Biotechnological Significance of Endophytic Actinobacteria an Intensive and Emerging Pursuance
Sudipta Roy*
Department of Biotechnology, Oriental Institute of Science and Technology, India
Submission: April 03, 2018; Published: June 07, 2018
*Corresponding author: Sudipta Roy, Department of Biotechnology, Oriental Institute of Science and Technology, Vidyasagar University, Midnapore, West Bengal 721102, India.
How to cite this article: Sudipta Roy. Biotechnological Significance of Endophytic Actinobacteria an Intensive and Emerging Pursuance. Adv Biotech & Micro. 2018; 9(4): 555767. DOI: 10.19080/AIBM.2018.09.555767
Abstract
The emerging field of drug discovery in recent decades has drawn much interest in using microbial natural products. These are fundamentally biodegradable, specific and generally have less toxicity. Finding of new bioactive active molecules, which are effective against harmful microbes, are an important area of antibiotic research. The prolife ration in number of drug-resistant microorganisms and the throttled isolation of combinatorial chemistry in providing new effective drugs direct uncertainty for future antimicrobial therapy. Thus finding new groups of microbes from unexplored or less explored habitats became mandatory by pursuing them as sources of novel bioactive compounds. Endophytes play mutual listic to their host; among which some act to be making returns for the nutrition from the plant by providing special substances, as secondary metabolites. This is the distinctive feature of endophytic research for pharmaceutically active compound.
Keywords: Actino bacteria; Endophyte; Bioactive; Microbial metabolites; Drug resistance
Introduction
Microbial cell factories are considered as the cheapest but the richest source of various molecules with immense application. There is always a definite need of potential remedial compounds to face the increasing health, agriculture or environment related problems. From the very ancient period we rely on plant as source of various potential bio-molecules. Moreover, pharmaceutical research was significantly dependent on exploration of plant derived bio-molecules from the ancient period. Among the 7,000 medicinal compounds documented in the modern pharmacopoeia are of plant origin. However, application microorganisms for pharmaceutical purpose was remarkably noticed among the ancient Egyptians and Greeks by using specific moulds and plant extracts to treat infection [1].
A revolution was initiated in the field of drug discovery after finding natural mold, Penicillium with antibacterial property by Sir Alexander Fleming and it was effectively used to treat bacterial infection [2]. Though a fungus was the first antimicrobial producer, soon actinomycetes have drawn most interest to be prolific sources of novel secondary metabolites with a range of biological activities that may ultimately find application as pharmaceutically or agriculturally useful compounds. Endophytic microorganisms are defined as, “microbes that colonize living, internal tissues of plant without causing any immediate overt negative effect” [3]. Evidences of plant associated microorganisms found in the fossilized tissues of stems and leaves have indicated that host - endophyte association may have evolved from the time that higher plants first appeared on the earth [4]. The most frequently encountered endophytes are representative of fungi; however, the existences of many endophytic actinobacteria are also documented well.
Such plant microbe interaction may be partially due to the endophytic production of phytohormones, such as Indole-3- Acetic Acid (IAA), cytokinine, or other plant growth promoting substances (vitamins) and/or partly owing to the fact that endophytes can enhance host absorption of nutritional elements such as nitrogen, phosphorus [5] and that they regulate nutritional qualities, nitrogen-carbon ratio [6]. Endophyte infected plants also get protection from herbivores or pathogens due to bioactive secondary metabolites that the endophytes produce in the plant tissues. Besides, some other beneficial features are also offered to plants by endophytes including drought acclimatization, improved resistance from pests, enhanced tolerance to stressful conditions like heavy metal presence, low pH, high salinity etc. [7].
Actino-Bacteria Biology
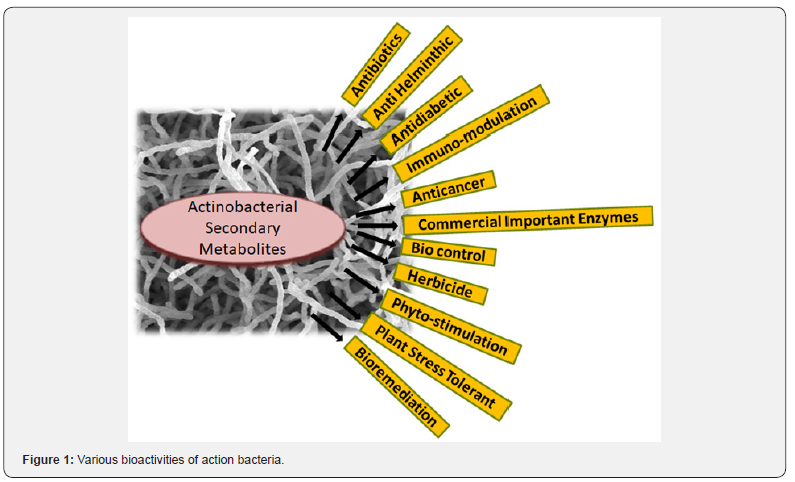
Actinobacteria are a group of high G-C (>55mol %) in DNA containing (with exception obligate pathogen Tropherymawhipplei, with less than 50% G+C) Gram positive bacteria mainly are soil residing. The phylum Actinobacteria represents one of the largest taxonomic positions among the 18 major domain Bacteria. It includes 5 subclasses and 14 suborders [8]. Actinobacteria reveal a wide range of morphologies; coccoid (Micrococcus) or oval (Arthrobacter) to fragmented hyphae (Nocardia) or highly differentiated branched mycelium (Streptomyces)). Various lifestyles of Actinobacteria are also notable such as the phylum includes pathogens (Mycobacterium, Nocardia, Tropherymaspp, Corynebacterium, and Propionibacterium), soil inhabitants (Streptomyces)), plant commensals (Leifsonia), nitrogen-fixing symbionts (Frankia) and gastrointestinal tract (GIT) inhabitants (Bifidobacterium). The divergence of action-bacteria from any other group of bacteria is so primordial that it is not possible to identify the phylogenetically neighbouring bacterial group to Actinobacteria with assurance [9]. Most Actino-bacteria are aerobic, but there are exceptions. They can be heterotrophic or chemoautotrophic, though most are chemo-heterotrophic and able to use a wide variety of nutritional sources, including various complex polysaccharides [10]. The Actinobacteria are rich in various metabolites important for human as sources of novel antibiotics, anti-fungal, anticancer agents, and other secondary metabolites that might be used in medicine or to improve plant growth and resistance to diseases. Actinobacteria are also very promising for bio-control of pests and as plant growth promoters (Figure 1). Keeping in mind the emerging growth in genomics, molecular biology and advanced instrumentation with the strong requirement for new bioactive molecules, the Actinobacteria is a highly dynamic research field.
Endophytic Actinobacteria and Future Possibilities
Traditional isolation of microorganisms from ecosystems, like soil and water results re-isolation of same species and metabolites. Endophytic microorganisms are defined as, microbes, that colonize living, internal tissues of plant without causing any immediate overt negative effect. Endophytic actinomycetes are relatively unexplored and now it is well documented to be a potential source of novel bioactive products.
Several secondary metabolites have been discovered from the endophytic actinomycetes from time to time. During the long co-evolution of endophytic microbes and host plants, they have adapted themselves to the distinctive microenvironment by genetic optimization; including recombination with plant DNA. This phenomenon may lead to some sort of recombination in endophyte’s genome and the ability of certain endophytes to biosynthesize some new class bioactive compound. It is believed that genetic recombination between endophytes and hosts that might be occurred in evolutionary timescale could result in some endophytes which would produce certain phytochemicals that were originally characteristic of the hosts [11]. Another important thing is, as the endophytic microorganisms are associated with eukaryotic organisms there will be minimum chance of getting any toxic products from them. This idea is the major concern of drug discovery, relating to the toxicity of a drug (Table 1).
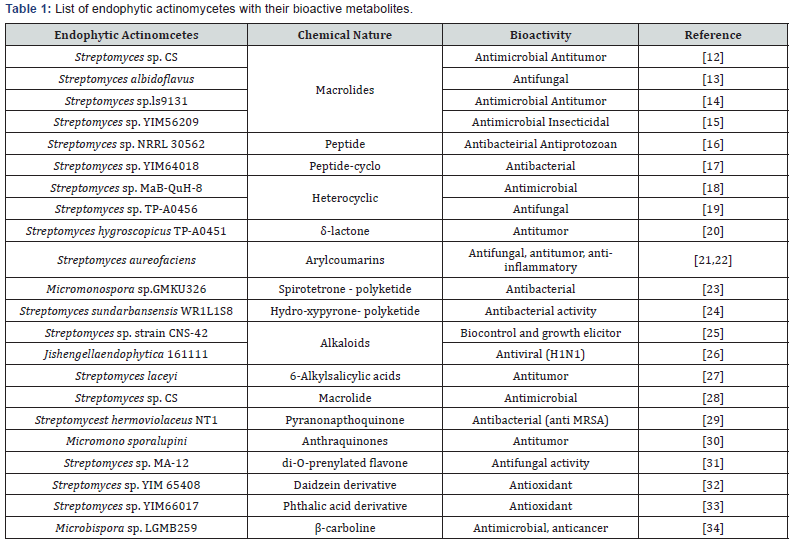
Based on this widely accepted hypothesis, there has been increasing research efforts focused on screening for novel natural products from endophytes. Actino-bacteria possess gene cluster for seconded metabolites and are frequently isolated as endophytes of all types of plants. Such microbes are being studied elaborately for their ecological as well bioactive potential to meet the basic evolutionary knowledge, ecological importance and serious global demands of pharmaceuticals. With the belief that endophytes belong to each plants there are huge scope for exploration of such group of bacteria as future source of novel natural products. Endophytic Actinobacteria are a promising source of new and interesting natural products that may have value in agricultural research and drug discovery programs [12-34].
References
- Wainwright M (1989) Moulds in ancient and more recent medicine. Mycologist 3(1): 21-23.
- Saxon W (1999) Anne Miller 90 first patient who was saved by penicillin. New York Times. New York, USA, Retrieved 29 August 2014.
- Stone JK, Polishook JD, White JF (2004) Endophytic fungi. In: Mueller GM, Bills GF, Foster MS (Eds.), Biodiversity of fungi. Inventory and monitoring methods, Elsevier Academic Press, USA, pp. 241-270.
- Carroll GC (1988) Fungal endophytes in stems and leaves: from latent pathogen to mutualistic symbiont. Ecology 69(1): 2-9.
- Gasoni L, deGurfinkel BS (1997) The endophyte cladorrhinum foecundissimum in cotton roots: phosphorus uptake and host growth. Mycological Res 101(7): 867-870.
- Raps A, Vidal S (1998) Indirect effect of an unspecialized endophtryic fungus on specialized plant-herbivorus insect interaction. Oecologia 114(4): 541-547.
- Zhang HW, Song YC, Tan RX (2006) Biology and chemistry of endophytes. Nat Prod Rep 23(5): 753-771.
- Stackebrandt SP (2000) The prokaryotes: an evolving electronic resource for the microbiological community. Springer-Verlag, New York, USA.
- Ventura MC, Canchaya A, Tauch G, Chandra G, Fitzgerald GF, et al. (2007) Genomics of Actinobacteria: tracing the evolutionary history of an ancient phylum. Microboil Mol Biol Rev 71(3): 495-548.
- Backhed F, Ley RE, Sonnenburg JL, Peterson DA, Gordon JI (2005) Hostbacterial mutualism in the human intestine. Science 307(5717): 1915- 1920.
- Germaine K, Keogh E, Garcia-Cabellos G, Borremans B, Lelie D, et al. (2004) Colonisation of poplar trees by GFP expressing bacterial endophytes. FEMS Microbiology Ecol 48(1): 109-118.
- Lu C, Shen Y (2003) A new macrolide antibiotic with antitumor activity produced by treptomyces) sp. CS, a commensal microbe of Maytenus hookeri. The Journal of Antibiotics 56(4): 415-418.
- Yan LL, Han NN, Zhang YQ, Yu LY, Chen J, et al. (2010) Antimycin A18 produced by an endophytic treptomyces) albidoflavus isolated from a mangrove plant. J Antibiot 63(5): 259-261.
- Zhao PJ, Fan LM, Li GH, Zhu N, Shen YM (2005) Antibacterial and antitumor macrolides from treptomyces) sp. ls9131. Arch Pharm Res 28(11): 1228-1232.
- Yu Z, Zhao LX, Jiang CL, Duan Y, Wong L, et al. (2011) Bafilomycins produced by an endophytic actinomycete treptomyces) sp. YIM56209. J Antibiot 64(1): 159-162
- Castillo UF, Strobel GA, Ford EJ, Hess WM, Porter H, et al. (2002) Munumbicins, wide spectrum antibiotics produced by treptomyces) munumbi, endophytic on Kennedianigricans. Microbiology 148(Pt 9): 2675-2685.
- Yang X, Peng T, Yang Y, Li W, Xiong J, et al. (2015) Antimicrobial and antioxidant activities of a new benzamide from endophytic treptomyces) sp. YIM 67086. Nat Prod Res 29(4): 331-335.
- Pullen C, Schmitz P, Meurer K, Bamberg DD, Lohmann S, et al. (2002) New and bioactive compounds from treptomyces) strains residing in the wood of Celastraceae. Planta 216(1): 162-167.
- Igarashi Y (2004) Screening of novel bioactive compounds from plantassociated actinomycetes. Actinomycetologica 18(2): 63-66.
- Igarashi Y, Miura SS, Fujita T, Furumai T (2006) Pterocidin, a cytotoxic compound from the endophytic treptomyces) hygroscopicus. J Antibiot 59(3): 193-195.
- Taechowisan T, Lu C, Shen Y, Lumyong S (2005) 4-arylcoumarins from endophytic treptomyces) aureofaciens CMUAc130 and their antifungal activity. Annals of Microbiolology 55: 63-66.
- Taechowisan T, Lu C, Shen Y, Lumyong S (2007) Antitumor activity of 4-arylcoumarins from endophytic treptomyces) aureofaciens CMUAc130. Journal of Cancer Research and Therapeutics 3: 86-91.
- Igarashi Y, Ogura H, Furihata K, Oku N, Indananda C, et al. (2011) Maklamicin, an antibacterial polyketide from an endophytic Micromonosporasp. J Nat Prod 74(4): 670-674.
- Djinni I, Defant A, Kecha M, Mancini I (2014) Metabolite profile of marine-derived endophytic treptomyces) sundarbansensis WR1L1S8 by liquid chromatographymass spectrometry and evaluation of culture conditions on antibacterial activity and mycelial growth. J Appl Microbiol 116(1): 39-50.
- Li X, Huang P, Wang Q, Xiao L, Liu M, et al. (2014) Staurosporine from the endophytic treptomyces) sp. strain CNS-42 acts as a potential biocontrol agent and growth elicitor in cucumber. Antonie Van Leeuwenhoek 106(3): 515-525.
- Wang P, Kong F, Wei J, Wang Y, Wang W, et al. (2014) Alkaloids from the Mangrove-Derived Actinomycete Jishengella endophytica 161111. Mar Drugs 12(1): 477-490.
- Kim N, Shin JC, Kim W, Hwang BY, Kim BS, et al. (2006) Cytotoxic 6 alkylsalicylic acids from the endophytic treptomyces) laceyi. J Anti Biot 59(12): 797-800.
- Lu C, Shen Y (2007) A novel ansamycin, naphthomycin K from treptomyces) sp. J Antibiot 60(10): 649-653.
- Roy S, Banerjee D (2015) Broad spectrum antibacterial activity of granaticinic acid, isolated from treptomyces) thermoviolaceus NT1; an endophyte in Catharanthusroseus(L.) G. Don. J Appl Pharm Sci 5(7): 6-11.
- Igarashi Y, Trujillo ME, Martínez-Molina E, Yanase S, Miyanaga S, et al. (2007) Antitumor anthraquinones from an endophytic actinomycete Micromonospora lupine sp. nov. Bioorg Med Chem Lett 17(13): 3702- 3705.
- Ding WJ, Zhang SQ, Wang JH, Lin YX, Liang QX, et al. (2013) A new di- O-prenylated flavone from an actinomycetetreptomyces) sp. MA-12. J Asian Nat Prod Res 15(2): 209-214.
- Yang X, Yang Y, Peng T, Yang F, Zhou H, et al. (2013) A new cyclopeptide from endophytic treptomyces) sp. YIM 64018. Nat Prod Commun 8(12): 1753-1754.
- Zhou H, Yang Y, Peng T, Li W, Zhao L, et al. (2014) Metabolites of treptomyces) sp., an endophytic actinomycete from Alpinia oxyphylla. Nat Prod Res 28(4): 265-267.
- Savi DC, Haminiuk CWI, Sora GTS, Adamoski DM, Kenski J, et al. (2015) Antitumor, antioxidant and antibacterial activities of secondary metabolites extracted by endophytic actinomycetes isolated from Vochysi adivergens. International Journal of Pharmaceutical 5(1): 347- 356.






























