices and recommendations.
Methods: Information was gathered through a survey enrolling 105 Tunisian intensivists with different expertise levels. The content of the questionnaire was guided by a literature review and the latest Clinical Practice Guidelines.
Results: In this series, many shortcomings in compliance with the guidelines were observed: early initiating enteral nutrition (58%), adapting the adequate osmolarity with the route of venous access (33%), using GRV to assess gastrointestinal dysfunction (60%), and administrating prokinetics in case of gastrointestinal intolerance (61%). Continuous feeding delivery was adopted by most of the respondents (88%) though intermittent feeding was only noted among a small part of junior anesthesiologists (p=0.01). Central venous access was indicated in the first intention by most respondents (74%). Physicians had blurred knowledge concerning parenteral nutrition indications. Concerning the obese patient’s nutritional management: 32% opted for a hypocaloric hyper protein diet and 11% had limited knowledge about the nutritional management particularities.
1.4. Conclusion: Among all groups, adherence to evidence-based guidelines was low. Nutritional support was found sometimes sporadic and multiple nutritional management particularities were ignored. To achieve the adequate practice of recommendations, more focus should be put on training, assistance, and a multidisciplinary implication program.
Keywords: Nutrition support; Critical care; Enteral nutrition; Parenteral nutrition; Clinical practice guidelines
Abbreviations: ICU: Intensive Care Unit; EN: Enteral Nutrition; PN: Parenteral Nutrition; GRV: Gastric Residual Volume
Introduction
Years ago, when artificial feeding was first developed, it was conceived as supportive care, as administering metabolic support while the patient recovered from their illness. Over the past 25 years, there have been significant scientific advances in the field of medical nutrition, leading to a better understanding of the role of nutrition in intensive care unit (ICU) patients’ management [1-3]. Evidence has demonstrated that nutrition therapy is associated with significant reductions in the length of hospital stays, infectious complications, and mortality rates. In addition to this, it is found to be linked to improved patient outcomes, decreased duration of dependence on mechanical ventilation, more rapid mobilization, increased survival, and better quality of life [1-4]. Accordingly, the role of nutrition in patient care has transitioned from supporting patients while they recover from their underlying injury to active therapy, modulating their disease response and improving their chances of survival [4-7]. Therefore, nutrition has evolved into an integral part of modern critical care for both medical and surgical patients. Initially, a neglected field that was considered peripheral to medical care, it is now recognized as a valid specialty with significant evidence to support current practice. Multiple Clinical Practice Guidelines have been published, providing evidence-based recommendations on how to optimally feed the critically ill patient [8,9]. Despite the availability of these guidelines to assist practitioners in making decisions related to the provision of nutrition therapy in the ICU, multiple observational studies of nutrition practices in critically ill patients have reported considerable variation in practices, demonstrating that guideline recommendations are not being uniformly applied at the bed-side [10-12]. To the best of our knowledge, this is the first multi-center study of its kind in Tunisia that has for aim:
i) First, to evaluate artificial feeding practices in multiple Tunisian intensive care units.
ii) Secondly, to determine the gap between these practices and recommendations about artificial feeding.
Methods
Subjects
This cross-sectional study was conducted through a survey over 6 months from January to August 2020. It was intended for all intensivists (certified specialists and residents) working in multiple university hospitals in various regions of Tunisia (Tunis, Ariana, Sousse, Monastir, and Mahdia). Respondents were provided with 2 options: Online via E-mails and via social networks. The second was a one-to-one interview where the physician in question was asked to fill in the questionnaire in paper form. All participants provided informed consent, and a statement of anonymity was included.
Survey content
The content of the survey was developed by a multidisciplinary group of nutrition support practitioners to determine nutrition support practices in multiple Intensive Care Units (ICUs) across Tunisia. The survey evaluates the delivery of artificial feeding methods including both enteral nutrition (EN) and parenteral nutrition (PN). Twenty-five multiple-choice questions were designed from evidence-based practice standards and the latest clinical guidelines related to adult nutrition support care. The questionnaire of the survey was written and explained in French and divided into 2 categories. The first concern the care provider characteristics (2 questions) and the second concern the nutrition support practices including 3 sections respectively: 8 questions on the prescription of enteral nutrition support; 5 questions specific to parenteral nutrition support practices in the ICU; and 5 questions on nutrition support practices in obese patients.
Statistical analysis
Data analysis was performed using IBM SPSS software version 25.0. Variables are expressed as mean ± standard deviation, significance analysis was performed with Student ‘s t-test or Fisher’s exact test. Statistical significance was set at p < 0.05.
Ethical statement
No ethics approval was considered necessary as the survey was anonymous and did not include any information regarding the health status of the participants.
Results
Population characteristics
A total of 170 physicians were requested to complete the questionnaire. Out of the 173 distributed questionnaires, 105 completed questionnaires (61%) were received. This study involved 105 physicians; including 79 anesthesiologists (75%) and 26 medical intensivists (25%) working in five different regions of the country; all were practicing in public health institutions. All Respondents were asked about their experience in their respective fields. Most of our study participants had less than 8 years of work experience (82%) and the rest were senior physicians with more than 8 years of experience (18%).
Enteral Nutrition (EN)
Initiation of EN: Nutritional support was carried out from the first hours of admission by 58% of respondents. Less experienced physicians were more conservative about early nutrition support rather than senior physicians (51% vs 90%) with a significant difference (p=0.02). In return, 37% of respondents opted for starting nutritional support within 3 days of admission. Junior physicians opted more for initiating nutrition within 3 days (11% vs 73%) with a significant p=0.008.
6 Modalities of EN: Continuous enteral nutrition was prescribed by most of the respondents (88%): all the medical intensivists and 84% of anesthesiologists with a significant difference (p=0.02). Meanwhile, intermittent enteral nutrition was only indicated by anesthesiologists (15%) with a significant difference of p=0.01. All senior physicians insisted on continuous enteral nutrition administration. Meanwhile, only junior physicians (18%) opted for intermittent feeding with p=0.048.
6 EN complications management: The measurement of gastric residual volume (GRV) to assess gastrointestinal dysfunction was reported by 60% of respondents. In the case of gastric feeding intolerance, 79% of the participants opted for reducing the EN delivery rate, 61% for administering prokinetics, 25% for withholding EN, 18% for switching to PN, and only 9% opted for considering post pyloric feeding. Erythromycin was used by 61% of respondents as a first-line prokinetic therapy.
Parenteral nutrition (PN)
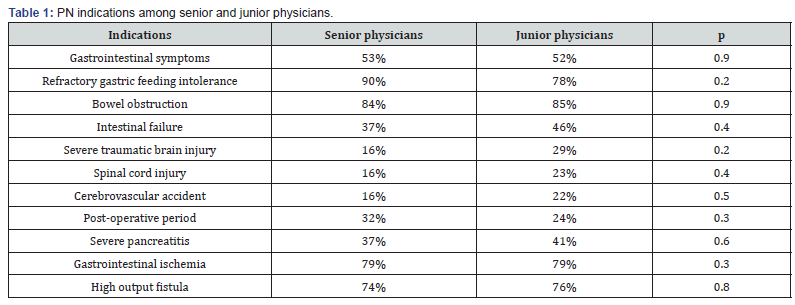
Indications: PN was recommended by 81% of respondents in case of failure of well-conducted enteral nutrition. PN indication was reported in several situations and no significant differences were found between senior and junior physicians as Table 1 shows. More than a third of respondents (39%) considered enteral nutrition insufficient within the fourth day. Most Senior physicians considered enteral nutrition insufficient after 4 days compared to less experienced physicians (38%) with a significant difference (p=0.05).
6 Route of access: Central venous access was indicated as the first intention by most respondents (74%). Peripheral venous access was more indicated by senior physicians (42%) than less experienced practitioners (30%) with no significant difference (p=0.3). Almost a third of respondents (31%) reported that they do not adapt the osmolarity of PN products to the chosen route of access. Only 33% of respondents recognized the adequate osmolarity for parenteral administration.
PN systematic supplementations: Systematic vitamin supplementations were reported by 35% of respondents. While trace elements and electrolytes prescriptions were more reported: phosphorus in 72%, magnesium in 76%, calcium in 75%, and potassium in 64%.
PN monitoring: Monitoring artificial feeding is mandatory for all respondents. In the early period of the acute phase, half of the respondents opted for enhanced monitoring (multiple times a day) while the other half opted for daily monitoring.
Situation in the intensive care: obese patients
For the obese patient’s management, 67% of respondents opted to calculate the ideal body weight while 27% had used the adjusted body weight to evaluate obese patients. A third of the respondents (32%) opted for hypocaloric nutrition while providing a larger dose of protein. Whereas providing an isocaloric intake was the choice of 60% of respondents.
Discussion
This survey highlights evidence-based guidelines that are already followed by physicians and focuses on weak practices that need to change among critical care providers. In terms of our study, we conclude that despite the basic knowledge of nutrition management among physicians, clinical practice is still far from applying bundles. Evidence-based guidelines that were poorly followed: early enteral nutrition initiation (58%) and prokinetics administration in case of gastrointestinal intolerance (61%). Physicians had blurred knowledge concerning parenteral nutrition indications, and nutrition management of obese patients. Differences between practices among junior and senior physicians were also mentioned: senior physicians insisted more on the early enteral nutrition (p=0.02) and continuous enteral nutrition (p=0.048).
Enteral nutrition
Initiation of EN: Early enteral nutrition was reported in 58%. These results are consistent with those of other studies where the EN initiation delay was commonly reported explaining the failure to reach the adequate caloric target [13,14]. Early EN, which is defined as the initiation of EN within 24 to 48 hours of ICU admission, has been shown to decrease the risk of infectious complications and ICU length of stay compared to delayed EN as well as early parenteral nutrition (PN) [8,15,16].
7.1.2. Modalities of EN: In our study, 88% of respondents opted for continuous enteral nutrition. All senior physicians insisted on continuous EN. These practices are in line with international guidelines where it is recommended that continuous EN should be used rather than bolus nutrition [8,9]. Though some studies had not found a difference in terms of morbidity or mortality [17-19]. A recent study showed that bolus and continuous tube feeding achieved the same gastric volumes, insulin requirements, time to goal therapy, or calorie intake in critically ill patients [20]. In other studies, bolus feeding was reported to provide a greater stimulus for protein synthesis [21,22]. However, limited data is investigating the benefit and safety of intermittent feeding.
. EN complications management: Though it is commonly used for the evaluation of gastrointestinal dysfunction, continued measurements of GRV during an established EN may not be necessary. Conversely, 60% of the respondents used the GRV. Given that there is no literature evidence to support both sides of the argument, the majority favors the idea that GRV is not an appropriate tool to assess EN tolerance [23-26]. A recent French study has reported that the omission of GRV monitoring may improve patient outcomes [23]. Another argument against the use of GRV is that it leads to premature withholding, ESPEN as well as ASPEN recommend withholding EN only if GRV >500 ml [8,9]. In our study, 25% opted to withhold EN and 18% to start PN in case of gastrointestinal dysfunction. The results are in concordance with another study where gastrointestinal dysfunction causing intolerance to enteral nutrition was found to be a common reason for not starting, or discontinuing, feedings. [27]. A protocol that includes the use of prokinetics, gradually initiates the volume of nutritional delivery, and tolerates a higher gastric residual volume (up to 500 ml), should be considered to optimize the delivery of enteral nutrition in critically ill adult patients [8,9]. In addition, the use of erythromycin as a first-line prokinetic for critically ill patients with gastric feeding intolerance is recommended since it has a significant advantage of a better enteral feeding tolerance and a reduced risk of pneumonia [28-31]. Alternatively, intravenous metoclopramide or a combination of metoclopramide and erythromycin could be used. If a large (>500 mL) GRV still persists, the use of post-pyloric feeding should be considered over withholding EN, unless a new abdominal complication (obstruction, perforation, severe distension…) is suspected [8].
Parenteral nutrition
PN indications: Results have shown that physicians seem to excessively indicate PN than EN without considering the benefit-risk of the intervention. In our study, physicians indicated PN in case: gastrointestinal symptoms, severe traumatic brain injury, spinal cord injury, cerebrovascular accident, post-operative period and severe pancreatitis. These are not the proper indications of PN, they are rather indications for EN [8]. Our results are in line with another study where it is found that enteral nutrition is commonly not started in all eligible ICU patients [27]. We should insist that the latest ESPEN guidelines recommend that for patients who do not tolerate full dose of EN during the first week in the ICU, the safety and benefits of initiating PN should be considered on a case-by-case basis and that PN should not be started until all strategies to maximize EN tolerance have been attempted [8].
It is important to highlight that PN is only indicated when the enteral route is not feasible or contraindicated (short gut syndrome, abdominal compartment syndrome, proximal high output fistula without distal feeding access, bowel ischemia, bowel obstruction, severe gastrointestinal bleed, gastric aspirate >500 ml/6 h) [8,32]. Supplemental PN (PN combined to EN) could be considered when EN cannot meet the daily nutritional requirements. However, the optimal time to start PN in supplement with EN to meet the energy target is not clear and remains controversial [33]. The European (ESPEN) and American (A.S.P.E.N.) guidelines were found to be conflicting regarding the timing of PN initiation [8,9]. The ASPEN/SCCM recommends that the use of supplemental PN should be considered if EN alone is unable to meet >60% of energy and protein requirements [9]. While the ESPEN recommends that in patients who are not expected to be at optimal nutrition within 2-3 days should initiate PN within 24 to 48 h [8]. Another study reported that Supplemental PN in critically ill adults; when initiated 4 days after admission; could be an effective way to reduce nosocomial infection and should be considered as a strategy to improve clinical outcomes when EN is insufficient [34]. The timing of Supplemental PN is still under debate and needs further research. That could explain the variable practices reported by our responders regarding this subject.
Route of access: PN could be administrated via a central venous catheter or peripheral venous cannulas. Choosing the appropriate route of access is tightly dependent to several criteria: patient condition (state of health, current illness…), accessibility of the venous system, composition and osmolarity of the infused solution and the planned duration of PN. According to the ESPEN guidelines [35], central venous PN should be indicated in first attention in case of:
i) Long term estimated nutritional support.
ii) Need for hyperosmolar solutions (>850 mosmol/l).
iii) Solutions with Ph<5 or Ph>9.
iv) High nutrients requirement with severe fluid restrictions.
v) Number of calories needed to be administrated.
vi) Need for multiple intravenous treatments for a patient with poor peripheral venous access.
Importantly, PN solution’s osmolarity is considered one of the most crucial factors that determine the feasibility of peripheral PN. Parenteral solutions with an osmolarity>850 mosmol/l should be administered into central veins with high blood flow since peripheral administration may cause thrombophlebitis [35]. Conversely, a third of our responders reported that they do not adapt osmolarity to the route of access of PN.
7.2.3. Systematic supplementations: Daily supplementation of micronutrients (vitamins and trace elements) with PN is an integral part of nutritional support as the ESPEN recommend [8]. Commercially available PN solutions do not contain micronutrients for stability reasons. Consequently, micronutrients separate prescription is required [8]. Standard ranges for electrolytes, minerals and vitamins are age specific and based on normal organ function and normal losses without considering the increased need of critically ill patients. Therefore, the exact dose of supplementary micronutrients dose depends on several parameters such as the kidney function, daily losses and actual condition needs.
Situations in the intensive care: Obese patient’s nutrition support
Obese critically ill patients experience more complications than their lean counterparts with normal BMI [36]. Therefore, nutritional status evaluation is both crucial and challenging. Opinions diverged regarding adjusted body weight calculation. While ESPEN recommends adjusted body weight calculation, it was not recommended in other studies since it has variable definitions and may overestimate caloric needs [8,9,37]. This data could explain why adjusted body weight was not commonly uses by physicians asked in our survey. Additionally, respondents were unsure about the obese nutrition support particularities. This could be related to the fact that guidelines differ regarding this subject: The latest ESPEN guidelines recommend an iso-caloric high protein diet in obese patients, preferentially guided by indirect calorimetry measurements and urinary nitrogen losses. And if indirect calorimetry is not available, energy intake can be based on “adjusted body weight” calculated: (actual body weight -ideal body weight) *0.25 + ideal body weight. If urinary nitrogen losses or lean body mass determination are not available, protein intake can be 1.3 g/kg “adjusted body weight”/d [8]. While the latest ASPEN guidelines recommend using 11-14 kcal/kg ABW/day for BMI of 30-50 and 22-25 kcal/kg IBW/day for BMI > 50 [9,38]. As for protein requirements, the ASPEN recommend using a weight-based simplistic equation of 2.0 g/kg IBW/day (for classes I and II: 30 < BMI ≤ 39.9) and 2.5 g/kg IBW/day (for class III: BMI ≥ 40) [9].
Limitations
A limitation of the study is the moderate sample of participating physicians. Because participation was voluntary, we are aware of this potential bias. Consequently, it is likely that the evidence–practice gap that we found in our study may be larger. The moderate response to our invitation to participate illustrates that critical care practitioners need to be encouraged to improve the provision of nutrition therapy in their ICU. However, the major strength of this study is the observation of actual nutrition practices compared to evidence-based recommendations in multiple ICUs across different regions of the country. Another limitation, the questionnaire was not formally validated; still, the aim of the study was to have a first estimation of the attitudes and self-perceived practices of critical care physicians regarding nutrition support, not to create a validated instrument. Indeed, we aimed to identify current practices to foresee a better implementation of clinical recommendations. The benchmarking aspect of this study, highlighting what is done compared to what is recommended, provides a powerful baseline for individual critical care practitioners and researchers alike.
Conclusion
This is the first Tunisian survey that has investigated the current practices of nutritional care to foresee a successful implementation of best practices in different Tunisian intensive care units. Results have shown that physicians responded differently regarding nutritional management. Adherence to evidence-based guidelines was low, which partly explains how nutritional status worsens before and after hospitalization. Nutritional support was found sometimes found sporadic and multiple nutritional aspects were blurred. We conducted this survey to collect baseline data that would serve as a solid ground to identify factors that limit or restrict the implementation of best practices so that interventions can be selected to address these barriers and improve nutritional care delivery. To achieve adequate practice of recommendations, many elements need to be tackled. Future interventions should be multi-levelled insisting on a training, assistance and multidisciplinary implication program. It was not the intent of the current study to explain why variation exists or to elucidate the low adherence. However, additional research should be encouraged to complement this initial work. Specifically, research targeting the importance of having a clinical nutrition physician per unit with an appropriate dietitian to maximize nutrition support practices in all its aspects.
Funding Statement
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Disclosure of Interest: None declared.
References
- Ridley E, Gantner D, Pellegrino V (2015) Nutrition therapy in critically ill patients- a review of current evidence for clinicians. Clin Nutr 34(4): 565-571.
- Heyland DK (1998) Nutritional support in critically ill patients. A critical review of the evidence. Crit Care Clin 14(3): 423-440.
- Heyland DK, Heyland J, Dhaliwal R, Madden S, Cook D (2010) Randomized trials in critical care nutrition: look how far we’ve come! (and where do we go from here. JPEN J Parenter Enteral Nutr 34(6): 697-706.
- Alberda C, Leah G, Naomi J, Khursheed J, Andrew GD, et al. (2009) The relationship between nutritional intake and clinical outcomes in critically ill patients: results of an international multicenter observational study. Intensive Care Med 35(10): 1728-1737.
- Mogensen KM, Clare MH, Steven WP, Takuhiro M, James DR, et al. (2018) Malnutrition, Critical Illness Survivors, and Postdischarge Outcomes: A Cohort Study. JPEN J Parenter Enteral Nutr 42(3): 557-565.
- Wray CJ, Mammen JMV, Hasselgren PO (2002) Catabolic response to stress and potential benefits of nutrition support. Nutrition 18(11): 971-977.
- Wischmeyer PE, Heyland DK (2010) The future of critical care nutrition therapy. Crit Care Clin 26(3): 433-441.
- Singer P, Annika Reintam B, Mette MB, Waleed A, Philip CC, et al. (2019) ESPEN guideline on clinical nutrition in the intensive care unit. Clinical Nutrition 38(1): 48-79.
- McClave SA, Beth ET, Robert GM, Malissa MW, Debbie RJ, et al. (2016) Guidelines for the Provision and Assessment of Nutrition Support Therapy in the Adult Critically Ill Patient: Society of Critical Care Medicine (SCCM) and American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.). JPEN J Parenter Enteral Nutr 40(2): 159-211.
- Cahill NE, Dhaliwal R, Day AG, Jiang X, Heyland DK (2010) Nutrition therapy in the critical care setting: what is “best achievable” practice? An international multicenter observational study. Crit Care Med 38(2): 395-401.
- De Jonghe B, Appere-De-Vechi C, Fournier M, Tran B, Merrer J, Melchior JC, et al. (2001) A prospective survey of nutritional support practices in intensive care unit patients: What is prescribed? What is delivered. Crit Care Med 29(1): 8-12.
- O’Meara D, Eduardo Mireles C, Fran F, A Christine H, Jeffrey H, et al. (2008) Evaluation of delivery of enteral nutrition in critically ill patients receiving mechanical ventilation. Am J Crit Care 17(1): 53-61.
- Yu A, Xie Y, Zhong M, Wang F, Huang H, et al. (2021) Comparison of the Initiation Time of Enteral Nutrition for Critically Ill Patients: At Admission vs. 24 to 48 Hours after Admission. Emerg Med Int 2021: 3047732.
- Kim H, Stotts NA, Froelicher ES, Engler MM, Porter C (2012) Why patients in critical care do not receive adequate enteral nutrition? A review of the literature. J Crit Care 27(6): 702-713.
- Li PF, Wang YL, Fang YL, Nan L, Zhou J, et al. (2020) Effect of early enteral nutrition on outcomes of trauma patients requiring intensive care. Chinese J Traumatol 23(3):163-167.
- McClave SA, Marsano LS, Lukan JK (2002) Enteral access for nutritional support: rationale for utilization. J Clin Gastroenterol 35(3): 209-213.
- Serpa LF, Kimura M, Faintuch J, Ceconello I (2003) Effects of continuous versus bolus infusion of enteral nutrition in critical patients. Rev Hosp Clin Fac Med Sao Paulo 58(1): 9-14.
- Tavares De Araujo VM, Gomes PC, Caporossi C (2014) Enteral nutrition in critical patients; should the administration be continuous or intermittent. Nutr Hosp 29(3): 563-567.
- Rhoney DH, Parker D, Formea CM, Yap C, Coplin WM (2002) Tolerability of bolus versus continuous gastric feeding in brain-injured patients. Neurol Res 24(6): 613-620.
- Evans DC, Rachel F, Christian J, Robert C, Chinedu N, et al. (2016) Continuous versus bolus tube feeds: Does the modality affect glycemic variability, tube feeding volume, caloric intake, or insulin utilization. Int J Crit Illn Inj Sci 6(1): 9-15.
- Patel JJ, Rosenthal MD, Heyland DK (2018) Intermittent versus continuous feeding in critically ill adults. Curr Opin Clin Nutr Metab Care 21(2): 116-120.
- Bear DE, Hart N, Puthucheary Z (2018) Continuous or intermittent feeding: pros and cons. Curr Opin Crit Care 24(4): 256-261.
- Reignier J, Emmanuelle M, Amelie Le G, Thierry B, Arnaud D, et al. (2013) Effect of not monitoring residual gastric volume on risk of ventilator-associated pneumonia in adults receiving mechanical ventilation and early enteral feeding: a randomized controlled trial. JAMA 309(3): 249-256.
- Moreira TV, McQuiggan M (2009) Methods for the assessment of gastric emptying in critically ill, enterally fed adults. Nutr Clin Pract 24(2): 261-273.
- Zaloga GP (2005) The myth of the gastric residual volume. Crit Care Med 33(2): 449-450.
- Kuppinger DD, Rittler P, Hartl WH, Rüttinger D (2013) Use of gastric residual volume to guide enteral nutrition in critically ill patients: a brief systematic review of clinical studies. Nutrition 29(9): 1075-1079.
- Heyland D, Cook DJ, Winder B, Brylowski L, Van deMark H, et al. (1995) Enteral nutrition in the critically ill patient: a prospective survey. Crit Care Med 23(6): 1055-1060.
- Chapman MJ, Fraser RJ, Kluger MT, Buist MT, De Nichilo DJ (2000) Erythromycin improves gastric emptying in critically ill patients intolerant of nasogastric feeding. Crit Care Med 28(7): 2334-2337.
- Berne JD, Scott HN, Clyde EM, Van LV, David V, et al. (2002) Erythromycin reduces delayed gastric emptying in critically ill trauma patients: a randomized, controlled trial. J Trauma 53(3):422-425.
- Reignier J, Bensaid S, Perrin-Gachadoat D, Burdin M, Boiteau R, et al. (2002) Erythromycin and early enteral nutrition in mechanically ventilated patients », Crit Care Med 30(6): 1237-1241.
- Yavagal DR, Karnad DR, Oak JL (2000) Metoclopramide for preventing pneumonia in critically ill patients receiving enteral tube feeding: a randomized controlled trial. Crit Care Med 28(5): 1408-1411.
- Reintam Blaser A, Joel S, Waleed A, Mette MB, Micheal PC, et al. (2017) Early enteral nutrition in critically ill patients: ESICM clinical practice guidelines. Intensive Care Med 43(3): 380-398.
- Aguilar-Nascimento JE, Bicudo-Salomao A, Portari-Filho PE (2012) Optimal timing for the initiation of enteral and parenteral nutrition in critical medical and surgical conditions. Nutrition 28(9): 840-843.
- Heidegger CP, Mette M Berger, Séverine G, Walter Z, Patrice D, et al. (2013) Optimisation of energy provision with supplemental parenteral nutrition in critically ill patients: a randomised controlled clinical trial. Lancet 381(9864): 385-393.
- Pittiruti M, Hamilton H, Biffi R, MacFie J, Pertkiewicz M (2009) ESPEN Guidelines on Parenteral Nutrition: Central Venous Catheters (access, care, diagnosis and therapy of complications). Clinical Nutrition 28(4): 365-377.
- Choban P, Dickerson R, Malone A, Worthington P, Compher C, et al. (2013) American Society for Parenteral and Enteral Nutrition, A.S.P.E.N. Clinical guidelines: nutrition support of hospitalized adult patients with obesity. JPEN J Parenter Enteral Nutr 37(6): 714-744.
- McClave SA, Robert K, Charles W Van, Matt C, Mark D, et al. (2011) Nutrition therapy of the severely obese, critically ill patient: summation of conclusions and recommendations. JPEN J Parenter Enteral Nutr 35(5 Suppl): 88S-96S.
- Robinson MK, Kris M, Jonathan DC, Caitlin KM, Takuhiro M, et al. (2015) The relationship among obesity, nutritional status, and mortality in the critically ill. Crit Care Med 4(1): 87-100.






























