Low Dose Sip Feeding and Glutathione - Results in Elderly Patients with Malnutrition
Weinrebe W1*, Stippler D2 and Karaman M3
1Hirslanden Salem Spital, Department of Geriatrics, Switzerland
2Nutricia, Erlangen, Germany
3Institute for Biostatistics, Berlin, Germany
Submission: June 10, 2023; Published: June 21, 2023
*Corresponding author: Weinrebe W Hirslanden Salem Spital, Department of Geriatrics, Switzerland. Email: Wolfram.weinrebe@hirslanden.ch
How to cite this article: Weinrebe W, Stippler D, Karaman M. Low Dose Sip Feeding and Glutathione - Results in Elderly Patients with Malnutrition. Nutri Food Sci Int J. 2023. 12(2): 555833. DOI: 10.19080/NFSIJ.2023.12.555833.
Abstract
Objectives: to evaluate the influence of a three-month low dose sip feeding on glutathione concentrations in blood cells in malnourished older patients.
Design: cohort therapeutic and diagnostic study with a treatment and a washout period in eight care facilities.
Participants: 60 geriatric patients with malnutrition.
Intervention: 1. three-month of low dose sip feeding (at least five Tetra Paks per week) 2. A three-month wash out phase without low dose sip feeding. Monitoring the patient’s metabolic and redox parameters.
Measurements: measurement of total and reduced glutathione concentrations by HPLC in erythrocytes and lymphocytes.
Results: A significant increase in the lymphocyte total glutathione concentration was observed during the sip feeding therapy (total GSH+0.672 nmol/106 cells; p=0.01) due to detectable less oxidation (red GSH +0.33 ± 0.78 nmol/106 cells, p=0.00). A pause in the sip feeding caused a significant decrease in the reduced glutathione concentration in the lymphocytes (red GSH -0.20 ± 0.61nmol/106 cells, p=0.00). The glutathione concentration in the erythrocytes showed no significant changes.
Conclusion: Low dose sip feeding can significantly reduce oxidation in lymphocytes leading to higher total GSH concentrations which were completely inversed in the sip free phase. This might be an important hint concerning support and nutrition in older malnourished patients.
Keywords: Low Dose Sip Feeding; Glutathione in Lymphocytes; Geriatric Patients; Malnutrition
Abbreviations: GSH: Glutathione; HPLC: High Performance pressure Liquid Chromatography; MNA: mini nutritional assessment; SGA: Subjective Global Assessment
Introduction
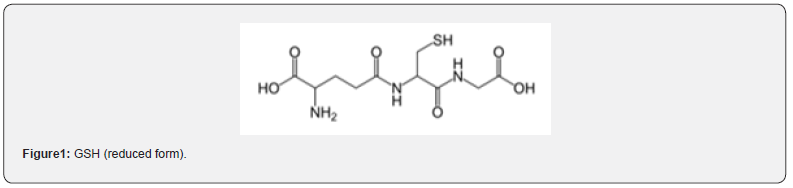
Glutathione (GSH, Figure 1) is the most important intracellular redox system to fight oxidative aggressors such as cellular hydroxyl peroxides [1]. Glutathione or gamma-gluthamyl-cysteine is built from glycine, cysteine, and glutamine. It’s an ingoing NADPH-dependent redox reaction to keep GSH in a reduced form (> 90%) and beware from being oxidized (GSSG or glutathione disulfide) [2, 3]. By this way the cell can protect the membranes from oxidation or to keep away from microsomal lipid peroxidation [4]. All cells possess different concentrations of glutathione [5]. The concentration may be depleted by medication, ongoing oxidative stress, or toxic products of metabolism [6]. The GSH concentration of the blood cells is depleted by growing age due to an increasing oxidative stress and necessary repair procedures consuming glutathione. Extern stressors as interventions in intensive care, chronic diseases, and malnutrition lead to a depletion of GSH [7]. Clinical examples are degenerative disorders as cataract [8], disturbance of liver function or cachexia and tumor induced weight loss. Centrally important is the metabolism and the high concentrations in the liver and the bile [9]. Increasing anti oxidative tasks augments GSH concentration. Especially active cells like tumor cells show extreme high concentrations of glutathione (e.g., neuroblastoma cells with an 8-10-fold higher GSH concentration). A higher GSH concentration leads to higher chemo resistance in these cells [10]. Low GSH levels are associated with autoimmune and inflammatory diseases as rheumatoid arthritis [11], Crohn`s disease [12] and M. Parkinson [13]. Similar results that demonstrate an important role of glutathione are described in degenerative diseases like M. Alzheimer [14] and even in schizophrenia [15] and age [16].
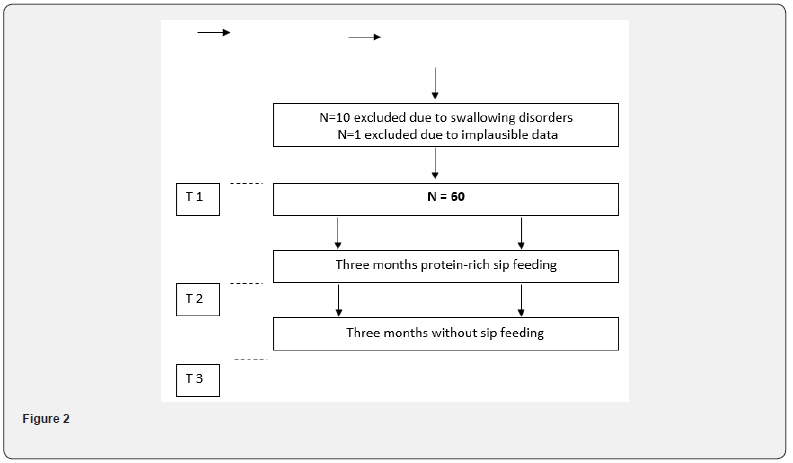
The study “Influence of low-dose sip feeding on malnutrition and cognition in elderly patients with malnutrition (ISOMACEPM)” explored the hypothesis that the administration of low-dose protein-rich sip feeding to malnourished individuals for 3 months would improve their nutritional, cognitive and or metabolic parameters. Malnutrition in the older patients is associated with an increase in morbidity and mortality [17]. Malnutrition is an important risk factor for complications like delirium [18]. In this study we focused on the metabolic redox pathway measured by glutathione concentrations in lymphocytes and erythrocytes while administering low dose sip feeding for three months.
Methods
The measurement of glutathione is sensible to external influences and to oxidation. In a pre-study phase, we tested different technical preparations of whole blood samples for the analysis of glutathione. We defined a test phase with a hundred samples to imitate the study conditions of taking and transporting blood and preparing for glutathione measurement in erythrocytes and lymphocytes.
Defined technical procedure
A layer of 2ml EDTA whole blood is given slowly over a 5ml Ficoll solution layer in a poly propylene tube, without producing any whirls. The separation of lymphocytes is done by centrifugation (800xg for 15 minutes). Lymphocytes build a small layer during the tube; the erythrocytes are found at the bottom of the tube. The small lymphocyte layer is isolated with the use of a special pipette. This probe is added to a tube filled up with 10ml phosphate buffered saline. A second centrifugation is done (800xg, 15 minutes). The supernatant is decanted. Shock freezing is done for lymphocytes. For the determination of glutathione in erythrocytes the centrifuge is filled up with 10ml saline after decanting the whole supernatant. After that a second centrifugation is done (800xg, 15 minutes). The supernatant is decanted. Shock freezing is done for erythrocytes. The shock frozen pellets are sent to a specialized lab for the determination of glutathione with the HPLC Kit [19]. HPLC is High Performance pressure Liquid Chromatography. In the first step a defined puffer and a derivate solution is given to the aliquot. After 20 minutes of incubation higher molecular weights are precipitated. To the second aliquot a reduction solution and an intern standard is added thereby transferring oxidized GSSG to GSH. In this step total GSH is measured. In the reaction of derivatization GSH is fluorescent. The separation is by HPLC in an isocratic procedure and a temperature of 30° using a “reversed phase “-column in two procedures following each other. Chromatograms are documented with a fluorescent detector. Quantification is done by an EDTA whole blood calibrator. The calculation of the results is done by using the intern standard method by integration of peak height / area.
The measurement of reduced GSH is done without internal standard to avoid mixing with other sulfides. Oxidized GSH (GSSG) is calculated by subtraction of reduced GSH from total GSH. One hundred malnourished residents in eight care facilities were selected. Twenty-nine of the residents were not included due to lack of consent. Ten of the 71 residents had to be excluded because of swallowing disorders. One resident was removed from the evaluation due to questionable records. 60 patients were selected to take part in the study performed based on a positive ethical approval (ethical review committee, chamber of physicians Rhineland palatinate No. 837.090.03(3752)) and an informed consent of every participant or his family members. Blood analyses were done to evaluate the GSH concentration in lymphocytes and erythrocytes in malnourished patients. The measurement was done in the beginning, after 3 months of sip feeding and after another 3 months without sip feeding.
For the low dose sip feeding, a normal caloric (=1 kcal/ ml, administered in Tetra Paks containing 200 ml=200 kcal each) protein-rich (20 percent of energy protein=40 kcal from protein=10 g additional protein) product (Fortimel from Nutricia, Erlangen, Germany) was used in various flavors at least 5 packs a week. This product contained - 3mg vitamin e, 34mg vitamin c. Branched chain amino acids (BCAA): 1.25g valine, 1.98g leucine, isoleucine 1.05 Isoleucine. Glutathione (GSH) synthesizing amino acids: 0.17g cystine /cysteine, 4.4g glutamine /glutamine acid, 0.39g glycine, sulfur containing amino acid Methionine 0.52g. With at least 5 packs the patients reached in one week the following 200mg vitamin C (2 x rda), 15mg vitamin E (1xrda), 0.085mg cysteine /cysteine (1xrda), 22g glutamine/glutamine acid (5 x rda), 1.95g glycine (2x rda), 2,37g methionine (1x rda). The other diet of the included patients was continued without change [20, 21]. Every patient was assessed for malnutrition. This was done by specially trained physicians by using the mini nutritional assessment (MNA) [22] and by subjective global assessment (SGA) [23]. Only patients tested positive for malnutrition were included in the study.
Results
The study group (n=60) was high-aged (average age was 84), showed low function (Barthel index was 0-50 points in 69%) and low weight (average weight was 48.87 ± 8.16 kg). Following MNA (Mini Nutritional assessment) and SGA (Subjective Global Assessment) the patients showed to be 100% malnourished. Data from literature show nearly the same results for concentration of glutathione in lymphocytes [24] and erythrocytes [25] as we found in the present study. In comparison to geriatric patients in clinical treatment, the study population young healthy persons the lymphocyte concentration of glutathione of is 1.47 GSH versus 1.43 versus 1.78 (nmol GSH/106 cells). The number of lymphocytes decreases with age and this malnourished group shows to have lymphopenia. For the GSH concentration in erythrocytes we couldn`t find great differences between the three groups. There was a trend to anemia in the elderly malnourished group. The pre-study results are given here in comparison to the study group. The total GSH concentration in blood decreased with age and malnutrition being highest in young and lowest in old, malnourished patients (1.978 versus 1.831 μmol/l) Table 1.

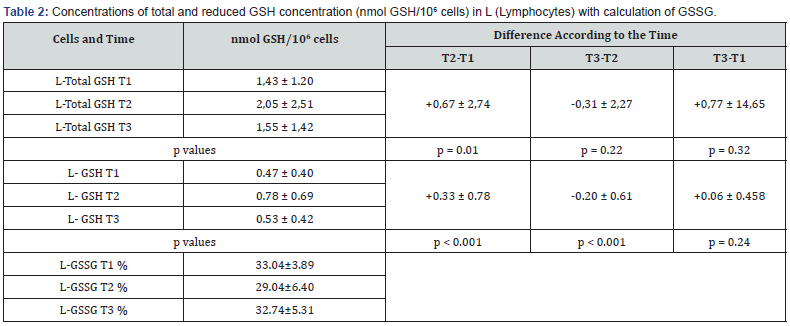
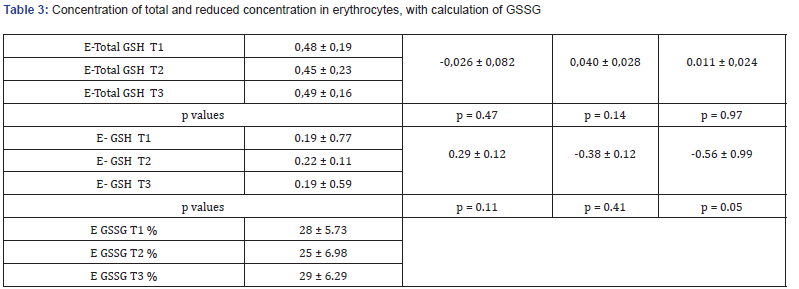
A significant increase in the lymphocyte glutathione concentration was observed during the sip feeding phase (+0,672 nmol GSH/106 cells; p=0.01). This was the result of less oxidation and a significant increase of reduced GSH. (+0.33 ± 0.78 nmol GSH/106 cells, p=0,.00). The sip feeding pause caused a decrease in the total glutathione concentration in the lymphocytes (-0,31 nmol/106 cells, p=ns) and a significant decrease of reduced GSH (-0.20 ± 0.61 nmol/106 cells, p=0.00) Table 2. The glutathione concentration in erythrocytes showed the same but no significant changes nor in total neither in reduced GSH concentration (Table 3).
Statistics
Statistical analyses were performed with the paired t-test and a chi-squared test. SPSS Statistics 15.0 was used for the data analysis.
Discussion
Protein supplementation is known to produce positive effects in elderly patients with malnutrition [26]. Low does sip feeding can also show positive effects on weight, triceps skin fold and muscle cross section [27]. We found that even low dose sip feeding can produce more GSH in lymphocytes. According to the data - GSH is directly affected by nutritional status and may be depleted due to the several factors. It is possible that the described disease related impairment of the glutamate-cysteine-ligase leads to a reduced production of GSH as also a reduced offer of amino acids in malnutrition can lead this situation. Amino acids are also known to have regulating functions in immune response with the best prototype like arginine, glutamine acid and cysteine.
In blood GSH concentrations belong to nearly 100% to the erythrocytes [28] and only a very small amount to the lymphocytes. GSH helps affect T-lymphocyte cell maturation [29] which has an important role in directing appropriate immune response as oxidative tasks in defending aggressors, infectious diseases, and oxidative burst in malnutrition [30]. Recent publications on immunosenescence [31] show the decreasing capacity of T-lymphocytes of elderly patients to provide active immune responses and suggest that e.g., death by Covid may be caused by a gutathione deficiency in lymphocytes of elderly patients [32]. The number of lymphocytes in our study patients was at the lower edge matching the fact that lymphopenia can be seen in malnutrition [33]. All the study patients proved to be malnourished. Whilst erythrocytes GSH concentration was not altered significantly, the lymphocytes significantly increased the cellular glutathione concentration under low dose sip feeding over the period of three month which was inversed without sip feeding after 3 months [34].
What could be the reason for the significant increase in GSH concentration in lymphocytes?
In the treatment phase the malnourished patients received 24 rda of vitamin c and 12 rda of vitamine E, both known to be strong antioxidatives, having a radical scavenging power, able to keep more GSH in reduced form. Additionally, the malnourished patients received 12 rda of cystin/cystein, 24 rda of glycine and 60 rda’s of glutamine/acid -a solid amount to rebuilt and resynthesize GSH. Zarka [32] could show in 2017 that a single dose of 2 or 4g Glutamylcystein increases 2 to 3-fold the basal levels in lymphocytes. Moreover, the sulfur containing amino acid methionine was administered in the exact rda (daily rda is 10mg/kg body weight, meaning 0.5g in 50kg about, which was exactly the dosage of methionine per day). This could deliver enough sulfur containing amino acids to rebuild GSH. Breitkreutz and Badaloo [35, 36] proved that high 1.8 -2.4g/day dose administration of (sulphur containing) cysteine stopped and inversed the GSH depletion. There was no significant effect in glutathione concentration in erythrocytes inspite of being a cell group of direct oxygen transport function. Since lymphocytes are increasingly in focus and the study showed GSH increase in lymphocytes, the well tolerated low dose sip feeding is likely to play an important role for the elderly.
Limitations
The number of study patients was less than that of the pilot study. This was due to the complex diagnostic structure (assessments, ultrasound, blood sampling) at the three time points. Also, for the homes themselves, the effort due to the study was high (training before participation, additional documentation, final interviews). In addition, some of the residents were not able to agree to participate themselves. Lymphocytes were not differentiated into B and T lymphocytes. Therefore, no statement can be made about which cell series benefited from the GSH increase, which would have been interesting.
Conclusion
We found that low dose sip feeding shows measurable changes in redox system of the lymphocytes in older patients with malnutrition. It augments GSH supporting lymphocytes to better manage oxidative stress. As malnourished patients are defined as having weight and muscle loss and to show a tendency for infectious problems this antioxidative effect on lymphocytes could be very important. A larger study should look for the different distributions of GSH elevation in different (T and B) lymphocytes and the outcome of patients presenting a higher GSH concentration. Then it would be possible to make more statements about the significance of this study result for elderly patients.
References
- Meister A, Anderson ME (1983) Annu Rev Biochem 52: 711-760.
- Griffith OW (1999) Biologic and pharmacologic regulation of mammalian glutathione synthesis. Free Radic Biol Med 27: 922–935.
- Shelly C, Lu MD (2013) Glutathions synthesis. Biochem Biophys Acta 1830(5): 3143-3153.
- Weinrebe W (1993) Der Einfluss reaktiver Sauerstoffverbindungen, Alkylantien und Hyperthermie auf das Glutathionssystem bei Neuroblastomzellen und Mononukleären Blutzellen, Inaugural Dissertation, Tü
- Meister A, Tate SS (1976) Glutathione and related gamma-glutamyl compounds: biosynthesis and utilization. Annu Rev Biochem 45: 559-604.
- DeLeve LD, Kaplowitz N (1990) Importance and regulation of hepatic glutathione. Semin Liver Dis 10: 251-266.
- Dringen R, Hirrlinger J (2003) Glutathione pathways in the brain. Biol Chem 384(4): 505-516.
- Calvin HI, Medvedovsky C, Worgul BV (1986) Near-total glutathione depletion and age-specific cataracts induced by buthionine sulfoximine in mice. Science 233: 553-555.
- Ballatori N, Truong AT (1992) Glutathione is a primary osmotic driving force in hepatic bile formation. Am J Physiol 263: G617-G624.
- Calvert P, Yao KS, Hamilton TC, O'Dwyer PJ (1998) Clinical studies of reversal of drug resistance based on glutathione. Chem Biol Interact 111-112: 213-224.
- Hassan MQ, Hadi RA, Al-Rawi ZS, Padron VA, Stohs SJ (2001) The glutathione defense system in the pathogenesis of rheumatoid arthritis. J Appl Toxicol 21: 69-73.
- Sido B, Hack V, Hochlehnert A, Lipps H, Herfarth C, et al. (1998) Impairment of intestinal glutathione synthesis in patients with inflammatory bowel disease. Gut 42: 485-492.
- Sian J, Dexter DT, Lees AJ, Daniel S, Agid Y, et al. (1994) Alterations in glutathione levels in Parkinson's disease and other neurodegenerative disorders affecting basal ganglia. Ann Neurol 36(3): 348-355.
- Viña J, Lloret A, Ortí R, Alonso D (2004) Molecular bases of the treatment of Alzheimer's disease with antioxidants: prevention of oxidative stress. Mol Aspects Med 25: 117-123.
- Oja SS, Janáky R, Varga V, Saransaari P (2000) Modulation of glutamate receptor functions by glutathione. Neurochem Int 37: 299-306.
- Lu Sc (2009) Regulation of glutathione synthesis. Mol Asp 30: 42-59.
- Inouye SK, Rushing JT, Foreman MD (1998) Does delirium contribute to poor hospital outcomes? A three-site epidemiologic study. J Gen Intern Med 13: 234-242.
- Sullivan DH, Walls RC, Bopp MM (1995) Protein-energy undernutrition, and the risk of mortality within one year of hospital discharge: A follow-up study. J Am Geriatr Soc 43(5): 507-512.
- Zur Bestimmung von Glutathion in EDTA Vollblut, ImmunDiagnostik.
- Guigoz Y (2006) The Mini-Nutritional Assessment (MNA) Review of Literature – What does it tell us? J Nutr Health Aging 2006 10: 466-487.
- Detsky AS, McLaughlin JR, Baker JP, Johnston N, Whittaker S, et al. (1987) What is subjective global assessment of nutritional status? JPEN J Parenter Enteral Nutr 11(1): 8-13.
- Richie JP Jr, Nichenametla S, Neidig W, Calcagnotto A, Haley JS, et al. (2015) Randomized controlled trial of oral glutathione supplementation on body stores of glutathione. Eur J Nutr 54(2): 251-263.
- van't Erve TJ, Wagner BA, Ryckman KK, Raife TJ, Buettner GR (2013) The concentration of glutathione in human erythrocytes is a heritable trait. Free Radic Biol Med 65: 742-749.
- Baldwin C, Weekes CE (2011) Dietary advice with or without oral nutritional supplements for disease related malnutrition in adults, Cochrans Database of systematic Reviews.
- Weinrebe W, Meister R, Stippler D, Füsgen I (2015) Low- dose sip feeding in individuals with malnutrition - effects in the nutritional parameters. JAGS 63(10): 2207-2208.
- Ballatori N, Hammond CL, Cunningham JB, Krance SM, Marchan R (2005) Molecular mechanisms of reduced glutathione transport: Role of the MRP/CFTR/ABCC and OATP/SLC21A families of membrane proteins. Toxicol Appl Pharmacol 204: 238-255.
- Peterson JD, Herzenberg LA, Vasquez K, Waltenbaugh C (1998) Glutathione levels in antigen-presenting cells modulate Th1 versus Th2 response patterns. Proc Natl Acad Sci 95: 3071-3076.
- Messina JP, Lawrence DA (1989) Cell cycle progression of glutathione-depleted human peripheral blood mononuclear cells is inhibited at S phase. J Immunol 143: 1974-1981.
- Fülöp T, Dupuis G, Witkowski JM, Larbi A (2016) The Role of Immunosenescence in the Development of Age-Related Diseases. Rev Invest Clin 68(2): 84-91.
- Polonikov A (2020) Endogenous Deficiency of Glutathione as the Most Likely Cause of Serious Manifestations and Death in COVID-19 Patients. ACS Infect Dis 6(7): 1558-1562.
- Smeyne M, Smeyne R (2018) Glutathione metiabolism and Parkinson disease. Free Rad Biol Med 62: 13-25
- Li P, Yin YL, Li D, Kim SW, Wu G (2007) Amino acids and immune function. Br J Nutr 98(2):237-252.
- Crétel I Veen, A Pierres, Y Binan, P Robert, AD Loundou, et al. (2011) Étude du profil immunitaire de sujets âgés hospitalisés en unité de court séjour gé La revue de médecine interne 32(5): 275-282.
- Zarka MH, Bridge WJ (2017) Oral administration of gamma-glutamyl-cysteine increase intracellular glutathione levels above hemeostasis in a randimised humant rail pilot study. Redox Bioil 11: 631-636
- Breitkreutz R, Pittack N, Nebe CT, Schuster D, Brust J, et al. (2000) Improvement of immune functions in HIV infection by sulfur supplementation: two randomized trials. J Mol Med (Berl) 78(1): 55-62.
- Badaloo A, Reid M, Forrester T, Heird WC, Jahoor F (2002) Cysteine supplementation improves the erythrocyte glutathione synthesis rate in children with severe edematous malnutrition. Am J Clin Nutr 76: 646-652.






























