Molecular Identification, Biological and Reproductive Characteristics of Drosophila Suzukii Developed in Six Host Plants of Economic Importance in Iran
Yousef Naserzadeh*1, Niloufar Mahmoudi1, Jaime Gonzalez Cabrera2 and Albert Engeribo1
1 Department of AgroBiotechnology, Peoples’ Friendship University of Russia (RUDN University), Russia
2 Entomophagous-insects Researcher at Centro Nacional de Referencia de Control Biologico, CNRF-DGSV, Mexico
Submission: September 07, 2021; Published: September 15, 2021
*Corresponding author: Yousef Naserzadeh, Department of AgroBiotechnology, RUDN University, Russia
How to cite this article: Yousef N, Niloufar M, Jaime G C, Albert E. Molecular Identification, Biological and Reproductive Characteristics of Drosophila Suzukii Developed in Six Host Plants of Economic Importance in Iran. Agri Res & Tech: Open Access J. 2021; 26 (2): 556332. DOI: 10.19080/ARTOAJ.2021.26.556332
Abstract
Currently, the spotted-wing drosophila, Drosophila suzukii Matsumura (Diptera: Drosophilidae), is threatening the worldwide berry and small fruit industries, but the current control methods have many disadvantages, among them are capital intensive, require high labour and not sustainable in the long term. Therefore, to develop alternative management strategies, there were investigated several biological and reproductive characteristics of D. suzukii developed in six host plants of economic importance in Tehran-Varamin province, Iran: Clementine mandarin, Golden apple smoothie, Pears, Persimmons, Red apples, and Valencia oranges. Among the six tested hosts, pear was identified as the most favorable host for D. suzukii since it registered high daily oviposition, high percentage of egg hatching, and both a short duration of larvae and pupa stages. These results could be useful to develop management strategies, in two ways. First, to implement the reproduction of this dipteran and its main parasitoid Trichopria drosophilae (Perkins) under a mass-rearing approach, and second, to help to predict the trend and pattern of infestation in Tehran-Varamin province. Due to the high sensitivity of pear fruits to this pest, it is recommended to exercise prudence in the planting of new pear orchards, especially mixed with citrus.
Keywords: Host Preference; Mass Rearing of Parasitoids; Augmentative Biological Control; Native Parasitoids; Spotted Wing Drosophila; Trichopria Drosophilae
Introduction
The spotted-wing drosophila, Drosophila suzukii Matsumura (Diptera: Drosophilidae), is native of Southeast Asia, but recently in 2008 invaded American, European, and Western countries. Its niche expansion represents a heavy burden for the worldwide berry and small fruit industries because it can cause economic losses up to 50 and 100% in conventional berry crops [1,2]. And organic fields [3,4]. Respectively. Worldwide, D. suzukii is controlled mainly through the use of insecticides and cultural practices, but these methods entails risks to human health and the environment, possible loss of its effectiveness through time, and they are not sustainable farming systems, in other words, they are not applied in lands surrounding the agricultural areas, and if these sites contain one of the 50 wild host plants of this dipteran, they could be a constant infestation source of D. suzukii [5-9]. In addition to the disadvantages previously mentioned, these current control methods are not 100% effective, since although all the recommended practices are applied, the pest still causes losses of 19%, moreover, these are capital intensive and require high labour input [10,1]. Therefore, the search for better and sustainable methods is a worldwide ongoing effort [3,11]. The use of native or local parasitoids is considered a sustainable alternative to control D. suzukii, since their effect would reach both agricultural and surrounding areas and would persist thorough time, and among the 20 worldwide collected species, Trichopria drosophilae (Perkins) (Hymenoptera: Diapriidae) has been reported with the highest potential to combat D. suzukii, mainly because augmentative field releases of this parasitoid have reduced the dipteran population up to 50% [4,9,13]. However, this parasitic has not been routinely used against this pest, probably due to the lack of effective mass production methods [14,15]. D. suzukii was first detected in Iran in 2015 (CABI, 2015), and despite the imminent economic threat of Drosophila suzukii in the small fruit industry, so far, limited studies have been conducted on the effect of the type of diet on its reproduction [16]. Therefore, this study was conducted to investigate biological and reproductive characteristics of D. suzukii developed in six host plants of economic importance in Tehran-Varamin province (Iran). Knowing how D. suzukii develops in different host plants could be useful to implement the mass rearing of both this dipteran and its parasitoid T. drosophilae, in addition, knowing such information could help to predict the trend and pattern of infestation in Iran, Tehran-Varamin province.
Materials and Methods
Place of study and source of biological materials
This work was carried out at the facilities of the Entomology Laboratory, Institute of agriculture. The rearing stock of the fruit flies was made up of four pairs of adults that originated from a laboratory belonging to the department of plant protection, PFUR in Russia (Picture 1). The dipteran colony as well as these experiments (described below) were maintained under laboratory conditions of 23 ± 1 °C and 40 ± 5 % relative humidity. All the tested fruit (described below) were collected in June 2021 in the Kashan province, which is a city in the northern part of Isfahan region, Iran (33°58'59.09"N, 51°26'11.18"E). Fruit that was reddish in color were collected directly from the trees and were immediately brought to the installations previously mentioned.
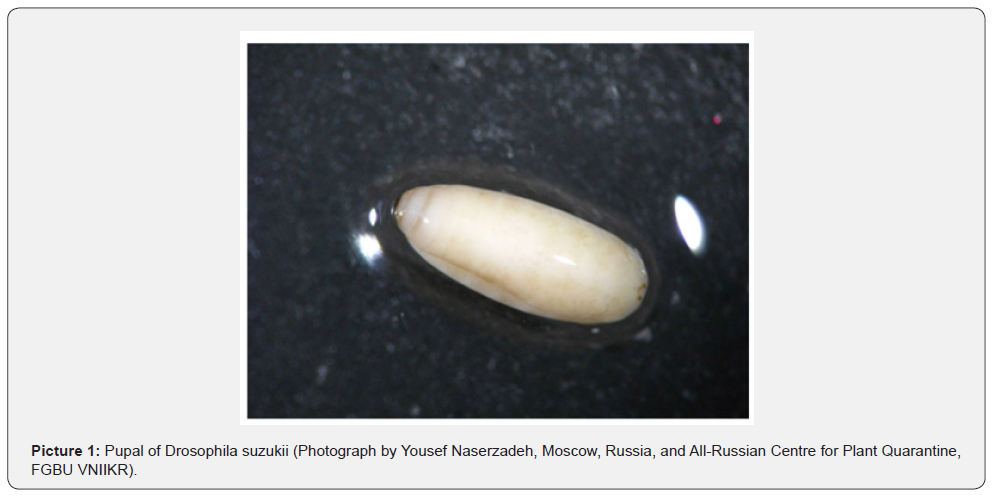
Molecular identification of the rearing stock
To verify the identity of D. suzukii, DNA was extracted from the offspring (larvae and adult) of the rearing stock, and it was performed by treating the specimens with Proteinase K followed by removal of proteins with no extraction with organic solvents and using «DNA Extran-2 Kit», set № NG-511-100 (“Synthol”, Russian Federation) as per manufacturer's instructions [17].
PCR-products purification
Added a 1:1 volume of Binding Buffer to completed PCR mixture (e.g., for every100 µL of the reaction mixture, add 100 µL of Binding Buffer). Mixed thoroughly. Transferred up the solution to the Gene JET purification column. Centrifuged for 30-60 s. discarded the flow-through. Added 700 µL of Wash Buffer to the Gene JET purification column. Centrifuged for 30-60 s. discarded the flow-through and place the purification column back into the collection tube. Then centrifuged the empty Gene JET purification column for an additional 1 min after that transferred the Gene JET purification column to a clean 1.5 mL micro centrifuged tube. Added 50 µL of Elution Buffer to the center of the Gene JET purification column membrane and centrifuged for 1 min.
Sequencing
DNA extracts were quantified on a NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific Inc., USA). Sequencing was done by the generally accepted protocol with the use of Genetic Analyzer AB- 3500 (Applied Biosystems, USA). The Drosophila spp. primers, S1859 and A2191, are targeting several Drosophila species and generate an amplicon of 220-bp length. PCR conditions were identical for both primer pairs: each 25 µL reaction included 2 µl of DNA extract (10 pmol), 5x PCR Master Mix, Screen-mix (HS-5x), 0.5 µM each primer, 17 µl water. Primer S1859 (5′-GGAACAGGATGAACAGTTTAACCGCC-3′) as a forward and A2191 (5́-CCCGGTAAAATTAAAATATAAACTTC-3́) as a reverse were used. And, in a VeritiTM thermocycler (Applied Biosystems, USA). The reaction mixture was as follows: ready-to-use PCR mixture Screen Mix-HS (Evrogen, Russia). PCR conditions: denaturation at 95 °С for 90 sec. followed by 39 cycles, including 30 sec. at 90 °С; primer annealing for 15 sec. at 61 °С; elongation for 30 sec. at 72 °С; final elongation for 5 min at 72°C.
Propagation of the rearing stock population
The initial population of the flies was multiplied first on Prusus persica L. for three generations and then on an artificial diet for two generations (five generations in total). The rearing in pear fruits was made as follows: the primary population was covered in wooden cages of 22×25×25 cm, with their ceilings and walls wrapped with wire holes 0.2mm in diameter (approximately 50 meshes). In order to access the interior of the cages on one side, a hole in the diameter of 10 cm was created and a piece of white shirt in the form of a sleeve was sewn and inserted into it. The cages were placed in a growing room of 1.5 × 1.5 m with a temperature of 25 ± 2 ° C, relative humidity of 70± 5 ° C, an optimal period of 12 hours of light and 12 hours of darkness. Every two to three days, three to four pear fruits were placed on the floor of the cage for the laying of female flies in order to feed adult insects in the cages, a diet based on hydrolyzed protein was used [17]. The infested fruit was gradually removed from the cages and placed in plastic pots with a diameter of 22 cm and a height of 15 cm with holes of 3 cm diameter on the floor. Pots containing infected fruits were blocked using a Lace fabric and the pots were placed on plastic containers containing a mixture of fine soil and sawdust (at a thickness of 3 cm and as a bedding for saplings). The pupas were placed in plastic containers containing a mixture of fine soil and sawdust and transferred to new cages to remove insects from them. Due to the lack of access to pear fruits all year round and also to create a colony with identical nutritional conditions, the rearing of these flies was continued using the artificial diets proposed by van der [18], as follows: artificial food was used to feed full flies in liquid form, and each unit contained warm gr of liquid hydrolyzed protein, 3 gr of sugar, and 25 ml of distilled water. Each artificial larvae unit was completely and sufficiently sustained by 100g of wheat bran, 25g of sugar, 25g of powdered beer yeast from (Health Aid Company of UK), one gram of hydrochloric acid, and one gram of sodium benzoate and 210 ml of distilled water.
Biological and reproductive characteristics
Daily oviposition
Two non-selective and selective tests were used to measure the daily oviposition. In the non-selective test, the persimmon fruits (Diospyros sp.), Citrus reticulata blanco and Valencia oranges (Citrus cinensis (L) were used as experimental treatments alone and without other fruits. For this purpose, approximately 20 pairs of full-day flies fed on artificial nutrition during the larval period were dispensed with a wooden cage with dimensions of 25 × 25× 25 cm, and the protein mixture was hydrolyzed, and water fed to them. Given that the average period before mating of this swarm was reported to be at least four days [19], after four days, the flies were placed inside the cages. A piece of the camellia fruit was cut from it within each cage given. After 24 hours, all fruits were removed from the cages and fresh fruits were replaced. This action lasted for 10 days. Having been removed from the egg cages, the fruits were carefully checked under the stereomicroscope, and the eggs were marked on them. In the next step, the marked sites were split using insect needles and the number of eggs laid in each of them was counted. By dividing the number of eggs laid per day by the number of flies in the cage, the average daily egg was calculated on different fruits. In the selective test, from apple fruits, Malus domestic (Borkh) (Golden smoothie and Red) and pear (Mashhad) were used as experimental treatments. At first, 20 pairs of swarms that had reached the laying period were released into the wooden log cabin. Then, all three fruits were placed in the cage simultaneously and together. After 12 hours, the fruits were removed from the cage and healthy fruits were replaced. Then, the number of eggs laid in them was counted as before. By dividing the total number of eggs counted on each fruit with the total number of flies in the cage (including the number of dead flies per day), the average daily egg was calculated on each fruit.
Egg hatching
According to the Hussain (1995) method [19], Clementin mandarin, apple (Golden smoothie and Red), and pear (Mashhad) cultivars were put in the wooden cages that were pre-purified for Mediterranean fruit (maximum 24 hours) during their growing season [20]. After 24 hours, the fruits were removed from the cage and the laying sites were marked under a stereomicroscope. The fruits were then placed in the germinator (25 ± 2 ° C, 70 ± 5%, 12 hours’ light, and 12 hours’ darkness). Given that the length of the embryonic period of the flies was reported to be 1.5 to three days and given the maximum length of time that it was possible to store split fruits inside the germinator, tangerines, persimmon, pear (Mashhad) were removed after four days, and Valencia orange fruits, yellow apples, and red apples were removed from the germinator after six days [21]. All the laying sites were split over them, and the number of hatching eggs remained (based on the number of eggshells) and the percentage of hatching eggs was calculated. Due to the low level of normal egg-laying in the Valencia oranges, a sharp knife was used to cut the skin in semi-circular orientation and a certain number of eggs were placed under the skin and on the fruit. Then, the cut was restored to its original location and tightened with tape.
Larvae and pupae developmental time
After determining the percentage of hatched eggs, each contaminated fruit was separately packaged in a plastic container with a diameter of 10 cm and a height of 7 cm and a layer of a centimeter mixture of fine soil and sawdust and multi-layer newspaper (as a substrate of pupae). The craters of the dishes were covered with lace fabric and then turned into germinator. The dishes were regularly visited until the pupae were formed. The time interval between egg hatching on each fruit and the formation of saplings was determined as the length of the period and the larval aspect. In order to determine the length of the pupae period, the soil in containers that had infested fruits was observed on a daily basis until the key removal of the larvae from the fruits and their transfer into the soil to form pupas. The pupas were collected per day depending on the type of fruit and placed in 8 cm diameter petri dishes that were covered with a centimeter layer of mixture containing fine soil and sawdust. Then, petri dishes containing pupas were kept until the insects were completely embedded in the germinator. The time interval between the formations of pupae to the complete extinction of insects was considered as the duration of the pupal period.
Percentage of adult emergence and female sex ratio
In order to calculate the total adult emergence rate and sex ratio of individuals, the number of pupae per fruit and the number of insects removed from them were counted on a daily basis, and the sex of all the flies was determined based on the presence or absence of egg yolks.
Statistical analysis
All experiments were conducted in a completely randomized design. The treatments in this study included persimmon, Tangerine, Valencia orange, yellow apples of Golden smoothie, red apples and pear (Mashhad) (sixty-day cultivar). Due to technical problems (for example, lack of sufficient swarms of flies and non-simultaneous possession of all fruits by reason of different growing seasons, some characteristics were not measured on some fruits. The number of replicates varied according to the type of character; the average daily egg, hatching percentage, total insect emergence percentage and female percentage in 10 replicates and length of larval and pupal periods in 25 replicates were measured. The data were analyzed using SAS (SAS 1999, Institute) software and the means were compared using Duncan's multi-domain test and at 5% probability level. Excel 2007 software was used to draw charts.
Results
Daily oviposition
Analysis of variance showed that in both selective and non-selective tests, the host plant type at the level of probability influenced the percentage of average daily laying of female flies (P ≤ 0.05). Based on comparison of the averages in the non-selective test (Fig. 1), the highest and lowest mean of eggs laid were observed on persimmon fruits (6.95 ± 0.68 eggs per female per day) and Valencia oranges (0.47± 0.57 eggs per female per day). The average daily average eggs of Clementin mandarin was lower than persimmons but more than Valencia oranges. In the selective test (Fig. 1), the mean daily egg laying of female flies on pear fruits was significantly higher than apple fruits (Lebanese yellow and Red varieties), but there was no significant difference between the two apple cultivars.
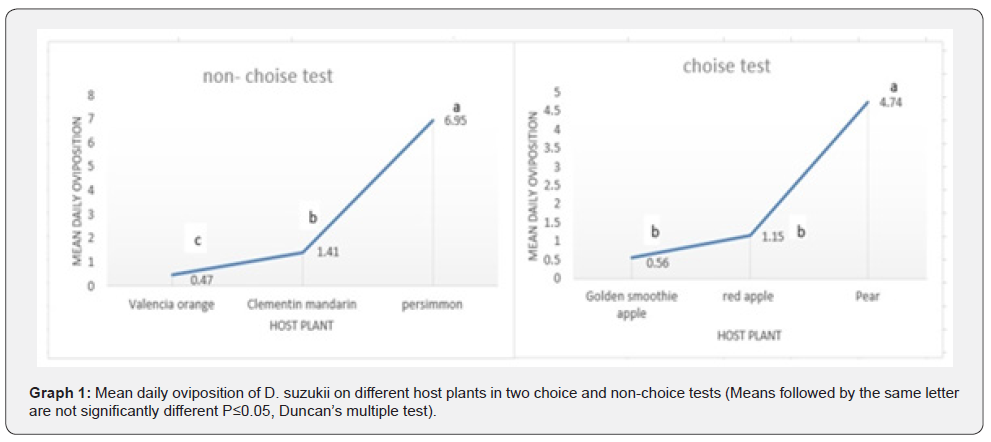
Larvae and pupae development time
The analysis of variance showed that the plant host type had a significant effect on the length of the larval period (P≤ 0.05, and pupae, P ≤ 0.05,) Mediterranean fly has a significant impact. Larval period and larval stage varied from 8.92 ± 0.33 days to 24.08 ± 1.67 days, respectively, on Valencia orange and red apple fruits. The lowest period of pupas (7.76 ± 0.3 days) was in Pear (Mashhad) and the highest in persimmons fruit (13.84 ± 0.59 days) (Table 1).
Hatched eggs
Analysis of variance showed that the effect of host plant on hatching percentage was significant at level of probability (P≤ 0.05) Based on the results of the comparison of the averages (Table 1), the highest number of eggs after feeding the larvae of the hatching Clementin mandarin fruit (93.65 ± 5.03 %), while the lowest egg hatching (67.49 ± 10.72%) was observed after larvae feeding from Valencia orange fruits.
Percentage of adult emergence and female sex ratio
Mean variance analysis showed that plant host type had no significant effect on the percentage of emergence of insects and the ratio of female subjects (P >0.05). Based on the comparison of the means, the average emergence rate of the adult insects was from 81.08 ± 8.6% in the red apple to 98.55±0.88% in the case of Valencia oranges and the mean the proportion of females varied from 43.25 ± 10.05% in red apples to 59.35 ± 3.01% in Valencia orange (Table 1).
Discussion
The results of this study showed that the daily oviposition, larval and pupae developmental time, and the percentage of hatched eggs were affected by the type of fruit, but the percentage of all adult emergence and sex ratio were not influenced. This was an expected result since it has been reported that the fruit type had affected the biological and reproductive traits of D. suzukii [11-20] as well as the traits of other fruit flies [6]. These characteristics are influenced by fruit traits, such as thickness of skin [18, 20], color [8], size [4], inner relative humidity [7], nutritional quality [13], and the oil concentration in the fruit juice [1,17]. In this regard, the biological difference of D. suzukii here reported may be due to some of the above-mentioned factors, which confirms that this particular aspect requires further research.
Effect of fruit type on daily oviposition
The findings of this research in non-selective tests indicated that the oviposition was low both on Valencia orange and Clementine mandarin fruit compared to persimmons fruit. This result was consistent with other researchers that also found low oviposition on citrus fruits (C. Limon L.) [9,21], and they attributed such effect to the amount and type of essential oils that the citrus fruit contains. In addition, the results of the selective tests showed that D. suzukii flies preferred for oviposition pear instead of red and yellow apple. So far, similar studies comparing these three fruits have not been reported, neither comparing apple and oranges. but Mahmoud et al. and El-Hawagry 2017 quantifying indirectly concluded that the apple fruits are less preferred for oviposition D. suzukii than orange fruits [12, 20]. In contrast, Manrakhan et al. 2018 found that the number of eggs laid by the D. suzukii on apple fruits (cultivars Golden smoothie) is much higher than Valencia oranges and Clementin mandarin [9].
Effect of fruit type on egg hatching
The results of this study showed that the percentage of hatched eggs on Valencia orange or persimmons fruits was lower than those of red and yellow apple fruits, Pear (Mashhad) and Clementine mandarin. The difference ratio in egg hatching among the tested fruits may be due to the different physical structure of the skin and the chemical nature of the essential oils contained in the fruit tissues, such as Pears, apples, nectarines moreover, probable the second-order metabolic compounds (e.g., terpenes and flavonoids) present in the skin could also a play a role in the hatching ratio; however, egg hatching differences of D. suzukii were not found among different species and varieties of citrus, despite the difference in skin thickness, acidity and pH [12]. Therefore, to explain thoroughly the egg hatching on different species and varieties of citrus, the role of other physical and chemical characteristics of the fruit should be further investigated.
Effect of fruit type on the developmental time of larvae and pupae
The results of this study showed that the larval developmental time of D. suzukii in apple fruits (red and yellow) was longer than other fruits. This corresponds to the results of [5]. Regarding the longer larval period of D. suzukii on apple fruits compared to pear and persimmon fruits. But the results of these researchers were slightly different regarding the length of the pupae period of our research; the results indicated that there was no significant difference between the pupae period in persimmon, apples, and pear (Mashhad), while our research findings showed no significant differences between Pear and apple fruits [13]. Nevertheless, the average of both fruits was significantly lower than persimmons. Given the long larval period on apple fruits, as well as the relative lack of oviposition egg on this host, yellow and red apple fruits, along with Valencia oranges (with a mean egg setting of 0.47± 0.57 eggs), was introduced to the most unviable host plants for the D. suzukii. Hauser et al. 2011 and Landolt et al. 2012, reported the mild and bitterness of the Mesopotamian carp apple fruits as the most important cause of larval mortality and prolonged periods. In the present study, the more rigid mesocarp of apple fruits than other fruits such as pear, persimmons, and citrus fruits, could have contributed to the prolonged larval period [22,23]. Concluding this discussion, the results of this study showed that the pear was the most favorable host for D. suzukii since it registered high daily oviposition, high percentage of egg hatching, and both a short duration of larvae and pupa stages. However, the results also showed that are other fruits that partially showed certain advantages, such as Valencia orange and persimmon. Although D. suzukii did not have eggs in Valencia oranges, but the larvae that was transferred by hand had the shortest developmental time among the tested fruits, and this suggests that Valencia orange fruits have a high nutritional value for D. suzukii larvae. If female flies overcome the barriers to egg laying on fruit, the possibility of mass rearing the Dipteran on this host is very high. Our results also showed that the registered daily oviposition was the highest in persimmon fruits, therefore, they can be categorized as the preferred oviposition substrate of these flies, and maybe these fruits should be used only for oviposition purposes, and subsequently, the larvae could rear in another more productive host. Although in the present study, the quantity and quality of the nutrients found in these tested five fruits were not evaluated, nonetheless, the literature reports differences among them, for example, the protein content of one hundred gr of apple juice (0.1-0.49 gr) is much lower compared to orange fruits (0.8-0.9 gr), NOORI et al. 2015, and such differences here reported may be due to some of those factors [23].
Conclusions and Suggestions
The results of this study that the pear was the most favorable host for D. suzukii could be useful to implement the rearing of this dipteran and its parasitoid T. drosophilae under the mass-rearing approach. If implemented this method (following the procedure outlined in materials and methods) would have a mass rearing approach since specialized technical personnel are not required, the substrate to reproduce the host is inexpensive and commonly found (i.e., pear), no sophisticated facilities are required, and no agar or preservatives are used, and consequently, such theoretical mass rearing would allow the groups of producers to self-supply their farms and control the pest using sustainable and low-cost technology. Considering the greater sensitivity of pear fruits than other hosts, as well as unpublished reports from plant protection experts in Tehran-Varamin province on the increased damage caused by this pest in citrus and citrus gardens, in the planting of gardens of fruit trees, including pear (Mashhad) in the province, especially in mixed crops, there should be more stringent measures. Likewise, the data generated in the evaluation of biological and reproductive characteristics of D. suzukii reared in six host plants could be inputted into climatic, demographic, and physiological models to help to predict the trend and pattern of infestation in Iran, Tehran-Varamin province.
References
- Asplen MK, Anfora G, Biondi A (2015) Invasion biology of spotted wing Drosophila (Drosophila suzukii ): a global perspective and future priorities 88(3): 469-494.
- Gonzalez Cabrera J, Moreno Carrillo G, Sanchez Gonzalez JA, Mendoza Ceballos MY, Arredondo Bernal HC (2019) Single and combined release of Trichopria drosophilae (Hymenoptera: Diapriidae) to control Drosophila suzukii (Diptera: Drosophilidae). Neotropical entomology 48(6): 949-956.
- Kruitwagen A, Beukeboom LW, Wertheim B (2018) Optimization of native biocontrol agents with parasitoids of the invasive pest Drosophila suzukii as an example. Evolutionary Applications 11(9): 1473-1497.
- DiGiacomo G, Hadrich J, Hutchison WD, Peterson H, Rogers M (2019) Economic impact of spotted wing drosophila (Diptera: Drosophilidae) yield loss on Minnesota raspberry farms: a grower survey. Journal of Integrated Pest Management 10(1): 11.
- Schetelig MF, Lee KZ, Otto S, Talmann L, Stokl J, et al. (2018) Environmentally sustainable pest control options for Drosophila suzukii. Journal of applied entomology 142(1-2): 3-17.
- Naserzadeh Y, Mahmoudi N, Pakina E, Zargar M (2020) Molecular identification and primer design for spotted wing drosophila (Drosophila suzukii). Research on Crops 21(2): 364-369.
- Burrack HJ, Fernandez GE, Spivey T, Kraus DAJPMS (2013) Variation in selection and utilization of host crops in the field and laboratory by Drosophila suzukii Matsumara (Diptera: Drosophilidae), an invasive frugivore 69(10): 1173-1180.
- Naserzadeh Y, Kartoolinejad D, Mahmoudi N (2018) Nine strains of Pseudomonas fluorescens and P. putida: Effects on growth indices, seed and yield production of Carthamus tinctorius L 19(4).
- Bolda MP, Goodhue RE, Zalom FG (2010) Spotted wing drosophila: potential economic impact of a newly established pest. Agricultural and Resource Economics Update 13(3): 5-8.
- Parchami Araghi M, Pont AC, Gilasian E, Basavand F, Mousavi HJZitME (2017) First Palaearctic record of the genus Pygophora Schiner 1868 (Diptera: Muscidae) from Iranian Baluchestan 63(3): 280-282.
- Rossi Stacconi MV, Grassi A, Ioriatti C, Anfora, G (2018) Augmentative releases of Trichopria drosophilae for the suppression of early season Drosophila suzukii populations. BioControl 64(1):9-19.
- Yousef N, Niloufar M, Elena P (2019) Antipathogenic effects of emulsion and nanoemulsion of cinnamon essential oil against Rhizopus rot and grey mold on strawberry fruits. Foods and Raw materials: 7(1).
- Walsh DB, Bolda MP, Goodhue RE, Dreves AJ, Lee J, et al. (2011) Drosophila suzukii (Diptera: Drosophilidae): invasive pest of ripening soft fruit expanding its geographic range and damage potential. Journal of Integrated Pest Management 2(1): G1-G7.
- Gress BE, Zalom FG (2019) Identification and risk assessment of spinosad resistance in a California population of Drosophila suzukii. Pest management science 75(5): 1270-1276.
- Krüger AP, Scheunemann T, Padilha AC, Pazini JB, Bernardi D, et al. (2021) Insecticide-mediated effects on mating success and reproductive output of Drosophila suzukii. Ecotoxicology 30(5): 828-835.
- Da Costa Oliveira D, Stupp P, Martins LN, Wollmann J, Geisler FCS, et al. (2021) Interspecific competition in Trichopria anastrephae parasitism (Hymenoptera: Diapriidae) and Pachycrepoideus vindemmiae (Hymenoptera: Pteromalidae) parasitism on pupae of Drosophila suzukii (Diptera: Drosophilidae). Phytoparasitica 49(2): 207-215.
- Martínez Ramírez A, Cicero L, Guillén L, Sivinski J, Aluja MJBc (2016) Nutrient uptake and allocation capacity during immature development determine reproductive capacity in Diachasmimorpha longicaudata (Hymenoptera: Braconidae: Opiinae), a parasitoid of tephritid flies 100: 37-45.
- Cuch Arguimbau N, Escudero Colomar LA, Forshage M, Pujade Villar J (2013) Identificadas dos especies de Hymenoptera como probables parasitoides de Drosophila suzukii (Matsumura,1931) en una plantacion ecologica de cerezos en Begues (Barcelona, Espa~na). Phytoma 247: 42-47.
- Naserzadeh Y, Bondarenko G, Kolesnikova E, Zargar M, Pakina E, et al. (2020) Molecular identification and design of specific primer for quarantine fruit fly (Drosophila suzukii). Research on Crops 21(3): 611-614.
- Mahmoudi N, Pridannikov M, Naserzadeh Y, Pakina E, Engeribo A, et al. (2020) Developing PCR primers for identification of potato rot nemato (Ditylenchus destructor) in the Russian Federation and Iran. Research on Crops 21(4): 813-816.
- Naserzadeh Y, Bondarenko GN, Kolesnikova EV, Pakina EN (2021) Phylogenetic analysis and designing new primers for molecular identification of Drosophila suzukii. RUDN Journal of Agronomy and Animal Industries 16(2): 137-145.
- De Pedro L, Tormos J, Asís JD, Sabater Muñoz B, Beitia FJCP (2018) Biology of Aganaspis daci (Hymenoptera: Figitidae), parasitoid of Ceratitis capitata (Diptera: Tephritidae): Mode of reproduction, biological parameters and superparasitism 108: 54-61.
- Dias N, Nava D, Garcia M, Silva F, Valgas RJBJoB (2018) Oviposition of fruit flies (Diptera: Tephritidae) and its relation with the pericarp of citrus fruits 78(3): 443-448.






























