Artichoke (Cynara Cardunculus L) Milk Clotting Enzyme Characteristics and its Impact in Cephalotyre (Ras) Cheese Production
Osama A Ibrahim1*, Howida A El Sayed2 and Diaa A Marrez3
1Dairy Science Department, Industries and Nutrition Research Institute, National Research Centre, Cairo, Egypt
2Dairy Research Department, Food Technology Research Institute, Agricultural Research Centre, Giza, Egypt
3Toxicology and Food Contaminants Department, Industries and Nutrition Research Institute, National Research Centre, Cairo, Egypt
Submission: June 30, 2023; Published: July 27, 2023
*Corresponding author: Osama A Ibrahim, Dairy Science Department, Industries and Nutrition Research Institute, National Research Centre, Cairo, Egypt. E-mail: osama_nrc@hotmail.com
How to cite this article: Osama A I, Howida A El S, Diaa A M. Artichoke (Cynara Cardunculus L) Milk Clotting Enzyme Characteristics and its Impact in Cephalotyre (Ras) Cheese Production. Nutri Food Sci Int J. 2023. 12(2): 555835. DOI: 10.19080/NFSIJ.2023.12.555835.
Abstract
Calf rennet substitutes from plants are still continuous with high milk clotting activity compared to their proteolytic activity especially in hard cheese which had long ripening period. Milk clotting enzyme (MCE) from artichoke (Cynara cardunculus L. ssp. scolymus) flowers as a plant coagulant was characterized and used in Cephalates (Ras) cheese production as the most popular hard cheese in Egypt in comparison of both calf rennet and microbial coagulant. Also, chemical, textural, sensorial, and microbiological properties of cheese during ripening period were investigated. MCE activity from artichoke flowers was decreased as the level of NaCl increased with 25% reduction at 5% NaCl level. It was characterized as a thermal stable coagulant with 90-95% residual activity after incubation at 40°C for 30-60 min; while pH stability showed their activity lies in acidic pH condition. It’s possible that artichoke MCE can coagulate various milk species. SDS-PAGE of casein proteolysis using artichoke MCE promoted extensive cleavage of k-CN, αs- and β-CN fractions within 120 min at the optimal reaction condition. Moreover, chemical, and microbiological characterization of Ras cheese coagulated with partial purified artichoke MCE was similar to Ras cheese made with commercial coagulants; while after 3 months of ripening the water-soluble nitrogen of Ras cheese coagulated with artichoke MCE was significantly (p≤0.05) higher than Ras cheese made with commercial coagulants. It could be concluded that artichoke MCE could be used in Ras cheese production without any defects during their ripening period for 4 months.
Keywords: Calf Rennet Substitute; Ras Cheese; Plant Coagulant; Artichoke Waste
Abbreviations: GLM: General Linear Model; MCA: Milk Clotting Activity
Introduction
Ras cheese as a most popular hard cheese in Egypt due to unique taste and aroma. Ras cheese which similar to Greek type “Cephalotyre” made from cow’s milk or a mixture of cow’s and buffalo’s milk. It is normally marketed after the ripening period of 4-6 months [1]. Conversion of milk into cheese with long shelf life was preserved the milk nutrients that have played a key role in human nutrition [2]. Calf rennet is the oldest known application of enzymes in cheese processing [3]. As a conventional MCE, it was extracted from the suckling calves’ fourth stomach (high level of chymosin against low level of pepsin), this enzymes proportion is inverted from adult animals [4]. Likewise, the overall expanding of cheese production with shorting of calf rennet providing are prompts looking for its alternatives from effectively and accessible resources [5]. Also, calf rennet may be restricted for religious reasons, diet (vegetarianism), or consumer concern regarding genetically engineered foods [6]. However, many scientific investigations have been focused on plant coagulant extracted from Solanum dubium [7], Bromelia hieronymi, Moringa oleifera & Carica papaya [8-11], and Calotropis procera [12] among others. Cheese made with vegetable coagulant can be found mainly in Mediterranean, West African, and southern European countries [13]. Also, in West African countries, like Nigeria and the republic of Benin, extracts from Calotropis procera have been used in traditional cheese making [6]. Albizia julibrissin seed extract has been used as a milk coagulant without bitterness developed after three months of cheese ripening [14]. Bruno et al. [8] reported that the cheese produced using coagulating extract from Bromelia hieronymi fruits was acceptable in appearance, body, texture, and flavor. On the other hand, some of these plant coagulants were found inappropriate due to excess proteolytic activity which leading to appear defects in cheese flavor and texture [15]. Thus, searching for calf rennet substitute from plants is still continuous with high milk clotting activity (MCA) in comparison of its proteolytic activity to overcome the above-mentioned problems. Also, cheese production with plant coagulants contributes significantly to the socioeconomic development of a locality, region and hence the whole country where it is produced [11]. Although, the wide spreading uses of artichoke as a food and medicine by the ancient Egyptians, Greeks and Romans, as well as in Egypt globe artichoke is becoming one of the most important vegetable crops grown for local consumption and export to European countries; while using of artichoke waste as a source of milk coagulant, MCE biochemical characteristics, suitability for coagulating of different milk species, as well as its effect on hard cheese making during their long ripening period are still limited.
Hence, the aim of the present study was to extract and partial purify MCE from artichoke (Cynara cardunculus L. ssp. scolymus) flowers as a suitable source, cheap and available; then biochemical characteristics were studied including pH stability, thermal stability, effect of NaCl on MCE activity, capability of MCE activity on different milk species coagulation, and In vitro hydrolysis of casein compared to other coagulants. Also, investigate its effect as a calf rennet alternative on chemical, rheological, sensorial, and microbiological properties of Cephalotyre (Ras) cheese during their ripening period in comparison of both calf rennet and microbial coagulant, in order to determine its technological suitability for Ras cheese production as the most popular hard cheese in Egypt.
Materials and Methods
Raw materials and chemicals
Artichoke (Cynara cardunculus L. ssp. scolymus) flowers were obtained from the Egyptian local market as a waste produced from cleaning and preparing artichoke. The flowers were collected and then dried for about three weeks at 25°C and shielded from light. Fresh cow’s and buffalo’s milk were obtained from Faculty of Agriculture, Cairo University. Fresh goat’s and sheep’s milk were obtained from the herd of Desert Research Center, Egypt. The starter culture used in the cheese manufacture consisted of the mixture of Lactococcus lactis subsp. lactis and Lactococcus lactis subsp. cremoris were obtained from Egyptian microbial culture collection, (MIRCEN), Ain Shams University. Skim milk powder was purchased from BIELMLEK Spoldzielnia mleczrska, Poland. Other chemicals were of analytical grade.
Methods
Extraction and partial purification of MCE
Extraction of MCE from the dried flowers (10%) of artichoke (Cynara cardunculus L. ssp. scolymus) was performed according to Nouani et al. [16] using different extraction buffers including 0.2 M sodium acetate pH 5.0, 0.2 M sodium acetate containing 5% NaCl, distilled water, and distilled water containing 5% NaCl. After macerating for 24 h/5 °C, the extract was filtrated through gauze and centrifugation (5,000 xg, 20 min, 5°C), the recovered fractions were pooled and considered as a crude enzymatic extract, and then MCE activity, and protein content were determined. MCE of crude extract was partial purified using ammonium sulphate (20- 40%) procedure according to Scopes [17]. The rich MCE active fraction was dialyzed against 0.2 M acetate buffer pH 5.0 for 24 h at 5°C. MCE activity and protein content of the recovered fractions were determined.
Milk-clotting activity (MCA)
MCA of all prepared fractions were measured according to the method of IDF [18]. MCA is expressed as Soxhlet unit (SU), which calculated using the following equation:
SU/ml = M×2400 / E× t
Where M is the substrate volume (ml)
E is the enzyme extract volume (ml)
t is the clotting time (sec).
One SU of MCA was defined as the amount of enzyme required to clot 1 ml of substrate within 40 min at 35°C. The specific activity is calculated by dividing the MCA to the protein content.
Protein content of MCE fractions
Protein content for all prepared artichoke enzymatic fractions were carried out as described by Bradford [19] procedure using coomassie brilliant blue G-250 dye (Bio-Rad, USA). The protein concentration was measured from the calibration curve of Bovine Serum Albumin (BSA) standard (Mallinckrodf, France).
Specific activity calculation
The specific activity is calculated by dividing the determined MCA to the protein content.
Biochemical characterization of MCE fractions
Biochemical characteristics of MCE from artichoke (Cynara cardunculus L. ssp. scolymus) flowers were determined as follows:
i. Effect of NaCl concentration on MCA: Effect of different concentrations of NaCl ranging from 1-5% on MCA was determined.
ii. pH stability:
the residual MCA was measured after preincubation. The enzymes in different buffers ranged in pH values of 5-8 using 0.2 M acetate buffer (pH 5.0), 0.2 M phosphate buffer (pH 6.0-7.0), and 0.2 M Tris-HCl buffer (pH 8.0) for 30 and 60 min.iii. Thermal stability: MCE pre-incubated at different temperature ranging from 50 to 80 °C for 30 and 60 min, and then the residual MCA was determined.
iv. Determination of suitability of MCA on different milk species: MCA was determined using different milk types such as: cow, buffalo, goat, and sheep milk.
In vitro hydrolysis of casein by different coagulants
The hydrolysis of whole casein (Sigma Chemical Co., USA) by artichoke MCE compared to calf rennet, microbial coagulant using Sodium Dodecyl Sulphate-Polyacrylamide Gel Electrophoresis (SDS-PAGE) method according to Laemmi [20]. The whole casein in 100 mM sodium phosphate buffer pH 6.5 was incubated at 60°C for 120 min with different coagulants. For the electrophoretic analysis, 300 μL of 0.125 M Tris-HCl buffer, pH 6.8, containing 0.1% (w/v) SDS, 5% (v/v) 2-mercaptoethanol, 10% (v/v) glycerol, and 0.01% (w/v) bromophenol blue were added to each hydrolysate solution. The latter was then boiled at 100°C for 3 min before electrophoretic analysis. The protein samples were loaded for electrophoresis at 50 V which performed in a vertical slab gel apparatus in 12.5% polyacrylamide separating gel, 4% stacking gel. Protein samples were visualized by Coomassie brilliant blue R-250. The pre-stained standard proteins in the range of 10–250 kDa (Thermo scientific) were used as a molecular weight marker.
Ras cheese manufacture
Ras cheese was made by the conventional method as described by Hofi et al. [21]. The first Ras cheese batch coagulated with calf rennet (Mifad, Misr food additives, Egypt, T1); the second batch was made with microbial coagulant (RENIPLUS, Caglio star, Spain, T2); while the third and fourth batches were coagulated with crude (T3) and partial purified MCE (T4) from artichoke (Cynara cardunculus L. ssp. scolymus) dried flowers, respectively. Fresh cheese samples and after 1, 2, 3 and 4 months of ripening at 12±1°C were taken for analysis. The whole experiment was repeated in duplicate and each analysis in duplicate and average results were tabulated.
Chemical analysis of cheese
Ras cheese samples were chemical analyzed for moisture, ash, protein, acidity and water-soluble nitrogen (WSN) contents were determined according to AOAC [22]. Total volatile fatty acids (TVFA) were determined according to Kosikowiski [23]. The pH on warm water macerates was determined using a digital pHmeter with glass electrodes, Ingold, Knick, Germany.
Texture profile analysis (TPA) of cheese
TPA of Ras cheese during their ripening period was performed with a Universal Testing Machine (Co metech, B type, Taiwan) using 25-mm-diameter perplex conical-shaped probe, and then the generated plot of force (N) versus time(s) was recorded. TPA parameters were determined according to the definition given by IDF [24] from the resulting force-time curve for textural attributes such as hardness, chewiness, cohesiveness, gumminess, and springiness were calculated.
Microbiological analysis of cheese
Ras cheese made with different coagulants was evaluated on the presence count of the key pathogenic and spoilage microbial populations after 0, 2, and 4 months of ripening. Representative Ras cheese samples (10 g) were taken and homogenized in 90 ml of 0.9% NaCl for 30 s. Serial 10-fold dilutions were prepared in saline tubes and 1 ml of solution was used for microbial counting. Microbial enumeration was performed using pour plate method according to APHA [25] and FDA [26]. Total viable counting was performed using the plate count agar medium and aerobically incubated at 35 °C for 48 h, the plates were analyzed for total growing population (cfu/g) as recommended by FDA [26]. Molds and yeasts counts were examined using malt extract agar and aerobically incubated at 30 °C for 3 days the plates were analyzed (cfu/g) for yeast and fungal population [27]. The psychrotrophic bacteria count was performed by adding samples (0.1 ml) in duplicate on the surface of plate count agar. The plates were inverted and incubated at 7°C for 10 days. Proteolytic bacteria were enumerated on milk plate count agar and incubated at 37°C/ 48hrs. The presence of clear zone around the colonies against an opaque with background indicated proteolytic colonies [28]. Total and fecal Coliforms counting, and E. coli detection were performed as described by El-Hadedy and Abu El-Nour [29]. For each sample, 9 ml Bromo-Cresol Purple MaCconkey broth tube was inoculated with 1 ml of the first three dilutions (3 tubes for each dilution). Positive tubes were indicated by the production of acid and gas upon incubation at 37°C for 24 hours, and the count was estimated statistically from the MPN index [30]. From each positive tube, a new MaCconkey broth tube was inoculated with one loopful, and incubated at 45.5°C for 24hr, to detect the fecal coliforms by the presence of acid and gas. EMB Agar plate was streaked from the positive tube (45.5°C), and E. coli was distinguished as purple colonies with green metallic sheen. Further biochemical behavior was confirmed by Catalase (+) and H2S (-), IMViC test, and microscopic examination (short rods, gram negative, non-spore forming). Detection of Salmonella and Shigella was performed according to APHA [25], and FDA [26]. Homogenized Ras cheese samples (25g) were added to 225 ml of sterile buffered peptone water for pre-enrichment. After incubation at 37°C/ 24hr, 10 ml of growth suspension were transferred to 90 ml boiling sterilized Selenite broth supplemented with 4 g/L sodium bi-selenite (Oxoid) and incubated at 37°C/ 24 h. After incubation, XLD plates were streaked from selenite broth [31,32]. Colonies were detected as Salmonella when appeared red with black centers or Shigella when appeared reddish. The presence of Salmonella was confirmed by recording Catalase positive; urease negative; gram negative short rods. Further confirmation was made through the Triple Sugar Iron test. The positive isolate showed Glucose fermentation (yellow button), H2S positive (Blackening), but negative gas production.
Sensory evaluation of Ras cheese
Sensorial attributes of Ras cheese during their ripening period at 12±1°C were assessed by means of regular panelists from the researcher’s staff of Dairy Research Department, Food Technology Research Institute, with a maximum score points of 50 points for flavor, 40 points for body and texture, and 10 points for the cheese appearance as described by Pappas et al. [33].
Statistical analysis
Statistical analysis of the results was performed using ANOVA procedure for analysis of variance, and the general linear model (GLM) procedure of SAS software [34]. The results were expressed as mean ± standard error and the difference between means were tested for the significance at (p≤0.05) using Duncan’s multiple range tests.
Results and Discussion
MCE in the present study was detected and characterized from artichoke (Cynara cardunculus L. ssp. scolymus) dried flowers as a plant coagulant for a model of hard cheese with long ripening period such as Cephalotyre (Ras) cheese as a most popular hard cheese in Egypt. Moreover, its technological suitability impact on chemical, textural and sensorial properties of Ras cheese compared to both commercial calf rennet and microbial coagulant were investigated to confirm their suitability as a calf rennet substitute.
Extraction of MCE from Artichoke Flowers
MCE activity was determined on dried flowers of artichoke (Cynara cardunculus L. ssp. scolymus) using different extraction buffers with/without NaCl (5%) as presented in (Figure 1). It might be noted that the highest MCA as well as its specific activity were observed in artichoke dried flowers dissolved in 0.2M acetate buffer (pH5.0). These results agree with Abebe and Getachew [11], and Nestor et al. [35] who detected MCE on different plants using sodium acetate pH 5.0. Hence, MCE was extracted from artichoke dried flowers using 0.2 M sodium acetate pH 5.0 in order to partial purify using ammonium sulfate precipitation procedure (20- 40%) as earlier described by Abd El-Salam et al. [36] (data not shown). However, for the purification of MCE from various plant sources, low saturation concentrations of ammonium sulfate are preferable to high concentrations [10,37]. Additionally, ASP makes it easier to concentrate the enzyme extract into a volume that can be used effectively for milk coagulation in cheesemaking [38]. When combined with the availability of plant waste, onestep ASP has been considered an economical purification method that makes it possible to produce MCE on a large scale.

Biochemical characterization of MCE
Sodium chloride is widely used as a preservative in food and dairy products [39]. (Figure 2) shows that the activity of MCE from artichoke violet flowers gradually decreased as the level of sodium chloride increased. It could be due to induce conformational changes in the enzyme molecule affected with high level of sodium chloride which leading to decrease of its MCA. Also, the addition of NaCl decreased the rate of enzymatic reaction and coagulation of renneted micelles [40]. These findings agree with Ahmed et al. [41] and Bakr et al. [42] who recorded the highest MCA at the lower level of NaCl for MCE extracted from Bacillus stearothermophilus and Pleurotus florida, respectively.

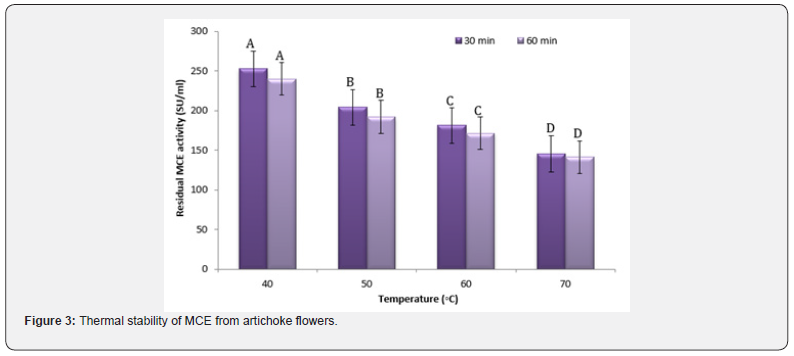
Thermal behavior of the enzyme is a useful property in cheese making process, as it can be quickly inactivated by moderate heating [43]. Thermal stability of MCE from artichoke dried flowers was shown in (Figure 3). The results revealed that the residual MCA decreased as both temperature and pre-incubation time was increased, which due to the conformational changes in the protein structure under high temperatures during the preincubation time. The results indicated that 90-95% retention of artichoke partial purified MCE after incubation at 40°C. Heating to 50°C reduced the MCA by 77-72% after pre-incubation for 30- 60 min, respectively, while its activity was quickly diminished after incubation at 70 °C. These results are similar with various plants MCE which have been mentioned by Tajalsir et al. [38], Raposo and Domingos [44]. Hence, partial purified MCE from artichoke flowers thermal stability at higher temperatures (40- 50°C) behaved like MCE extracted from other plant (30-60°C) and microbial sources [35, 38]. However, it was characterized as a thermal stable coagulant compared to calf rennet, since calf rennet reached its maximum activity at 45°C, followed by a sharp decline when the temperature exceeded 50°C [45].
(Figure 4) shows the pH stability of artichoke MCE. The results showed that the residual activity of the partial purified MCE from artichoke flowers decreased as both pH and pre-incubation time were increased. It could be mainly due to the conformational changes in the protein structure of MCE under alkaline pH condition, since pH changes alter the conformation of the enzyme structure including the binding of the substrate and the catalytic activity of the group at the active site of the enzyme [46]. However, Abdeen et al. [10], and Nouani et al. [16] reported that the optimal pH of MCE activity form various plants lies in acidic pH condition. Also, MCE isolated from Cyanara scolymus L. flowers showed 87% loss of its activity at pH 7.0 [47]. This is a great advantage for dairy industrial application as the partial purified MCE extracted from artichoke flower shows a high specificity of action in the acidic pH range that could be easily controlled in the industrial application as designed [48].

The suitability of partial purified MCE from artichoke dried flowers towards different milk species (e.g., cow, buffalo, goat, and sheep milk) were shown in (Figure 5). Artichoke MCE may be able to coagulate the various examined milk species with different coagulation time started with goat milk followed by cow, buffalo and sheep milk. It could be due to the different compositional components of milk types, as well as the difference of casein fractions content among the studied milk types which affects their coagulation time [49]. Park et al. [50] mentioned that the casein micelles diameter, hydration, and mineralization generally leads to shorter coagulating time, weaker gel consistency, and decreased yield of the cheese curd. Similar results were mentioned by Barbagallo et al. [51] who reported the caprine, ovine and bovine milk.
coagulated with (Cynara cardunculus L.) flowers. Hence, the effect of artichoke MCE towards different milk types supports their suitability as a calf rennet alternative in cheese processing.

In vitro casein hydrolysis by different coagulants
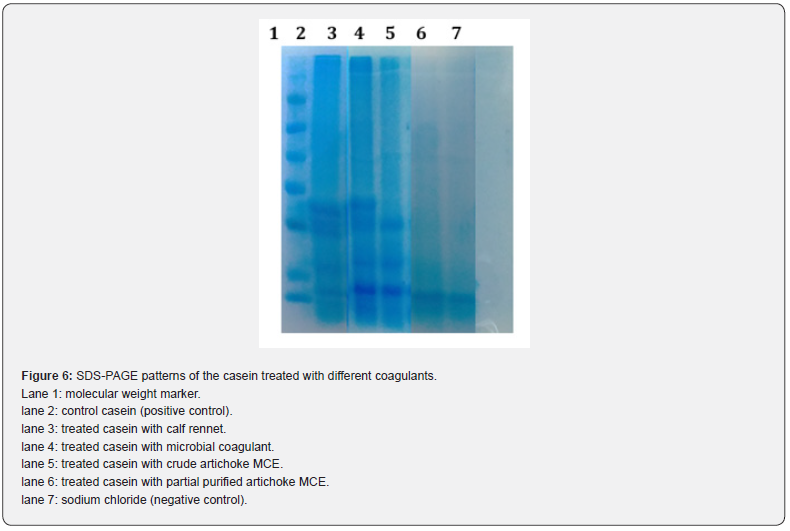
The SDS-PAGE of casein proteolysis by MCE from artichoke (Cynara cardunculus L. ssp. scolymus) flowers compared to calf rennet and microbial coagulant as positive controls (lanes 2-3) and 0.15M NaCl as a negative control (lane 6) showed that the order of hydrolysis obtained with artichoke MCE was the following: ᴋ-CN, αs-CN, and β-CN (Figure 6). Most of ᴋ-CN, β-CN and αs-CN components disappeared after 120 min of hydrolysis by artichoke MCE (Figure 7), lanes 4-5). Similar casein hydrolysis rate observed with Tavaria et al. [52] and Ordiales et al. [53] who found higher hydrolysis rate of αs-CN than on β-CN by dry flowers of C. cardunculus. Also, Cavalli et al. [54] reported that S. marianum extract induced extensive degradation of αs-CN, but only a slight degradation of β-CN. Pontual et al. [37] found that the partial purified MCE from Moringa oleifera flowers promoted extensive cleavage of ᴋ-CN and low level of αs- and β-CN hydrolysis. However, the untreated casein (lane 1) shows three main bands corresponding to αs-, β-and ᴋ-CN whose apparent molecular weights are ~34 kDa, 28 kDa and 28 kDa, respectively. This order of hydrolysis was similar to the order of susceptibility of the different casein components in whole bovine, caprine, and ovine caseins toward the action of animal rennet [55].
Chemical changes of Ras cheese
The chemical changes of Cephalotyre (Ras) cheese during their ripening period which coagulated with artichoke (Cynara cardunculus L. ssp. scolymus) flowers in the form of crude (T3) and partial purified (T4) MCE compared to both calf rennet (T1) and microbial coagulant (T2) were showed in (Table 1). The results showed that the moisture content of Ras cheese coagulated with artichoke MCE were slightly higher than those of cheese coagulated by calf rennet or microbial coagulant with significantly (p≤0.05) differences after 4 months of ripening. These findings are in accordance with El-Siddig et al. & El-Kholy [9, 56] who observed the higher moisture content of cheese using vegetable coagulant than cheese manufactured with calf rennet. Moreover, there is no significant difference in the moisture content of Ras cheese coagulated with partial purified artichoke MCE (T4) in comparison of commercial coagulants-made cheese. In addition, the moisture contents of all resulting Ras cheese treatments were decreased as the ripening period prolonged with significantly differences after 4 months of ripening.


T1: liquid calf rennet; T2: microbial coagulant powder; T3: crude MCE form artichoke flowers; T4: partial purified MCE form artichoke flowers. All parameters are represented as means of replicating ±standard error. Means with different small superscript letters in the same row and different capital superscript letters in the same column are significantly different at p ≤0.05.
Also, (Table 1) shows that significantly (p≤0.05) differences in the protein/dry matter (P/Dm) ratio of Ras cheese made with crude artichoke MCE coagulant (T3) in comparison of those coagulated with commercial coagulants. No significant differences of P/Dm ratio of Ras cheese made with partial purified artichoke MCE coagulant (T4) compared to commercial coagulants-made cheese. Similar results concerning the cheese P/Dm ratio were observed by Pino et al. [57] who found no significant differences between cheese treatments coagulated with calf rennet or plant coagulant. Moreover, the P/Dm ratio of all resulted Ras cheese were significantly (p≤0.05) increased as their ripening period progressed which mainly due to lose the moisture and increase of the cheese protein content during the cheese ripening.
It could be seen from (Table 1) that there were no significant differences of fat/dry matter (F/Dm) ratio of Ras cheese made with artichoke coagulant in comparison of those coagulated with both calf rennet and microbial coagulant. Also, the results showed that F/Dm ratio was increased in all Ras cheese treatments throughout their ripening period progressed which mainly due to change of the moisture content of cheese and increase of fat content of cheese during their ripening period [56]. The ash content of Ras cheese coagulated with artichoke MCE was slightly higher than commercial coagulants-made Ras cheese without significantly differences as illustrated in (Table 1). It could be due the minerals content of the added artichoke extracts especially in their crude extract. These results were in harmony with those observed by Abd El-Salam et al. & Tejada et al. [36, 58]. Moreover, the ash contents of all resulted Ras cheese were increased as their ripening period prolonged which mainly due to the moisture reduction during cheese ripening [59]. Moreover, (Table 1) shows that the titratable acidity (%) of artichoke Ras cheese with opposite trends in its pH values were higher than those coagulated with commercial coagulants without significantly differences. It could be probably due to the proteolytic activity of artichoke MCE extracts as mentioned in earlier study [36], combined with other acidic components resulted from the fermentation of residual lactose and degradation of cheese fat through their ripening period [56,59]. These results are in accordance with those noticed by El-Siddig et al. [9], and Al-Jasser and Al-Dogan [60]. Also, Abu- Zeid [61] observed the higher acidity level in cheese made with plant coagulant from Sonchus olerceus L. with long coagulation time which possibly favored microbial growth and consequently leads to higher acidity in the curd from plant origin coagulant.
Ripening indices of Ras cheese
WSN as a ripening index of cheese reflects the extent of proteolysis through their ripening period [62]. WSN content of Ras cheese coagulated with artichoke (Cynara cardunculus L. ssp. scolymus) MCE in comparison of control Ras cheese coagulated with both calf rennet and microbial coagulant were presented in (Figure 7). The results showed that WSN of artichoke-made cheese were higher than those coagulated with commercial coagulants during their ripening period with significantly (p≤0.05) differences after 3 months of ripening. It could be attributed due to intense proteolytic activity of plant coagulant compared to animal or microbial coagulants. Moreover, WSN content of Ras cheese coagulated with crude artichoke MCE was higher than Ras cheese coagulated with partial purified MCE from artichoke during their ripening period mainly due to the higher proteolytic activity of crude extract than partial purified MCE [36]. These findings agree with those reported by El-Kholy [56], and Pino et al. [57] who indicated that cheese made with artichoke extract as coagulant exhibited higher levels of WSN than cheese made with calf rennet. In addition, WSN contents of all resulted Ras cheese were increased as their ripening period prolonged which mainly due to the moisture reduction during ripening [59]. However, WSN in cheese is primarily formed by enzymatic coagulants, plasmin or cell-wall envelope proteases during the early stage of proteolysis. It is well recognized that protein degradation breakdown is an essential factor for equally flavors and texture of cheese throughout its ripening period [63,64].
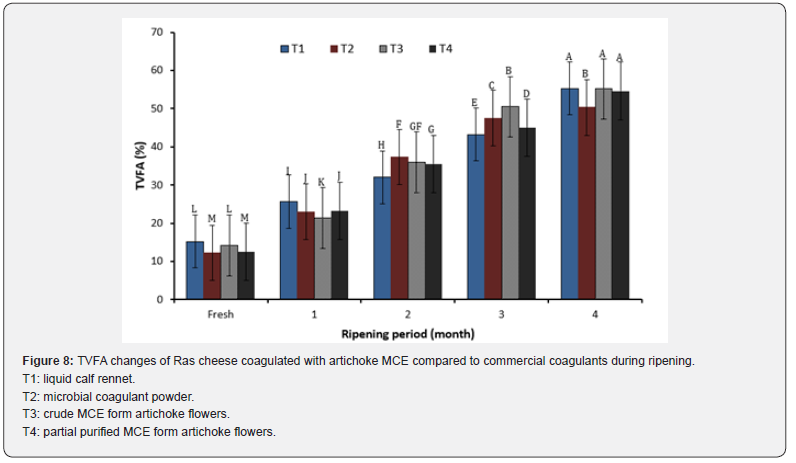
(Figure 8) shows TVFA content changes of Ras cheese coagulated with different coagulants during their ripening period for 4 months. It could be seen that no significant difference on TVFA contents of artichoke Ras cheese and control cheese coagulated with both animal and microbial coagulants throughout their ripening period. Moreover, TVFA contents of all resulted Ras cheese were increased as their ripening period prolonged which mainly due to the moisture reduction during ripening [59].
Textural profile of Ras cheese
TPA parameters of Ras cheese made by different coagulants are presented in (Table 2). It could be noted that Ras cheese coagulated with crude artichoke MCE (T3) recorded the lowest hardness value in comparison of other coagulants, while the hardness of Ras cheese coagulated with partial purified MCE (T4) from artichoke was close to commercial coagulants of calf rennet or microbial coagulant without significantly differences. It could be probably due to high proteolytic activity of artichoke crude extract compared to partial purified MCE [38], which leads to produce soluble nitrogenous compounds (Figure 6) because of proteolysis of cheese proteins [65]. Also, the higher moisture content of cheese coagulated with crude artichoke than partial purified MCE (Table 1). Ras cheese coagulated with crude artichoke MCE recorded the highest cohesiveness value among other coagulants without significantly differences. It could be due to the negative correlation between cohesiveness and hardness [57]. Also, (Table 2) shows not significantly (p≤0.05) increase for springiness, chewiness, and gumminess in Ras cheese coagulated with artichoke MCE (crude/partial purified) in comparison of commercial (calf rennet/ microbial coagulant) during their ripening period. These findings agree with those reported by Prados et al. [65] who indicated that the cardoon cheese exhibited softer texture and higher creaminess in comparison of cheese made with calf rennet and microbial coagulant. Overall, the results showed that the all-textural parameters of all resulted Ras cheese were increased as the ripening period prolonged which might be due to reduce of their moisture content and degradation of protein and fat contents during the ripening period [66-68].

T1, liquid calf rennet; T2, microbial coagulant powder; T3, crude MCE form artichoke flowers; T4, partial purified MCE form artichoke flowers. All parameters are represented as mean±standard error. Means with different small superscript letters in the same row and different capital superscript letters in the same column are significantly different at p ≤0.05.
Microbiological analysis of Ras cheese
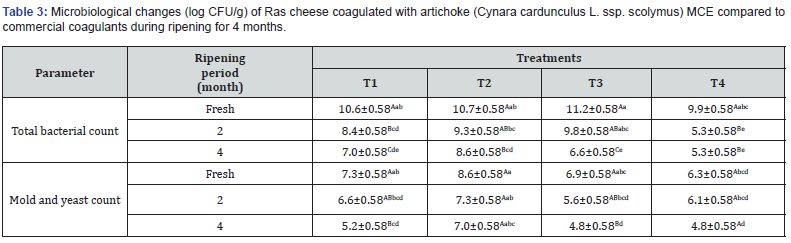
The results of microbiological examination of Ras cheese coagulated with different coagulants during their ripening period for 4 months are illustrated in (Table 3). It could be noted that the total bacterial count (TBC) of Ras cheese coagulated with crude artichoke MCE (T3) was higher than Ras cheese made with other coagulants which might be due to use of crude extract form of artichoke. Ras cheese (T3) had the highest initial TBC count (11.2 log cfu/g) followed by 10.7 log cfu/g (T2), 10.6 log cfu/g (T1), and T4 is 9.9 log cfu/g without significantly differences. Moreover, Ras cheese coagulated with partial purified MCE (T4) is close to both calf rennet and microbial coagulant during their ripening period. It means the partial purification of MCE from artichoke reduces the TBC of the resulting Ras cheese. Also, (Table 3) shows no significant difference of the molds and yeasts counts of Ras cheese since it initially 8.6, 7.3, 6.9, and 6.3 log cfu/g for T2, T1, T3, and T4, respectively. After 4 months of ripening, the remaining counts were 7.0 log cfu/g (T2), 5.2 log cfu/g (T1), and 4.8 log cfu/g for both T3 and T4. However, TBC, molds and yeasts decreased with the ripening period progressed could be due to the acidity development of cheese during ripening. Notably, coliform, psychotropic bacteria, proteolytic bacteria, E. coli, salmonella and shigella counts were not detected of Ras cheese coagulated with artichoke MCE coagulant compared to both calf rennet and microbial coagulant during 4 months of ripening period.
T1: liquid calf rennet; T2: microbial coagulant powder; T3: crude MCE form artichoke flowers; T4: partial purified MCE form artichoke flowers. All parameters are represented as means of replicating ±standard error. Means with different small superscript letters in the same row and different capital superscript letters in the same column are significantly different at p ≤0.05.
Ras cheese sensory properties
Total sensorial attributes score including the collective scores of individual flavor, appearance, body, and texture which coagulated with different coagulants during their ripening period for 4 months were presented in (Figure 9). After one month of ripening, the overall results showed that the total sensory scores of Ras cheese coagulated with crude artichoke MCE (T3) were higher than other Ras cheese treatments until 4 months of their ripening period without significantly differences. Moreover, it could be noted that Ras cheese coagulated with the partial purified MCE from artichoke (T4) was close to commercial coagulantsmade Ras cheese without significantly differences during the ripening period. It could be mainly due to decrease of proteolytic activity of partial purified MCE than crude MCE extract [36] which leads to limited soluble nitrogenous compounds (Figure 6) as an important factor for both flavor and texture development of cheese during its ripening period [60]. It makes the sensory attributes of Ras cheese (T4) close to Ras cheese made with both calf and microbial coagulants. These results are in accordance with Abd El-Salam et al. [36] who indicated that no significant differences in the sensory characteristics of cheese made with animal or microbial coagulants and purified artichoke MCE as a plant coagulant. Also, Abdul-Rahman [69] reported that no significant differences in sensory properties of cheese were made with both animal and safflower coagulants.
Conclusion
MCE was extracted from artichoke (Cynara cardunculus L. ssp. scolymus) flowers using 0.2 M acetate buffer pH 5.0, and then partial purified using 20-40 % of ammonium sulfate. The isolated and partial purified MCE from artichoke flowers showed thermal and pH stability, as well as being able to coagulate various milk species including goat milk followed by cow, buffalo and sheep milk. Also, it decreased as the level of NaCl increased with 25% reduction at 5% NaCl level. SDS-PAGE of casein proteolysis using artichoke MCE promoted extensive cleavage of k-CN and low level of αs- and β-CN hydrolysis. Also, chemical and microbiological properties of Ras cheese coagulated with partial purified MCE were close to Ras cheese made with commercial coagulants. Similar textural and sensorial properties of Ras cheese coagulated with artichoke MCE compared to both calf rennet and microbial coagulant were observed. It could be concluded that MCE extracted from artichoke flowers could be applied in cheese production as a calf rennet alternative without any defects of the resultant cheese during their ripening for 4 months.
Ethical Approval
The present study has been carried out according to the Medical Research Ethics Committee, National Research Centre, Cairo, Egypt (Registration No. 20-360).
References
- Abou-Donia SA (2002) Recent development in Ras cheese research: A review. Egyptian Journal Dairy Science 30(2): 155-166.
- Fox PF, McSweeney PLH (2004) Cheese: An Overview in Cheese: Chemistry, Physics and Microbiology. 3rd (Edn), Chapman and Hall London, UK.
- Barry Law A, Tamime AY (2010) In: Law, BA, AY Tamime (eds.), Technology of cheese making. Wiley-Blackwell, London, UK.
- Agudelo RA, Gauthier SF, Pouliot Y, Marin J, Savoie L (2004) Kinetics of peptide fraction release during in vitro digestion of casein. Journal of Science Food andAgriculture 84(4): 325-332.
- Mahajan RT, Badgujar SB (2012) Biological aspects of proteolytic enzymes: A Review. J Pharm Res 3: 2048-2068.
- Roseiro LB, Barbosa M, Ames JM, Wilbey RA (2003) Cheese making with vegetable coagulants—the use of Cynara L for the production of ovine milk cheeses. Int J Dairy Technology 56(2): 76-85.
- Mohamed Ahmed IA, Morishima I, Babiker EE, Mori N (2009) Characterization of partially purified milk- clotting enzyme from Solanum dubium Fresen seeds. Food Chemistry 116(2): 395-400.
- Bruno MA, Lazza CM, Errasti ME, López LMI, Caffini NO, et al. (2010) Milk clotting and proteolytic activity of an enzyme preparation from Bromelia hieronymi fruits. LWT–Food Science and Technology 43(4): 695-701.
- Siddig EEEI, Abdelgadir WS, Kabeir BM, Koko MYF, Ibrahim RA (2018) Quality of white cheese made using Moringa oleifera leaf extract. Journal of Academia and Industrial Research 7(1): 7-17.
- Abdeen ESMM, Ibrahim OA, Kholif AMM (2021) Utility of Moringa oleifera waste as a coagulant in goat soft cheese production. Heliyon 7(7): e07536.
- Abebe GD, Getachew GD (2019) Isolate and extract for milk clotting enzymes from the leaves of Moringa oleifera, Carica papaya and Mangifera indica and use in cheese making: the case of western hararage region, Ethiopia. Journal of Food and Nutrition Research 7(3): 244-254.
- Abebe B, Emire S (2020) Manufacture of fresh cheese using east African Calotropis procera leaves extract crude enzyme as milk coagulant. Food Science and Nutrition 8(9): 4831-4842.
- Sanjuán E, Millán R, Saavedra P, Carmona MA, Gómez R, et al. (2002) Influence of animal and vegetable rennet on the physicochemical characteristics of Los Pedroches cheese during ripening. Food Chemistry 78(3): 281-289.
- Otani H, Matsumori M, Hosono A (1991) Purification and some properties of a milk clotting protease from the young seeds of Albizia julibrissin. Animal Sci Techno 62(5): 424-432.
- Anusha R, Singh MK, Bindhu OS (2014) Characterisation of potential milk coagulants from Calotropis gigantea plant parts and their hydrolytic pattern of bovine casein. European Food Res. Technology 238: 997-1006.
- Nouani A, Dako E, Morsli A, Belhamiche N, Belbraout S, et al. (2009) Characterization of the purified coagulant extracts derived from artichoke flowers (Cynara scolymus) and from the Fig tree latex (Ficus carica) considering their use in the manufacture of traditional cheeses in Algeria. J Food Technol 7: 20-29.
- Robert Scopes K (1982) Separation by precipitation. In: Protein purification Springer, New York, p. 39-66.
- IDF (1992) Bovine rennets: determination of total milk-clotting activity. Bulletin No. 157 Brussels, Belgium, International Dairy Federation.
- Bradford MM (1976) A rapid and sensitivity method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72: 248-354.
- Laemmi UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriofage T4. Nature 227(5259): 680-685.
- Hofi AA, Youssef EH, Ghoneim MA, Tawab A (1970) Ripening chances Cephalotyre (Ras) cheese manufactured from raw and pasteurized milk with special reference to flavour. J Dairy Sci 53: 1207.
- AOAC Association of Official Analytical Chemists (2007), 18th edn, Benjamin Franklin Station Washington, DC, USA.
- Kosikowski FV (1982) Cheese and fermented milk food, 2nd edn Brooktonal, New York, USA.
- IDF (1991) Rheological and fracture properties of cheeses. International Dairy Federation.
- APHA (1976) American Public Health Association, Compendium methods for the microbiological examination of foods. 1st ed. Marvin specked. Washington, DC, USA.
- FDA Food and Drug Administration (2002) Bacteriological Analytical Manual 9th ed., AOAC International Arlington, VA, USA.
- Tassew A, Seifu E (2011) Microbial quality of raw cow’s milk collected from farmers and dairy cooperatives in Bahir Dar Zuria and Mecha District. Agric Biol Jour North America 2(1): 29-33.
- Rosso AL, Azam F (1987) Proteolytic activity in coastal oceanic waters: Depth distribution and relationship to bacterial populations. Marine ecology progress series 41(3): 231-240.
- El-Hadedy D, El-Nour SA (2012) Identification of Staphylococcus aureus and Escherichia coli isolated from Egyptian food by conventional and molecular methods. Journal of Genetic Engineering and Biotechnology 10(1): 129-135.
- Bartram J, Balance R (1996) Water quality monitoring: a practical guide to the design and implementation of freshwater quality studies and monitoring programmes. CRC Press.
- Taylor WI, Harris B (1965) Isolation of shigellae. II. Comparison of plating media and enrichment broths. American Journal of Clinical Pathology 44(4): 476-479.
- McCarthy MD (1996) The use of XLD agar as a medium for the isolation of intestinal pathogens. New Zealand Journal Medicine Laboratory Technologic p. 20.
- Pappas CP, Kondly E, Voustsinas LP, Mallatou H (1996) Effect of starter level, draining time and aging on the physicochemical, organoleptic and rheological properties of Feta cheese. J Society Dairy Technology 49(3): 73-78.
- SAS (1999) SAS User's Guide statistics Ver. 8, SAS Inst, Cary, NC, USA.
- Néstor GM, DelyRubí CG, Héctor JC (2012) Exploring the Milk-Clotting Properties of a Plant Coagulant from the Berries of S. elaeagnifolium var. Cavanilles. J Food Sci 77(1): C89-C94.
- Abd El-Salam BA, Hamid Ibrahim OAEI, l-Razek El-Sayed HA (2017) Purification and characterization of milk clotting enzyme from artichoke (Cynara cardunculus L) flowers as coagulant on soft white cheese. Int J Dairy Science 12: 254-265.
- Pontual EV, Carvalho BEA, Bezerra RS, Coelho LCBB, Napoleão TH, et al. (2012) Caseinolytic and milk-clotting activities from Moringa oleifera flowers. Food Chemistry 135(3): 1848-1854.
- Tajalsir AE, Ebraheem AS, Abdallah AM, Khider FJ, Elsamani MO, et al. (2014) Partial purification of milk-clotting enzyme from the seeds of Moringa oleifera. J Microbiol Biotechnol Food Science 4(1): 58-62.
- Guinee TP, Fox PF (2004) Salt in cheese: Physical, chemical, and biological aspects, Cheese: chemistry, physics, and microbiology. 3rd edn, Fox PF, McSweeney PLH, Cogan TM, Guinee TP, editors, San Diego, Elsevier, Academic Press, CA 1: 207-259.
- Zoon P, Van Vliet T, Walstra P (1989) Rheological properties of rennet-induced skim milk gels. Neth Milk Dairy J 43: 17-34.
- Ahmed SA, Wehaidy HR, Ibrahim OA, El Ghani SA, El-Hofi, MA (2016) Novel milk-clotting enzyme from Bacillus stearothermophilus as a coagulant in UF-white soft cheese. Biocatalysis and Agriculture Biotechnology 7: 241-249.
- Bakr A, Ibrahim O, El-Sattar El-Ghandour A, El-Deeb N (2022) Purification and characterization of milk clotting enzyme from edible mushroom (Pleurotus florida). Letters in Applied NanoBioscience 11(2): 3362-3373.
- Llorente BE, Brulti CB, Natalucci CL, Caffini NO (1997) Partial Characterization of a Milk Clotting Proteinase isolated from Artichoke (Cynara scolymus L, Asteraceae). Acta Farmaceutica Bonaerense 16(1): 37- 42.
- Raposo S, Domingos A (2008) Purification and characterization milk-clotting aspartic proteinases from Centaurea calcitrapa cell suspension cultures. Process Biochemistry 43(2): 139-144.
- Shehata AE, Fayet EA, Ismail AA, Salem MM (1996) Production and characterization of bacterial coagulants as calf rennet replace for Egyptian cheese making. Egyptian Journal of Food Science 24(3): 417-449.
- Tubesha ZA, AL-Delaimy KS (2003) Rennin-like milk coagulant enzyme produced by a local isolate of Mucor. International Journal of Dairy Technology 56(4): 237-241.
- Chazara S, Sidrach L, Lόpez-Molina D, Rodrίguez-Lόpez JN (2003) Characterization of the milk clotting properties of extracts from artichoke (Cynara scolymus, L) flowers. International Dairy Journal 17(12): 1393-1400.
- Rao MB, Tanksale AM, Ghatge MS, Deshpande VV (1998) Molecular and biotechnological aspects of microbial proteases. Microbiology and Molecular Biology Reviews 62(3): 597-635.
- Medhammar E, Wijesmha-Bettoni R, Stadlmayr B, Nilsson E, Charrondiere UR, et al. (2012) Composition of milk from minor dairy animals and buffalo breeds: a biodiversity perspective. Journal of the Science of Food and Agriculture 92(3): 445-474.
- Park YW, Juarez M, Ramos M, Haenlein GFW (2007) Physico-chemical characteristics of goat and sheep milk. Small Ruminant Research 68(1-2): 88-113.
- Barbagallo RN, Chisari M, Spagna G, Ierna A, Patanè A, et al. (2007) Casinolytic activity expression in flowers of Cynara cardunculus. Acta Horticulturae 730: 195-199.
- Tavaria FK, Sousa MJ, Malcata FX (2001) Storage and lyophilization effects of extracts of Cynara cardunculus on the degradation of ovine and caprine caseins. Food Chemistry 72(1): 79-88.
- Ordiales E, Mart A, Benito MJ, Ruiz-Moyano S, Gallardo G, et al. (2016) Characterisation of the vegetable rennets used for Torta del Casar cheesemaking by a protein profile method. International Journal of Dairy Technology 69(2): 272-281.
- Cavalli SV, Claver S, Priolo N, Natalucci C (2005) Extraction and partial characterization of a coagulant preparation from Silybum marianum flowers. Its action on bovine caseinate. Journal of Dairy Research 72(3): 271-275.
- Pintado AI, Macedo AC, Teixeira G, Pais MS, Clemente A, et al. (2001) Caseinolytic activity of fruit extract from Opuntia ficus-indica on bovine, caprine, and ovine sodium caseinates. Biotechnology Progress 17(4): 643-646.
- El-Kholy AM (2015) The use of artichoke (Cynara scolymus L) extracts to produce Tallaga cheese. Int J Food Nut Sci 4: 34-41.
- Pino A, Prados F, Galan E, McSweeney PLH, Fernandez-Salguero J (2009) Proteolysis during the ripening of goats’ milk cheese made with plant coagulant or calf rennet. Food Research International 42(3): 324-330.
- Tejada L, Abellan A, Prados F, Maria Cayuela J (2008) Compositional characteristics of Murcia al Vino goat's cheese made with calf rennet and plant coagulant. Int J Dairy Technology 61(2): 119-125.
- Fahmy MA (2003) Effect of using different starters on the chemical, bacteriological and sensory properties of Ras cheese. Assiut Journal of Agricultural Science 34: 241-253.
- Al-Jasser MS, Al-Dogan AA (2009) Properties of soft white cheese produced using rennet substitute extracted from Solanum dubium seeds. Egypt J Dairy Sci 37(2): 237-243.
- Abu-Zeid NA (1994) Utilization of weed (Sonchus oleraceus L) as vegetable rennet for Domiati cheese. Ind J Dairy Sci 47: 140-144.
- Chen S, Agboola S, Zhao J (2003) Use of Australian cardoon extract in the manufacture of ovine milk cheese – a comparison with commercial rennet preparations. International Journal of Food Science and Technology 38(7): 799-807.
- El-Hofi MA, Ismail AA, AbdRabo FHR, El-Dieb SM, Ibrahim OA (2010) Studies on acceleration of Ras cheese ripening by aminopeptidase enzyme from buffaloes’ II Utilization of Buffaloes' pancreas aminopeptidase in acceleration of Ras cheese ripening. J American Science 6(9): 575-581.
- El-Sayed SM, Ibrahim OA, Kholif AMM (2020) Characterization of novel Ras cheese supplemented with Jalapeno red pepper. Journal of Food Processing and Preservation 44(7): e14535.
- Prados F, Pino A, Fernandez-Salguero J (2007) Effect of a powdered vegetable coagulant from cardoon Cynara cardunculus in the acclerated of ripening of Manchego cheese. Int J Food Sci Technology 42(5): 556-561.
- Calvo MV, Castillo I, Díaz-Barcos V, Requena T, Fontecha J (2007) Effect of a hygienized rennet paste and a defined strain starter on proteolysis, texture and sensory properties of semi-hard goat cheese. Food Chemistry 102(3): 917-924.
- Youssef AM, Assem FM, Abdel-Aziz ME, Elaaser M, Ibrahim OA, et al. (2019) Development of bionanocomposite materials and their use in coating of Ras cheese. Food Chem 270: 467-475.
- Sayadi A, Madadlou A, Khosrowshahi A (2013) Enzymatic cross-linking of whey proteins in low fat Iranian white cheese. International Dairy Journal 29(2): 88-92.
- Abdul-Rahman SM (2013) Using safflower (Cartamus tinctorius) Seeds extract for soft white Cheese manufacturing. In: The first scientific conference of the Department of Food Science. Journal of University of Tikrit of Agricultural Sciences 39-44.






























