Effect of Impregnation Solution Concentration on Vacuum Impregnation Efficiency of Potato Tuber
Erihemu1*, Jia Yu1, Dongsheng Wang2, Guoqin Li1, Xiaoqing Song1, Hongmei Zhu1 and Kazunori Hironaka3
1Laboratory of Potato Product Processing and Storage, College of Food Science, Shanxi Normal University, China
2College of Life Science, Shanxi Normal University, China
3Department of Regional Agricultural Engineering, University of the Ryukyus, Japan
Submission: September 29, 2021; Published: October 02, 2021
*Corresponding author: Erihemu, Laboratory of Potato Product Processing and Storage, College of Food Science, Shanxi Normal University, China
How to cite this article: Erihemu, Jia Y, Dongsheng W, Guoqin L, Xiaoqing S, et al. Effect of Impregnation Solution Concentration on Vacuum Impregnation Efficiency of Potato Tuber. Nutri Food Sci Int J. 2021. 11(2): 555809. DOI: 10.19080/NFSIJ.2021.11.555809.
Abstract
Vacuum impregnation (VI) with adequate solutions can be an interesting technique in order to develop functional food products, by introducing nutrition ingredients in their highly porous structure. However, the VI efficiency of food matrix is significantly affected by the VI conditions such as impregnation solution concentration. In this study, the effects of changes in the iron solution (IS) concentration upon volume fractions (impregnation and deformation), effective porosity, and the iron content of VI whole potatoes (Toyoshiro and Dejima) were evaluated. The potatoes were immersed in different concentrations of IS, vacuum pressure of 800 Pa was applied for 1 h, and atmospheric pressure was restoration for 3 h. Results demonstrated that the moisture content, total mass variation, and iron content of the VI-potatoes increased with IS concentration; further, at 3.0% of IS concentration, the VI-potatoes reached maximum iron content; the porosity of meanwhile changed to 3.75 and 2.75% for the Toyoshiro and Dejima potatoes, respectively. However, the impregnation, deformation, and effective porosity values decreased with IS concentration levels by the end of VI process, but not by the end of the vacuum step. This study generally indicated that the impregnation, deformation, effective porosity and iron content of the VI-potatoes are greatly dependent upon IS concentration levels.
Keywords: Vacuum impregnation; Whole potato; Impregnation and deformation; Effective porosity; Iron concentration
Introduction
Anemia due to iron deficiency is the most common and widespread nutritional anemia in the world and significantly afflicts individuals of all ages and economic groups in both developing and developed countries. The World Health Organization (WHO) estimates that some 2 billion peoples are anaemic, and the majority due to iron deficiency [1]. So, in recent years, scientific and technological attempts have been focused on overcoming mineral undernourishment in the world, because of the deficiency of essential minerals, including iron [2]. Foods today are not intended to only provide necessary nutrients, but to prevent nutrition related diseases and to improve the physical and mental well-being of peoples. In this regard, functional foods play a prominent role. The functional food is a part of human diet and is demonstrated to provide health benefits and to decrease the risk of chronic diseases beyond those provided by adequate nutrition [3-4]. In fact, vacuum impregnation (VI) is employed in a wide range of food processing. It is used to improve product qualities [5], to stabilize structural changes and cell walls [6], to improve sensory properties such as taste [7] and color [8], and to enrich functional ingredients [9-10]. Furthermore, the VI is particularly useful in developing of fruit and vegetable products that have internal pores within structure. It is in this pores structure where functional ingredients such as essential minerals can be introduced and supported [11].
The VI of porous food-matrices such as fruits and vegetables with adequate external solutions or suspensions of functional ingredients has been claimed as a useful way to obtain functional food without destroying their initial matrices, filling only the initial porous fractions with a liquid phase [12]. In particular, plant tissues possess incompact structural matrices, and this defines both their VI response and their mechanical properties as functional food. Gras et al. [13] has evaluated changes in the microstructure of different vegetables by means of Cryo-scanning electron microscopy (Cryo-SEM), and has found that VI can be used to fill intercellular spaces (ICS) in vegetable matrices. The functional ingredients supported in the pores of these structures interact with the food matrix at different levels. Consequently, the development of functional food by means of VI requires analysis of interaction levels in order to optimize process and food properties. The hydrodynamic mechanism enhancing liquid permeation in food matrix has been described and modelled in previous study by Fito [14]. The VI operation consists of three steps in food matrix. The first is of immersing a given product into an external solution containing functional ingredient. The second step is applying vacuum pressure (p1) for a short period of time (t1) in order to promote the outflow of internal gas. The third step restores the atmospheric pressure (p2) for a period of time (t2); it is during this time that in which the hydrodynamic inflow is promoted from the external solution into the pores. The amount of external solution which has impregnated in the food structure (X), due to the action of the hydrodynamic mechanism, has been modelled as a product of the effective porosity (εe), the applied compression ratio (r ≈ p2/p1) and the sample volume deformation (γ). This is all provoked by changes in pressure of VI system [15,16].
Potatoes are an essential food crop occupying a remarkable position within the human diet, and are thus an important food for the ingestion of minerals, vitamins, and other bioactive compounds [17]. With regards to the VI treatment of potatoes, some authors have already investigated the fortification of their nutrient contents [12,18], while others have investigated the effects of VI processing conditions [19,20]. Nevertheless, relationship between the changes in impregnation solution concentration and the sample behavior parameters (impregnation, deformation, and effective porosity) and physicochemical properties were not established in earlier studies. Because the impregnation solution concentration will directly affect the solution viscosity, it effects on mass transfer rate and composition of final food products [16]. Therefore, the purpose of this present study has been to investigate the effect of iron solution (IS) concentration via VI upon the impregnation, deformation, and effective porosity, and iron content of whole potato tubers. Furthermore, the physicochemical properties (moisture content, pH value, total mass variation and density) of whole potato tuber have also been evaluated.
Materials and Methods
Materials
Two potatoes cultivars, the Toyoshiro and the Dejima, were provided by the Hokkaido Agricultural Research Center (Hokkaido, Japan); we took into account homogeneity of size and shape. Potatoes weighing between 130 g and 150 g were selected and stored at 4 oC and 90% of relative humidity before the experiments took places. Ferric pyrophosphate was purchased from Kanto Chemical (Tokyo, Japan). It was used for iron fortification, and is widely used in food enrichment [10, 21-22], as it neither changes colour nor palatability [23,24]. It was prepared with low (0.5 g/100 g), middle (1.5 g/100 g), and high (3 g/100 g) concentrations of IS (Table 1).
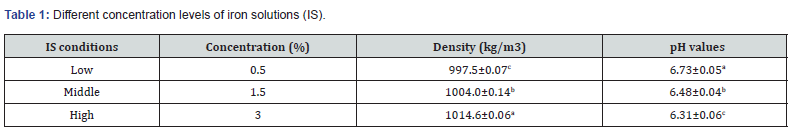
Source: Values are the means ± SD (n = 6). Different letters show significant differences at 0.05 probability.
Vacuum impregnation treatment
Potatoes were taken from storage, washed and dried with tissue papers, then placed at room temperature for 2 h. Whole potato tubers (unpeeled) were measured for initial weight (M0) and volume (V0), then placed into a 2 L beaker containing a desiccator with 0.75 L of IS. Vacuum meter (PM-12, Shimadzu Corporation, Kyoto, Japan) was used to measure pressure, and was placed between vacuum pump (GDH-362, Shimadzu Corporation, Kyoto, Japan) and the desiccator. A vacuum pressure of 800 Pa was then applied to the system for 1 h (vacuum step; t = t1). After this period within the vacuum, atmospheric pressure was restored, and the potato tuber was maintained in the IS for a 3 h of restoration time (restoration step, t = t2) [20]. After the VI process, final potato tuber weight (M2) and volume (V2) were measured.
Description of sample behavior parameters
The impregnation (X) and deformation (γ) levels of VI whole potato at the end of the vacuum step and end of the VI process were calculated by the equations (Eqs. 1 and 2) proposed by previous study [25].
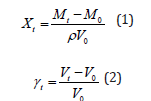
Where: ρ, density of the IS; Mt, mass of liquid impregnated in the sample at the end of the vacuum step (M1) or at the end of the VI process (M2); γt, sample volume deformed at the end of the vacuum step (γ1) or at the end of the VI process (γ).
The effective porosity (εe) of VI whole potato available to the hydrodynamic mechanism action can be calculated, according to the reported mathematical model (Eq. 3) [25].
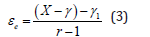
Where r ≈ P2/P1 (compression ratio), P2 = atmospheric pressure and P1 = vacuum pressure.
Physicochemical analysis
Total mass variation (ΔM) of the VI-potatoes was calculated by the following equation:
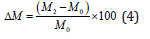
Where: M0 and M2 are the weight of the potato before and after the VI treatment, respectively.
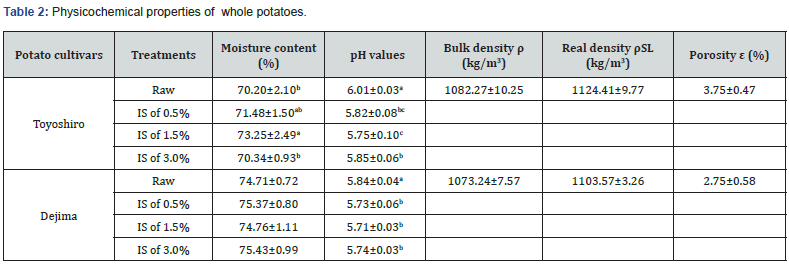
The moisture content of the VI or raw (control; without VI treatment) potatoes was determined using air-drying oven DS-42 (DS-42, Yamato Scientific, Tokyo, Japan) at 105 oC for at least 16 h [26]. The solid contents were then determined based on individual moisture content. 100 g of VI or raw potato were homogenized by a laboratory blender in order to measure pH values [27]. The pH values were measured using pH meter (D-12, Horiba, Kyoto, Japan), having been previously calibrated with buffer solutions (pH 7.0). Bulk density of the raw potatoes was calculated by Blahovec’s equations [28]. This was, determined by weighing the potato tuber both in air and in water at room temperature (25 oC). However, the samples degasifying led to some difficulties, in particular inconsistent solid-liquid values. Thus, real solid-liquid density of raw potato was calculated according to the water-mass fraction (Xw), and previously used in a study by Gras’s [13]. The porosities (ε) of the raw potatoes were calculated from density values: ε = 1-(ρ/ρSL) (Eq. 5).
Cooking
The cooking portion of the experiment was performed according to the methods described in Erihemu’s study [20], and with some modifications. The unpeeled whole potato tubers (raw or VI-potatoes) were placed in a stainless steam-cooker, covered with a lid, and steamed over boiling water at atmospheric pressure for 25 min. This was cooking experiments were done in order to investigate the effects upon their on content of raw and VI-potatoes.
Iron determination
The potato tubers were washed, dried with tissue paper, weighed and diced. A portion was oven-dried at 70 oC (DPS-41, Yamato Scientific Co., Ltd., Tokyo, Japan), until a constant weight was reached. Then, the dehydrated material was ground and passed through a 1 mm sieve, and used for the determination of iron content. Approximately 1 g of the oven-dried sample was put in a muffle furnace at 550 (± 2) oC for 24 h. Subsequently, the incinerated sample was dissolved with hydrochloric acid at 70 oC by a hotplate. Then, the dissolved solution was diluted to 50 mL in a volumetric flask with distilled water. The iron content was determined with an atomic absorption spectrometer system (Z-2010, Hitachi, Tokyo, Japan) [29]. The quantification of iron in the tubers was performed by the calibration curve. Data are expressed as mg 100 g-1 of fresh weight.
Statistical analysis
One tuber was used per treatment, and each treatment was assayed six times. Duncan’s multiple range test of IBM SPSS 20.0 (Chicago, USA) was used to obtain mean values and standard deviation, and to determine the differences between physicochemical property means, total mass variation, volume fractions of impregnation and deformation, effective porosity, and iron content.
Results and Discussion
Effect of IS concentration on physicochemical properties of the VI-potatoes
Table 2 shows the physicochemical properties of the VI-potatoes. The results demonstrate that the moisture content of VI-potatoes (Toyoshiro) increases with IS concentration levels; in particular, at 0.5% of IS concentration, higher moisture content was observed, and reached 73.25%. On the other hand, no significant differences in moisture content were found in the Dejima potatoes (p>0.05). It is important to note that when comparing, the pH values of each potatoes, a lower pH value was found in the VI-potatoes rather than their raw counterparts (p<0.05). Sun and Li [30] observed that the cells of raw potato tissue are in almost perfect contact, although some small intercellular voids exist. These voids are the ICS, which are common in parenchymous tissue of potato. The ICS volumes in potatoes are generally estimated at 1% of their total volume [31]. However, in this study, the porosity/ICS (Table 2) was greater in both potato cultivars, Toyoshiro at 3.75%, and Dejima at 2.75%. This suggests that different potato cultivars may have different porosity/ICS. Fito et al. [32] conducted structural analysis on eggplant and orange peels, and impregnated them via Cyro-SEM with an isotonic solution containing iron and calcium salts. They reported that the higher the porosity, the greater the ICS. The VI efficiency has been reported to depend on a given foods matrix, particularly its porosity/ICS [33]. Therefore in this study, the external iron solution gradually penetrated into the ICS of the whole potato during the atmospheric step (t2). Figure 1 shows the total mass variation (ΔM) values of VI-potatoes with decreased IS concentration levels. In both potato cultivars, the higher ΔM value was found at 0.5% of IS concentration rather than 1.5% and 3.0% (p<0.05). Moreover, at 0.5% of IS concentration, the ΔM values of the Toyoshiro and Dejima were at 4.0% and 5.4%, respectively. Concerning VI in general, Zhao and Xie [16] reported that the conditions of VI plays a significant role in the VI of vegetables, particularly impregnation solution concentration. Yang and Maguer [34] reported that stabilization or decrease in mass transfer occurred when the impregnation solution concentration reached 50-60%. Furthermore, Garrote and Bertone [35] found that an increase in solution viscosity along with increased solution concentration resulted in a decrease of solute transfer rate, which canceled out for the increase in the concentration gradient. Therefore, an increase in IS concentration demonstrates decrease of the ΔM of VI-potatoes, as shows in Figure 1.
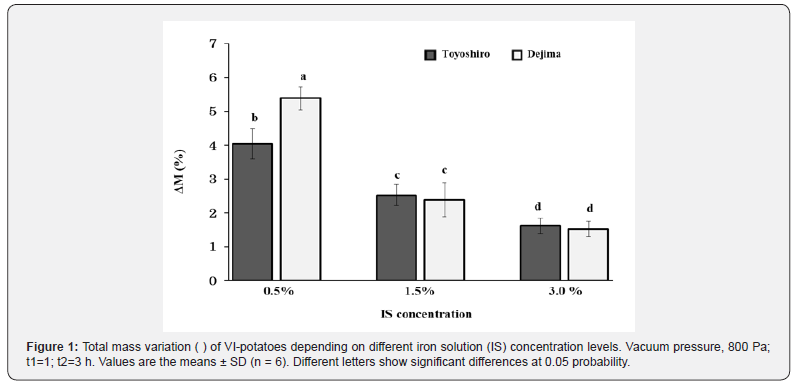
Source: Values are the means ± SD (n = 6). Different letters show significant differences at 0.05 probability. IS: Iron solution.
Effect of IS concentration on the impregnation, deformation and effective porosity levels of VI-potatoes
Figure 2A shows the X value of VI-potatoes at different IS concentration levels. At the end of VI process, the VI-potatoes reached maximum X values at 0.5% of IS concentration (4.4% of Toyoshiro and 5.8% of Dejima), followed by 1.5% and 3.0% of IS concentrations. For the γ value of VI-potatoes (Figure 2B), the greatest γ value increase again occurs at 0.5% of IS concentration (4.2% of Toyoshiro and 5.6% of Dejima), followed again by 1.5% and 3.0%. As mentioned previously, the porosity of the food matrix is the most relevant structural property in VI effectiveness. In plant tissues, this porosity may be high (20-30%) such as in apples, eggplants, or orange peels [36]; thus, and so VI can be highly effective. Gras et al. [6] found that the X value of an eggplant and oyster mushroom at the end of VI was increased, although the carrot’s wasn’t. This is because the carrot has a lower porosity at about 2.2-3.5%, depending on sample thickness. A similar result was found in this study, the potatoes were found to have a lower relative porosity (Table 2). However, the X and γ values of the VI-potatoes at was greatly dependent on IS concentration levels (Figure 2).
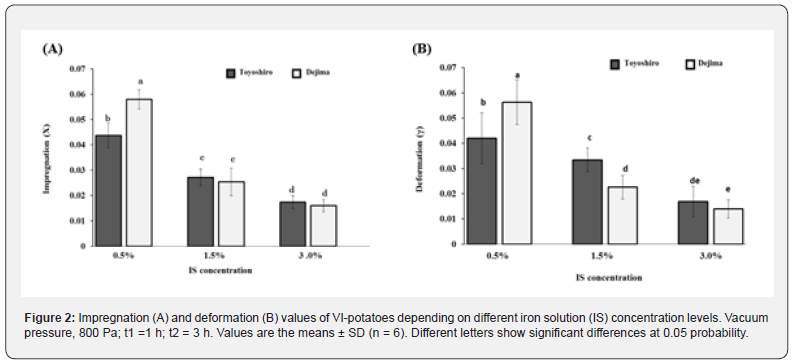
At the end of t1 step, no notable differences were found for the X1 and γ1 values (data not shown). As Gras et al. [6] indicated that the X1 and γ1 values of carrot slices at the end of t1 was very small, and also depended upon slice thickness, size, structural properties, and porosity of samples. For the potato sample, the ICS of potato is very small (about 2.75-3.75%), particularly as in present study compared with that of an apple (25%), peach (15%) and mushroom (37-45%) [37]. Moreover, potatoes have a thick periderm, impermeable to water and gases [38], which protect the mechanical and structural properties of the potatoes tissues, particularly when compared with other studies [6,39]. Therefore, all the internal gas and native liquid (including pores’ native liquid) is difficult to release from the ICS of potato tissue. We reason that this is why no notable difference of X1 and γ1 values at end of the t1 step was found in this study. Table 3 shows that the εe value of VI-potatoes decreased with IS concentration levels. In both potato cultivars, the higher εe value was found at 0.5% of IS concentration (p<0.05): 4.4% for the Toyoshiro and 5.8% for the Dejima potatoes. Previous research has been done into the coupling of the hydrodynamic mechanism with deformation-relaxation phenomena of products solid-matrix [25], this study demonstrated a significant reduction in the volume of gas in vegetable tissues during t2. This is probably due to compression, as atmospheric pressure is restored during this phase. Hence, the external liquid inflows into the porous structure of fruits and vegetables [15], and the εe affects the volume fraction [40]. As previously mentioned, Garrote and Bertone [35] found that an increase in the impregnation solution concentration resulted in a decrease of solute transfer rate in food matrix. Therefore, the lower values of εe at the higher concentration imply that the mechanical equilibrium was not reached, and that the IS penetration did not occur at the maximum level.
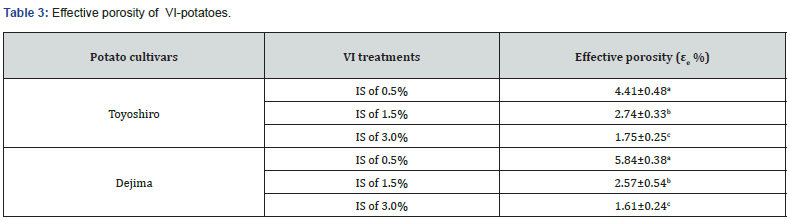
Source: Values are the means ± SD (n = 6). Different letters show significant differences at 0.05 probability. IS: Iron solution.
Effect of IS concentration on the iron content of VI-potatoes
Figure 3A shows the relation between the iron contents of VI-potatoes (Toyoshiro) and IS concentration levels. As shown in this figure, the iron content increased with IS concentration levels; in particular, maximum iron content was reached at the 3.0% of IS concentration, containing 5.7 mg iron per 100 g. Moreover, the VI-cooked potatoes contained higher iron content than the raw-cooked potatoes (p<0.05). At the 3%, 1.5%, and 0.5% of IS concentrations; the VI-cooked potatoes had 6.3, 7.4, and 6.1 times higher iron content than the raw-cooked potatoes, respectively. Also, Figure 3B likewise shows that iron content of the VI-potato (Dejima) also increased with IS concentration levels. It too reached maximum iron content at 3.0% of IS concentration, containing 6.1 mg iron per 100 g. Moreover, at 3%, 1.5%, and 0.5% of IS concentration, the VI-cooked potatoes had 9.7, 9, and 8 times higher iron content than their raw-cooked potatoes. Similarly, Gras et al. [6] indicated that the final X levels of carrot slices (10, 15, and 25 mm) depended on slice thicknesses; the thinner the slice, the greater the X level. Also previously mentioned, Zhao and Xie [16] indicated that the impregnation solution concentration plays a significant role in VI operation; the VI solutions with lower concentrations can be impregnated to a greater extent into the porous tissue of fruits and vegetables, particularly when compared to solutions with higher concentrations and a lowered transfer rate. Furthermore, similar results were observed in this study, although the highest iron content in VI-potatoes was obtained at 3% of IS concentration (Figure 3). Therefore, the IS concentration is an important factor in VI of whole potatoes, as iron content greatly depended upon IS concentration levels. Further study, however, is needed on the iron distribution in the VI-potato tissues.
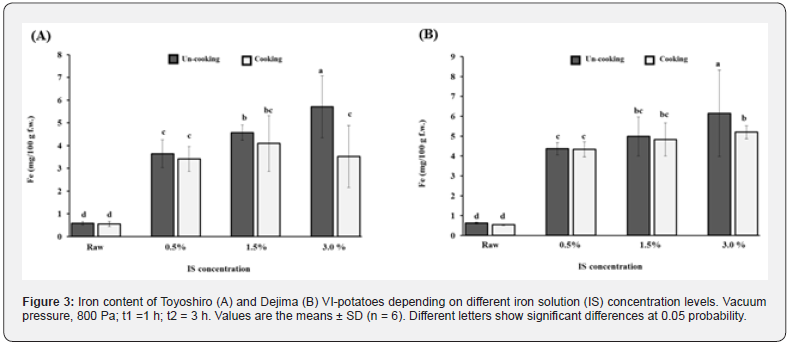
The VI is a useful tool in developing fruit or vegetable products that modify their original composition without disrupting their cellular structure. Gras et al. [6] have shown that many of the calcium ions of VI-products exist only inside the ICS, not inside of cells. Furthermore, in VI-potatoes, vacuum pressure appears to be higher in the outer sections than the inner sections, this is because the outer section is closer to the potato surface. Hironaka et al. [19] indicates that impregnation occurred near the central pith, and the areas between the vascular ring and the periderm of whole potato tuber (at same conditions of t1 and t2). Therefore, the iron content mainly exists in the outer sections of the VI-potatoes. Moreover, Peterson et al. [38] confirms this. They indicate that the lenticels of potatoes hold excess water lost from the internal tissues of the tuber. Heat applied by steam-cooking causes an expansion of internal gas in the ICS of the potato tuber, which may be leads to a loss of iron content from the ICS [41]. Thus, we posit that this is the reason for large losses of iron content in VI-cooked potatoes at 3% of IS concentration (Figure 3).
Conclusion
The results of the present study indicates that the values of X, γ, and εe of the VI-potatoes showed decreased with IS concentration levels by the end of VI process; however, no notable differences in the X and γ values of the VI-potatoes were found at the end of t1 step. Moreover, the iron content of VI-potatoes increased with IS concentration levels; in particular, the VI-potatoes reached maximum iron content at 3.0% of IS concentration. Finally, this study generally indicated that the X, γ, εe, and iron content of VI-potatoes are greatly dependent upon IS concentration levels. These results provide useful information for controlling IS concentration, and thus developing a fortified potato via VI technique. As the colour, taste, and texture are important to consumers, VI-potatoes will need to be further evaluated in future studies.
Acknowledgment
Financial supports from the National Natural Science Foundation of China (NSFC) (Grant No. 31701667) are gratefully acknowledged. Also, the authors are grateful to the Hokkaido Agricultural Research Center (Hokkaido, Japan) for kindly providing the potato samples and the University of the Ryukyus, Okinawa, Japan, for their research facilities.
References
- Radlowski E, Johnson RW (2013) Perinatal iron deficiency and neurocognitive development. Front Hum Neurosci 7: 585.
- Gharibzahedi SMT, Jafari SM (2017) The importance of minerals in human nutrition: Bioavailability, food fortification, processing effects and nanoencapsulation. Trends Food Sci Tech 62: 119-132.
- Al Sheraji SH, Ismail A, Manap MY, Mustafa S, Yusof RM, et al. (2013) Prebiotics as functional foods: A review. J Funct Foods 5(4): 1542-1553.
- Galanakis CM (2021) Functionality of food components and emerging technologies. Foods 10(1): 128.
- Assis FR, Rodrigues LGG, Tribuzi G, de Souza PG, Carciofi BAM, et al. (2019) Fortified apple (Malus spp., var. Fuji) snacks by vacuum impregnation of calcium lactate and convective drying. LWT-Food Sci Technol 113: 108298.
- Gras ML, Vidal D, Betoret N, Chiralt A, Fito P (2003) Calcium fortification of vegetables by vacuum impregnation interactions with cellular matrix. J Food Eng 56(2-3): 279-284.
- Chiralt A, Fito P, Barat JM, Andrés A, González MC, et al. (2001) Use of vacuum impregnation in food salting process. J Food Eng 49(2-3): 141-151.
- Jeon M, Zhao Y (2005) Honey in combination with vacuum impregnation to prevent enzymatic browning of fresh-cut apples. Int J Food Sci Nut 56(3): 165-176.
- Sanzana S, Gras ML, Vidal BD (2011) Functional foods enriched in Aloe Vera. Effects of vacuum impregnation and temperature on the respiration rate and the respiratory quotient of some vegetables. Procedia Food Sci 1(11): 1528-33.
- Mashkour M, Maghsoudlou Y, Kashaninejad M, Aalami M (2018) Iron fortification of whole potato using vacuum impregnation technique with a pulsed electric field pretreatment. Potato Res 61(4): 375-389.
- Yılmaza FM, Bilek SE (2018) Ultrasound-assisted vacuum impregnation on the fortification of fresh-cut apple with calcium and black carrot phenolics. Ultrason Sonoche 48: 509-516.
- Duarte-Correa Y, Granda RD, Cortés M, Vega CO (2020) Potato snacks added with active components: effects of the vacuum impregnation and drying processes. J Food Sci Technol 57: 1523-1534.
- Gras ML, Vidal BD, Betoret N, Chiralt A, Fito P (2002) The response of some vegetables to vacuum impregnation. J Inno Food Sci Emer Technol 3(3): 263-269.
- Fito P (1994) Modelling of vacuum osmotic dehydration of food. J Food Eng 22(94): 313-328.
- Fito P, Chiralt A, Barat JM, Andrés A, Martínez MJ, et al. (2001) Vacuum impregnation for development of new dehydrated products. J Food Eng 49(4): 297-302.
- Zhao Y, Xie J (2004) Practical applications of vacuum impregnation in fruit and vegetable processing. Trends Food Sci Tech 15(9): 434-51.
- Amoroso L, Rizzo V, Muratore G (2018) Nutritional values of potato slices added with rosemary essential oil cooked in sous vide bags. Int J Gastro Food Sci 15: 1-5.
- Joshi A, Kar A, Rudra SG, Sagar VR, Varghese E, et al. (2016) Vacuum impregnation: a promising way for mineral fortification in potato porous matrix (potato chips). J Food Sci Technol 53(12): 4348-4353.
- Hironaka K, Kikuchi M, Koaze H, Sato T, Kojima M, et al. (2011) Ascorbic acid enrichment of whole potato tuber by vacuum impregnation. Food Chem 127(3): 1114-1118.
- Erihemu, Hironaka K, Oda Y, Koaze H (2014) Iron enrichment of whole potato tuber by vacuum impregnation. LWT-Food Sci Technol 59(1): 504-509.
- Almeida CAN, Crott CC, Ricco RG, Del Ciampo LA, Dutra JE, et al. (2003) Control of iron-deficiency an aemia in Brazilian preschool children using iron-fortified orange juice. Nutr Res 23(1): 27-33.
- Tripathi B, Platel K (2011) Iron fortification of finger millet (Eleucine coracana) flour with EDTA and folic acid as co-fortificants. Food Chem 126(2): 537-542.
- Zimmermann MB, Hurrell RF (2007) Nutritional iron deficiency. The Lancet 370(9586): 511-520.
- Navas CS, Pérez AM, Sarriá B, Vaquero MP (2009) Iron absorption from meat pate fortified with ferric pyrophosphate in iron-deficient women. Nutr 25(1): 20-24.
- Fito P, Andrés A, Chiralt A, Pardo P (1996) Coupling of hydrodynamic mechanism and deformation-relaxation phenomena during vacuum treatments in solid porous food-liquid systems. J Food Eng 27(3): 229-40.
- Ihns R, Diamante LM, Savage GP, Vanhanen L (2011) Effect of temperature on the drying characteristics, colour, antioxidant and beta-carotenes of two apricot varieties. Int J Food Sci Technol 46(2): 275-283.
- Angós I, Vírseda P, Fernández T (2008) Control of respiration and color modification on minimally processed potatoes by means of low and high O2/CO2 Posth Bio Technol 48(3): 422-430.
- Blahovec J, Kuroki S, Sakurai N (2007) Cooking kinetics of potato tubers determined by vibration techniques. Food Res Int 40(5): 576-584.
- Brandão GP, Campos RC, Castro EVR, Jesus HC (2007) Determination of copper, iron and vanadium in petroleum by direct sampling electrothermal atomic absorption spectrometry. Spectrochimica Acta Part B 62: 962-969.
- Sun DW, Li B (2003) Microstructural change of potato tissues frozen by ultrasound-assisted immersion freezing. J Food Eng 57(4): 337-345.
- Aguilera JM (1999) Microstructural principles of food processing and engineering. 2nd ed, Aspen Publishers, Gaithersburg, USA.
- Fito P, Chiralt A, Betoret N, Gras M, Cháfer J, et al. (2001) Vacuum impregnation and osmotic dehydration in matrix engineering: Application in functional fresh food development. J Food Eng 49(2-3): 175-183.
- Mújica PH, Valdez FA, López MA, Palou E, Welti CJ (2003) Impregnation properties of some fruits at vacuum pressure. J Food Eng 56(4): 307-314.
- Yang DC, Maguer ML (1992) Osmotic dehydration of strawberries in a batch recirculation system. J Food Qual 15(6): 387-397.
- Garrote RL, Bertone RA (1989) Osmotic concentration at low temperature of frozen strawberry halves. Effect of glycerol, glucose and sucrose solutions on exudates loss and thawing. Lebensm Wissens Technol 22(5): 264-267.
- Fito P, Chiralt A (2000) Vacuum impregnation of plant tissues. In: Alzamora SM, Tapia MS, López MA (Eds.), Design of minimal processing technologies for fruit and vegetables. Aspen Publishers, Gaithersburg, Maryland, USA, pp. 189-201.
- Alzamora SM, Tapia MS, Leunda A, Guerrero SN, Rojas AM, et al. (2000) Minimal preservation of fruits: a cited project. Technomic publishing, Lancaster, Pennsylvania, USA.
- Peterson RL, Barker WG, Howarth WJ (1985) Development and structure of tubers. Academic Press, London, UK.
- Betoret N, Puente L, Díaz MJ, Pagan MJ, García MJ, et al. (2003) Development of probiotic-enriched dried fruits by vacuum impregnation. J Food Eng 56(2-3): 273-277.
- Salvatori D, Andrés A, Chiralt A, Fito P (1998) The response of some properties of fruits to vacuum impregnation. J Food Process Eng 21(1): 59-73.
- Erihemu, Hironaka K, Koaze H, Oda Y, Shimada K (2015) Zinc enrichment of whole potato tuber by vacuum impregnation. J Food Sci Technol 52(4): 2352-2358.






























