Comparative study on some in vitro biological activities of freeze-dried leaves extracts of six advanced accessions of Ipomoea batatas (L.) Lam
Shahidul I*, Z Adam and Ishrar Islam1
1Department of Agriculture, University of Arkansas at Pine Bluff, USA
2Hendrix college, USA
Submission: April 09, 2020; Published: May 01, 2020
*Corresponding author: Shahidul Islam, Department of Agriculture, University of Arkansas at Pine Bluff, 1200 North University Drive, Mail Slot 4913, 153 Woodard Hall, Pine Bluff, AR 71601, USA
How to cite this article: Shahidul I, Z Adam, Ishrar I. Comparative study on some in vitro biological activities of freeze-dried leaves extracts of six advanced accessions of Ipomoea batatas (L.) Lam. Nutri Food Sci Int J. 2020. 10(1): 555776. DOI: 10.19080/NFSIJ.2020.10.555776.
Abstract
Sweet potatoes (Ipomoea batatas L.) are an excellent source of bio-active phytochemicals. In recent years, polyphenols and other naturally occurring compounds have become essential research targets for non-insulin-dependent diabetes. Precisely, substances and plant extracts that occur readily have been checked for α-glucosidase (AGH) enzyme inhibition. This study investigated the anti-diabetic, antimutagenicity, and antioxidant activity from sweet potato leaves in vitro. The anti-diabetic activities were tested using the enzyme –α-glucosidase obtained from the rat intestine using the p-nitrophenyl-α-D-glucopyranoside (PNP-G) substrate for inhibitory activities. The α-glucosidase inhibition assay evaluated the anti-diabetic activity, and the extract showed a considerable α-glucosidase inhibitory activity. Among the genotypes, AN-6 exhibited the highest a-glucoside inhibitory activity, followed by AN-4, and AN-2. The leaf extract showed the inhibitory activity ranging from 22.33% to 74.98 on a-glucosidase from 10 to 1000 mg/ml, which was increased steadily with increasing sample concentrations. The antimutagenicity in the leaves explored using the Salmonella typhimurium TA 98. The Ipomoea batatas genotypes effectively decreased the reverse mutation induced by Trp-P-1, and the mutagenic activities were dose-dependent. Furthermore, the extract also capable of reducing the reverse mutation persuaded by Trp-P-2, IQ, and DEGB extract of grilled beef. The AN-6 showed higher antimutagenicity followed by AN-5 at 100 μL concentrations. The fallouts demonstrate that antioxidant capacity (4.42 to 10.98 μmole Trolox g-1 DW) and total phenolic contents (7.68 to 16.96 μmole TA. g-1 DW) broadly fluctuate among the genotypes. Our data demonstrate that all the genotypes have the physiological functions studied, and AN-6 and AN-4 exhibited the highest activities. The sweet potato leaves extract showed a more potent inhibitory activity for all the physiological functions studied, which might have values in anticipation of certain human health conditions.
Keywords: Antioxidant; Polyphenol; Anti-diabetic activity; Antimutagenicity; Sweet potato tops
Introduction
Sweet potato (Ipomoea batatas L. Lam) is the sixth most important food crop in the world, and new uses for this crop have been identified [1]. It is one of the diversified crops supplying vitamins and minerals such as vitamin A, B, C, beta-carotene, iron, calcium, zinc, protein and has high energy [2]. Fresh leaves contain vitamin A on an average of 1600 IU 100g-1 [3]. Leaves are very nutritious compared to leaves of cassava, amaranth, mushrooms, taro and pumpkin [4]. It is also one of the plants selected by the US National Aeronautics and Space Administration to be grown in a controlled ecological life support system as a primary food source [5]. Recent studies show that it contains bio-active compounds as polyphenols, anthocyanins, flavonoids, dietary fiber, etc., which are essential for human health. Sweet poato storage roots are a source of carbohydrates, while its leaves and green stems contain nutritional compounds in higher than many commercial vegetables [6-8]. Sweet potato leaves are cooked as vegetables in different locations of the world. The eating of Ipomoea batatas L. leaves as a vegetable in many parts of the world indicates that they are acceptable as edible like other traditional leafy vegetables. They are rich in phytonutrients and are further tolerant of diseases and pests than many other green plants [9-12]. Phytonutrients act as bioactive composites and a diverse group of secondary metabolites commonly present in higher plants [7,13-19]. They play important roles and contribute to the structure of the plants and complicated by a significant number of metabolic pathways [20,21]. Thus, the phenolic plant complexes, because of their diversity and widespread distribution, are the most exceptional talented group of natural antioxidants and add to the organoleptic and nutritional qualities of fruit and vegetables.
Diabetes mellitus is a common disease with many complications, such as atherosclerosis, cardiac dysfunction, retinopathy, neuropathy, and nephropathy [22]. α-glucosidase (EC 3.2.1.20) catalyzes the final step in the digestive process of carbohydrates. Its inhibitors can retard the uptake of dietary carbohydrates and suppress postprandial hyperglycemia and could be useful for treating diabetic and obese patients [23]. α-Glucosidase inhibitors such as acarbose, miglitol, and voglibose are known to reduce postprandial hyperglycemia primarily by interfering with the carbohydrate digestive enzymes and by delaying glucose absorption. Free radicals can lead to a variety of physiological and biochemical lesions [24] and induce degenerative diseases such as coronary disease, diabetes, stroke, and cancer [25]. The new expansion of screening approaches for environmental carcinogens by determining their mutagenicity has allowed detecting numerous types of mutagens and carcinogens in foods [2,24-27]. On the other hand, it is now recognized that several types of inhibitors act against mutagens and carcinogens in food. They show a substantial part in plummeting the dangers of mutagenesis and carcinogenesis [28]. Several authors described that the nutritive constituents of sweet potato tops are comparable to those of commercial leafy vegetables [2,6,14-18,26,29-30]. However, the physiological function of sweet potato leaves has not yet been deliberate synthetically. In the current article, the effects of the extracts of the selected sweet potato accessions with the diverse polyphenolic levels on the antidiabetic activity, mutagenicity, and antioxidant capacity are explored.
Materials and Methods
The leaves from six Ipomoea batatas L. (sweet potato) advanced accessions, namely AN-1, AN-2, AN-3., AN-3, AN-4, AN- 5, and AN-6, were used for this study. Sweet potato roots were planted 2 inches deep and about 2 inches apart (density of 5 cm x 5 cm) in a greenhouse and field conditions in late February (greenhouse) to March/April in the University of Arkansas at Pine Bluff’s Agricultural Research Farm, Pine Bluff, AR. After two months, tips were harvested every 10-15 days. Chemical fertilizer (N: P: K = 8: 8: 8) was used at a rate of 500 lbs/acre, and compost was used at a rate of 8000lbs/acre in volume. After each harvest, 150 lbs/acre of ammonium sulfate was applied as additional fertilizer. After harvest, the leaves were washed softly, moved into pre-labeled separate vinyl bags, and directly frozen at -85 ℃. The next day all the frozen samples were freeze-dried for 48 h in a freeze dryer. The freeze-dried samples were ground in a blender and used for laboratory analysis. The extract was prepared from the lyophilized flour (1g) using 20 mL of ice-cold water for 1h. The suspension was centrifuged at 18000 x g for 20 min, and the resultant precipitate was re-extracted under the same conditions. The collected supernatant was lyophilized and used for analysis.
α-Glucosidase inhibitory assay
The α-Glucosidase inhibitory assay was performed according to a slightly modified method described by Islam [31]. One hundred microliters of 3 mM pNPG in 0.2 M sodium phosphate buffer (pH 6.8) was added as a substrate to the mixture of 50 μl of α-glucosidase (0.15 unit/ml) and 50 μl of sample to start the reaction. The reaction was conducted at 37 ℃ for 15 min and stopped by the addition of 750 μl of 0.1 M Na2CO3. The α-Glucosidase activity assessed by measuring the release of p-nitrophenol from pNPG at 405 nm. Acarbose used as a positive control. All tests were performed in independent triplicate (n=3), and data were expressed as mean ± SD.
Extraction and Measurement of Total Phenolic
The total phenolic contents of the extracts were measured according to a slightly modified method described by Islam et al. [18]. The lyophilized sweet potato leaf extract was forcefully mixed with ten times its equivalent volume of 80% ethanol. The mixture was boiled for 5 min and centrifuged at 5000g for 10 min, and the supernatant was composed. The residue was mixed with an additional 80% ethanol and boiled for 10 min to re-extract the phenolic and centrifuged under similar conditions. The extracts were pooled and made up to 10 mL and used for to quantity of total phenolic. The alcohol extract was diluted to achieve an absorbance reading at the range of the standard tannic acid (TE). The results were stated as μmol TE g-1 DW (dry weight).
Antioxidant capacity in the DPPH assay
The radical-scavenging activity of the extracts was measured according to a slightly modified method described by Islam et al. [17]. A stock solution of DPPH (6 mM) was prepared by dissolving 0.0263g in 10 ml of ethanol (or methanol). The stock solution is diluted to develop a 60 μM working solution. Again, a ten mM stock solution of Trolox was ready for every sample tested. Dilutions were made for each sample tested. Dilution strength was dependent upon each extract’s relative antioxidant capacity. For each dilution, 20 μL were added to 2.5ml of DPPH solution and incubated in a dry bath at 37 ˚C for 30 min. Absorbances were measured at 520 nm on an ASYS UVM 340 plate reader. TEAC values were measured by comparing the slope of sample plots to the slope of Trolox. Antioxidant activity was reported as μmoles Trolox equivalent per gram dry weight sample (μmol TE/g DW).
Assay of antimutagenicity
The antimutagenicity assay was performed as described in earlier papers [27]. The antimutagenic activity was assessed for Salmonella typhimurium TA 98 using a mutagen, Trp-P-1. These mutagens need metabolic activation to induce mutation in TA 98. The s-9 mix containing 50 μmol of sodium phosphate buffer (pH 7.4), 4 μmol of MgCl2, 16.5 μmol of KCl, 2.5 μmol of glucose-6- phosphate, two μmole of NADH, 2 μmol of NADPH, and 50 μL of the S-9 fraction in a total volume of 0.5 mL. For the inhibition test, 0.1 mL of mutagen, 0.1 mL DMSO-dissolved polyphenolics solution, and 0.5 mL of S-9 mix or phosphate buffer were concurrently incubated with 0.1 mL of a bacterial suspension at 37 ℃ for 20 min and then dispensed into minimal-glucose-agar plates with 2 mL of soft agar. The colony number of each dish was accounted after 48 h cultivation at 37 ℃.
Statistical analysis
A randomized complete block design with three replications was adopted. Data for the different parameters were analyzed by analysis of variance (ANOVA) procedure, and the level of significance was calculated from the F value of ANOVA.
Results and Discussion
α-Glucosidase inhibitory effect
The α-glucosidase inhibition assay evaluated the antidiabetic activity, and the extract showed a considerable α-glucosidase inhibitory activity (Table 1). The leaf extract demonstrated a moderate to high inhibitory activity on α-glucosidase, among the genotypes, AN-6 (80% inhibition at 1000 μg/ml) exhibited the highest a-glucoside inhibitory activity, followed by AN-4 (75% inhibition at 1000 μg/ml) and AN-2 (80% inhibition at 1000 μg/ml). The results also suggested that the α-Glucosidase inhibitory effect in the sweet potato tops is dose-dependent (Table 1), and increasing the doses resulted in a higher rate of inhibition percentage. The leaf extract showed the inhibitory activity ranging from 22.33% to 74.98% on a-glucosidase from 10 to 1000 mg/ml, which was increased steadily with increasing sample concentrations. On the other hand, the control treatment (Acarbose) showed 98.01% inhibitory activity at the strength of 0.1 μg/ml.
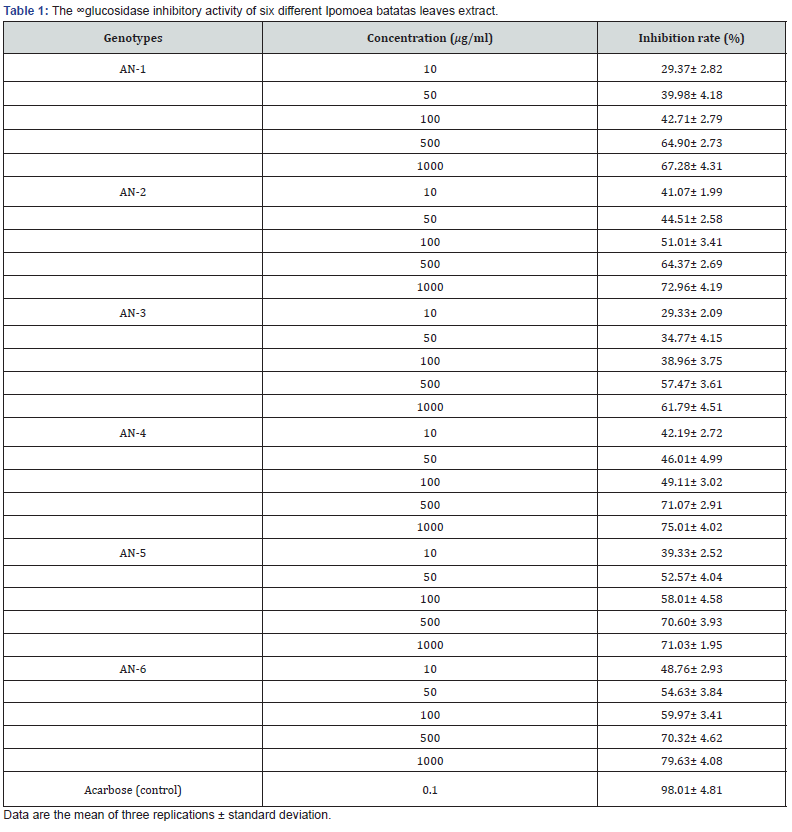
One therapeutic approach for treating diabetes is to increase postprandial hyperglycemia. This is done by retarding the absorption of glucose through the inhibition of the carbohydrate hydrolyzing enzyme α-glucosidase in the digestive tract. Inhibitors of these enzymes delay carbohydrate digestion, causing a reduction in the rate of glucose absorption and consequently blunting the postprandial plasma glucose rise (Rhabasa and Chiasson, 2004) [32]. The results suggest that the Ipomoea batatas leaf extracts have the potentiality for treating diabetes by inhibiting α-glucosidase activity.
Total polyphenol content and antioxidant capacity
The antioxidant capacity (μmole Trolox/mg dry leaf powder) and total polyphenol (μmol TE g-1 DW) in the leaves of three genotypes are presented in Table 2. The genotypes fluctuated extensively in their total polyphenolic contents. The highest total phenolic found was 16.98 μmol TE g-1 DW, and the lowest was 7.68 μmol TE g-1 DW. The accessions AN-6 had higher content (16.98 μmol Trolox.g-1 DW) flowed by AN-4 (16.52 μmol Trolox.g-1 DW), and AN-5 (15.63 μmol Trolox.g-1 DW). The results showed that sweetpotato leaves had higher or similar content of total polyphenolics than other vegetables [12,17-18,21,29]. The data also suggested that there was a positive correlation between polyphenol contents and antioxidant activity. Because the accessions higher in total phenol contents also exhibited higher antioxidant activity. The above results also agree with the observations of Islam [6], where he added that sweet potato leaves, could serve as a new leafy vegetable. Acceptable tops should be tender, glabrous, and purplish. Those eating tops prefer the top 10 cm of tips, including both stem and leaves. Heads with the most significant number of leaves with petioles less than 4/10 of 1cm long are considered desirable because they are tender and suitable for vegetables. Petiole length varies widely with genotype and may range from approximately 10 to 40 cm [33]. (Table 2)
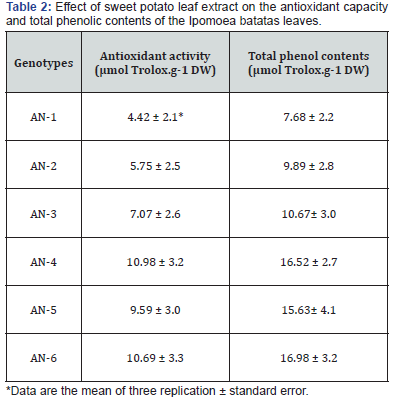
The antioxidant capacity of the genotypes ranges from 4.42 to 10.98 μmol Trolox g-1 DW. The accessions AN-4 (10.98 μmol Trolox.g-1 DW) had the highest contents of phenolic, followed by AN-6 (10.69 μmol Trolox.g-1 DW). The accessions AN-1 showed the lowest (4.42 μmol Trolox.g-1 DW) antioxidant activity followed by AN-2 (5.75 μmol Trolox.g-1 DW). The phenolic is pervasive bioactive compounds found in plant foods and beverages. The polyphenolic compounds show numerous biological functions, sweet potato leaves might also be expected to have physiologically active possessions because they comprise higher contents phytonutrients. The antioxidative substances contained in plant parts have attracted much consideration all over the world. Several researchers [17,27,34] have reported the radical scavenging and antioxidant activities of sweet potato leaves. The polyphenolics contents and antioxidant activity in sweet potato leaves, other different plants and foods showed a high correlation [6,16,17,26]. Usually, the antioxidant capacity of various plants is influenced by the genetic factor. Therefore, the extent of the antioxidant capacity may be a critical tool for use in plant breeding programs intended to improve antioxidant components available for human consumption. This result will be valuable for some chemical breeding programs to develop needed organoleptic and nutritional quality characteristics of crop plants.
Effects of water extract of leaves on the mutagenicity
The antimutagenic impact of the water extracts from sweet potato leaves of three genotypes were determined by antimutagenicity assays using Trp-P-1 at a dose of 0.075 μg/plate, and using three different doses of the sweet potato leaves extracts such as 100, 50 and 10 μg/plate (Table 3).
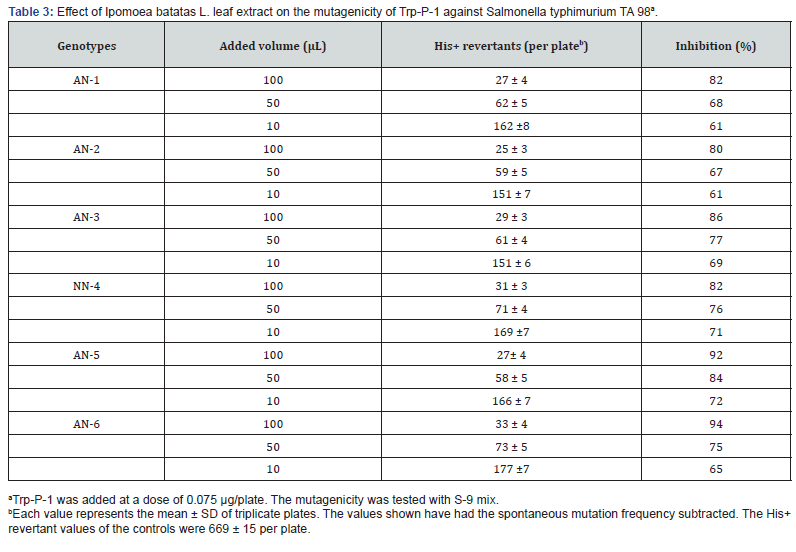
The results found that inhibitory activity was higher at higher doses in all genotypes studies. The inhibitory activity (%) ranged from 69 to 94 at 100μg/plate, 68 to 84 at 50μg/plate, and 61 to 73 at ten μg/plate doses. The highest activity found in the accessions AN-6 (90% inhibition at 100 μL) followed by AN-5 (92% inhibition at 100 μL) while AN-2 (80% inhibition at 100 μL) had the lowest. Therefore, the results propose a wide disparity of antimutagenicity among the genotypes, and the extracts showed dose-dependent inhibitory activities. Similar trends were also found by several researchers [6,17,35,36]. The antimutagenic effect of the extract at low doses is relatively minor compared with the one from higher doses. (Table 4)
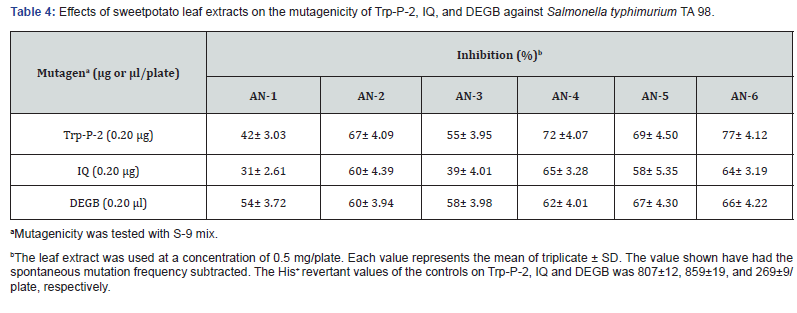
We also evaluated the antimutagenic activity of the extract using several mutagens, such as Trp-P-2, IQ, B[a] P, and DEGB (Table 4). The DEGB was utilized at a dose of 100 μL/plate without dilution. The s-9 mix was added for the assay using Trp-P-1, Trp-P-2, IQ, B[a] P, and DEGB to cause mutations in TA 98. The extract used in doses of 50, 10, and 5 μL/plate. The extract inhibited Trp-P-2 induced mutation by 14%, IQ by 88%, b[a] P by 27%, and Trp-P-1 by 71%, respectively, at the concentration of 10 μL/plate. Thus, the sweet potato leaf extract effectively decreased the reverse mutations induced by all purified mutagens tested. This study exhibited that Ipomoea batatas L. tops could be an outstanding source of natural active compounds with numerous biological functions with the aptitude to defend in contradiction of certain sorts of human illnesses. We tested several physiological functions of the leaves extracts in six advanced accessions. All the accessions tested accumulated higher physiological functions. The high biological activity in the leaves extracts, which might have values in the prevention of specific human conditions. Therefore, sweet potato leaves can be considered as a potential source of functional food and a pharmaceutical agent. Furthermore, the leaves with high phytonutrient content may be used as herb, tea, food ingredient, and a nutritional supplement that could be demanded to have a positive impact on human health.
Acknowledgments
The authors would like to express great appreciation to Dr. Lee for her valuable and constructive suggestions during this study. The authors also wish to extend their thanks to the technicians, graduate students, and greenhouse manager Mr. H. Sorker of the laboratory for their help in offering us the resources in running the study. The authors are also owed to the National Institute of Food and Agriculture, United States Department of Agriculture for funding (USDA-NIFA Award number 2018-38821-27740) to complete this research.
References
- Food and Agriculture Organization (1997) FAO production yearbook. FAO (United Nations), Rome, Italy, 51(2).
- Islam S (2014) Medicinal and Nutritional Qualities of Sweet potato Tips and Leave. Published by Cooperative Extension Service. FSA6135, pp. 1-4.
- Bhuiyan MKR, Alam MS, Islam ATMT, Hossain M, Begum SN (2008) Production technology of newly released sweet potato varieties (In Bangla), Tuber Crops Research Centre, Bangladesh Agricultural Research Institute (BARI), Gazipur-1701. 8p. DAE. 2014. Dept. of Agricultural Extension. Annual Report, 2013-14.
- Oduro I, Elis WO, Owusu D (2008) Nutritional evaluation of two leafy vegetables: Moringa olifera and Ipomoea batatas SRE 3(2): 57-60.
- Hoff JE, Howe JM, Mitchell CA (1982) Nutritional and cultural aspects of plant species selection for a controlled ecological life support system. NASA Contractor Rep. 166324, Moffett Field, CA.
- Islam S (2006) Sweet potato (Ipomoea batatas ) Leaf: Its Potential Effect on Human Health and Nutrition. J Food Sci 71: 13-21.
- Islam S (2016) Some bioactive constituents, antioxidant, and antimutagenic activities in the Leaves of Ipomoea batatas Genotypes. American Journal of Food Science & Technology 4(3): 70-80.
- Islam S (2019) Antimutagenicity of the water extracts, radical scavenging activity, and phenolic acids in the tops of diverse Ipomoea batatas (L.) Lam. Advances in Biomedical Sciences. 4: 46-51.
- Pace RD, Sibiya TE, Phills BR, Dull GG (1985) Ca, Fe and Zn content of ‘Jewel’ sweetpotato greens as affected by harvesting practices. J Food Sci 50: 940-941.
- AVRDC (1985) Composition of edible fiber in sweet potato tips. 1985. Asian Vegetable Research and Development Center Progress Report 1985: 310-313.
- Woolfe JA (1992) Sweet potato. An untapped food resource, Cambridge University Press, Cambridge, UK, pp. 118-187.
- Yoshimoto M (2001) New trends of processing and use of sweet potato in Japan, Farming Japan, 35: 22-28.
- Harborne JB (1994) The flavonoids: Advances in research since 1986. London, U.K.: Chapman & Hall.
- Islam S, Yoshimoto M, Yamakawa O, Ishiguro K, Yoshinaga M (2002). Antioxidative compounds in the leaves of sweetpotato cultivars. Sweetpotato Res Front 13: 4.
- Islam S, Yoshimoto M, Yahara S, Terahara N, Yamakawa O (2002) Anthocyanin compositions in sweetpotato (Ipomoea batatas ) leaves. Biosci Biotechnol Biochem 66: 2483-2486.
- Islam S, Yoshimoto M, Yahara S, Okuno S, Ishiguro K, et al. (2002) Identification and characterization of foliar polyphenolic composition in sweetpotato (Ipomoea batatas ) genotypes. J Agric Food Chem 50: 3718-3722.
- Islam S, Yoshimoto M, Yamakawa O (2003) Distribution and physiological function of caffeoylquinic acid derivatives in sweetpotato genotypes. J Food Sci 68: 111-118.
- Islam S, Yoshimoto M, Ishiguro K, Yamakawa O (2003) Bioactive and Functional properties of Ipomoea batatas Leaves. Acta Hortic 628: 693-699.
- Islam S, Jalaluddin M, Garner JO, Yoshimoto M, Yamakawa O (2005) Artificial shading, and temperature influence on anthocyanin compositions in sweetpotato leaves. Hort Science 40: 176-1801.
- Harborne JB (1980) Plant phenolics, In: Bell EA, Charlwood BV (Eds.). Encyclopedia of plant physiology. New series, vol. 8. Springer Verlag, Berlin, Germany. pp. 329-402.
- Heim KE, Tagliaferro AR, Bobilya DJ (2002) Flavonoid antioxidants: chemistry, metabolism and structure activity relationship. J Nutri Biochem 13: 572-584.
- Sowers JR, Epstein M, Frohlich ED (2001) Hypertension and cardiovascular disease: an update. Hypertension 37: 1053-1105.
- Toeller M (1994) α-Glucosidase inhibitors in diabetes: efficacy in NIDDM subjects. Eur J Clin Invest 24(Suppl 3): 3l-35.
- Ames BN, McCann J, Yamasaki E (1975) Methods for detecting carcinogens and mutagens with Salmonella/mammalian microsome mutagenicity test. Mutat Res 31: 347-364.
- Halliwell B, Gutteridge JMC, Cross CE (1992) Free radicals, antioxidants, and human disease: where are we now? J Lab Clin Med 119: 598-620.
- Islam S (2008) Potential Chemo-preventative properties isolated from Ipomoea batatas In: Functional Foods and Chronic Diseases. Publisher: Functional Food Center at D & A Inc., TX., USA, pp. 96-109.
- Islam I, Shaikh A, Islam S (2009) Antimutagenic and antioxidative potential phytochemicals from sweetpotato. Int J Cancer Res 5: 83-94.
- Shinohara K, Kurogi M, Miwa M, Kong Z, Hosoda H (1988) Antimutagenicity of dialyzates of vegetables and fruits. Agric Biol Chem 52: 369-1375.
- Yamakawa O, Yoshimoto M (2002) Sweet potato as food materials with physiological functions. Acta Hortic 583: 179-185.
- Islam S, Jalaluddin M (2004) Sweet potato- a potential nutritionally rich multifunctional food crop for Arkansas: J Arkansas Agric Rural Dev 4: 3-7.
- Islam S (2015) Potential antidiabetic activity of naturally occurring compounds by enzyme α-glucosidase inhibition. Acta Hortic 1106: 169-174.
- Rhabasa LR, Chiasson JL (2004) Alpha-glucoside inhibitors. In: Defronzo RA, et al. (Eds.). International textbook of diabetes mellitus (vol. 1). John Wiley., UK.
- Huaman Z (1992) Systematic Botany and Morphology of the Sweet potato Plant. Technical Information Bulletin 25. International Potato Center, Lima, Peru. pp.22.
- Islam S, Yoshimoto M, Ishiguro K, Okuno S, Yamakawa O (2003) Effect of Artificial shading and temperature on radical scavenging activity and polyphenolic composition in sweet potato (Ipomoea batatas ) leaves. J Amer Soc Hort Sci 128: 182-187.
- Yoshimoto M, Okuno S, Kumagai T, Yoshinaga M, Yamakawa O (1999) Distribution of antimutagenic components in colored sweet potatoes. J. Japan Agric Res 33: 143-148.
- Islam S (2009) Polyphenol contents and caffeic acid derivatives from leaves of Ipomoea batatas Acta Hortic 841: 527-530.






























