Nitrogen-Containing Heterocycles in Agrochemicals
Eman A Al-Harbi* and Wafaa Ahmed Gad
Department of Chemistry, Taibah University, Saudi Arabia
Submission: May 06, 2018; Published: May 22, 2018
*Corresponding author: Eman A Al-Harbi, Department of Chemistry, Taibah University, Al-Madinah Al-Minawara, Saudi Arabia, Email: eman.a.alharbi@hotmail.com
How to cite this article: E A Al-Harb, Wafaa A G. Nitrogen-Containing Heterocycles in Agrochemicals. Agri Res & Tech :Open Access J. 2018; 16(2): 555986. DOI: 10.19080/ARTOAJ.2018.16.555986
Introduction
Nitrogen-Containing heterocyclic compounds are the most abundant and integral scaffolds that occur in a variety of synthetic drugs, bioactive natural products, pharmaceuticals and agrochemicals.
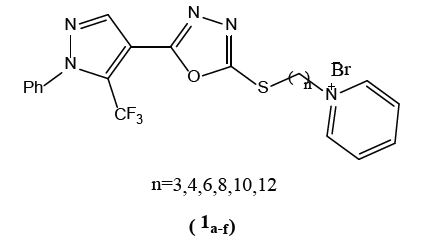
Lei Zhou et al. [1] reported the synthesis and the bacterial activity of pyridinium -5- trifluoro-methylpyrazoles (1a-f) bearing 1,3,4 oxadiazole moiety . These thioethers exhibited significant inhibitory activity against pathogenic bacteria Xanthamonas oryzae pv. oryzae (Xoo), Ralstonia solanacearum and Xanthomonas axonopodis pv.citri (Xac).
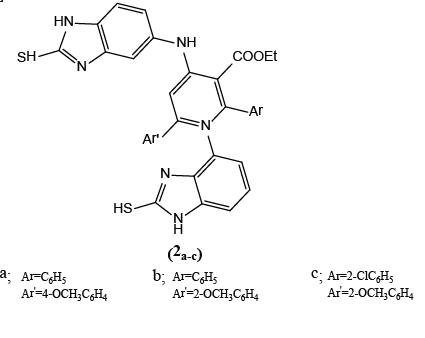
Benzo[d] imidazolyl tetrahydropyridine carboxylates (2a-c) have been prepared and were evaluated for their antifungal activity against Aspergillus niger (ATCC 16404). These compounds are significally toxic toward the fungi. Compounds with the electron- donating methoxy group on the phenyl ring are highly toxic [2].
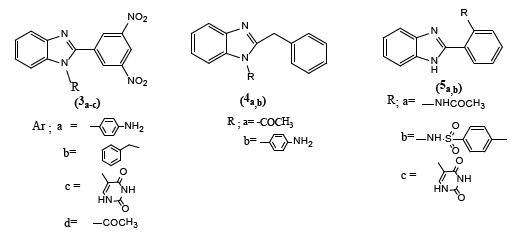
Olyinka O Ajani et al. [3] synthesized benzimidazole derivatives by ecofriendly method and examined their antimicrobial properties for future drug and agrochemical developments. large zones of inhibition were noticed for compounds (3a-c) and (4a) against Bacillus Licheniformis, compounds (3b,d), (4b) and (5a,b) against Proteus Vulgaris and compounds (3a-c), (4a) and (5c) against Pseudomnas acruginosa.
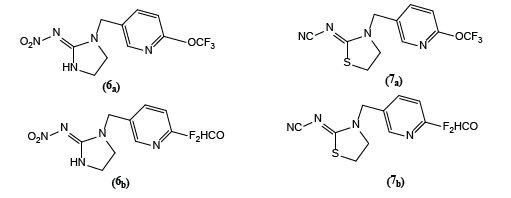
Gregory Landellea et al. [4] reported the synthesis of imidacloprid and thiacloprid containing either 6- trifluoromethoxy or 6-difluoromethoxy –pyridin-3-yl methyl moieties (6a,b) and (7a,b), respectively. F2HCO-Imidacloprid (6b) is more active in vivo against green-peach aphid (M. persicae) than F3CO derivatives (6a), both derivatives are less active than imidacloprid. F3CO-Thiacloprid (7a) is more active in vivo against M. persicae than (7b). Both derivatives (7a,b) are much less active than thiacloprid.
Compounds (2a,b) inhibit 14-α demethylase in sterol biosynthesis pathway of the fungi 2018.
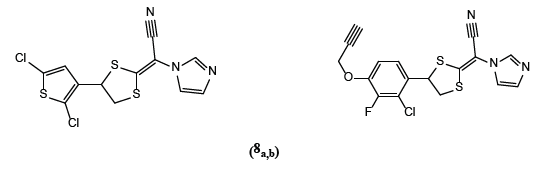
Stephan et al. [5] synthesized 4-(2,5-dichlorothiophen-3-yl)-1,3-dithiolan-2-ylidene)methyl)-1H-imidazole derivatives ( 8a,b) inhibited 14-α-demethylylase in the sterol biosynthesis pathway of the fungi.
The above investigations open the route to a new acess to bioactive ingredients and their further evaluation, which could possibly lead to large activity enhancements.
References
- Lei Zhou, Pei-Yi Wang, Jian Zhou, Wu-Bin Shao, He-Shu Fang, et al. (2017) Antimicrobial activities of pyridinium-tailored pyrazoles bearing 1,3,4-oxadiazole scaffolds. J Saudi Chem Soc 21(7): 852-860.
- Anisetti Ravindernath, Malladi S Reddy (2017) Synthesis and evaluation of anti-inflammatory, antioxidant and antimicrobial activities of densely functionalized novel benzo[d] imidazolyl tetrahydropyridine carboxylates. Arabian J Chem 10(Supplement 1): s1172-s1179.
- Olayinka O Ajani, Olayinka O Tolu-Bolaji, Shade J Olorunshola, Yuxia Zhao, Damilola V Aderohunmu (2017) Structure-based design of functionalized 2-substituted and 1,2-disubstituted benzimidazole derivatives and their in vitro antibacterial efficacy. J Adv Research 8(6): 703-712.
- Gregory Landellea, Etienne Schmitta, Armen Panossiana, Jean-Pierre Vorsb, Sergiy Pazenokc, et al. (2017) Tri- and difluoromethoxylated N-based heterocycles-Synthesis and insecticidal activity of novel F3COand F2HCO-analogues of Imidacloprid and Thiacloprid. J Fluorine chem 203: 155-165.
- Stephane Jeanmart, Julien Gagnepain, Pulakesh Maity, Clemens Lamberth, Fredrik Cederbaum, et al. (2018) Synthesis and fungicidal activity of novel imidazole-based ketene dithioacetals. Bioorg Med Chem 268(8): 2009-2016.






























