Influence of oligochitosans and highly molecular chitosan on Lactobacillus bulgaricus cultivation
Shramko MI1, Berezueva EV1, Alieva LR1, Lodygin AD1, Evdokimov IA1, Tikhonov VE3, Sushinskaya NV2, Halavach TN2 and Kurchenko VP*1,2
1North-Caucasian Federal University, Stavropol, Russia
2 Belorussian State University,Minsk, Belarus
3A.N. Nesmeyanov Institute of Organoelement Compounds of Russian Academy of Sciences, Moscow, Russia
Submission: April 20, 2020; Published: April 30, 2020
*Corresponding author: Kurchenko Vladimir Petrovich, Faculty of Biology, Head of the Laboratory of Applied Problems of Biology, Belarusian State University, 220030, Minsk, Independence Avenue, 4, Belarus
How to cite this article: Shramko M, Berezueva E, Alieva L, Lodygin A, Evdokimov I, et al. Influence of oligochitosans and highly molecular chitosan on Lactobacillus bulgaricus cultivation. Nutri Food Sci Int J. 2020. 9(5): 555773. DOI: 10.19080/NFSIJ.2019.09.555773.
Abstract
It was established that decrease of oligochitosans with molecular masses 7.0, 25.4, 45.3 kDa concentration in the process of Lactobacillus bulgaricus cultivation leads to fermented dairy product pH reduction and titratable acidity increase. Further increase in titratable acidity and decrease of lactic acid microorganisms’ amount was determined during the fermented dairy product storage process. Oligochitosans with molecular masses 7.0, 25.4, 45.3 kDa in concentrations interval from 0.0025 to 0.01 per cent did not exhibit prebiotic properties. Active acidity elevation and titratable acidity depression was observed at the chitosan with molecular mass 350 kDa concentration rises. Also increase of highly molecular chitosan concentration leads to elevation of lactic acid microorganisms’ total amount, which was more than three degree as many as total count of lactic acid bacteria in control sample.
Keywords:Chitosan; Oligochitosan; Lactic acidbacteria; Lactose, Lactic acid fermentation; Lactic acid
Introduction
Starters of the Lactobacillus bulgaricus species pure cultures are widely used for manufacturing of functional fermented dairy products with dietary and health-promoting properties. The prospective way of fermented milks production technological development is enrichment with chitosan [1-3]. Chitosan is a biogenic heteropolymer consists of N-acetylglucosaminaine and glucosaminresidues [2,4]. Chitosan has high molecular mass and soluble in organic acids [5,6]. Low-molecular derivatives of chitosan are represented byolygochitosans with a molecular mass from 2 to 50 kDa, which are well soluble in water. Chitosan and olygochitosans are able to interact with Lactobacillus bulgaricus cells by a different mechanism depending of their molecular mass [7-9]. Teichoic acid negatively charged molecules of lactic acidbacteria cells are capable to multi-point ion binding with positively charged high-molecular chitosan, whereas their cytoplasmic membrane proteins interact with oligochitosans [4,9]. The consequence of this process may be a change in metabolic processes in lactic acid bacteria cells. The goal of research was to study the effect of different concentrations of high-molecular chitosan and oligochitosans with varying molecular mass on lactic acid fermentation process driven by Lactobacillus bulgaricus.
Materials and Methods
Targets of research were skim milk, starter culture of lactic acid bacteria Lactobacillus bulgaricus (producer: Dairy Plant “Stavropolsky”, Russia), chitosan with a molecular mass of 350 kDa and a 95 per cent degree of deacetylation (manufacturer: “Bioprogress LLC”, Russia). Oligochitosans with molecular masses of 7.0, 25.4, 45.3 kDa and 96 per cent degree of deacetyration was prepared by the previously described technique [5]. Dry skim milk was reconstituted to a dry mass concentration of (10 ± 0.2) % by dissolving in distilled water at temperature 30 to 35 °C. Reconstituted skim milk after recombination was characterized by the following parameters: mass concentration of fat 0.15 per cent, mass concentration of protein 3.2 per cent,mass concentration of lactose 5 per cent. The solution of chitosan with molecular mass 350kDa in 2 per cent concentration lactic acid aqueous solution with mass concentration 1 per cent was added into skim milk experimental samples for preparation of mixture with final concentration of chitosan 0.0025, 0.005, 0.0075 and 0.01 per cent respectively. Similar experiments were carried out using oligohitosans with molecular masses of 7.0, 25.4, 45.3 kDa in above mentioned concentrations. The starter culture of Lactobacillus bulgaricus was inoculated in the amount of 3 per cent of the total samples volume after pasteurization of the mixture and cooling to the fermentation temperature of (43 - 45) °C. The end of the fermentation process was determined by organoleptic curd density, as well as by titratable and active acidity. Experimental and control samples were stored during 17 days at 4 ± 2 °С after completion of fermentation process. Following parameters were tested in triplicate during storage of control and experimental samples: pH by potentiometry, titratable acidity by titrimetric analysis and total count of lactic acid bacteria (CFU per gram).
Results and Discussions
The effect of highly molecular chitosan and oligohitosans with a molecular weight of 7.0, 25.4, 45.3 kDa various concentrations on fermented dairy products physical and chemical properties during the cultivation of Lactobacillus bulgaricus and long-term storage process was studied.
As shown in Tables 1 &2, decrease in the concentration of oligochitosans leads to significant decrease in pH and increase of titratable acidity after 20 hours of cultivation.
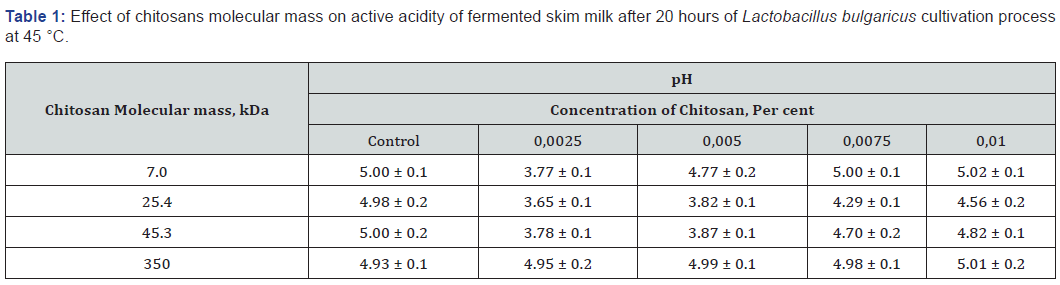
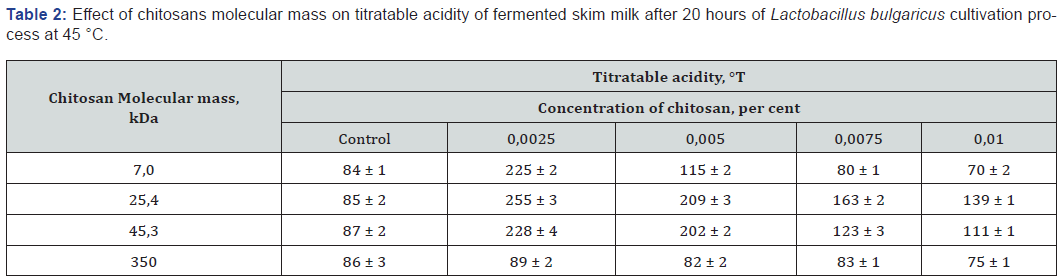
This is explained by the fact that oligohitosans with molecular masses of 7.0, 25.4, 45.3 kDa in concentrations of 0.0025 and 0.005 percent effectively interact with the proteins of the lactic acid bacteria cytoplasmic membrane. This interaction induces bacterial stress [10]. Consequently, lactose enzymatic hydrolysis and lactic acid production are accelerated resulting in titratable acidity increase. The elevation of oligohitosans concentration leads to promotion of their interaction with bacterial cells teichoic acid molecules. This type of interaction influences on lactic acid bacteria cells cytoplasmic membrane permeability and as a result inhibit rate of lactose metabolism. Highly molecular chitosan concentration variation did not lead to significant changes of pH and titratable acidity of fermented skim milk in comparison with control samples. Chitosan with a molecular mass of 350 kDa puts into effective multi-point ion binding with negatively charged teichoic acid molecules of Lactobacillus bulgaricus cells. This is due to the presence into highly molecular chitosan structure of about 1850 amino groups. Lactose assimilation and lactic acid formation rates are changed depending on highly molecular chitosan concentration.
Physical and chemical properties of fermented dairy products during long-term storage at 4 ± 2 °С were studied after the completion of the Lactobacillus bulgaricus cultivation process. It was established that optimal organoleptic attributes (taste and odor) of fermented product control sample are achieved after 5 days of storage at pH 4.2 - 4.5 and titratable acidity 70 - 140 °T. Organoleptic attributes of this product deteriorated during the further storage.
As shown in Table 3, optimal titratable acidity of fermented milks experimental samples containing oligochitosans at a concentration of 0.01 per cent persisted for up to 17 days. Further increase of titratable acidity of experimental samples containing oligochitosans at a concentration 0.0025, 0.005 and 0.0075 per cent was observed during the storage after the completion of the fermentation process.
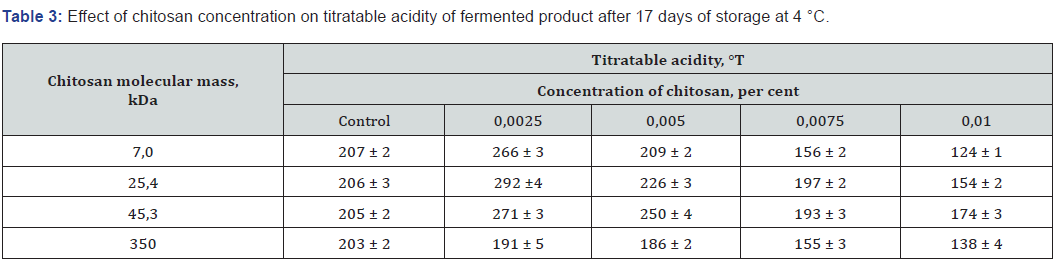
Decrease in titratable acidity of fermented dairy product experimental samples was detected when concentration of chitosan with molecular mass 350 kDa increased in interval from 0.0025 to 0.01 percent. Therefore high-molecular chitosan concentration elevation reduces the intensity of lactic acid fermentation in experimental samples. The most powerful process of lactose homo fermentative fermentation inhibition occurred in a sample containing high-molecular chitosan in concentration of 0.01 per cent. The decrease of lactose assimilation intensity by Lactobacillus bulgaricus cells may be propelled by two reasons. The interaction between chitosan molecules and lactic acid bacteria cells cytoplasmic membrane leads to disturbance of membrane permeability for β-galactosidase enzyme, which catalases the reaction of lactose into glucose and galactose hydrolysis. At the same time structural changes in cell cytoplasmic membrane cause retardation of lactose hydrolysis products active transport into bacterial cells.
Thus, there is an inhibition of lactic acid formation in the process of fermented dairy product containing high-molecular chitosan storage, which stimulates the preservation of a large number of lactic acid bacteria. This is confirmed by the data of lactic acid microorganisms ‘quantitative accounting in control and experimental samples after 17 days of storage, as shown in Table 4.
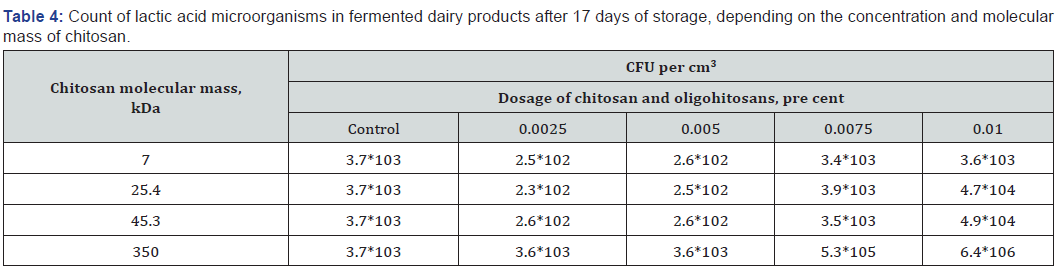
The data presented in Table 4 indicates that oligohitosans with molecular masses of 7.0, 25.4, 45.3 kDa did not affect significantly on Lactobacillus bulgaricus grows rates during fermented dairy products storage process. Addition of highly molecular chitosan in concentrations of 0.0075 and 0.01 per cent in fermented milks increased the content of lactic acid microorganisms,which was more than three degree as many as total count of lactic acid bacteria in control sample.
Thus, tested samples ofoligohitosans with varying degrees of polymerization did not exhibit prebiotic properties and did not prolong the shelf life of fermented dairy products. High-molecular chitosan in a concentration of 0.01 per cent can be recommended as a prebiotic, prolonging the shelf life of fermented milks, manufactured with application of Lactobacillus bulgaricus starter cultures.
References
- Evdokimov IA, Alieva LR, Varlamov VP, Kharitonov VD, Butkevich TV, et al. (2015) Usage of chitosan in dairy products production. Foods and raw materials 3: 29 -39.
- Kean T, Thanou M (2010) Biodegradation, biodistribution and toxicity of chitosan. Adv Drug Deliv 62(1): 3-11.
- Aranaz I, Mengíbar M, Harris R, Paños I, Miralles B, et al. (2009) Functional characterization of chitin and chitosan. Current Chemical Biology 3(2): 203-230.
- Raafat D, Bargen K, Haas A, Sahl HG (2008) Insights into the Mode of Action of Chitosan as an Antibacterial Compound. Appl Environ Microbiol 74(12): 3764-3773.
- Kulikov S, Tikhonov V, Blagodatskikh I, Bezrodnykh E, Lopatin S, et al. (2012) Molecular weight and pH aspects of the efficacy of oligochitosan against methicillin-resistant Staphylococcus aureus (MRSA). Carbohydrate Polymers 87(1): 545 - 550.
- Bezrodnykha EA, Blagodatskikha IV, Kulikovb SN, Zelenikhinb PV, Yamskova IA (2018) Consequences of chitosan decomposition by nitrous acid: Approach to nonbranched oligochitosan oxime. Carbohydrate Polymers 195: 551-557.
- Hoven VP, Tangpasuthadol V, Angkitpaiboon Y, Vallapa N, Kiatkamjornwong S (2007) Surface-charged chitosan: Preparation and protein adsorption. Carbohydr Polym 68: 44-53.
- Kurchenko VP, Radzevich TV, Rizevskya SV, Varlamov VP, Yakovleva IV, et al. (2018) Influence of Molecular Weight of Chitosan on Interaction with Casein. Applied Biochemistry and Microbiology 54: 501-504.
- Raafat D, Sahl HG (2009) Chitosan and its antimicrobial potential - A critical literature survey. Microb Biotechnol 2: 186-201.
- Olicón-Hernández DR, Uribe-Alvarez C, Uribe-Carvajal S, Pardo JP, G Guerra-Sánchez (2017) Response of ustilago maydis against the stress caused by three polycationic chitin derivatives. Molecules 22: 1745.






























