Antiproliferative Effect of Green Papaya on Lymphocytic Leukemic Cells
Zainab Allqmani1,3, Alexus Greene1,3, Haiwen Li1, Rafie Reza2, Andrea R Beyer3, Sarah M Witiak3, Paul A Kaseloo3 and Rafat A Siddiqui1*
1 Food Chemistry and Nutrition Science Research Laboratory, Virginia State University, USA
2Cooperative Extension, Agricultural Research Station, College of Agriculture, Virginia State University, USA
3 Department of Biology, College of Natural and Health Sciences, Virginia State University, USA
Submission: May 22, 2020; Published: June 22, 2020
*Corresponding author: Rafat A Siddiqui, Food Chemistry and Nutrition Science Research Laboratory, Agricultural Research Station, College of Agriculture, Virginia State University, Petersburg, VA 23806, USA
How to cite this article: Zainab A, Alexus G, Haiwen L, Rafie R, Andrea B, et al. Antiproliferative Effect of Green Papaya on Lymphocytic Leukemic Cells. Nutri Food Sci Int J. 2020. 10(2): 555781. DOI: 10.19080/NFSIJ.2020.10.555781.
Abstract
Numerous edible plants have been reported to interfere with the carcinogenic process, and therefore, the regular consumption of these plant products may reduce the risk of developing cancer. We investigated the effect of papaya fruit and leaves on the cell proliferation of Jurkat T-lymphocytic and Daudi B-lymphocytic leukemia cells. Cells were treated with aqueous or methanolic extracts from leaves, skin, pulp, and seeds from green papaya. The papaya fractions were tested for total phenolic content, total flavonoids content, and anti-oxidation activity using chemical assays. Cell proliferation was measured using a WST-1 assay. Our data indicate that methanol and water extracts of seeds and leaves contained higher concentrations of total phenolic compounds and higher anti-oxidation activity than that of extracts from skin and pulp.
Both methanol and water extracts from leaves and skin potently inhibited the proliferation of leukemic Jurkat T-cells and Daudi B-cells. However, the effect was more potent on Jurkat T-cells, and the leaf extracts were more effective than that of skin extracts. None of the pulp or seed extracts showed inhibitory activity on leukemic cell proliferation. Although papaya leaves are not consumed as a food, leaf extracts have been used for the treatment of various conditions, including dengue and malaria fevers, gastric ulcers, low platelet counts, and cancers of the breast, lung, and cervix. Our data suggest that the consumption of papaya leaf extracts may also be beneficial in preventing and/or treating lymphocytic cancer. Isolation of active compounds from papaya leaves will also help in developing new drugs for cancer treatment.
Keywords: Polyphenols; Antioxidants; Anticancer; Jurkat cells; Daudi cells; Leukemia
Introduction
Leukemia is a cancer of the blood or bone marrow, as described by an anomalous multiplication of immature clonal hematopoietic cells. Leukemia is one of the cancers that can affect all races in the United States. The American Cancer Society estimates that there will be around 60,530 new cases of leukemia in the United States in 2020, resulting in 23,100 deaths [1]. Leukemia is classified based on its onset (acute or chronic), the affected blood cell type (lymphoblastic/lymphocytic or myeloid/myelogenous), the maturity stage of the blood cell, and phenotypic expression of the disease [2]. Leukemia, like other forms of cancer, can be treated by using a number of methods, including chemotherapy, radiation therapy, targeted therapy, or stem cell therapy, though it is easier to treat acute leukemia than chronic leukemia [3]. Although leukemia remains a common medical issue, there has been a decrease in incidence due to preventive measures, and death rates have also declined since 1991 due to recent advances in treatment strategies [4]. The 5-year survival rate for children with acute lymphocytic leukemia has greatly increased over time to about 90% [5]. Although there are improved treatment strategies and survival rates, the cost of such therapies remains an issue that hinders access for all patients [6]. There is a need for less expensive alternative strategies to increase treatment access and survival outcomes for all patients.
Recent advances in alternative and complementary medicine have shown that various herbs, fruits, and vegetables have compounds that could act as inhibitors of cancer formation, blockers of carcinogen interaction, and suppressors of tumor progression. For example, Korean red ginseng extract treatment inhibited the growth of U937 leukemia cells by downregulating the expression of human telomerase reverse transcriptase [7]. Another study confirmed that Kenaf seed oil causes the death of human leukemia cells HL60 and K562 by induction of apoptosis [8]. Studies demonstrated that a hot water extract of Euphorbia formosana (EFW) restrained the growth of THP-1 human leukemic cells by inducing apoptosis through caspase-dependent cell death via Fas and mitochondrial pathways [9]. Water, ethanol, and methanol extracts of garlic have exhibited lethal cytotoxic effects of 50-75% on cells harvested from 3 patients with acute lymphoblastic leukemia (ALL) and three patients with acute myeloid leukemia (AML) [10]. Typhonium flagelliforme (TF), a herbal plant in Malaysia, exhibited cytotoxic effects in P388 [11] and WEHI-3 [12] murine leukemia cells. Further, in vivo studies showed a significant decline in the cell count of immature monocytes and immature granulocytes, when the TF extract was orally administered for 28 days in a BALB/c leukemic mouse model [12]. Studies with grape seed extracts reported induction of apoptosis in Jurkat leukemic cells through a process that involves sustained JNK activation and Cip1/p21 up-regulation, culminating in caspase activation [13].
Papaya fruit has also been studied for its anticancer activities for colorectal [14], prostate [15,16], cervical [17], breast [18- 20], and skin cancers [21]. Papaya also effectively inhibits cancers of hematopoietic cell lines, including T cell lymphoma (Jurkat), plasma cell leukemia (ARH77), Burkitt’s lymphoma (Raji), anaplastic large cell lymphoma (Karpas-299), and human promyelocytic leukemia (HL-60) [22-23]. A study demonstrated that Benzyl isothiocyanate (BITC), a promising chemo preventive agent found in papaya fruits, when given followed by cisplatin (BITC/cisplatin) caused significant death in HL-60 cells [24]. Furthermore, a recent study reported that papaya leaf extract improved bone marrow blast counts and stabilized the hematological parameters of a 76-year-old male with untreated chronic myelomonocytic leukemia [25].
It is well Known that fruits, vegetables, and other medicinal plants contain polyphenols and flavonoids that exhibit various biological and pharmacological properties [26], which are often mediated by their anti-oxidation activity [27]. Based on these earlier studies, we evaluated water and methanol extracts from leaves, skin, pulp, and seeds of green papaya on T-lymphocytic leukemia and B-cell lymphocytic leukemia using Jurakt and Daudi cell lines, respectively, and hypothesized that the effect of papaya fractions on cell proliferation of leukemic cells is correlated to their phenolic contents and/or anti-oxidation activity.
Materials and Methods
Materials
The Jurkat T-cell line and Daudi B-cell line were purchased from ATCC (Manassas, VA). RPMI 1640 medium was purchased from Gibco (Grand Island, NY). Fetal Bovine Serum (FBS) was purchased from RAMBIO (Missoula, MT). Penicillin and streptomycin antibiotics (BP2959) and Phosphate Buffered Saline (PBS; BP399-550) were purchased from Fisher Scientific (Fair Ln, NJ). Folin Ciocalteu, sodium carbonate, aluminum chloride, Diphenyl-1-picrylhydrazyl (DPPH), quercetin, gallic acid, and trolox were purchased from Sigma Chemical Co. (St Louis, MO). WST-1 (MK400) was purchased from Takara (Kusatsu, Shiga, Japan). Green papaya was obtained from Randolph Farm at Virginia State University, Petersburg, VA.
Isolation of papaya leaves, skin, pulp, and seeds
The papaya samples were isolated as described previously [28]. Briefly, the papaya was washed with distilled water and blotted dry with a paper towel. The skin was peeled off using a kitchen peeler. An unskinned papaya was cut into half to remove seeds, and then the pulp was cut into small pieces. The leaves and seeds were washed with distilled water. All fractions (leaves, skin, pulp, and seeds) were freeze-dried. The dried leaves, skin, pulp, and seeds were ground to a fine powder using a mortar and pestle with liquid nitrogen added to keep the powder frozen. The dried powder was flash-frozen with liquid nitrogen and stored at -80 ℃ until used.
Preparation of extracts from papaya leaves, skin, pulp and seeds
The methanol and water extracts of papaya fractions were prepared as described previously [28]. Briefly, a known quantity (5g) of different fractions of freeze-dried papaya powder was homogenized with 200ml of 80% methanol or distilled water. The mixtures were placed on a shaker at room temperature overnight. The next day, the mixture was centrifuged at 2000 x g for 20 minutes using a Thermo Scientific centrifuge (Waltham, MA). The supernatants were collected, and the residues were washed two times by resuspending in the respective solutions, mixing, and placing on a shaker overnight. The collected supernatants were pooled, and the insoluble residues were discarded. The methanol extracts were dried in a nitrogen evaporator (Organomation Associates, Inc, Berlin, MA), whereas the water extract was freezedried. The dried extracts were stored in a -20℃ freezer. The dried extract was suspended into DMSO at a concentration of 250mg/ ml and was further diluted into media for cellular studies.
Characterization of anti-oxidation activity of papaya extracts
The oxidative properties of papaya were determined by assaying their total polyphenolic content (TPC), total flavonoid content (TFC), and anti-oxidation capacity (DPPH) as previously described [29].
Cell culture
Jurkat T-cells and Daudi B-cells were maintained in RPMI 1640 media supplemented with penicillin (100 units/ml), streptomycin (100μg/ml) and 10% FBS. All cell cultures were incubated in a humidified incubator at 37 °C and 5% CO2. Media was changed every three days, and cells were subcultured when they became confluent.
Cell proliferation assay
The effects of papaya leaf, skin, pulp, and seed extracts on cell proliferation were determined using a WST-1 assay as per manufacturer instructions and described previously [28]. Briefly, cells were isolated from a confluent flask, and about 10,000 cells were placed in each well of a 96 well plate and incubated in a CO2 incubator for 24 hours. The next day, media was removed, and cells were incubated in serum-free media containing varying concentrations of papaya leaf, skin, pulp, or seed extract for 24 hours. After treatment with papaya extract, 10 μl of WST-1 was added in each well, and the plate was read at 420 nm in a 96 well plate reader for the background reading. The plate was then incubated for 3 hours at 37 ℃ in a CO2 incubator, and the reduction of WST-1 dye due to mitochondrial oxidation was recorded at 420 nm. The change in absorbance was calculated by subtracting the initial reading from the final reading. The results are expressed as % change from control (untreated cells).
Data analysis
The data are expressed as mean ± SD for at least three replicates. All comparisons were made by one-way ANOVA with Tukey’s -HSD-post-hoc test using SPSS Statistics 20 software (IBM, Armonk, NY). All significant differences are reported as P < 0.05 and indicated by “*” whereas non-significant differences are not marked.
Results
Characterization of papaya extract for anti-oxidation activity
The anti-oxidation potentials of papaya extracts were determined by assessing their total polyphenol content, total flavonoid content, and by assaying their anti-oxidation capacity. The data showing the total phenolic contents are presented in Table 1 and reported previously [28]. The highest amounts of polyphenols were found in the seed extracts that ranged 14 -16 mg/g dry weight. There was no significant difference in total phenolic contents between water and methanol extracts from papaya seeds. The leaves were second highest in total phenolic content but had a significantly lower amount of total phenolic content than that of seeds (P < 0.05). The leaves contained 1- 4 mg/g dry weight total phenolic content. Water extracts of leaves contained less total phenolic content (~ 1 mg/g dry weight) compared to that of methanol extracts. The skin extracts contained total phenolic content ~ 0.5 - 1 mg/g dry weight, whereas the pulp extracts have very small amounts of total phenolic content (0.05 - 0.5 mg/g dry weight).
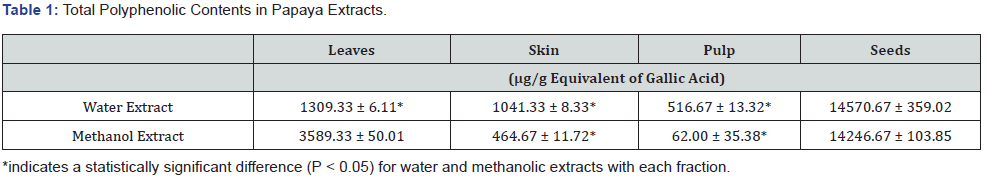
The data for the total flavonoid contents in various papaya fractions are presented in (Table 2) and reported previously [28]. The water extract of seeds contained the highest amounts of total flavonoid content (5 mg/g dry weight), whereas the methanol extracts contained lower amounts (2-2.5 mg/g dry weight, P < 0.05). In leaves, the methanol extract contained more total flavonoid content (~ 5mg/g dry weight) than that of water (~2 mg/g dry weight). The total flavonoid contents in pulp and skin were less than those of seeds and leaves. In the skin, higher total flavonoid contents were present in the methanol extracts (~ 3.2 - 3.5 mg/g dry weight) than in water extracts (1 mg/g dry weight). The pulp contained the lowest total flavonoid contents compared to other fractions. The total flavonoid content in pulp ranged from 1 - 1.5 mg/g dry weight.
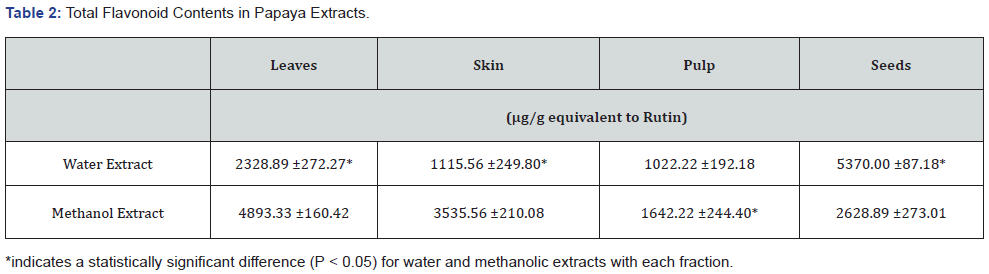
The anti-oxidation capacities of papaya fractions were measured by assaying the inhibition of DPPH oxidation and are shown in (Table 3) and reported previously [28]. The seeds and leaves contained significantly more (P < 0.05) anti-oxidation capacity than that of the skin and pulp. The methanol fractions of seeds and leaves contained more anti-oxidation activity than water extracts. The methanol fractions of seeds and leaves inhibited DPPH oxidation by 75 - 85%, whereas the water extracts of these fractions inhibited DPPH oxidation by 50 - 70%. The skin and pulp inhibited DPPH oxidation from 25 - 35%. There were no significant differences between water or methanol extracts of skin and pulp.
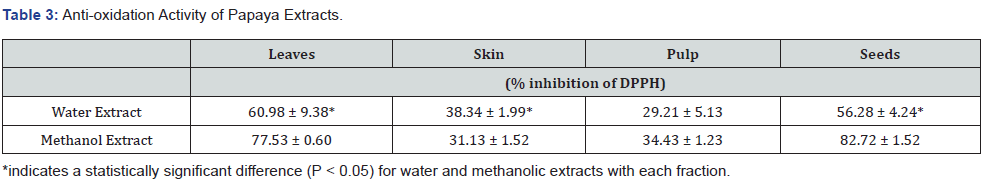
Effect of papaya water extracts on T- cell leukemia
The effects of papaya water extracts from different fractions are presented in Figure 1. The treatment with leaf extract caused a dose-dependent decrease in cell viability. A concentration of the extract as low as 50 μg/ml caused significant cell death by 30% (P < 0.05). Upon increasing the extract concentration more cells died, reaching a plateau of almost 70% cell death (P < 0.05) at 150 μg/ml. Additional increases to 250 μg/ml concentration resulted in a very small change in cell viability (Figure 1).
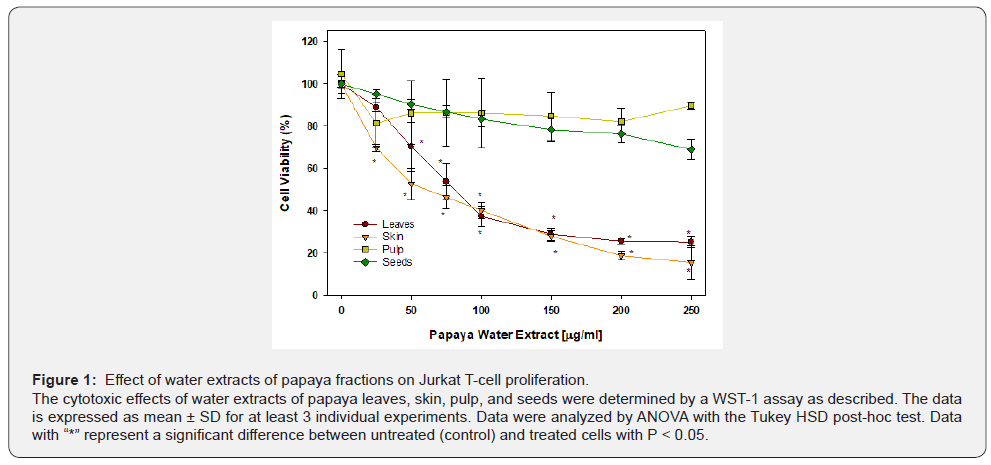
The treatment with water extract from papaya seeds caused a dose-dependent decrease in cell viability. Extract concentrations as low as 150 μg/ml caused a significant 20% increase in cell death (P < 0.05). On further increasing the extract concentration, no further decrease in cell viability occurred, which reached a plateau at 250 μg/ml with 25% cell death (P < 0.05) (Figure 1)
The water extract of papaya skin started to significantly (P < 0.05) inhibit leukemic cell proliferation at 25 μg/ml, causing about 30% cell death. However, the leukemic cells progressively died in a dose-dependent manner reaching to about 80% cell death at 250 μg/ml of the dried papaya skin extract.
The water extract from papaya pulp was unable to cause significant cell death of Jurkat leukemic T-cells.
Effect of papaya methanol extracts on T- cell leukemia
The effects of papaya methanol extracts from different fractions are presented in Figure 2. The methanolic extract of papaya leaves appeared to be less effective than water extract. At 25 μg/ml a significant (P < 0.05) 30% decrease in cell viability resulted; however, the cell viability was decreased by only 60% (P < 0.05) on further increasing the methanolic extract concentration to 250 μg/ml. Similarly, the methanolic extract of papaya seeds appeared to be less effective than water extract. At 200 μg/ml, a significant P < 0.05) decrease in cell viability resulted (10%); however, the further decrease in cell viability was not significant at 250 μg/ml. The methanolic extract of skin also caused significant cell death by 25% at 25 μg/ml (P < 0.05). Further increase of the extract concentration resulted in a dose-dependent response reaching 80% cell death at 250 μg/ml .
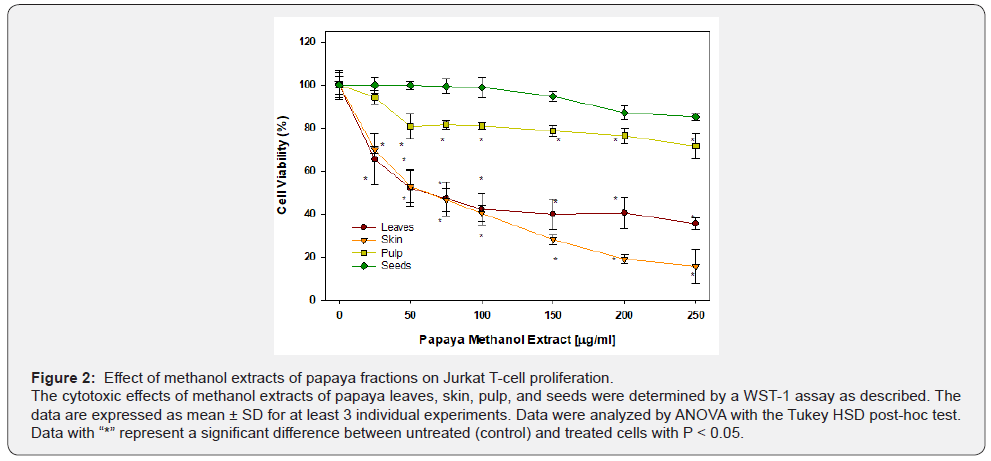
The methanol extract from papaya pulp caused a modest significant effect by decreasing cell viability by 20% at 50 μg/ml ; however, on increasing the concentration of pulp extract to 250 μg/ml , there was no further increase in cell death.
Effect of papaya water extracts on B- cell leukemia
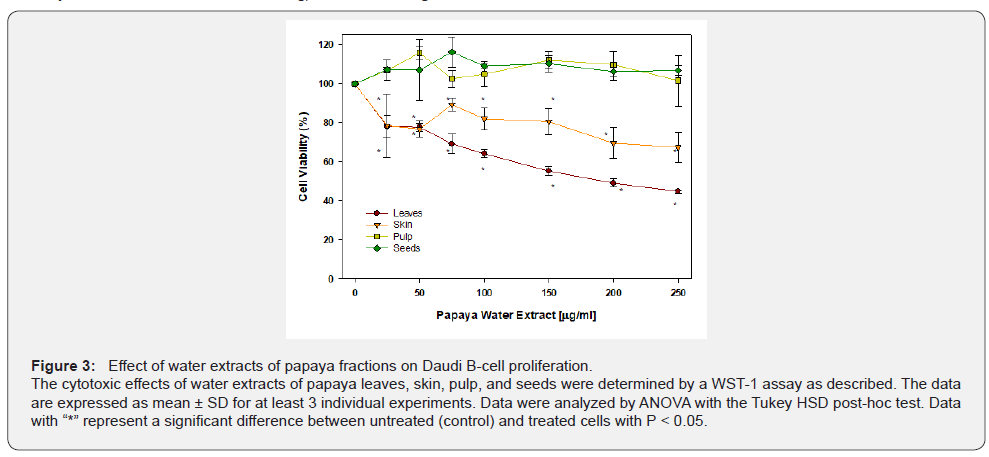
The effects of water extract from different papaya fractions on Daudi leukemia B-cells are presented in Figure 3. Treatment with leaf extract caused a dose-dependent decrease in cell viability. Concentrations as low as 25 μg/ml caused a significant 20% cell death (P < 0.05). On further increasing the extract concentration, significantly more cells (P < 0.05) died in a dosedependent manner, with approximately 50% cells dead at the highest concentration of 250 μg/ml. The water extracts from seeds and pulp have no significant effect on cell viability at any concentration. The extract of papaya skin started to significantly (P < 0.05) inhibit leukemic cell proliferation at 25 μg/ml causing about 25% cell death. However, on further increasing the extract concentration, no change in cell death was observed
Effect of papaya methanol extracts on B- cell leukemia
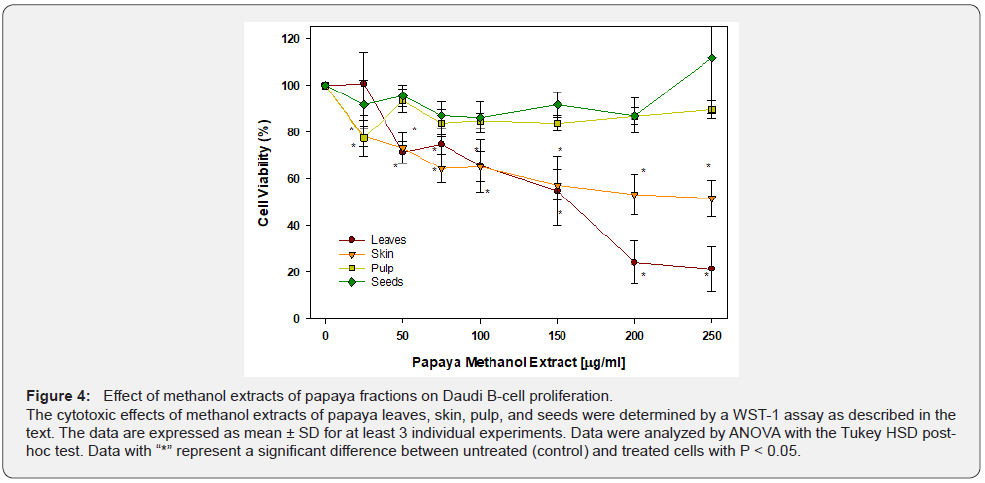
The effects of methanol extracts from different papaya fractions on Daudi leukemia B-cells are presented in Figure 4. The methanolic extract of papaya leaves appeared to affect Daudi B-cells, similar to water extracts. At 50 μg/ml, a significant 30% (P < 0.05) of cells died and the cell death was further increased in a dose-dependent manner to 80% (P < 0.05) at 250 μg/ml. Consistent with the water extract, the methanol extract of papaya pulp has no effect on Daudi B-cells. The methanolic extract of skin had a similar effect on Daudi B-cells as the water extract. It caused significant cell death by 25% at 25 μg/ml (P < 0.05). Increasing the concentration of extract resulted in a dose-dependent effect, reaching 50% cell death at 250 μg/ml.
Discussion
The present study was carried out to investigate the effect of water and methanol extracts of papaya leaves, skin, pulp, and seeds on Jurkat leukemia T- cells and Daudi leukemia B-cells. Water and methanol were used because phenolic compounds have different chemical characteristics and polarities, and because of these properties, compounds in different fractions of papaya may or may not be soluble [30]. We initially tested these fractions for their phenolic contents and their anti-oxidation activity. Most of the anti-oxidation activity in fruits and vegetables is due to their total polyphenolic contents [31]. Our data indicate that papaya seeds have higher amounts of polyphenols than that of other fractions (Table 1). Papaya leaves also contained substantial amounts of total phenolic contents but were 1/3 that of seeds. Other fractions have very little total phenolic content. In seeds, the amount of total phenolic content was similar in both extracts, but the leaf water extract has less total phenolic content than that of methanol. However, the water extract of skin contained almost twice the polyphenols than that of the methanol extract. Organic solvents are generally used to extract bioactive agents from fruits and vegetables. The most suitable solvents for the extraction of polyphenols are aqueous mixtures containing ethanol, methanol, acetone, and ethyl acetate. Ethanol has been known as a good solvent for polyphenol extraction and is safe for human consumption. Methanol has been generally found to be more efficient in the extraction of lower molecular weight polyphenols, whereas aqueous acetone is good for the extraction of higher molecular weight flavonoids [32]. Currently, over 8000 phenolic structures have been identified in fruits and vegetables [33], and flavonoids are one of the major phenolic classes comprising almost 4000 compounds present in different edible plants [33]. We also determined the total flavonoids in the papaya extracts. Our data show that water extracts of seeds and methanol extracts of leaves contained the highest amount of total flavonoid content, which represented about 30- 40% of total phenolic compounds in seeds and leaves (Table 2). The data suggest that most of the polyphenols in seeds are water-soluble flavonoids, whereas a higher proportion of polyphenols in leaves are less water-soluble flavonoids.
Next, the anti-oxidation activities were determined in these fractions. The leaves and seeds possess a higher anti-oxidation activity compared to that of other fractions (Table 3). Although leaves and seeds have different phenolic and flavonoid contents, they have similar profiles for their anti-oxidation activity. It was interesting to note that methanol extracts exhibited higher antioxidation activity than the water extracts. This difference could be due to the different natures of polyphenols.
Next, we analyzed the anticancer properties of papaya fractions on Jurkat cells and Daudi cells. The Jurkat cells are an immortalized line of human T lymphocyte cells that are often used to study acute T cell leukemia. The Jurkat cell line was established in the late 1970s from the peripheral blood of a 14-year-old boy with T cell leukemia [34] whereas the Daudi cell line is a wellcharacterized B lymphoblast cell line which was derived from a 16-year-old black male with Burkitt’s lymphoma [35].
A large number of phytochemicals from vegetables, fruits, and grains have been reported to possess anticancer properties and are promoted for cancer prevention and treatment [36-40]. Recently, multiple scientific reports have emerged about the medicinal benefits of papaya [16,41-43].
Our data indicated that none of the extracts from seeds or pulp possess significant inhibitory activity for cell proliferation in Jurkat leukemic T-cells or Daudi leukemic B-cells. The extracts from leaves and skin, however, showed significant inhibition of both Jurkat leukemic T-cells and Daudi B-cell growth. This effect was stronger on Jurkat T-cells and modest on Daudi B-cells. During the present study, we found that papaya seeds contained the highest amounts of phenolic compounds; however, none of the extracts from papaya seeds have any inhibitory activity for Jurkat leukemic cells. Our data clearly indicated that the inhibitory activity of these extracts was neither dependent on their phenolic contents, nor was it dependent on their anti-oxidation activity. It appears from this observation that the inhibitory compounds in papaya leaves and skin extracts acted on cellular mechanisms linked to cell growth. For example, phytochemicals have been shown to interfere with the stabilization of the microtubule structure, thereby inhibiting mitosis and cancer cell propagation [44]. Vincristine and vinblastine, isolated from the leaves of Madagascar periwinkle, were the first phytochemicals to be used clinically in combination with other anticancer agents in lymphomas, leukemias, and breast, lung, and ovarian cancers [45,46].
Epigallocatechin-3-gallate (EGCG), a polyphenol from the leaves of Camellia sinensis (processed to green tea), possesses anti-oxidation activity but it limited cancer cell proliferation by reducing DNA methylation through the inhibition of DNA methyltransferase together with reactivation of the silenced tumor suppressor genes [47]. Curcumin (diferuloylmethane), a polyphenol isolated from the rhizome of the turmeric plant, has also shown therapeutic efficacy on numerous disorders, including cancer [48]. Curcumin is reported to inhibit NF-κB signaling that triggers the intracellular inflammatory response as well as cellcycle- associated genes. By arresting the cell cycle and inducing apoptosis through the relaying pathways, curcumin interferes with angiogenesis and reduces tumor invasion.
An additional group of anticancer phytochemicals functions as inhibitors of topoisomerase I or II, which are the nuclear enzymes that control DNA supercoiling, eliminate tangles in the chromatin structure, and allow DNA to be replicated and transcribed. Thus, topoisomerase inhibitors can act as anticancer agents by inducing a delay of the cell cycle, followed by cell death [36]. β-Lapachone from the bark of the Lapacho (Handroanthus impetiginosus) plant, camptothecin from the bark/stem of Camptotheca acuminata (the Chinese happy tree), and podophyllotoxin from the root of the Mayapple (Podophyllum peltatum) plant are examples of phytochemicals inhibiting topoisomerases in cancer cells [49- 51]. We have not yet characterized the chemical composition of aqueous and methanolic extracts of leaves and skin or explored the molecular mechanisms that may be involved in inhibition of Jurkat leukemic cell growth. Clearly, additional experiments are required for isolation and identification of active compounds in papaya leaves and skin to determine the cellular mechanisms of their anticancer activity.
Our data suggest that consumption of papaya leaves or skin may be beneficial for preventing and/or treating leukemia, particularly T-cell leukemia. Historically, papaya leaves have been used in different cultures around the world, which suggests that the consumption of leaves extract is not toxic [52]. It is possible that anticancer compounds from leaves can be extracted by boiling them in water. However, the skin is not typically consumed because of its bitter taste. The skin can be dried and made into a powder that can be consumed in the form of pills. Alternatively, the active compounds from leaves and skin can be extracted and isolated in pure form, which can be the basis for new therapeutic leukemia drugs. In conclusion, this study suggests that leaves and skin of papaya contained anticancer activity for leukemic cells.
Acknowledgments
Financial support to Zainab Allqmani was provided by the Saudi Arabia Culture Mission, Kingdom of Saudi Arabia. The research was supported by a Capacity Building Grant from NIFA (USDA) to Dr. Rafat Siddiqui (Grant # 12405879). Institutional support from the Virginia State University Agricultural Research Station by Dr. Wondi Mersie and Dr. M. Ray McKinnie is gratefully acknowledged.
References
- World Health Organization (2015) Cardiovascular diseases (CVDs).
- Siegel RL, Miller KD, Jemal A (2020) Cancer statistics, 2020. CA: A Cancer Journal for Clinicians 70: 7-30.
- Estcourt LJ, Bain BJ (2013) WHO Classification of leukemia. Brenner's Encyclopedia of Genetics 2: 329- 336.
- Hallek M, Cheson BD, Catovsky D, Caligaris-Cappio F, Guillaume Dighiero, et al (2008) Guidelines for the diagnosis and treatment of chronic lymphocytic leukemia: a report from the International Workshop on Chronic Lymphocytic Leukemia updating the National Cancer Institute-Working Group 1996 guidelines. Blood 111: 5446-5456.
- Howlader N, Noone AM, Krapcho M, Miller D, et al. (2019) SEER Cancer Statistics Review, 1975-2016, National Cancer Institute. Bethesda, MD, based on November 2018 SEER data submission, posted to the SEER web site, April 2019.
- ACS (2018) Cancer Facts & Figures (2018) Atlanta, GA: American Cancer Society, Inc. pp. 71.
- Marjanvoic G (2017) The use of inexpensive broad-spectrum lower toxicity therapeutics in chronic lymphocytic leukemia. Journal of the Balkan Union of Oncology 22(2): 288 - 294.
- Park SE, Yoo HS, Jin CY, Hong SH, Yeon W, et al (2009) Induction of apoptosis and inhibition of telomerase activity in human lung carcinoma cells by the water extract of Cordyceps militaris. Food Chem Toxicol 47(7): 1667-1675.
- Foo JB, Yazan LS, Chan KW, Tahir PM, M Ismail, et al (2011) Kenaf seed oil from supercritical carbon dioxide fluid extraction induced G1 phase cell cycle arrest and apoptosis in leukemia cells. African Journal of Biotechnology 10: 5389-5397.
- Hsieh YJ, Chang CJ, Wan CF, Chen CP, Yi Han C, et al (2013) Euphorbia formosana root extract induces apoptosis by caspase-dependent cell death via Fas and mitochondrial pathway in THP-1 human leukemic cells. Molecules 18(2): 1949-1962.
- Khalafalla MM, Abdellatef E, Daffalla HM, Nassrallah A, Aboul Enein KM, et al. (2009) Antileukemia activity from root cultures of Vernonia amygdalina. Journal of Medicinal Plants Research 3: 556-562.
- Choo CY, Chan KL, Sam TW, Hitotsuyanagi Y, et al (2001) The cytotoxicity and chemical constituents of the hexane fraction of Typhonium flagelliforme (Araceace). J Ethnopharmacol 77: 129-131.
- Mohan S, Abdul AB, Abdelwahab SI, Al-Zubairi AS, Mohamed A, et al. (2010) Typhonium flagelliforme inhibits the proliferation of murine leukemia WEHI-3 cells in vitro and induces apoptosis in vivo. Leuk Res 34(11): 1483-1492.
- Gao N, Budhraja A, Cheng S, Yao H, et al (2009) Induction of Apoptosis in Human Leukemia Cells by Grape Seed Extract Occurs via Activation of c-Jun NH 2 -Terminal Kinase. Clinical Cancer Research 15: 140-149.
- Lohsoonthorn P, Danvivat D (1995) Colorectal cancer risk factors: a case-control study in Bangkok. Asia Pac J Public Health 8: 118 - 122.
- Shahar S, Shafurah S, Shaari NSAH, Rajikan R, et al. (2011) Roles of diet, lifetime physical activity and oxidative DNA damage in the occurrence of prostate cancer among men in Klang Valley, Malaysia. Asian Pacific Journal of Cancer Prevention 12(3): 605 - 611.
- Pandey S, Walpole C, Cabot PJ, Shaw PN, et al. (2017) Selective anti-proliferative activities of Carica papaya leaf juice extracts against prostate cancer. Biomed Pharmacother 89: 515-523.
- Siegel EM, Salemi JL, Villa LL, Ferenczy A, et al (2010) Dietary consumption of antioxidant nutrients and risk of incident cervical intraepithelial neoplasia. Gynecol Oncol 118(3): 289-294.
- Zhang CX, Ho S, Chen YM, Fu JH, Cheng SZ et al (2009) Greater vegetable and fruit intake is associated with a lower risk of breast cancer among Chinese women. International Journal of Cancer 125: 181-188.
- García-Solís P, Yahia EM, Morales-Tlalpan V, Díaz-Muñoz M (2009) Screening of antiproliferative effect of aqueous extracts of plant foods consumed in México on the breast cancer cell line MCF-7. Int J Food Sci Nutr 60 Suppl 6: 32-46.
- Gayosso-García Sancho LE, Yahia EM, García-Solís P, González-Aguilar GA (2014) Inhibition of Proliferation of Breast Cancer Cells MCF7 and MDA-MB-231 by Lipophilic Extracts of Papaya (Carica papaya L. var. Maradol) Fruit. Food and Nutrition Sciences 5(21): 2097-2103.
- Nguyen TT, Parat MO, Hodson MP, Pan J, et al (2015) Chemical Characterization and in Vitro Cytotoxicity on Squamous Cell Carcinoma Cells of Carica Papaya Leaf Extracts. Toxins (Basel) 8.
- Otsuki N, Dang NH, Kumagai E, Kondo A, Satoshi I, et al. (2010) Aqueous extract of Carica papaya leaves exhibits anti-tumor activity and immunomodulatory effects. J Ethnopharmacol 127(3): 760-777.
- Morimoto C, Dang N (2008) Compositions for Cancer Prevention, Treatment, or Amelioration Comprising Papaya Extract. In Y S Therapeutics Co Ltd TJ.
- Lee Y, Kim YJ, Choi YJ, Lee JW, Sunyeong L, et al (2012) Enhancement of cisplatin cytotoxicity by benzyl isothiocyanate in HL-60 cells. Food Chem Toxicol 50(7): 2397-2406.
- Rahmat LT, Damon LE (2018) The Use of Natural Health Products Especially Papaya Leaf Extract and Dandelion Root Extract in Previously Untreated Chronic Myelomonocytic Leukemia. Case Rep Hematol 2018: 7267920.
- Bravo L (1998) Polyphenols: chemistry, dietary sources, metabolism, and nutritional significance. Nutr Rev 56(11): 317-333.
- Zhou K, Wang H, Mei W, Li X, et al (2011) Antioxidant activity of papaya seed extracts. Molecules 16(8): 6179-6192.
- Hadadi SA, Li H, Rafie R, Kaseloo P, Sarah M, et al. (2018) Anti-oxidation properties of leaves, skin, pulp, and seeds extracts from green papaya and their anti-cancer activities in breast cancer cells. J Cancer Metastasis Treat 4: 25.
- Al-Shaya HM, Li H, Beg OU, Hamama AA, Sarah M, et al. (2020) Phytochemical profile and antioxidation activity of annona fruit and its effect on lymphoma cell proliferation. Food Sci Nutr 8(1): 58-68.
- Turkmen N, Velioglu YS, Sari F, Polat G (2007) Effect of extraction conditions on measured total polyphenol contents and antioxidant and antibacterial activities of black tea. Molecules 12(3): 484-496.
- Miguel-Chávez RS (2017) Phenolic Antioxidant Capacity: A Review of the State of the Art London, UK: IntechOpen Limited.
- Dai J, Mumper RJ (2010) Plant phenolics: extraction, analysis and their antioxidant and anticancer properties. Molecules 15(10): 7313-7152.
- Lima GPP, Vianello F, Corrêa CR, da Silva Campos RA, et al (2014) Polyphenols in Fruits and Vegetables and Its Effect on Human Health. Food and Nutrition Sciences 5(11): 1065-1082.
- Schneider U, Schwenk HU, Bornkamm G (1977) Characterization of EBV-genome negative "null" and "T" cell lines derived from children with acute lymphoblastic leukemia and leukemic transformed non-Hodgkin lymphoma. Int J Cancer 19(5): 621-626.
- Klein E, Klein G, Nadkarni JS, Nadkarni JJ, et al (1968) Surface IgM-kappa specificity on a Burkitt lymphoma cell in vivo and in derived culture lines. Cancer Research 28(7): 1300-310.
- Nobili S, Lippi D, Witort E, Donnini M, Enrico M, et al. (2009) Natural compounds for cancer treatment and prevention. Pharmacol Res 59(6): 365-378.
- Graham JG, Quinn ML, Fabricant DS, Farnsworth NR (2000) Plants used against cancer - an extension of the work of Jonathan Hartwell. J Ethnopharmacol 73(3): 347-377.
- Amin A, Gali-Muhtasib H, Ocker M, Schneider-Stock R (2009) Overview of major classes of plant-derived anticancer drugs. Int J Biomed Sci 5(1): 1-11.
- Diederich M, Cerella C (2016) Non-canonical programmed cell death mechanisms triggered by natural compounds. Semin Cancer Biol 40-41: 4-34.
- Shanmugam MK, Lee JH, Chai EZ, Kanchi MM, et al (2016) Cancer prevention and therapy through the modulation of transcription factors by bioactive natural compounds. Semin Cancer Biol 40-41: 35-47.
- Saha S, Giri TK (2019) Breaking the Barrier of Cancer through Papaya Extract and their Formulation. Anticancer Agents Med Chem 19(13): 1577-1587.
- Nguyen TT, Shaw PN, Parat MO, Hewavitharana AK (2013) Anticancer activity of Carica papaya: a review. Mol Nutr Food Res 57(1): 153-164.
- Aruoma OI, Somanah J, Bourdon E, Rondeau P, et al (2014) Diabetes as a risk factor to cancer: functional role of fermented papaya preparation as phytonutraceutical adjunct in the treatment of diabetes and cancer. Mutat Res 768: 60-68.
- Oh J, Hlatky L, Jeong YS, Kim D (2016) Therapeutic Effectiveness of Anticancer Phytochemicals on Cancer Stem Cells. Toxins 8(7): 199.
- Wani MC, Taylor HL, Wall ME, Coggon P, et al (1971) Plant antitumor agents. VI. The isolation and structure of taxol, a novel antileukemic and antitumor agent from Taxus brevifolia. J Am Chem Soc 93(9): 2325-2327.
- Schiff PB, Fant J, Horwitz SB (1979) Promotion of microtubule assembly in vitro by taxol. Nature 277: 665-667.
- Nandakumar V, Vaid M, Katiyar SK (2011) (-)-Epigallocatechin-3-gallate reactivates silenced tumor suppressor genes, Cip1/p21 and p16INK4a, by reducing DNA methylation and increasing histones acetylation in human skin cancer cells. Carcinogenesis 32(4): 537-544.
- Anand P, Sundaram C, Jhurani S, Kunnumakkara AB, et al. (2008) Curcumin and cancer: an "old-age" disease with an "age-old" solution. Cancer Lett 267: 133-164.
- Li YZ, Li CJ, Pinto AV, Pardee AB (1999) Release of mitochondrial cytochrome C in both apoptosis and necrosis induced by beta-lapachone in human carcinoma cells. Mol Med 5: 232-239.
- Pommier Y (2006) Topoisomerase I inhibitors: camptothecins and beyond. Nat Rev Cancer 6: 789-802.
- Hartmann JT, Lipp HP (2006) Camptothecin and podophyllotoxin derivatives: inhibitors of topoisomerase I and II - mechanisms of action, pharmacokinetics and toxicity profile. Drug Saf 29(3): 209-230.
- Hettige S (2008) Salutary effects of Carica papaya leaf extract in dengue fever patients - a pilot study. Srilankan Family Physician 29: 17-19.






























