Difference of Soil Microbial Populations under Long-Term Different Fertilizer Application and Impact of Herbicide Acetochlor on Them
Hong Ding1*, Jing Zhang1, Xiangzhou Zheng1, Jusheng Gao2, Yue Zou1 and Yushu Zhang1
1Institute of Soil and Fertilizer, Fujian Academy of Agricultural Sciences, China
2Hengyang Red soil Experimental Station, Chinese Academy of Agricultural Sciences, China
Submission: December 11, 2017; Published: February 02, 2018
*Corresponding author: Hong Ding, Institute of Soil and Fertilizer, Fujian Academy of Agricultural Sciences, Fuzhou 350013, China, Tel:+8659187573597; Fax: +8659187572514; Email: hongding@china.com
How to cite this article: Hong D, Jing Z, Xiangzhou Z, Jusheng G, Yue Z, Yushu Z. Difference of Soil Microbial Populations under Long-Term Different Fertilizer Application and Impact of Herbicide Acetochlor on Them. Agri Res & Tech: Open Access J. 2018; 13(5): 555897. DOI: 10.19080/ARTOAJ.2018.13.555897
Abstract
Soil microorganism plays an important role in quality and health of soil, long term different fertilizer application patterns would have differential effect on microorganism populations. The experimental results indicated that manure and chemical fertilizer + manure treatments could maintain higher fertility levels and microbial populations than chemical fertilizer and control treatments, increment of soil organic matter is benefit to growth of microorganisms. Application of herbicide acetochlor had no significant positive or negative effects on the populations of soil microorganisms and species, and also did not display inconsistent results among the soils with different fertilizer application patterns.
Keywords: Microbial population; Paddy soil; Long-term fertilizer application; Herbicide
Introduction
Fertilizer application is an essential measure to improve crop production in modern agriculture. However, different fertilization patterns have obvious effect on soil property and fertility, and optimum pattern can maintain a high soil fertility level and crop yield [1]. Soil microorganism is one of indicators of soil health and fertility, and long-term fertilizer application experiments indicated that different fertilizers lead to great impact on soil microbial population [2-5]. It is thought that organic fertilizer application has a promoting effect on increasing soil microorganism community.
In the meantime, currently herbicides are commonly applied to control weed in crop systems. Differential toxicity of herbicides in soil may cause changes in microbial community structure and function, and concomitantly influence soil health and ecosystem processes [6]. But, there is a serious lack of information that herbicide application whether has differential effects on microbial population of soils with long-time different fertilizer application treatments so far.
Materials and Methods
Experimental soil
The paddy soils sampled from a long-term fertilizer application experimental site of over 35 years at Qiyang red soil experimental station, Hunan province, China (111°52’32” E,26°45’12” N). The basic properties of the soil are shown in Table 1.
Experimental herbicide
The herbicide acetochlor is commonly applied in agriculture. The acetochlor used in this experiment is 50% active ingredient emulsifiable concentrate (EC), which was produced by Qingfeng Agro-chemical Co. Ltd. Hangzhou, China.
Experimental design
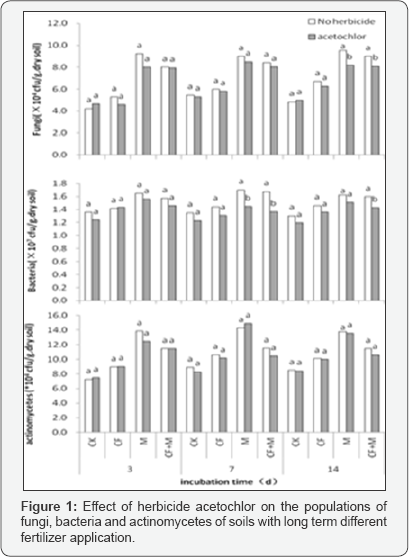
Our investigation on the influence of different fertilizer treatments and acetochlor application on soil microorganisms was conducted in the laboratory with a 14-day incubation period. Ten mg active ingredient (a.i) kg-1 was applied to paddy soil with four replicates. The fresh soils sampled from the experimental site were placed into a 300mL wide-mouth bottle. Subsequently, soil samples were adjusted to 60% of water holding capacity with double deionized water in which acetochlor could be dissolved. Control soils were also prepared only with double deionized water. All soil samples were incubated at 25 °C, and 10g soil was sampled each time by destructive sampling from each bottle at 3, 7 and 14 days after incubation.
Methods for soil microbial populations
Microbial numbers were determined using conventional methods and calculated. Ten grams of each soil sample incubated was suspended in 90mL sterile water and shaken in a shaker for 30min. Then, these soil suspensions were diluted in 10-fold series.
The Colony counting method
0.05mL soil suspensions of 10-3 - 10-7 dilutions were used to inoculate in PDA medium, gauserime synthetic agar medium, beef extract peptone medium [7] and acetamide agar medium[8] respectively, which determined the numbers of fungi, actinomycete, aerobic bacteria and heterotrophic nitrifying bacteria. Each microorganism was cultured with four dilutions and every concentration had four parallel culture dishes. All the dishes were incubated in dark at 2 5 0c. Total number of microorganisms on the agar mediums just need to be counted.
The Most probable number (MPN) method
One mL soil suspensions of 10-3 - 10-7 dilutions were used to inoculate tubes containing selective liquid media. Ammonifier were cultured in peptone ammonification medium[9], nitrosomonas medium reported by Kihn et al. [10], autotrophic nitrifying bacteria medium described by Lin et al. [11], and denitrifying bacteria medium followed by Johns et al. [12]. Each microorganism was cultured with four dilutions and every concentration has four parallel tubes. All the tubes were incubated in dark at 25 °C. After incubation, the pattern of positive and negative tubes is noted, and a standardized MPN table was consulted to determine the most probable number of organisms (causing the positive results) per unit volume of the original sample.
Statistical analysis
All statistical analyses were performed using SPSS19.0 (SPSS for Windows, Version 13.0). The differences of microbial populations among treatments were analyzed using ANOVA, and the differences were considered significant at p<0.05.
Results and Discussion
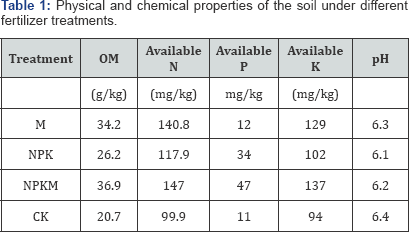
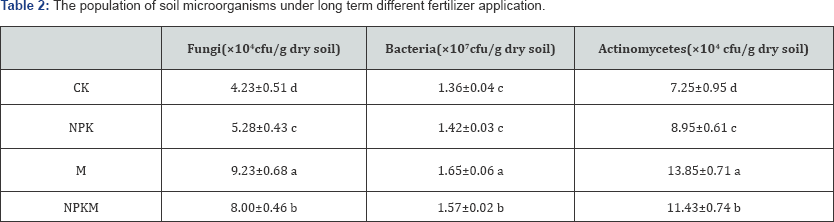
The experimental data in Table 2 showed that fungi, bacteria and actinomycetes populations in soils appeared great differences among fertilizer application treatments, their numbers were in order M>NPKM>NPK>CK. Soil organic matter (OM) is benefit to growth of microorganism, the higher soil OM are the higher the populations relatively are (Table 1 & 2). And, chemical fertilizer application also could increase the populations compared to control. The long term mineral fertilizer increases microbial biomass in cropping systems [4].
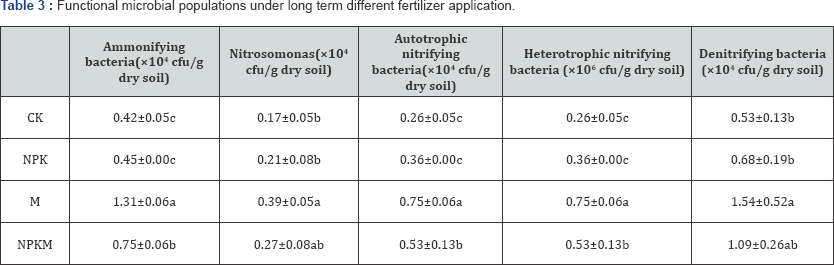
Similarly, the numbers of functional microbe (Table 3) included ammonifying bacteria, nitrosomonas, autotrophic nitrifying bacteria, heterotrophic nitrifying bacteria, denitrifying bacteria also showed M>NPKM>NPK>CK, and significantly greater in two manure application treatments (P<0.05), however, have no apparent differences between chemical fertilizer treatment and control.
Microorganisms. However, these researches had drawn different conclusions that the impacts depended on herbicide types [6,13], applied concentrations [14,15], soil types [16,17], and microbial species [18, 19]. The results in Figure 1 & 2 indicated that herbicide acetochlor application has no influence on microbial populations and species in four long term fertilization treatments under this experimental condition.
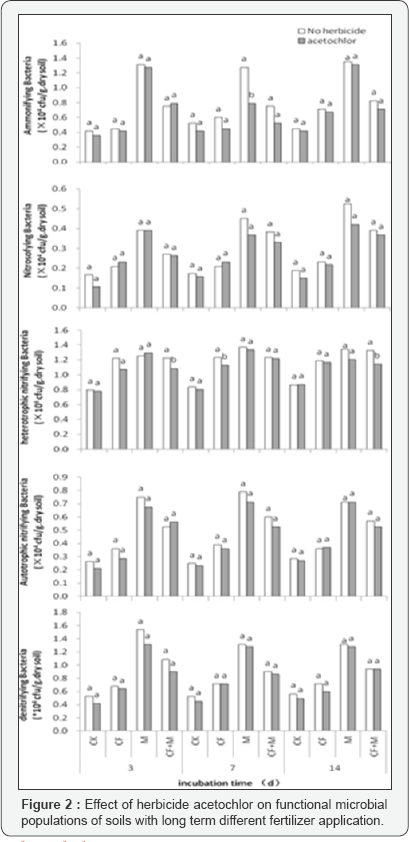
Conclusion
Soil microbial populations, include fungi, bacteria and actinomycetes, as well as ammonifying bacteria, nitrosomonas, autotrophic nitrifying bacteria, heterotrophic nitrifying bacteria and denitrifying bacteria, showed great variation among long term different fertilizer application treatments, the order of numbers displayed as fellows M>NPKM>NPK>CK. Herbicide acetochlor did not significantly inhibit microbial populations, and also had no differences of responses to soils with long term different fertilizer application.
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (grant No.31270556), Fujian Provincial Natural Science Foundation (2015J01159; 2015J01111) and Innovation Team Project of Fujian Academy of Agricultural Sciences (ST1T2017-1-9).
References
- Meena SC, Singh DP, Meena SR (2017) Effect of Long Term Application of Fertilizer and Manure on Establish Relationship between Soil Organic Carbon Fractions with Yield of Maize and Wheat Crops under Maize-Wheat Sequence in Heplustepts. 1nt J Curr Microbiol App Sci 6(9): 600-606.
- Chakraborty A, Chakrabarti K, Chakraborty A, Ghosh S (2011) Effect of long-term fertilizers and manure application on microbial biomass and microbial activity of a tropical agricultural soil. Biol Fertil Soils 47(2): 227-233.
- Ndubuisi-Nnaji UU, Adegoke AA, Ogbu HI, Ezenobi NO, Okoh AI (2011) Effect of long-term organic fertilizer application on soil microbial dynamics. Afric J Biotech 10(4): 556-559.
- Geisseler D, Scow KM (2014) Long-term effects of mineral fertilizers on soil microorganisms - A review. Soil Biol Biochem 75: 54-63.
- Geisseler D, Linquist BA, Lazicki PA (2017) Effect of fertilization on soil microorganisms in paddy rice systems - A meta-analysis. Soil Biol Biochem 115:452-460.
- Zabaloy MC, Garland JL, Gomez MA (2008) An integrated approach to evaluate the impacts of the herbicides glyphosate, 2, 4-D and metsulfuron-methyl on soil microbial communities in the Pampas region, Argentina. Appl Soil Ecol 40: 1-12.
- Smith EA, Mayfield CI (1977) Effect of paraquat on select microbial activities in soil. Microbiol Ecol 3(4): 333343.
- Kidd PS, Prieto-Fernandez A, Monterroso C, Acea MJ (2008) Rhizosphere microbial community and hexachlorocyclohexane degradative potential in contrasting plant species. Plant Soil 302(1-2): 233247.
- Fargo LL, Fleming RW (1977) Effects of chromate and cadmium on most probable number estimates of nitrifying bacteria in activated sludge. B Environ Contam Tox 18: 350354
- Kihn A, Laurent P, Servais P (2000) Measurement of potential activity of fixed nitrifying bacteria in biological filters used in drinking water production. J Indus Microbiol Biotech 24(3): 161166.
- Lin Y, Kong HN, He YL, Yan L, Li CJ (2006) Isolation and characterization of heterotrophic nitrifying bacteria. Environ Sci 27(2): 324328.
- Johns D, Williams H, Farrish K, Wagner S (2004) Denitrification and soil characteristics of wetlands created on two mine soils in east Texas, USA. Wetlands 24(1): 5767.
- Martens DA, Bremner JM (1993) Influence of herbicides on transformations of urea nitrogen in soil. J Environ Sci Health 28(4): 377395.
- Haney RL, Senseman SA, Hons FM, Zuberer DA (2000) Effect of glyphosate on soil microbial activity and biomass. Weed Sci 48(1): 8993.
- Sofo A, Scopa A, Dumontet S, Mazzatura A, Pasquale V (2012) Toxic effects of four sulphonylureas herbicides on soil microbial biomass. J Environ Sci Health B 47(7): 653659.
- Banks ML, Kennedy AC, Kremer RJ, Eivazi F (2014) Soil microbial community response to surfactants and herbicides in two soils. Appl Soil Ecol 74: 12 20.
- Paul S, Chakravarty A, Patra PK, Paul N, Sukul P, et al. (2015) Microbe- chloroacetanilide herbicide interaction across soil type. Open J Soil Sci 5(4): 8799.
- Moorman TB (1989) A review of pesticide effects on microorganisms and microbial processes related to soil fertility. J Product Agric 2(1): 1423.
- Cycon, M, Borymski S, Piotrowska-Seget Z (2013) Short-term effects of the herbicide napropamide on the activity and structure of the soil microbial community assessed by the multi-approach analysis. Appl Soil Ecol 66(8): 818.






























