Impact of Aerobicity on Mollicute Colony Growth on Agar
Shayn Armstrong1*, Jamie Strough1 and Raymond Nims2
1Bionique Testing Laboratories Inc, USA
2RMC Pharmaceutical Solutions Inc, USA
Submission: November 25, 2017; Published: February 07, 2018
*Corresponding author: Shayn Armstrong, Bionique Testing Laboratories, Inc., 156 Fay Brook Drive, Saranac Lake, NY 12983, USA.
How to cite this article: Shayn A, Jamie S, Raymond N. Impact of Aerobicity on Mollicute Colony Growth on Agar. Adv Biotech & Micro. 2018; 8(2): 555733. DOI: 10.19080/AIBM.2018.08.555733
Abstract
The impact of aerobicity (oxygen concentration in the incubation atmosphere) on colony growth of various mollicutes (mycoplasmas, acholeplasmas, spiroplasmas, etc.) on agar was investigated in order to confirm the optimal oxygen content for colony growth. The organisms evaluated included known cell culture contaminants and positive control organisms typically employed in compendial mycoplasma detection assays (Acholeplasma laidlawii, Mycoplasma arginini, M. galliseptum, M. hyorhinis, M. orale, M. pneumoniae, M. salivarium, and M. synoviae). The incubation atmospheres evaluated included microaerophilic (95% N2, 5% CO2), aerobic (21% O2, 5% CO2, 74% N2 or ambient/room air), and strictly anaerobic (<0.1% O2). Colony growth on commercial fortified agar or heart infusion agar was evaluated after a 14-day incubation in the specified atmosphere. For M. arginini, M. pneumoniae, M. galliseptum, A. laidlawii, and M. synoviae, equivalent growth occurred under oxygenated, microaerophilic, and strictly anaerobic conditions. For M. orale, M. salivarium, and M. fermentans, low oxygen atmospheres favored colony growth. M. hyorhinis did not grow well under strictly anaerobic conditions, but grew equally well under the aerobic and microaerophilic atmospheres. The results of the prospective study described above generally agree with our experience in recovery of positive control organisms under ambient vs. microaerophilic atmospheres, with the one exception that M. pneumoniae positive controls tested in the past displayed higher recovery under microaerophilic than ambient conditions. The frequency of contaminant isolation from actual test samples has, over the period 2002 to present, been higher under microaerophilic than aerobic conditions.
Keywords: Acholeplasma; Aerobic; Aerobicity; Anaerobic; Microaerophilic; Mycoplasma
Introduction
Mycoplasma detection assays employing the direct culture (broth and agar) approach are described in various regulations and compendia (e.g., 21 CFR 610.30 [1], United States Pharmacopeia <63> [2], United States FDA 1993 Points to Consider [3], and European Pharmacopoeia 2.6.7 [4]). The earliest approach (21 CFR 610.30) specified incubation under both aerobic and anaerobic conditions, while the other approaches specified incubation under low oxygen (microaerophilic) conditions only. The decision as early as 1993 to use only microaerophilic conditions was based on empirical studies conducted by the FDA demonstrating that mollicute (e.g., mycoplasma, acholeplasma, spiroplasma, ureaplasma) growth in broth and agar under such conditions was approximately equivalent to aerobic and therefore no significant advantage was conferred by using both incubation conditions [5].
The literature on this topic is not extensive, and if a number of empirical studies were indeed performed to determine optimal incubation conditions, these may not have been published. Gardella & DelGiudice [6] articulated in 1983 the difficulties in establishing the optimal oxygen requirements of mollicutes in view of various associated factors that were not at that time typically controllable as isolated variables (pH, growth medium, redox potential of medium components, and humidity). Further, these authors recognized that the requirements for primary isolation of mollicutes from tissue samples might differ from the requirements for cultivation and subculture of organisms. For instance, isolation was viewed as optimal in an anaerobic environment, while subculture of acholeplasmas, mycoplasmas, and ureaplasmas was thought to be favored in an aerobic (albeit low oxygen) environment such as 5% CO2 in nitrogen or hydrogen [5]. Similar findings were reported for laboratory strains of M. hyorhinis by Polak-Voglezang et al. [7] in 1983. These authors demonstrated the inability to isolate this species or to subcultivate laboratory adapted strains in strictly anaerobic atmospheres [7].
With the availability of defined cultivation media and ability to closely control temperature and gas composition, it is now possible to evaluate the impact of atmosphere on colony growth for various mollicutes in a manner which perhaps was not possible in the late 1900’s. While the isolation of wild type organism from animal tissues is not of interest to our testing laboratory, the question of the atmospheric requirements for subcultivation of wild type vs. laboratory-adapted mollicute species is of greater interest. The requirements for cultivation of laboratory-adapted species are most easily addressed, as such organisms are available for use as positive controls for routine mycoplasma detection assays. We therefore have concentrated our initial efforts toward characterizing the growth of laboratory-adapted acholeplasmas and mycoplasmas in various atmospheres. These included two aerobic atmospheres (ambient or room air, and a defined atmosphere of 21% O2, 5% CO2, 74% N2) and two anaerobic atmospheres (microaerophilic and strictly anaerobic). The question of isolation of contaminants from cell culture samples is addressed in less of a systematic manner by providing information on frequency of isolation on agar incubated under ambient vs. microaerophilic atmospheres.
Materials and Methods
Mollicutes
Laboratory-adapted mollicute species were obtained initially from the American Type Culture Collection (Manassas, VA). These included A. laidlawii (ATCC #23206), M. arginini (ATCC #23838), M. fermentans (ATCC #19989), M. galliseptum (ATCC #19610), M. hyorhinis (ATCC #23839), M. orale (ATCC #23714), M. pneumoniae (ATCC #29085), M. salivarium (ATCC #23064), and M. synoviae (ATCC #25204). Parental stocks were prepared and were used to prepare low titer working stocks at Bionique.
Media and gases
Fortified commercial (FC) and heart infusion (HI) agar plates were formulated, poured, and growth promotion tested at Bionique per proprietary recipes. Bottled gas mixtures (5% CO2, 95% N2; 5% CO2, 21% O2, 74% N2) were obtained from Airgas (Plattsburgh, NY). BD GasPak™ EZ Gas Generating Container System catalytic packages were obtained from Becton, Dickinson and Company (Sparks, MD).
Prospective atmosphere impact study
Working stocks of the various mollicute species were inoculated onto agar plates at 0.1mL per plate, with the targeted inocula being <100 colony forming units (CFU) per plate. Triplicate plates were inoculated per treatment group, and the resulting plates were incubated for 14 days at 36±1 °C in one of the following atmospheric conditions:
A. In Plas-Lab chambers (805 LCSP/117, Clear Acrylic Lab Chamber; Lansing, MI) purged with 95% N2/5% CO2 (microaerophilic) every three days
B. In Plas-Lab boxes purged with 21% O2, 5% CO2, 74% N2 (defined aerobic) every three days
C. In sealed plastic bags (aerobic, ambient air)
D. In Plas-Lab boxes initially purged with 95% N2/5% CO2 down to <0.1% oxygen and then maintained at or below this level with oxygen-depleting GasPak catalytic packages (strictly anaerobic).
During the course of incubation, oxygen levels were monitored in the various chambers using portable oxygen monitors (Dräger Pac 3500 O2 Detector; Dräger, Telford, PA). After 14 days of incubation, the plates were scored for mycoplasma colony counts and the results were recorded. Three trials were performed using similar methodologies. The second trial was conducted to confirm the results of the initial trial, and to include two additional species (M. galliseptum and M. synoviae). The third trial was conducted to confirm results obtained for M. hyorhinis and M. salivarium in the initial two trials).
Historical positive control database
Positive control cultures are routinely included in mycoplasma detection assays conducted at Bionique to enable assessment of system suitability. For certain assay types, these positive controls include M. orale, M. pneumoniae, M. hyorhinis, M. arginini, and M. salivarium, and both aerobic (sealed plastic bags) and microaerophilic (chambers purged with 95% N2/5% CO2) incubations. For a subset of these organisms (i.e., M. pneumoniae and M. salivarium) two agar types (FC and HI) are inoculated for each atmosphere, allowing the impact of agar type on atmosphere preference to be assessed. Colony counts for the various controls are normalized to the CFU inoculated to provide % recovery values. The % recovery following incubation under aerobic conditions vs. microaerophilic conditions in one or more agar types may therefore be compared to assess impact of incubation atmosphere and medium.
Historical positive sample isolation database
Testing data from 2002 to the present were mined to determine the relative frequencies of detection of mycoplasma in test samples from agar plates incubated under microaerophilic vs. aerobic (sealed plastic bags) conditions. These isolates were not submitted for species determination.
Statistical analyses
Colony counts from the various treatment groups of the prospective atmosphere impact study were analyzed by singlefactor ANOVA. Where significant differences (p<0.05) were noted in the ANOVA, two-tailed t-tests were performed to determine significant differences (p<0.05) between specific treatment groups. For the historical positive control data, raw colony counts were expressed relative to the CFU inoculated to obtain % recovery values. The % recovery values for positive control organisms incubated in aerobic vs. microaerophilic atmospheres and on FC vs. HI agar were compared using single-factor ANOVA. Where significant differences (p<0.05) were noted in the ANOVA, two-tailed t-tests were performed to determine significant differences (p<0.05) between specific treatment groups. The statistical significance of differences in mycoplasma detection frequency data was determined by Fisher Exact test.
Results
Prospective atmosphere impact study
On the basis of impact on colony growth of the incubation atmospheres evaluated, the mollicute species can be divided into two groups. The first includes species for which no impact was detected (Table 1). These species included M. pneumoniae, M. arginini, M gallisepticum, and M. synoviae. No significant differences in colony growth were observed for these species when incubated for 14 days in aerobic, anaerobic, or microaerophilic atmospheres.
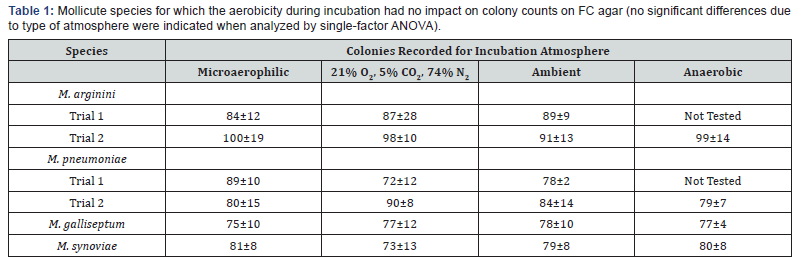
The second group includes those species for which significant differences were observed among one or more of the atmospheres evaluated (Table 2). These included A. laidlawii, M. hyorhinis, M. orale, M. salivarium, and M. fermentans. In the case of A. laidlawii, a significant difference (p<0.05) in colony growth between the ambient (room air) atmosphere and the microaerophilic atmosphere was detected in the second trial, but a significant difference was not observed for these conditions in the initial trial performed (Table 2). The biological significance of the colony count difference between these two conditions is likely minimal.
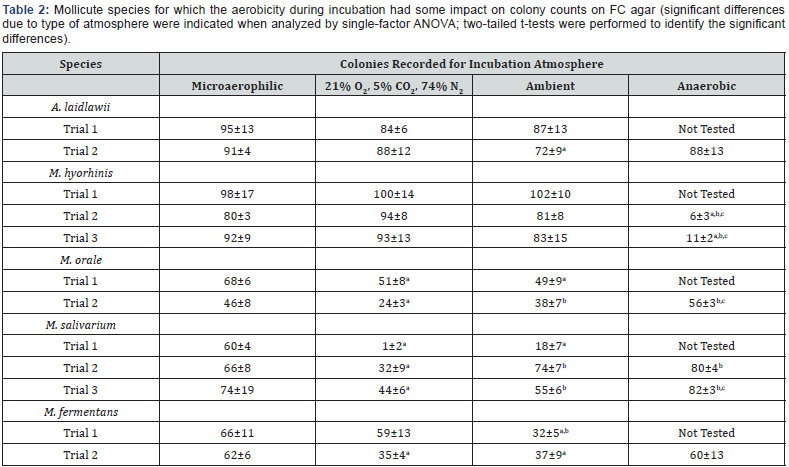
In the case of M. orale and M. fermentans, the aerobic incubations resulted in lower colony counts relative to the microaerophilic or anaerobic conditions. These differences were, in some cases, statistically significant (p<0.05) (Table 2). For M. salivarium, somewhat discrepant results were obtained between trials 1 and 2. In the initial trial, both aerobic conditions resulted in significantly (p<0.05) fewer colonies relative to the microaerophilic condition, with the greatest reduction occurring for the 21% O2, 5% CO2, 74% N2 atmosphere. In the second trial, the colony counts for the 21% O2, 5% CO2, 74% N2 group were significantly lower than the other atmospheres. A third trial was therefore performed in order to clarify the discrepancy in results. The results of this trial confirmed that the 21% O2, 5% CO2, 74% N2 atmosphere yielded the lowest colony counts, in certain cases significantly lower (p<0.05). Taken together, the three trials for this species indicated that the anaerobic and microaerophilic atmospheres reproducibly resulted in similar colony counts, while the aerobic atmospheres resulted in variable and generally lower colony counts (Table 2).
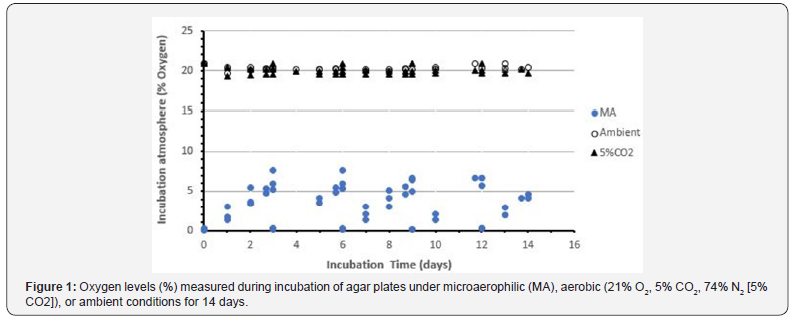
The oxygen levels achieved during the various aerobic and microaerophilic atmospheres were monitored continuously in trials 1 and 2 using an oxygen meter placed in the incubation chambers/sealed bags. The oxygen levels are displayed in Figure 1. Only the microaerophilic atmosphere was monitored during the third trial, since the trial-1 and -2 data indicated that oxygen levels were routinely maintained in the range of 19-22% in the aerobic conditions. The microaerophilic atmosphere ranged from <0.1% to ~8% oxygen. This range reflects the low oxygen levels achieved upon purging with 95% N2/5% CO2 every three days, and the subsequent linear rise in oxygen content due to leakage of room oxygen into the chambers (Figure 1). The effectiveness of the GasPak catalytic packages for maintaining a strictly anaerobic state in the anaerobic chamber was verified by placing an oxygen meter in the chamber for a few days. Oxygen levels achieved and maintained were lower than 0.1% (the limit of detection of the oxygen meters) (data not shown).
Historical positive control database
Table 3 displays the recovery of the expected inoculated positive control CFU under microaerophilic vs. ambient (room air) atmospheres during routine mycoplasma testing conducted from 2012 to 2013. Colony counts observed in agar plates were normalized to the numbers of CFU inoculated (determined from the inoculation volume and the positive control stock titer) in order to obtain % recovery values that could be directly compared among positive control species (M. orale, M. pneumoniae, M. arginini, M. hyorhinis, M. salivarium, and A. laidlawii) and atmospheres (microaerophilic vs. ambient). The ratios of recovery in microaerophilic vs. aerobic atmosphere were ~1.0 for M. hyorhinis, M. arginini, and A. laidlawii, and the minor differences in absolute recoveries in the two atmospheres for these species were not statistically significant. In the cases of M. orale, M. pneumoniae, and M. salivarium, absolute recovery values were significantly different (p< 0.05) between microaerophilic and ambient atmospheres, with recovery ratios (recoverymicroaerophilic/recoveryambient) of 2.4, 1.4, and 1.3 for these three species, respectively (Table 3).
A similar analysis was performed for M. pneumoniae and M. salivarium, with data mined from assays utilizing these two species in both FC and HI agar (Table 4). These more recent testing data were analyzed to confirm that the atmosphere preferences observed in Table 3 were not unique to the FC agar type alone. For M. salivarium, the percent recoveries under microaerophilic conditions were significantly (p<0.05) different from the recoveries under ambient conditions. The recovery of the expected inoculated positive control CFU in the two atmospheres differed, with the ratio (recoverymicroaerophilic/ recoveryambient) calculated as 1.3 on FC agar and 1.5 on HI agar. This dataset did not indicate a preference for atmosphere for M. pneumoniae in either agar type (Table 4). Both species displayed a preference for FC agar over HI agar, regardless of incubation atmosphere.
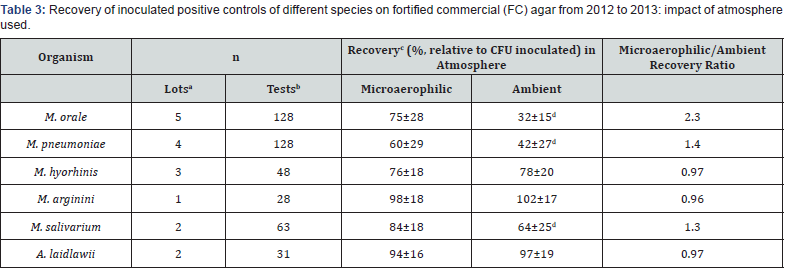
aPositive control stock lots used
b Individual tests in which positive controls were inoculated
cMean±standard deviation, target inoculum based on stock titer was less than 100 CFU per plate
d Significantly different than microaerophilic recovery (p<0.05)
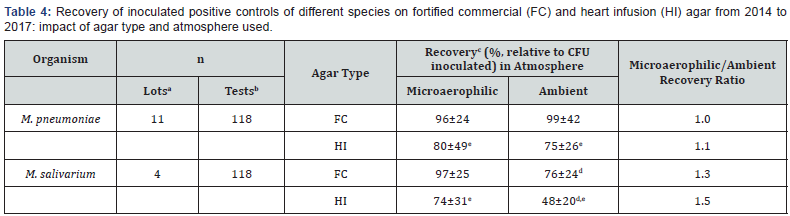
aPositive control stock lots used
b Individual tests in which positive controls were inoculated
cMean±standard deviation, target inoculum based on stock titer was less than 100 CFU per plate
d Significantly different than microaerophilic recovery (p<0.05)
e Significantly different from corresponding FC agar condition (p<0.05)
Historical positive sample isolation database
The frequency of isolation of mycoplasma from test samples incubated under microaerophilic vs. ambient atmospheres is shown in Table 5. Over the time period 2002-2008, a greater number of isolates were recovered from samples, compared to the period from 2009-2017. The ratio (1.1) of isolates recovered under microaerophilic vs. ambient incubation conditions was the same over both time periods. Regardless of time period evaluated, the frequency of isolation under microaerophilic conditions was statistically greater (p<0.05) than that under ambient conditions.
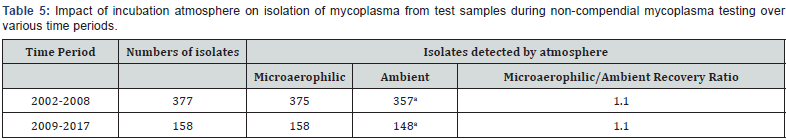
aSignificantly different from microaerophilic value (p<0.05) by Fisher Exact test.
Discussion
The simplification of the test design with respect to incubation atmosphere in the 1993 Points to Consider mycoplasma method [3] resulted from studies performed at the FDA. The results of these studies were not reported in the literature, although the comment was made in Chandler et al. [5] that “The Mycoplasma Laboratory at FDA had evaluated the relative recovery of cell culture contaminants using anaerobic vs. aerobic conditions and found that increased recovery of cell culture contaminants using both aerobic and anaerobic incubation was minimal compared to anaerobic incubation alone, as also seen in other laboratories…”. Since the FDA data were not published, one must go to the cited papers [6-9] and others from that time period [e.g. 10] to understand the rationale for the decision to use only the anaerobic (95% N2, 5% CO2, referred to here as microaerophilic so as not to confuse with strictly anaerobic conditions achieved using GasPak packages) atmosphere in the 1993 Points to Consider test. The assessment of optimal incubation atmosphere by comparing the detection of culture contaminants in one vs. the other atmosphere was utilized in these studies [6-10].
It should be acknowledged that much attention was placed, in previous studies reported in the literature, on the optimal requirements for isolation of mycoplasma from cell cultures or animal tissues and less on the requirements for routine subcultivation of lab-adapted mycoplasmas. Our investigation, described above, has evaluated the incubation atmosphere requirements for sub cultivation of lab-adapted mollicute species, in what appears to be the first study designed prospectively to address this topic. We also have evaluated the atmosphere requirements for isolation of mollicutes directly from test samples.
Subcultivation of lab-adapted mollicutes
For M. arginini, M. pneumoniae, M. galliseptum, A. laidlawii, and M. synoviae, equivalent growth occurred under ambient, microaerophilic, and strictly anaerobic conditions in our prospective study. For M. orale, M. salivarium, and M. fermentans, low oxygen atmospheres favored colony growth. This result is in agreement with our positive control recovery experience and with the opinion of other investigators [6,8]. M. hyorhinis did not grow well under strictly anaerobic conditions, but grew equally well under the aerobic and microaerophilic atmospheres. The inability to cultivate M. hyorhinis under strictly anaerobic conditions was reported previously by Polak-Vogelzang et al. [7]
The results of our prospective study agree with our experience in recovery of positive control organisms under ambient vs. microaerophilic atmospheres, with the one exception that M. pneumoniae positive controls have in the past (2012 to 2013) been recovered at significantly greater levels under microaerophilic than ambient conditions. This preference for microaerophilic atmosphere by M. pneumoniae has not, however, been observed in more recent assays (2014 to 2017). The reason for this discrepancy is not clear.
Isolation of mollicutes from test samples
Gardella & DelGiudice [6] reported that isolation from animal, plant, and insect tissues was favored by strictly anaerobic conditions. For isolation from primary or continuous cell cultures, the consensus view of the time appeared to be that a strictly anaerobic or microaerophilic atmosphere was optimal in the case of M. hyorhinis, M. orale, M. fermentans, and M. hominis [6,8,9]. Isolation of A. laidlawii and M. arginini from cell cultures under microaerophilic vs. aerobic conditions was found to be equivalent [6,9]. Gabridge & Lundin [10] reported that they were able to isolate cell culture contaminants more often under aerobic (49/60 isolates) than anaerobic (42/60 isolates) conditions, though the difference in detection frequency is not statistically significant by Fisher Exact test.
Our results on isolation of contaminants from cell culture and other samples is in agreement with those described above for the previous investigators. Over the time period from 2002 to present, we found that the frequency of isolation under microaerophilic conditions (533/535 isolates in total) slightly exceeded the frequency under aerobic conditions (505/535 isolates in total). The ratio of isolation under microaerophilic conditions to that under aerobic conditions was found to be 1.1 in our detection data analysis. This ratio likely reflects the fact that these isolates represented a mix of mollicute species for which microaerophilic conditions favored growth and other species for which approximately equivalent growth would be expected regardless of atmosphere. The isolation results suggest that our subcultivation results with lab-adapted mollicute species may be considered to be applicable also to wild-type species that might contaminate cell cultures.
Conclusion
Taken together, our results indicate that the decision to use only microaerophilic conditions in the 1993 Points to Consider mycoplasma test [3], and subsequently in the European Pharmacopoeia 2.6.7 [4] and USP <63> [2] compendial mycoplasma tests, is supported by empirical data derived from both prospective as well as historical positive control recovery and mycoplasma isolation data from our own and various other laboratories. While it is not possible to extrapolate our data to all mollicute species, our data include most of the mollicutes of concern [11] from a cell culture contamination point of view.
References
- (1998) United States Code of Federal Regulations Title 21 Part 610.30 Test for mycoplasmas, 1973, amended in 1998.
- United States Pharmacopeia <63> Mycoplasma tests.
- United States FDA (1993) Points to Consider in the characterization of cell lines used to produce biologicals.
- European Pharmacopoeia 2.6.7 Mycoplasmas.
- Chandler DKF, Volokhov DV, Chizhikov VE (2011) Historical overview of mycoplasma testing for production of biologics. Am Pharm Rev 14: 2011.
- Gardella RS, DelGiudice RA (1983) Optimal temperature and atmospheric conditions for growth. In: Razin S (Ed.), Methods in Mycoplasmology, Volume 1, Mycoplasma Characterization, Academic Press, pp. 211-215.
- Polak-Vogelzang AA, de Haan HH, Borst J (1983) Comparison of various atmospheric conditions for isolation and subcultivation of Mycoplasma hyorhinis from cell cultures. Antonie van Leewenhoek 49(1): 31-40.
- McGarrity GJ, Sarama J, Vanaman V (1979) Factors influencing microbiological assay of cell-culture mycoplasmas. In Vitro 15: 73-81.
- Polak-Vogelzang AA, Brugman J, Reijgers R (1987) Comparison of two methods for detection of mollicutes (mycoplasmatales and acholeplasmatales) in cell cultures in The Netherlands. Antonie van Leeuwenhoek 53: 107-118.
- Gabridge MG, Lundin DJ (1990) Optimal culture conditions for detection and isolation of mycoplasma contaminants from cell cultures. In: Stanek G, Cassell GH, Tully JG, Whitcomb RF, (Eds.), Recent Advances in Mycoplasmology. Stuttgart, Gustav Fischer Verlag, pp. 943-944.
- Drexler HG, Uphoff CC (2002) Mycoplasma contamination of cell cultures: incidence, sources, effects, detection, elimination, prevention. Cytotechnology 39: 75-90.






























