Association of The Antitumoral Effect of Bursera Fagaroides with The Inhibition of Kidney Ornithine Decarboxylase in The L5178y Lymphoma Model
Ramón Reynoso-Orozco1, Anne Santerre1*, Jorge Iván Delgado-Saucedo2, Salvador Velázquez-Magaña1 and Ana María Puebla-Pérez2
1Department of Cellular and Molecular Biology, Laboratory of Molecular Biomarkers in Biomedicine and Ecology, University of Guadalajara, México
2Department of Pharmacobiology, University of Guadalajara, México
Submission: May 25, 2020; Published: August 07, 2020
*Corresponding author: Anne Santerre, Centro Universitario De Ciencias Biologicas Y Agropecuarias Camino Ramón Padilla Sánchez No. 2100, Las Agujas, Nextipac, Zapopan, Jalisco, México
How to cite this article: Ramón R-O, Anne S, Jorge I D-S, Salvador V-M, Ana M P-P. Association of The Antitumoral Effect of Bursera Fagaroides with The Inhibition of Kidney Ornithine Decarboxylase in The L5178y Lymphoma Model. Nutri Food Sci Int J. 2020. 10(3): 555786. DOI: 10.19080/NFSIJ.2020.10.555786.
Abstract
Ornithine decarboxylase and polyamines overexpression are key events for cancer growth, and potential markers of cancer outcome, as well as critical targets for cancer prevention or therapy. Bursera Fagaroides is a native plant to Mexico with antitumoral properties in the BALB/c mice L5178Y lymphoma model; however, its mechanism of action is not fully understood.
Thus, to better comprehend the antitumoral effect of B. fagaroides in this model, we evaluated ornithine decarboxylase activity, polyamines profile, total proteins, and tumor growth.
Lymphoma was induced by a single intraperitoneal injection of 2 x 104 L5178Y cells. The hydroalcoholic plant extract was given, daily, intraperitoneal or orally, at 100 mg/Kg body weight for 15 days. Kidney ornithine decarboxylase activity was determined by using a specific radiometric test, polyamines levels by liquid chromatography, and total proteins with the Bradford assay.
Experimental data showed that kidney ornithine decarboxylase activity increased in L5178Y mice compared to healthy mice. Remarkably, the intraperitoneal administration of B. fagaroides induced an 87% drop of kidney ornithine decarboxylase activity compared to untreated L5178Y mice. The oral administration of the plant extract did not show this positive effect. Biologically active polyamines: spermidine and spermine, levels in the kidney were unchanged between groups.
These data highlight that ornithine decarboxylase activity is a biomarker of L5178Y lymphoma growth in BALB/c mice, providing a model of great potential to search for new antitumoral compounds. In this model, the antitumoral property of B. fagaroides involves the inhibition of ornithine decarboxylase activity and the maintenance of polyamines homeostasis.
Keywords: Antitumoral effect; L5178Y lymphoma; Ornithine decarboxylase; Polyamines; Bursera Fagaroides
Abbreviations: ODC: Ornithine Decarboxylase (EC 4.1.1.17)
Introduction
Polyamines (PA), mainly putrescine, spermidine, and spermine are small aliphatic basic molecules that play important roles in cell physiology [1]. They are distributed ubiquitously in all cells and organs and are generally abundant in rapidly dividing cells. PA are necessary for the completion of the cell cycle of normal cells as well as hyperproliferative cancer cells [2-10]. PA homeostasis is the product of complex regulatory mechanisms and signal transduction events that involve de novo biosynthesis, active uptake, and efflux, and complex feedback mechanisms [11-13]. Ornithine decarboxylase (ODC) is the first rate-limiting enzyme for the PA biosynthetic pathway. Although mandatory for PA synthesis, ODC is expressed in small amounts in almost all normal cells and its half-life is one of the shortest of mammalian enzymes (10-30 min), allowing an efficient regulation of its activity and PA biosynthesis [14]. PA levels change rapidly upon modification in the rates of synthesis or degradation of ODC (Graphical abstract).
Because ODC overexpression participates in the transformation of a normal cell to a malignant one, it is a critical target for the prevention and therapy of cancer and other hyperproliferative diseases [6,15]. For example, 2-difluoro-methylornithine (DFMO) is a synthetic molecule that irreversibly binds to ODC active sites, inhibiting enzymatic activity and PA biosynthesis [15,16]. Its effects have been documented in animal models and several human clinical trials, especially for hyperproliferative and infective diseases [10], as well as colon cancer chemoprevention and neuroblastoma [17-19]; however, its clinical use may present cytostatic and noncytotoxic secondary effects [20]. Thus, on an intention to treat hyperproliferative diseases, it is important to pursuit new molecules of natural or synthetic origin that target ODC activity and PA metabolism.
Bursera Fagaroides, a native plant from Mexico, traditionally used in religious and medicinal contexts, presents antitumoral effects in BALB/c mice with L5178Y lymphoma [21]. In a previous study, we showed the utility of urinary PA, especially the diamine putrescine as a biomarker of L5178Y lymphoma growth. The antitumoral activity of B. fagaroides was associated with a decrease in putrescine levels in the urine of treated animals compared to the urine of untreated ones [22]. However, no decisive conclusion could be drawn on the mechanisms of action of the extracts, and if it targets ODC activity and PA metabolism.
The main goals of the present study were two-fold: to evaluate ODC activity and PA levels in the BALB/c L5178Y lymphoma model, and to determine the contribution of both parameters to the antitumoral effect of B. fagaroides.
Materials and Methods
Plant material
Bursera Fagaroides was originally collected during January 1995 in Michoacán State, Mexico (19°40′25″N 101°23′36″O). Identification was performed both at the Instituto Mexicano del Seguro Social´s Herbarium, by A. Aguilar (registry number IMSSM-12051) and by Jaqueline Reynoso-Dueñas, Institute of Botany, University of Guadalajara (registry number IBUG 140748) as previously reported [21]. The hydroalcoholic extract of B. fagaroides was prepared according to Puebla-Pérez et al. [21], and administered to BALB/c mice intraperitoneally (i.p.) or orally at 100 mg/Kg body weight, daily and for 15 days, starting 24 h after tumor cells inoculation [21].
Animals
Thirty male adult BALB/c mice [haplotype] H2d of six to eight weeks of age were used. Animals were maintained in a specific pathogen-free animal care facility in a regulated environment (22±1°C, 50–60% relative humidity, and 12-h light cycle). All mice were fed a commercial diet Lab Chow (Purina Lab Chow–Mexico) with autoclaved tap water ad libitum. All experimental procedures were performed following the protocols approved by the Research Center’s animal care committee and in compliance with national guidelines on animal welfare (Official Mexican standard NOM- 062-ZOO-1999, Technical specifications for production, care and use of laboratory animals) [23].
Study groups
The mice were randomly placed in one of six groups (n=5):
Group (1), mice without treatment (Healthy).
Group (2), healthy mice that received B. fagaroides i.p. (Healthy Bf ip).
Group (3), healthy mice that received B. fagaroides orally (Healthy Bf oral).
Group (4), mice inoculated with L5178Y cells (L5178Y).
Group (5), mice inoculated with L5178Y cells and treated with B. fagaroides i.p. (L5178Y Bf ip).
Group (6), mice inoculated with L5178Y cells and treated with B. fagaroides orally (L5178Y Bf oral).
On the 17th day, mice were weighed, anesthetized in an ether chamber, and then sacrificed.
L5178Y lymphoma and body weight gain
The L5178Y murine lymphoma is a thymic tumor conserved by weekly i.p. transplants in BALB/c mice. It is an excellent experimental model of lymphoma for its rapid growth inside the peritoneal cavity, which provokes weight gain, and the absence of metastatic potential. Therefore, average body weight gain at the end of the experimental period has proven adequate to follow lymphoma growth. L5178Y is very aggressive, and BALB/c mice inoculated i.p. with 20 x 106 L5178Y cells die within 15 ± 2 days [21]. In the present work, the tumor was induced on the first day of the experiment by a single peritoneal injection of 2 x 104 L5178Y cells.
ODC radiometric assay
ODC is expressed in small amounts in almost all cells, but its expression has been reported higher in the kidney [24,25]. In a recent report, we confirmed higher levels of PA in the kidney than in the intestine of L5178Y mice [26]. Additionally, in a preliminary evaluation, the ODC activity showed to be 1000 times higher in kidney than in the liver, mesothelium, and small intestine (data not shown); thus, based on these preliminary data kidneys were used here to evaluate ODC activity. At the moment of dissection, a fragment of the right kidney was obtained and immediately processed for ODC enzyme activity assay. The rest of the kidney was stored at -80 °C for later PA extraction and HPLC analysis. For the determination of the ODC activity, a crude extract was prepared from homogenized kidney, as described by Arteaga- Nieto et al. [27], except that the buffer was 100 mM Tris-HCl, pH 7.5. The specific enzymatic activity was assayed using labeled ornithine (L-[1-14Carboxyl]-ornithine from Amersham Biosciences, Little Chalfont, Buckinghamshire, England, specific activity: 1.92 GBq/nmol, concentration 0.56 mM), and measuring the radioactive 14CO2 generated by ODC. Radioactivity was measured in a Beckman beta counter and results were expressed as 14CO2 released in pmols / mg total protein / 90 min. Total protein concentration was determined according to Bradford [28]. ODC activity was expressed as pmols 14CO2 released / mg total protein / 90 min.
Polyamines extraction from kidney
The frozen kidney sample was thawed on ice and homogenized mechanically in a solution containing 5% trichloroacetic acid solution, and 0.05N hydrochloric acid (TCA-HCl; 2 μL per mg of tissue). After centrifugation (10,000 rpm, 20 min, 4°C), the supernatant was transferred to a fresh tube and stored at -80 °C until analysis. For the chromatographic analysis, 20 μL of the supernatant, 60 μL of TCA-HCl, and 20 μL of diamine-heptane (0.1 nmol/μL; control peak) were mixed, vortexed, pulse-centrifuged and injected onto the chromatograph.
Ion exchange Chromatography
The separation of the three main biogenic polyamines: putrescine (Pu, diamine), spermidine (Spd, triamine), and spermine (Spm, tetraamine) was based on ion exchange using a BCX12, 7-10 μm cationic exchange resin (Benson CO, Reno, Nevada, USA). This method was carried out by HPLC according to Villanueva and Adlakha [29], using a Shimadzu fluorometer (excitation 345 nm, emission 456 nm) and a 100 μL sample loop automatic injector AS3000 (Spectra-Physics) with o-phthalaldehyde (OPA, ICN Biomedical, Eschwege, Germany) post-column detection. All analyses were duplicates. Raw data visualization, treatment, and analysis were made possible thanks to the creation of a special extensive object-oriented instrument controller based on Linux [30]. Data are expressed as pmols/mg of tissue.
Statistical analysis
Results are expressed as the mean ± standard error of the mean (SEM). Data were analyzed by one-way analysis of variance (ANOVA), and for the comparison of significance between groups Tukey post-hoc test was used. The probability level of p˂0.05 was considered significant. Data analyses were performed using the GraphPad Prism 5.0 software.
Results
Ornithine decarboxylase activity
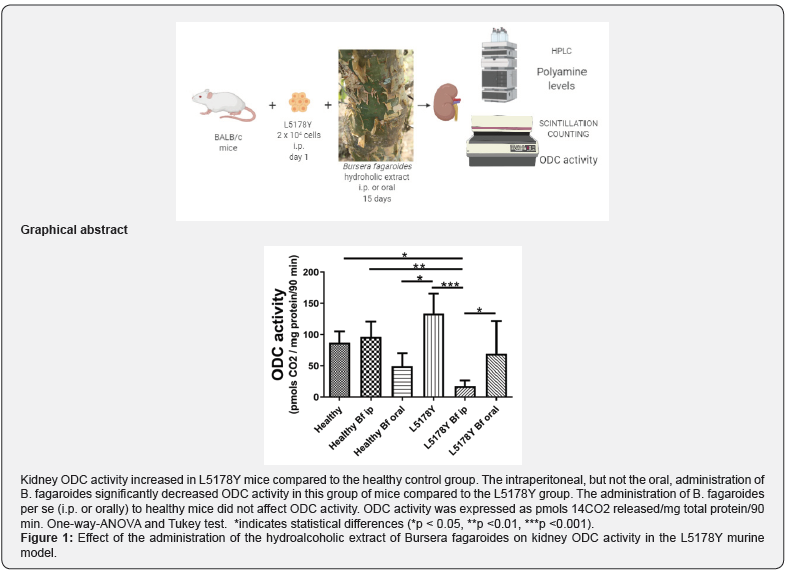
Figure 1 presents kidney ODC activity, expressed as pmols 14CO2 release / mg total protein / 90 min. Kidney ODC activity, increased in L5178Y mice (134.11 ±31.64) compared to healthy mice (87.59 ±17.44) even though the difference did not quite reach significance. Remarkably, kidney ODC activity dropped 87% in L5178Y mice that received B. fagaroides i.p. (17.67±9.3, L5178Y Bf ip group) compared to untreated L5178Y mice. However, the oral administration of B. fagaroides to L5178Y mice (L5178Y Bf oral group) did not significantly decrease ODC activity (95.10±72.97) compared to the L5178Y animals. The administration of B. fagaroides to healthy mice, either i.p. (96.29 ±24.75, Healthy Bf ip) or orally (49.38±20.95, Healthy Bf oral), did not significantly affect ODC activity.
Body weight gain
The L5178Y mice (L5178Y group) presented a noticeable increase in body weight compared to healthy mice (Healthy group). Remarkably the i.p. administration of B. fagaroides maintained the weight of L5178Y mice to that of healthy animals. However, the oral administration of B. fagaroides to L5178Y mice lost that positive effect, and mice from the L5178Y Bf oral group suffered an important body weight gain, compared to the healthy group, and similar to that of the L5178Y group of mice. The chronic (15 days) administration of B. fagaroides, either i.p. or orally, did not affect the body weight of healthy mice. These results coincide with previous report in the same model [22].
Correlation between ODC activity and body weight gain
The dispersion plot presented in figure 2 highlights the relationship between ODC activity and animals’ weight gain among the different experimental groups. Figure 2 evidences two clusters: one formed by the groups of healthy mice, the mice that received B. fagaroides alone (i.p. or orally), and the L5178Y mice successfully treated with B. fagaroides i.p. This cluster presented normal body weight, low/mid-ODC activity at the end of the experimental period, and a good life expectancy. In contrast, the other cluster is formed by the L5178Y mice and the L5178Y mice that received B. fagaroides orally. These groups of mice presented tumor growth (reflected in important weight gain: + 5.2 g and + 4.83 g, respectively), increased ODC activity, and low life expectancy.

Polyamines determination in kidney
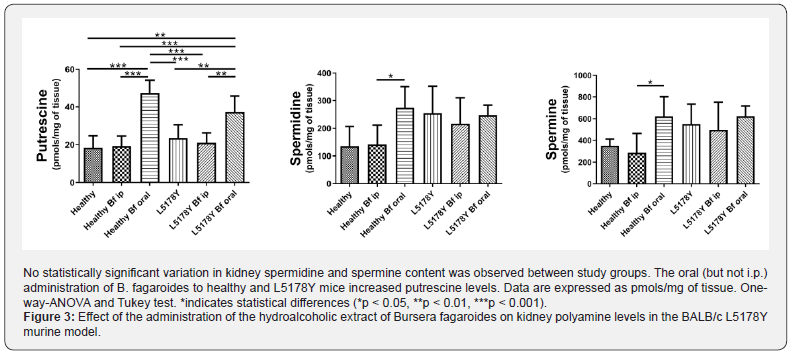
As shown in Figure 3, the oral administration of B. fagaroides (Healthy Bf oral and L5178Y Bf oral groups), significantly increased putrescine levels in the Healthy (p<0.001) as well as L5178Y (p<0.01) groups of mice. Meanwhile the i.p. administration of the plant extract did not present this effect, neither in Healthy nor in L5178Y mice. The levels of putrescine were similar in the four groups of mice that did not receive B. fagaroides orally. No statistically significant variation in kidney spermidine and spermine content was observed between study groups, except between the groups of healthy mice that received B. fagaroides orally (p<0.05).
Total protein concentration in kidney
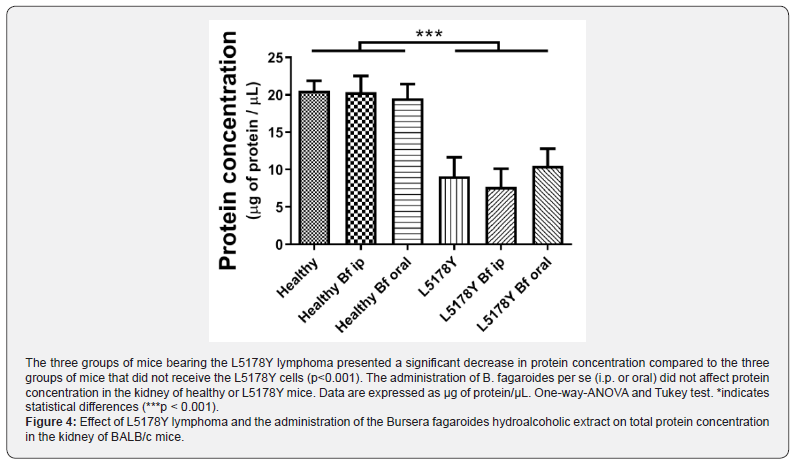
Figure 4 shows that the presence of L5178Y tumor cells significantly decreased the concentration of total kidney proteins in the three groups of mice injected L5178Y cells, compared to animals that did not receive the tumor cells. The administration of B. fagaroides either i.p. or orally to healthy mice did not affect protein concentration.
Discussion
ODC activity is a biomarker of L5178Y lymphoma growth. ODC enzyme is the first rate-limiting enzyme of PA biosynthesis; it is responsible for the synthesis of the diamine putrescine, the precursor of the biologically active spermidine and spermine. ODC activity is essential for normal cell growth; nevertheless, an increase of its activity beyond a certain threshold exacerbates cell proliferation [1,14,17].
Our experimental data revealed an elevated kidney ODC activity in L5178Y mice, which matches with the increase in body weight due to noticeable peritoneum growth in this group of mice. However, the levels of spermidine and spermine in the kidneys did not reflect the increase of ODC activity in this organ. Taking into account that biosynthesis, transport regulation (uptake and outtake), and catabolism are important for the maintenance of PA homeostasis [4], we hypothesize that the podocytes used compensatory mechanisms to keep adequate PA levels within the kidney. These mechanisms probably include the conversion of excesses of spermidine and spermine back to putrescine (the precursor of spermidine and spermine), through the activation of polyamine oxidases, spermine oxidases and acetylpolyamine oxidases [11,14,20], followed by an enhanced excretion of putrescine to body fluids, as shown in a previous report in which we observed high levels of putrescine in the urine of L5178Y mice [22]. Other mechanisms apart from stimulation of PA conversion and outtake, may involve a decreased in PA uptake [5,14], or activation of ODC antizymes to down-regulate ODC expression [2- 4].
In the present work, the data from the Bradford assay showed that mice injected with L5178Y tumor cells contained lower levels of total protein in the kidney than the healthy BALB/c mice. This finding may indicate that the lymphoma growth and the high ODC activity, affected the kidney’s physiology. As mentioned above, this tissue presented normal levels of spermidine and spermine, probably thanks to the activation of oxidases involved in PA catabolism; these enzymes are a source of reactive oxygen species (ROS, mainly in the form of H2O2) which increase oxidative stress and contribute to cellular damage [20] which may have induced renal failure [31]. Thus, our findings suggest that in their intent to keep PA homeostasis through the activation of catabolic enzymes, the podocytes also contribute to the production of ROS which in turn provokes renal impairment and proteinuria. This observation also coincides with an earlier report from de Blaauw et al. who reported that proteins are chronically wasted during cancer process in a tumor-bearing rat model induced with methylcholantrene [32].
ODC inhibition is a possible mechanism of the antitumoral effect of B. fagaroides
The main contribution of the present work is to put forward that the antitumoral effect of B. fagaroides given i.p. involves the inhibition of kidney ODC activity. We showed that L5178Y mice treated i.p. with the plant extract presented an 87 % decrease in ODC activity compared to untreated L5178Y mice. This decrease coincides with lower levels of urinary putrescine and no weight gain compared to healthy mice [21,22]. Thus, ODC inhibition by B. fagaroides prevented the accumulation of PA and therefore tumor growth. Two mechanisms of actions can be debated. On one hand, the extract may contain secondary metabolites that directly inhibit ODC activity as a way to control cell proliferation. To further prove this point, studies are in progress and the extract is under chemical characterization in order to isolate and identify fractions or molecules, in particular triterpenoids and lignins (podophyllotoxin, pers. com. de JIDS) that have been linked to reducing the risk of cancer and other major chronic diseases, per se or in synergistic with other phytonutrients or drugs [33]. On the other hand, based on the observation of scar-like structure within the peritoneum as part of the wound healing process after mechanical action of the needle during the administration of the extract, we hypothesize that the extract may have favored the development of fibrosis within the peritoneal cavity and the formation of a physical barrier that isolated the L5178Y cells inside the peritoneum (abdominal cavity) and inhibited their proliferation due to the lack of nutrients.
The administration of B. fagaroides either i.p. or orally to healthy mice (control mice) for 15 consecutive days did not significantly affect body weight, ODC activity, spermidine, spermine or total protein levels, indicating that the plant extract did not present secondary effects on the global metabolism of BALB/c mice, especially in kidney.
The evaluation of body weight gain and ODC activity highlighted the loss of the antitumoral activity when the extract was given orally. The oral ingestion of the extract and its travel through the gastrointestinal tract, which is an aggressive environment, probably affected its biological activity by changing its composition.
We also observed that the oral (but not i.p.) administration of the plant extract significantly increased the levels of putrescine in kidney of healthy and L5178Y mice. More investigation is now needed to investigate the relationship between the potential anti-inflammatory effect of putrescine [34] and the known anti-inflammatory effect of the oral administration of Bursera fagaroides [35,36].
Conclusion
Kidney ODC activity (this work), and urinary putrescine [22] are useful biomarkers of L5178Y lymphoma growth in BALB/c mice. This is an important point for future projects that foresee the search for novel cancer therapies that target PA metabolism. Focusing then our attention on B. fagaroides, we showed that the antitumoral effect of the crude extract from this native plant of Mexico, involves, at least in part, the inhibition of ODC activity in kidney and the control of PA homeostasis. Further work is now needed to point out the particular groups of metabolites responsible for this effect.
Acknowledgments
This study was supported by fundings from the P3E Program of the Universidad de Guadalajara and the Programa de Fortalecimiento de la Calidad Educativa - Secretaria de Educación Pública (PFCE-SEP), Mexico. Our special thanks to PhDs Victor Raul Villanueva† who inspired this work, and to Pablo Arteaga- Nieto and Carlos Calvo-Mendez, from the University of Guanajuato, Mexico, for their support with the ODC protocol.
Author Disclosure Statement
The authors declare that they have no conflicts of interest with the content of this article.
References
- Bae DH, Lane DJR, Jansson PJ, Richardson DR (2018) The old and new biochemistry of polyamines. BiochimBiophys Acta Gen Subj 1862(9): 2053-2068.
- Pegg AE, Feith DJ, Fong LYY, Coleman CS, O´Brien TG, et al.(2003) Transgenic mouse models for studies of the role of polyamines in normal, hypertrophic, and neoplastic growth. Biochem Soc Trans 31(2): 356-360.
- Pegg AE (2006) Regulation of ornithine decarboxylase. J Biol Chem 281(21): 14529-14532.
- Pegg AE (2018) Introduction to the Thematic Minireview Series: Sixty plus years of polyamine research. J Biol Chem 293(48): 18681-18692.
- Babbar N, Gerner EW (2011) Targeting polyamines and Inflammation for cancer prevention. Recent Res Cancer 188: 49-64.
- Nowotarski SL, Woster PM, Casero Jr RA (2013) Polyamines and cancer: implications for chemotherapy and chemoprevention. Expert Rev Mol Med 15: e3.
- Meyskens FL, Simoneau AR, Gerner EW (2014) Chemoprevention of prostate cancer with polyamine synthesis inhibitor difluoromethylornithine. In Cuzick J, Thorat M (eds) Prostate Cancer Prevention. Recent Results in Cancer Research vol 202 Springer, Berlin, Heidelberg.
- Kusano Tomonobu, Suzuki Hideyuki (2015)Polyamines, a universal molecular nexus for growth, survival, and specialized metabolism, Springer,Tokyo, Japan.
- Irecta-Nájera CA, Huizar-López M, Casas-Solís J, Castro-Félix P, Santerre A (2017) Protective effect of Lactobacillus casei on DMH-induced colon carcinogenesis in mice. Probiotics Antimicro Proteins 9(2): 163-171.
- LoGiudice N, Linh Le, Abuan I, Leizorek Y, Roberts SC (2018) Alpha-difluoromethylornithine, an irreversible inhibitor of polyamine biosynthesis, as a therapeutic strategy against hyperproliferative and infectious diseases. MedSci(Basel) 6(1): 12.
- Mandal S, Mandal A, Johansson HE, Orjalo AV, Park MH (2013)Depletion of cellular polyamines, spermidine and spermine, causes total arrest in translation and growth in mammalian cells. Proc Natl Acad Sci U S A. 110(6): 2169-2174.
- Miller-Fleming L, Olin-Sandoval V, Campbell K, Ralser M (2015) Remaining mysteries of molecular biology: The role of polyamine metabolites in the cell. J Mol Biol 427(21): 3389-3406.
- Casero Jr RA, Murray Stewart T, Pegg AE (2018) Polyamines metabolism and cancer: treatment, challenges, and opportunities. Nat Rev Cancer 18: 681-695.
- Flynn AT, Hogarty MD (2018) Myc, oncogenic protein translation, and the role of polyamines. Med Sci 6(2):41.
- Murray-Stewart TR, Woster PM, Casero Jr RA (2016) Targeting polyamines metabolism for cancer therapy and prevention. Biochem J 473(19): 2937-2953.
- Gerner EW, Meyskens Jr FL, Goldschmid S, Lance P, Pelot D (2007) Rationale for, and design of, a clinical trial targeting polyamine metabolism for colon cancer chemoprevention. Amino Acids 33(2): 189-195.
- Arruabarrena-Aristorena A, Zabala-Letona A, Carracedo A (2018) Oil for the cancer engine: The cross-talk between oncogenic signaling and polyamine metabolism. Sci Adv 4(1): eaar2606.
- Bachmann AS, Geerts D (2018) Polyamine synthesis as a target of MYC oncogenes. J Biol Chem 293(48): 18757-18769.
- Sholler GLS, Ferguson W, Bergendahl G, Bond P, Neville K, et al. (2018) Maintenance DFMO increases survival in high risk neuroblastoma. Sci Rep 8: 14445.
- Park MH, Igarashi K (2013) Polyamines and their metabolites as diagnostic markers of human diseases. BiomolTher (Seoul) 21(1): 1-9.
- Puebla‐Pérez AM, Huacuja‐Ruiz L, Rodríguez‐Orozco G, Villaseñor‐García MM, Miranda‐BeltránML, et al. (1998) Cytotoxic and antitumour activity from Bursera fagaroides ethanol extract in mice with L5178Y lymphoma. Phytotherapy Res 12(8): 545-548.
- Reynoso-Orozco R, Santerre A, Delgado-Saucedo JI, Casas-Solis J, Velásquez-Magaña S, et al. (2008)Polyamines as biomarkers of the antitumoral activity of Bursera fagaroides. Interciencia 33(5): 384-388.
- NOM-062-ZOO-1999 (2001) Secretaria de Agricultura Ganadería, Desarrollo Rural, Pesca y Alimentación: Norma oficialmexicana NOM-062-ZOO-1999, Especificacionestécnicas para la producción, cuidado y uso de animales de laboratorio, Edited by DiarioOficial de la Federación.
- Tovar A, Sánchez-Capelo A, Cremades A, Peñafiel R (1995). An evaluation of the role of polyamines in different models of kidney hypertrophy in mice. Kidney Int 48(3): 731-737.
- Hayashi T, Tsujino T, Iwata S, Nonaka H, EmotoN, et al. (2002)Decreased ornithine decarboxylase activity in the kidneys of Dahl Salt-sensitive rats. Hypertens Res 25(2): 787-795.
- Reynoso-Orozco R, Santerre A, Puebla-Pérez AM, Noa-Pérez M (2018) Effect of a hydroalcoholic extract of Bursera fagaroides on intestinal and kidney polyamine levels in the murine lymphoma model L5178Y. Revista de Ciencias de la Salud 5(14): 1-8.
- Arteaga-Nieto P, Villagómez-Castro JC, Calvo-Méndez C, López-Romero E (1996) Partial purification and characterization of ornithine decarboxylase from Entamoeba histolytica. Int J Parasitol 26(3): 253-60.
- Bradford M (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein dye binding. Anal. Biochem. 72(1-2): 248-254.
- Villanueva V, Adlakha R (1978) Automated analysis of common basic amino acid, mono-di and polyamines, phenolocamines, and indoleamines in crude biological samples. Anal Biochem 91(1): 264-275.
- Carr DW, Ruelas R, Reynoso-Orozco R, Santerre A (2002) An extensive object-oriented instrument controller for Linux.
- Yaribeygi H, Farrokhi FR, Rezaee R, Sahebkar A (2018) Oxidative stress induces renal failure: a review of possible molecular pathways. J Cell Biochem 119(4): 2990-2998.
- de Blaauw I, Deutz NEP, Hulsewe KW, von Meyenfeldt MF (2003) Attenuated metabolic response to surgery in tumor-bearing rats. J Surg Res 110(2): 371-377.
- Kale A, Gawande S, Kotwal S (2008) Cancer phytotherapeutics: role for flavonoids at the cellular level. Phytother Res 22(5): 567-577.
- Lagishetty CV, Naik SR (2008) Polyamines: Potential anti-inflammatory agents and their possible mechanism of action. Indian J Pharmacol 40(3): 121-125.
- Alonso-Castro AJ, Villarreal ML, Salazar-Olivo LA, Gómez-Sanchez M, Domínguez F, et al. (2011) Mexican medicinal plants used for cancer treatment: Pharmacological, phytochemical and ethnobotanical studies. J Ethnopharmacol 133(3): 945-972.
- Acevedo M, Nuñez P, Gónzalez-Maya L, Cardos-Taketa A, María Luisa Villarreal, et al.(2015) Cytotoxic and anti-inflammatory activities of Bursera species from Mexico. J Clin Toxicol 5(1): 232.






























