Enhanced Acidogenesis by the Degenerated Clostridium Sp. Strain on a Continuous Membrane Cell Recycle Reactor
Hiral S, Abhishek M, Aruna GA, Annamma AO* and Arvind Lali
DBT-ICT Centre for Energy Biosciences, Institute of Chemical Technology (Formerly UDCT), India
Submission: July 13, 2017; Published: November 28, 2017
*Corresponding author: Annamma AO, DBT-ICT Centre for Energy Biosciences Institute of Chemical Technology (Formerly UDCT), Nathalal Parekh Marg, Mumbai-400019, India, Tel: 02233612310; Fax: 02233611020; Email: a.dbtceb@gmail.com
How to cite this article: Hiral S, Abhishek M, Aruna G, Annamma A, Arvind L. Enhanced Acidogenesis by the Degenerated Clostridium Sp. Strain on a Continuous Membrane Cell Recycle Reactor. Adv Biotech & Micro. 2017; 7(4): 555716. DOI: 10.19080/AIBM.2017.07.555716.
Abstract
Microbial organic acids production is being extensively studied to achieve success at industrial scale. Commercial organic acid production employs homofermentative microbes while the use of heterofermentative organisms has been restricted due to expensive downstream processes. With development on appropriate inexpensive separation technology for separation of a mixture of organic acids, heterofermentative organic acid fermentation may soon become feasible at large scale. Present work aims at enhancing organic acid production by the degenerated strain of Clostridium acetobutylicum ATCC4259. At batch mode, pH 6 resulted in the highest total organic acid concentration of 10.22g.L-1 and total organic acid productivity of 0.58g.L-1.h-1. The constant specific productivity of 1.1g.g-1.h-1 obtained for total organic acid at different pH values proved that total organic acid productivity is not governed by pH of the medium but is dependent on cell growth. However, the metabolic shifts in the cell were proved to be subject to external pH values. Substrate kinetics revealed 30-50g.L-1 of glucose concentration as optimum for acidogenesis.The optimization of continuous cell recycle fermentations at different dilution rates and different glucose concentrations positively impacted the total organic acid productivity and complete glucose utilization and resulted in the highest total organic acid productivity of 3.6±0.05g.L-1.h-1 at an optimum dilution rate of 0.16h-1.
Keywords: Cell recycle; Clostridium; Organic acids
Introduction
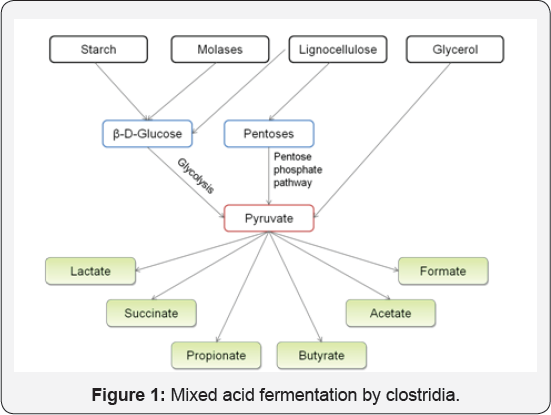
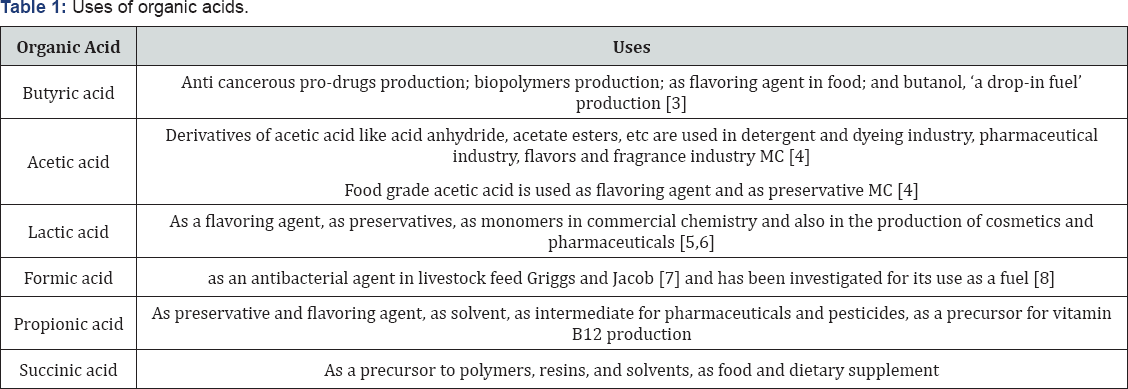
Clostridia belong to firmicutes which is a vast phylum of saprophytes with the ability to produce endospore, resist desiccation and survive in extreme environments [1]. Their exceptional properties are exemplified by their versatility in substrates utilization and their diversity in product formation. Heterofermentative Clostridium species are known to produce a mixture of organic acids in the first phase of growth (acidogenesis) and a mixture of solvents in the second phase (solventogenesis). As depicted in Figure 1, butyric acid, acetic acid, lactic acid, formic acid, etc. are the major organic acids produced by species of Clostridium during acidogenesis [2]. The potential to produce organic acids as intermediates makes clostridia a bacterium of great importance for the commercial production of organic acids. These organic acids are then used as precursors to various essential commodity chemicals (Table 1) (Figure 1).
For a bioprocess, diversion of significant carbon sources towards product formation is a major challenge for any biobased technology. Reports are available for the vast research done using clostridial strains in terms of the study of factors affecting the production of organic acids, strain improvement, process optimizations, in situ product removal, etc. for organic acid production at the industrial scale [9,10]. These reports target independent objectives like 'butyric acid production' or 'acetic acid production' while lack a detailed investigation on by-products formed eg. The maximum butyric acid yield of 0.49g.g-1 of glucose consumed by Clostridium thermobutyricum has been reported by Liang Wang et al. [10] while total product (organic acid) yield is 0.52g.g-1 of glucose consumed [10]. This figure for the yield of total organic acids proves loss of nearly 48% of available carbon during product formation. For organic acids production with a market price slightly higher than sugar, to become economically viable, an efficient conversion of the maximum amount of available carbon to organic acids is a prerequisite. Apart from the yield of the organic acid, the expense of downstream processes too dictates the feasibility of the bioprocess at industrial scale. Clostridial species being heterofermentors have never been encouraged for use at industrial scale. However, development of appropriate inexpensive separation technology for separation of a mixture of organic acids, heterofermentative organic acid fermentation may soon become feasible at large scale. This kind of development may demand research on mixed-acid fermentations by clostridial strains to achieve maximum yield and productivity.
The interdependency of acidogenesis with physicochemical conditions like pH of the medium and glucose concentration opens up new strategies for redesigning clostridial fermentations and thereby improving the overall productivities. The influence of pH on growth and product profile of the bacteria has been signified by the decrease in specific growth rate from 0.86h-1 to 0.20h-1 along with three times decrease in total organic acid content for Clostridium sporogenes 3121 when transferred from pH value of 6.50 to 5.00 [11]. Moreover, Clostridium propionicum has been reported to exhibit a change in propionic acid to acetic acid ratio from 0.45: 1 to 1.8: 1 when grown at pH values of 7 and 6 respectively [12]. Besides pH values, the medium constituents and substrate concentration have a noteworthy effect on organism's behavior in a controlled environment as well as on economy of the bioprocess [13]. With higher initial concentrations of substrate, higher titers of products are formed that aid in downstream processing. However, for every microbe, a threshold of substrate concentration exists, beyond which the substrate itself becomes inhibitory due to osmotic dehydration of the microbe. This phenomenon has been well illustrated during fermentation by C. tyrobutyricum CNRZ 596 wherein, the titers of organic acids produced increased with increasing initial glucose concentration but the productivity of organic acids decreased beyond 80g.L-1 of glucose concentrations used due to longer initial lag [14]. On similar lines, the increase in the doubling time for Clostridium thermobutyricum with the use of glucoseconcentration more than 100g.L-1 [15] supports the fact of substrate inhibition.
An industrially viable process demands sufficiently high productivity. Reports on the use of higher cell density at batch [15], fed-batch [16] and continuous mode [17] to increase the productivity of organic acids at near neutral pH values are available. Apart from the traditional continuous fermentation approach where cell wash out limits productivity, strategies with continuous cell recycle and cell immobilization have shown increased organic acids productivities. At pH 6.5, Clostridium tyrobutyricum fermentation on the biofilm based fibrous bed bioreactor was reported to have a maximum productivity of 4.13g.L-1.h-1 of butyric acid which is four times more as compared to that achieved with low cell density batch fermentation [18]. High cell density fermentation by C. tyrobutyricum on cell-recycle mode using hollow fiber membranes has also been reported and an increase in productivity (six-fold) with maximum butyric acid productivity of 1.13g.L-1.h-1 at pH 6.8 when compared to low cell density batch fermentations [17]. Additionally, with the use of these techniques, problems associated with product related inhibitions have also been addressed as the increase in productivity has been achieved at comparatively lower concentrations of organic acids.
In this work, we present an investigation of pH values and glucose concentrations that influence acidogenesis and can affect improved productivities of organic acids. The work also illustrates the use of continuous cell recycle to achieve an increase in total organic acid productivity.
Materials and Methods
Bacterial strain and inoculum development
Clostridium acetobutylicum NRRL B 527 and Clostridium butyricum NRRL B 1024 were obtained from Northern Regional Research Lab, USA. Clostridium acetobutylicum ATCC 4259 was obtained from the American Type Culture Collection which eventually got degenerated in the lab. The cultures were sub-cultured in Reinforced Clostridial Broth (RCB) (HiMedia Laboratories Pvt. Ltd, Mumbai) at 30 °C. All cultures were preserved at 4 °C as clean/pure spore preparations in RCB.
For inoculum development, spores were activated by heat shock at 80 °C for 10 minutes followed by cooling on ice followed by inoculation into sterile RCB. 10% of developed inoculums from RCB were used for inoculation in all the sterile semi-synthetic media. 10% of culture grown in the semi-synthetic medium was used as inoculum for a sterile semi-synthetic medium which was considered for experimental purpose. All incubations were done under stringent anaerobic conditions in an anaerobic chamber (Model Bug Box Plus, Ruskinn Technology Ltd., UK.) supplied with anaerobic gas (10% H2, 10% CO2 and 80% N2).
Media components
Medium components used included glucose, Yeast Extract, 1g.L-1 of NH4(SO4)2, 0.2g.L-1 of NaHCO3, 0.2g.L-1 of MgSO4.7H2O, 0.1g.L-1 of MnSO4.H2O and 0.01g.L-1 CaCl2. These medium components were selected by referring the semi-synthetic medium composition reported in the literature [16]. All abovementioned chemicals were obtained from Sisco Research Laboratories Pvt. Ltd., Mumbai.
Bioreactor operation
Batch and continuous runs were carried out in 3L glass reactors (Biostat® B plus, Sartorius Stedim biotech, Pvt. Ltd.) equipped with turbidity probe (Fundalux II, Sartorius Stedim biotech, Pvt. Ltd.), temperature probe and pH probe (Easyferm Plus K8 200, Hamilton Pvt. Ltd.) for continuous monitoring of temperature, pH and turbidity as well as automated controlling of temperature and pH. pH was controlled by automated addition of 3N NH4OH wherever required. Reactors were sterilized by autoclaving at 121 °C for 20min. All fermentation runs were carried out at 1L working volume. During fermentation, the temperature and agitation were maintained at 30 °C and 150rpm respectively and the anaerobicity was maintained by sparging sterile N2 gas. N2 gas was filter sterilized by using 0.2|im pore sized PTFE filters (Midistart® 2000, Sartorius Stedim biotech, Pvt. Ltd.).
Cell recycle operation
The bioreactor was integrated with a hollow fiber microfiltration membrane of 850cm2 of 0.2|im pore size and 0.7mm lumen I.D. (Model no CFP-2-2E-4X2MA, GE Healthcare Bio-sciences Corp). Fermentation broth from the bioreactor was pumped through the hollow fiber membrane using a Watson Marlow 520 S pump. Two pressure gauges (Tipco) were connected at the two ends of the membrane to monitor trans-membrane pressure. Permeate was collected in separate 2L product collection vessel while the cells were recycled back to the bioreactor vessel. Sterile medium was pumped in the bioreactor continuously at a constant flow rate.
Analytical methods
Samples were filtered through 0.2|im pore sized nylon filters (AxivaSichem Biotech, Delhi) to separate cells. Glucose utilized and OA produced were determined using HPLC (Agilent 1100 series, Agilent Technologies, New Castle DE), with an Aminex 87H column (Bio-Rad, Richmond, CA) at 50 °C and RI detector (G1362A) at 35 °C with 5mM H2SO4 as mobile phase at the flow rate of 0.6ml/min; ChemStation software was used for data processing.
Optical measurements were done using spectrophotometer at 600nm (Multiscan, Thermoscientific). Scanit 2.4.2 software was used for data processing.
Calculation
Yield = g of organic acids produced/ g of glucose utilized
Volumetric productivity = g of organic acids produced/ L of fermentation broth/hour
Specific productivity = g of organic acids produced/ g of dry cell biomass/hour
For cell recycle experiments-
Productivity (g.L-1.h-1) = organic acids concentration (g.L-1) * Dilution rate (h-1)
Dilution rate (h-1) = Feed flow rate (L.h-1)/ Working volume of the bioreactor (L)
Hydraulic Retention Time (HRT) = 1/Dilution rate (h-1)
Results
Acidogenesis at various pH values
Fermentation was performed by the degenerated ATCC 4259 in batch mode maintained at various pH values in semisynthetic medium with 20g.L-1 glucose as carbon source. Runs were performed in triplicates. In the case of the fermentation run without pH control, glucose utilization was seen to stop within 24 hours due to cell sporulation resulting in a final total organic acid titer and yield of 3.5g.L-1 and 0.51g.g-1 respectively at the rate of 0.17g.L-1.h-1. No complete glucose utilization was observed. This limitation on complete glucose utilization was not observed in the fermentations carried out at various pH values above pH 5. It was observed that runs above pH 5 continued with no sporulation until complete utilization of glucose occurred.
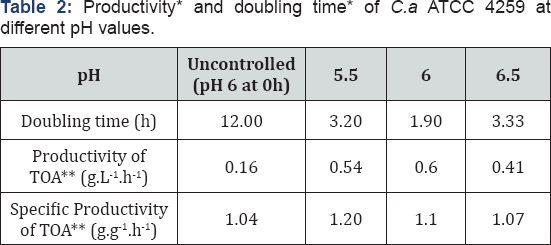
Differences in doubling time of the bacterium (growth rates) were observed with differences in the pH value of the medium Table 2. It was noticed that productivity of the total organic acids varied with differences in pH values while no major differences in specific productivity of the total organic acids at various pH values were observed (Table 2) (Figure 2).
*Results are indicated as the mean of three independent fermentation runs. ** Total Organic Acid.
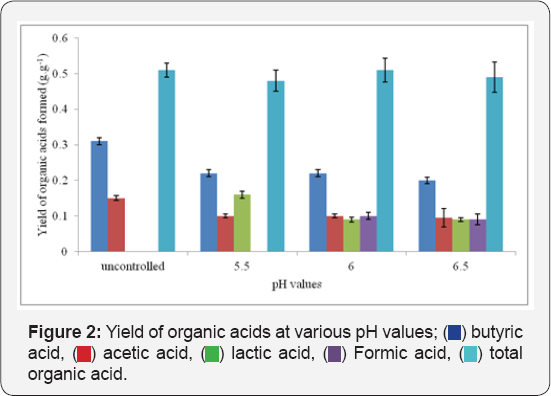

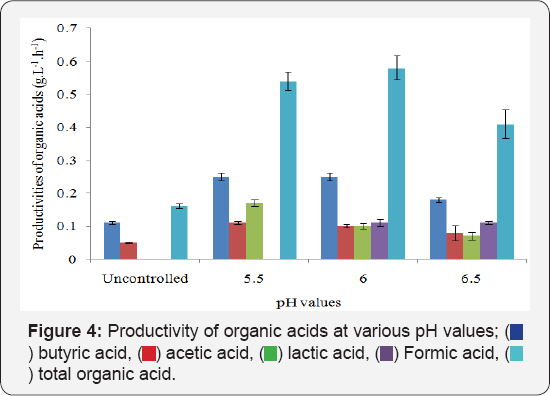
It is evident from Figure 2 that no major differences in yields of the total organic acids were observed but yields of individual organic acids varied significantly at various pH values studied. In this case, it was observed that for runs without pH control, butyric acid and acetic acid were formed. Lactic acid was found to be co-produced with butyric acid and acetic acid in the case of fermentation runs maintained at pH 5 while formic acid was another product detected along with the other three at pH 6 and pH 6.5 (Figure 2 & 3). Variation titers as well as in volumetric productivities were observed at different pH values (Figure 3 & 4).
Organic acid production at various glucose concentrations
Prevention of sporulation of the degenerated strain during fermentation in semi-synthetic medium resulted in complete utilization of 20g.L-1 glucose and made possible use of increased glucose concentrations for fermentation (Figure 5).

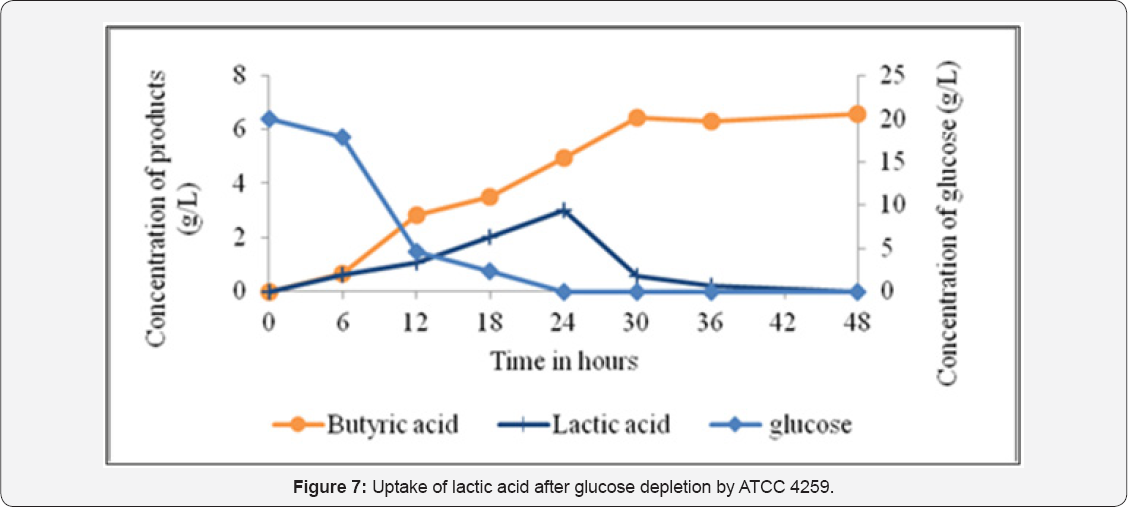
Figure 5 explains the kinetics of clostridial growth under different glucose concentrations ranging from 5g.L-1 to 80g.L-1 in the semi-synthetic medium at pH 6. Specific growth rates declined at 70g.L-1 and 80g.L-1 of glucose concentrations. Compilation of specific growth rates for various glucose concentrations resulted in derivation of the maximum specific growth rate value as well as the Ks value (Figure 5) that can form the basis of a model for clostridial growth during evaluation of its potentials at large scale. It was predictable from Figure 5 that for this system at any given time, decrease in glucose concentration below the Ks value (0.5g.L-1) or increase in glucose concentration beyond 60g.L-1 will impede the growth of the bacteria. Similarly, for any continuous runs, dilution rates more than maximum specific growth rate (0.5h-1) will result in washout of the cells. An examination of growth curve with respect to organic acid production profile provided evidence for total organic acid inhibition beyond 20g.L -1 at controlled pH of 6 (Figure 6). Additionally, utilization of lactic acid as carbon source was detected after glucose depletion (Figure 6 & 7).

The study of clostridial growth kinetics resulted in derivation of the Ks value and maximum specific growth rate value as well as offered the MIC values for glucose and total organic acids.
Fermentation on continuous cell recycle mode
It was considered that fermentation runs with high cell density and total cell recycle could increase the productivity of total organic acids. Hence, continuous runs with cell recycle were operated with various glucose concentrations at various dilution rates at pH value of 6, results of which are depicted in (Table 3).
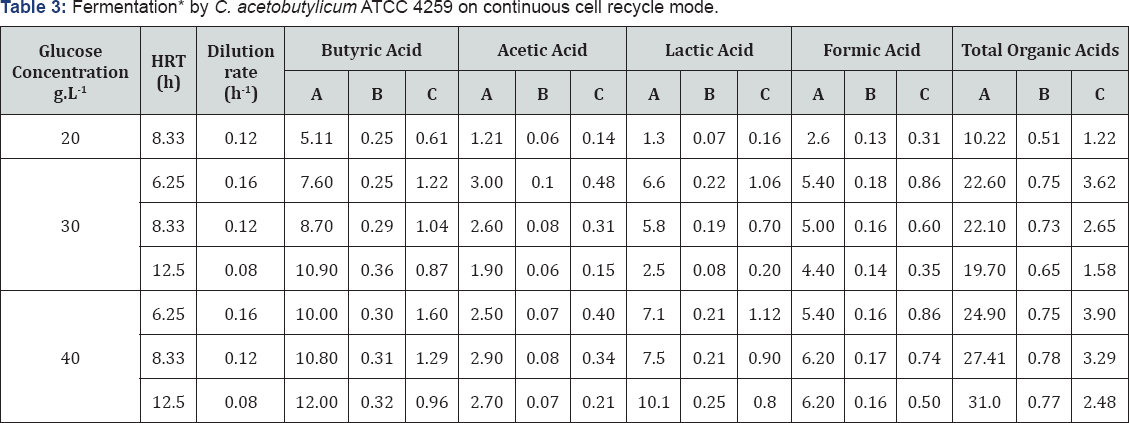
*Results are indicated as mean of four independent samples withdrawn during steady state of fermentation
Key:
A=concentration g.L-1
B=Yield g.g-1
C=productivity g.L-1.h-1
Each run was extended up to 168 hours of steady state operation. The experiment was observed to take almost three hydraulic retention times (HRT) to achieve steady stage at a cell density of 100g.L-1 (wet cell mass) after every change incorporated in the experimental conditions. No data of the initial HRTs was incorporated in any of the measurements. At the dilution rate of 0.12h-1, the glucose concentration of 20g.L-1 and 30g.L-1 in feed resulted in complete glucose utilization while glucose concentrations of 40g.L-1 in feed resulted in residual glucose in the permeate stream. It was observed that increase in feed glucose concentration not only increased productivity but also increased the yield of total organic acids.
For 30g.L-1 feed glucose concentrations, all the dilution rates resulted in complete glucose utilization. The lowest dilution rate of 0.08h-1 was seen to result in a total organic acid yield of 0.65g.g-1 which was lower than the total organic acid yields obtained for the dilution rates 0.12h-1 and 0.16h-1. For 40g.L-1 feed glucose concentration, the lowest dilution rate of 0.08h-1 exhibited complete utilization of glucose. However, the runs with higher dilution rates were ended with the incomplete utilization of glucose indicating that 40g.L-1 of feed glucose concentration was more than enough at dilution rates of 0.12h-1 and 0.16h-1 for organic acid production. It was observed that the yields of total organic acids as well as for lactic acid were maintained almost constant for all the dilution rates examined which proved that no cell death or no lactic acid uptake had occurred. Moreover, similar to the runs with feed concentrations of 30g.L-1, in this case, increase in productivity observed for dilution rate of 0.16h' 1 as compared to that with a dilution rate of 0.12h-1 indicated that the productivity was a function of dilution rates under conditions at constant biomass wherein glucose starvation had not occurred. The maximum total organic acid concentration under optimum conditions obtained using this organism with the batch mode of fermentation was 10g.L-1 with productivity of 0.55g.L-1.h-1.
Discussion
Acidogenesis at various pH values
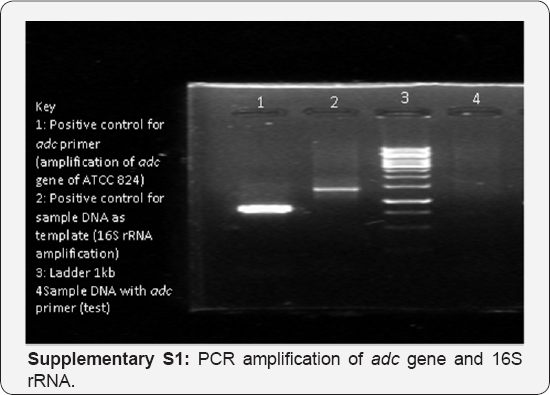
To confirm degeneration of Clostridium acetobutylicum ATCC 4259, plasmid isolation and PCR analysis of solvent specific genes like ‘adc’ was performed. Unsuccessful plasmid isolation and no amplification of ‘adc’ gene (Figure S1) proved that the strain was a degenerated strain of Clostridium acetobutylicum ATCC 4259 which had undergone a plasmid loss and its DNA contained no genes specific for solventogenesis. Amplification of adc gene of solventogenic ATCC 824 was done and was used as a positive control (lane 1); Amplification of 16S rRNA of the sample strain was done and used as a positive control for sample DNA as template (Lane 2). Degeneracy of strains has been reported to occur due to curing of plasmid while performing several serial passages of solventogenic cells. The sol plasmid which gets cured has been known to carry the majority of the solventogenesis specific genes like ‘ctf B’, ‘adc’ and 'adhE'.
Clostridium acetobutylicum is biphasic in nature that performs acidogenesis followed by solventogenesis and simultaneous sporulation to overcome the end product inhibition incurred due to the accumulation of organic acids at lower in pH values. However, the strain under this study was a degenerated strain (supplementary material 1) and hence did not perform solventogenesis. Unlike other solventogenic wild type clostridial strains, this strain underwent only sporulation as a stress response against the accumulation of the organic acids in un-dissociated form or due to the accumulation of H+ ions.
It has been well established that pH of the medium plays a detrimental role during growth and biosynthesis of organic acids by Clostridial strains. Variations have been observed in the growth of Clostridium thermocellum when grown at different pH values [19]. Such variations can be attributed to the fact that different pH values correspond to different anion specific effects. It has been reported that anion specific effect dictates metabolic reactions like decarboxylation (consumption of protons), ion transportation (removal of protons), changing membrane composition, expression of stress genes etc. [20]. In the case of the degenerated clostridial strain under this study, a successful extension of fermentation runs beyond the inhibitory organic acid concentration of 3.5g.L-1 at pH values above 5 indicated that the accumulation of dissociated form of organic acids produced was not inhibitory to the bacterium. Above pH 5, the organic acids produced by clostridia have been known to be present in the non protonated form or in the dissociated form (pKa value of butyric acid 4.82; pKa value of acetic acid 4.75).
Organic acids have been known to be primary products or exponential phase products and their production has been linearly proportional to the cell growth. Thus it appeared that productivity of the total organic acids was subject to the growth rate of the bacterium and not directly to the pH values of the medium. This was further supported by the values of the specific productivity of the total organic acids Table 2. Similar total organic acid yield for all pH values suggested that the ratio of the carbon directed towards biomass generation to the carbon directed towards product formation was independent of the pH value of the medium. Differences in yields of individual organic acids obtained Figure 2 at different pH values proved that the ratio of carbon fluxes diverted to individual organic acids differed with differences in pH values. Similar variation in product profile has been observed in the case of Clostridium thermocellum where pH of the medium has been proved to govern the metabolic flux shifts the bacterium Rydzak et al. [19] and Sparling et al. [21]. A constant specific productivity for total organic acid for all the Ph studied proved that the total organic acid production per unit cell was independent of pH values and changes in volumetric productivity of the total organic acids are due to changes in growth rate of the cells.
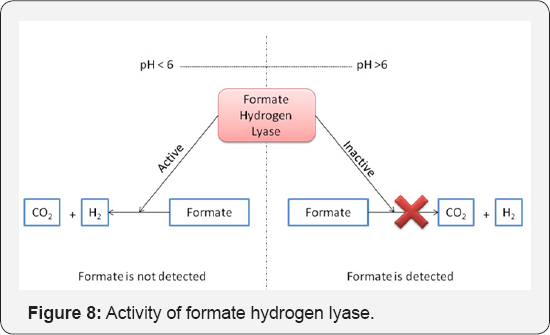
The production of lactic acid at pH values higher than 5.5 can be attributed to the need of the cell to balance the NADH to NAD+ ratio. Excess glycolysis has been reported to result in increased intercellular NADH concentration which if not directed towards solvent production becomes toxic to the cell. It has been known that lactate dehydrogenase utilizes NADH for lactic acid production [2]. Hence the cell has been reported to divert carbon fluxes towards lactic acid production while continuing glycolysis at a higher rate. Formate production by Clostridium at near neutral pH has been reported by various researchers Sparling et al. and Rydzak et al. [19,21] but the reason underlying its production being induced only at higher pH values has not been justified. An explanation of formate hydrogen lyase activity as shown in Figure 8 may give a justification for detection of formate production at higher pH values. There is a possibility of formate formation at lower pH values. However, the formate thus formed must have gone undetected. Formate hydrogen lyase which has been reported to convert formate to CO2 and H2 as soon as it is formed is active only at lower pH values [22]. Thus detection of formate produced at lower pH becomes practically impossible. At higher pH values, formate hydrogen lyase has been reported to remain inactive and hence production of formate was detected in this work (Figure 8).
The metabolic shifts in individual organic acid production observed in the cell due to pH changes Figure 3 notify that the specific productivity of individual organic acids was different at different pH values. This proved that individual organic acid production per unit cell was subject to pH values. Unlike volumetric productivity of total organic acids, the differences in volumetric productivity of individual organic acids Figure 4 at different pH values were subject to pH values as well as to the microbial growth rate. Of all the pH values studied, pH 6 supported maximum metabolic activities with the maximum total organic acid of 10.22 ±0.033g.L-1.h-1, the total organic acid productivity of 0.58±0.036g.L-1.h-1 as well as the least doubling time of 1.9h-1. Hence, pH 6 was used for further experiments.
Organic acid production at various glucose concentrations
The decline of specific growth rate beyond 60g.L-1 of glucose concentration indicated substrate inhibition beyond 60g.L-1. Such inhibitions have been widely studied for various microbes and have been always associated to osmotic dehydrations. Substrate inhibition observed in this system is common with C. tyrobutyricum CNRZ 596 [9], C. acetobutylicum ATCC 824 [23] and C. thermobutyricum [5].
It was predictable from Figure 5 that for this system at any given time, decrease in glucose concentration below the Ks value (0.5g.L-1) or increase in glucose concentration beyond 60g.L-1 will impede the growth of the bacteria. Similarly, for any continuous runs, dilution rates more than maximum specific growth rate (0.5h-1) will result in washout of the cells.
The concept of end product inhibition observed in our case beyond 20g.L-1 of total organic acid concentration results was in agreement with previous reports on clostridial fermentation controlled at near neutral values of pH where the ionized forms of organic acids has been mentioned as end product inhibitors of growth [24,25]. Though the present study did not generate any novelty regarding the concept of end product inhibition, the study was essential because the minimum inhibitory concentration (MIC) values determined have been known to be different for different strains. Nevertheless, the MIC values for this strain have practical significance while considering its use for large-scale production.
An unusual phenomenon of uptake of lactic acid after depletion ofglucose by the strain was observed in this case. Uptake of lactic acid in presence of glucose during stationary phase has been a known phenomenon of solventogenic (Clostridium acetobutylicum) [25]. No reports have been available for lactic acid uptake after glucose depletion. In this case, increase in butyric acid concentrations after glucose depletion Figure 7 was indicative of diversion of lactic acid carbon towards butyric acid. Reverse action of lactate dehydrogenase has been known to generate reducing power in form of NADH. Butyric acid production has been known to require NADH. An inference can be drawn that lactic acid uptake along with continued butyric acid formation balances the reducing power.
Fermentation on continuous cell recycle mode
For all the glucose concentrations studied at continuous cell recycle mode, increased productivity as well as yield of total organic acids observed with increase in dilution rate indicated a possibility that depletion of glucose as C-source resulted in uptake of organic acids thus decreasing the total organic acids yield and productivity. The ratios of organic acids obtained in this case at various glucose concentrations cannot be attributed to the 'selectivity of production' by the organism due to difference in growth rates as in case of other clostridial strains [26] but were due to the amount of medium nutrients or glucose available for fermentation.
At 30g.L-1 of glucose concentration in the feed, the dilution rate of 0.08h-1 was too low for the complete substrate conversion. The low dilution rate resulted in death due to glucose depletion as well as the uptake of lactic acid after glucose depletion ultimately decreasing the yield of total organic acids. This can also be supported with the data of lactic acid yield for the different dilution rates. It was observed that the yield of lactic acid was lowest for the dilution rate of 0.08h-1 while it increased with increase in dilution rates. For dilution rates 0.12h-1 and 0.16h-1, no major differences in yield of total organic acid were observed indicating no cell death and no uptake of organic acid upon glucose depletion. The increase in productivity observed for dilution rate of 0.16h-1 as compared to that with a dilution rate of 0.12h-1 indicated that the productivity was a function of dilution rates at constant biomass under conditions wherein glucose starvation had not occurred.
Considering the total organic acid concentrations and yields for the dilution rate of 0.08h-1 with 40g.L-1 feed glucose concentrations, it can be stated that these conditions were optimum for organic acid production. However, the productivity obtained under these conditions was exceptionally low as compared to that obtained for other dilution rates. This behavior can be the consequence of inhibitory effects due to the accumulation of total organic acids at concentrations higher than 20g.L-1.
This pattern of increase in productivity observed is in general agreement with results obtained by various researchers not only for clostridial species [12,26] but also for (Enterococcus) Lee et al. [27] and (Lactobacillus) Wee & Ryu [28] species which have been used for continuous lactic acid production. Continuous cell recycle at higher dilution rates resulted in almost two times increase in the total organic acid concentration (22.60g.L-1) and almost seven times increase in the productivity of total organic acids (3.62g.L-1.h-1) was observed. This proved that continuous fermentation with cell recycle at higher dilution rates increases productivity even at lower organic acid concentrations. The total organic acid concentration obtained in this work is lower than the reported high organic acid concentrations obtained by various researchers with batch or fed-batch mode, but the productivity obtained in this work is higher than the corresponding reported productivities (Table 4).

The maximum productivity of total organic acids obtained in this work was 3.6±0.05g.L-1.h-1 at the dilution rate of 0.16h-1 with 30g.L-1 feed glucose concentration is better than the reported productivities* of 2.13 g.L-1.h-1 obtained at the dilution rate of 0.21h-1 [12] and 2.94g.L'1.h-1 obtained at the dilution rate of 0.2 h-1 [31]. However, productivity, in this case, is lesser as compared to the productivity of 10.11g.L-1.h-1 Michel-Savin et al. [21] that has been obtained using Clostridium tyrobutyricum wherein the concept of cell bleeding and cell growth along with cell recycle has been employed. The volumetric productivity can be increased with continuous cell bleeding and cell growth because it is a function of the growth rate of the cell.
*The total organic acid concentration was calculated from the concentrations of organic acids mentioned in the graphs (for productivity of 2.13g.L-1.h-1, total organic acids = 10.65g.L -1 considering butyric acid =3.76g.L-1, acetic acid =4.3g.L-1, lactic acid =2.57g.L-1 and for productivity of 2.94g.L-1.h-1, total organic acids =14.7 considering butyric acid =9.7g.L-1 and acetic acid =5g.L-1). Mass balances arrived from the experimental data for the complete cell recycle runs with dilution rate of 0.16h'1 and 30g.L-1 feed glucose concentrations considering 2 moles of CO2 formed per mole of butyric acid [9] gave the following stoichiometry
0.16 C6H12O6→ 0.086 C4H8O2 + 0.05 C2H4O2 + 0.073 C3H6O3 + 0.11 CH2O2 + 0.172 CO2 + BIOMASS (corresponding to 0.04mol of C)
The mass balances reflected diversion of significant carbon flux towards organic acids synthesis under given operating conditions.
At continuous recycle mode, the physiology of organism was seen to be changed in terms of autolysin activity as well as tolerance to organic acid concentration as compared to the batch mode. No autolysis was observed at cell recycle mode. This can be attributed to the dilution of broth occurring at continuous mode. Unlike at batch mode, no decrease in productivity was observed with organic acid concentrations slightly higher than 20g.L-1 at continuous cell recycle mode at pH value of 6. A possible reason for this tolerance can be an increase in cell density at cell recycle mode. Similar results on tolerance to higher concentrations of end products with increase in cell density at continuous mode have been reported by various reporter [32-34], but reports on entire product profile for fermentation by clostridial strains on cell recycle mode have not been available. With the recent development of appropriate inexpensive separation technology for separation of a mixture of ammonium salts of organic acids [35], heterofermentative organic acid fermentation of this kind may soon become feasible at large scale. To summarize, fermentation runs on continuous cell recycle mode by the degenerate strain of Clostridium acetobutylicum ATCC 4259 resulted in a maximum productivity of 3.6±0.05g.L-1.h-1 at pH 6. Continuous dilution of broth resulted in fermentation without autolysis and increased cell mass rendered an advantage of increased tolerance to higher concentrations of organic acids.
Conclusion
The work proved that variations in product profile can be achieved by variations in pH of the medium. A saturation curve for growth and organic acid production obtained with increasing glucose concentration concluded that maintenance of an optimum concentration of glucose is essential to achieve maximum possible growth without end product inhibitions. Continuous fermentation runs at high cell density with cell recycle have the potentials to increase productivity at comparatively lower total organic acid titers as compared to the batch fermentation runs. The continuous runs render an additional advantage of increased organic acid tolerance without detectable autolysis of cells.
References
- Tracy BP, Jones SW, Fast AG, Indurthi DC, Papoutsakis ET (2012) Clostridia: The importance of their exceptional substrate and metabolite diversity for biofuel and biorefinery applications. Curr Opin Biotechnol 23(3): 364-381.
- Jones DT, Woods DR (1986) Acetone-butanol fermentation revisited. Microbiol Rev 50(4): 484-524.
- Dwidar M, Park JY, Mitchell RJ, Sang BI (2012b) The Future of Butyric Acid in Industry. Sci World J 2012: 471417.
- MC (2009) Acetic Acid Production. Appl Microbiol Ind 1: 144-149.
- Lee RK, Ryu HW, Oh H, Kim M, Wee YJ (2014) Cell-recycle continuous fermentation of Enterococcus faecalis RKY1 for economical production of lactic acid by reduction of yeast extract supplementation. J Microbiol Biotechnol 24(5): 661-666.
- Gibbs PA (1987) Novel uses for lactic acid fermentation in food preservation. J Appl Microbiol 63(s16): 51s-58s.
- Griggs JP, Jacob JP (2005) Alternatives to antibiotics for organic poultry production. J Appl Poult Res 14(4): 750-756.
- Sauer M, Porro D, Mattanovich D, Branduardi P (2008) Microbial production of organic acids: expanding the markets. Trends Biotechnol 26(2): 100-108.
- Green EM (2011) Fermentative production of butanol-the industrial perspective. Curr Opin Biotechnol 22(2): 337-343.
- Wang L, Ou MS, Nieves I, Erickson JE, Vermerris W, et al. (2015) Fermentation of sweet sorghum derived sugars to butyric acid at high titer and productivity by a moderate thermophile Clostridium thermobutyricum at 50 °C. Bioresour Technol 198: 533-539.
- Montville TJ, Parris N, Conway LK (1985) Influence of pH on organic acid production by Clostridium sporogenes in test tube and fermentor cultures. Appl Env Microbiol 49(4): 733-736.
- Stinson EE, Naftulin KA (1991) Effect of pH on organic acid production by Clostridium propionicum in test tube and fermentor cultures. J Ind Microbiol 8(1): 59-63.
- Vandak D, Tomaska M, Zigova J, Sturdik E (1995) Effect of growth supplements and whey pretreatment on butyric acid production by Clostridium butyricum. World J Microbiol Biotechnol 11(3): 363.
- Michel-Savin D, Marchal R, Vandecasteele JP (1990b) Control of the selectivity of butyric acid production and improvement of fermentation performance with Clostridium tyrobutyricum. Appl Microbiol Biotechnol 32(4): 387-392.
- Canganella F, Kuk SU, Morgan H, Wiegel J (2002) Clostridium thermobutyricum: growth studies and stimulation of butyrate formation by acetate supplementation. Microbiol Res 157(2): 149-156.
- He G, Kong Q, Chen Q, Ruan H (2005) Batch and fed-batch production of butyric acid by clostridium butyricum ZJUCB. J Zhejiang Univ Sci B 6(11): 1076-1080.
- Du J, McGraw A, Lorenz N, Robert RB, Edgar CC, et al. (2012) Continuous Fermentation of Clostridium tyrobutyricum with Partial Cell Recycle as a Long-Term Strategy for Butyric Acid Production. Energies 5(8): 2835-2848.
- Jiang L, Wang J, Liang S, Wang X, Cen P, et al. (2009) Butyric acid fermentation in a fibrous bed bioreactor with immobilized Clostridium tyrobutyricum from cane molasses. Bioresour Technol 100(13): 34033409.
- Rydzak T, Levin DB, Cicek N, Sparling R (2011) End-product induced metabolic shifts in Clostridium thermocellum ATCC 27405. Appl Microbiol Biotechnol 92(1): 199-209.
- Warnecke T, Gill RT (2005) Organic acid toxicity, tolerance, and production in Escherichia coli biorefining applications. Microb Cell Fact 4: 25.
- Sparling R, Islam R, Cicek N, Carere C, Chow H, et al. (2006) Formate synthesis by Clostridium thermocellum during anaerobic fermentation. Can J Microbiol 52(7): 681-688.
- Sawers RG (2005) Formate and its role in hydrogen production in Escherichia coli. Biochem Soc Trans 33(Pt 1): 42-46.
- Monot F, Martin JR, Petitdemange H, Gay R (1982) Acetone and butanol production by Clostridium acetobutylicum in a synthetic medium. Appl Environ Microbiol 44(6): 1318-1324.
- Wang G, Wang DI (1984) Elucidation of Growth Inhibition and Acetic Acid Production by Clostridium thermoaceticum. Appl Environ Microbiol 47(2): 294-298.
- Sonomoto K, Oshiro M, Hanada K, Tashiro Y (2010) Efficient conversion of lactic acid to butanol with pH-stat continuous lactic acid and glucose feeding method by Clostridium saccharoperbutylacetonicum. Appl Microbiol Biotechnol 87(3): 1177-1185.
- Michel-Savin D, Marchal R, Vandecasteele JP (1990a) Butyric fermentation: Metabolic behaviour and production performance of Clostridium tyrobutyricum in a continuous culture with cell recycle. Appl Microbiol Biotechnol 34(2): 172-177.
- Wee YJ, Ryu HW (2009) Lactic acid production by Lactobacillus sp. RKY2 in a cell-recycle continuous fermentation using lignocellulosic hydrolyzates as inexpensive raw materials. Bioresour Technol 100(18): 4262-4270.
- Michel Savin D, Marchal R, Vandecasteele JP (1990c) Butyrate production in continuous culture of Clostridium tyrobutyricum: effect of end-product inhibition. Appl Microbiol Biotechnol 33(2): 127-131.
- Sjöblom M, Matsakas L, Christakopoulos P, Rova U (2015) Production of butyric acid by Clostridium tyrobutyricum (ATCC25755) using sweet sorghum stalks and beet molasses. Ind Crops Prod 74: 535-544.
- Song H, Eom MH, Lee S, Julia L, Jung HC, et al. (2010) Modeling of batch experimental kinetics and application to fed-batch fermentation of Clostridium tyrobutyricum for enhanced butyric acid production. Biochem Eng J 53(1): 71-76.
- Rani KS, Swamy MV, Sunitha D, Haritha D, Seenayya G (1996) Improved ethanol tolerance and production in strains of Clostridium thermocellum. World J Microbiol Biotechnol 12(1): 57-60.
- Zhu Y, Yang ST (2003) Adaptation of Clostridium tyrobutyricum for Enhanced Tolerance to Butyric Acid in a Fibrous-Bed Bioreactor. Biotechnol Prog 19(2): 365-372.
- Jiang L, Wang J, Liang S, Cai J, Xu Z, et al. (2011) Enhanced butyric acid tolerance and bioproduction by Clostridium tyrobutyricum immobilized in a fibrous bed bioreactor. Biotechnol Bioeng 108(1): 31-40.
- Lali AM, Maurya RR (2016) Separation of organic acids from mixtures containing ammonium salts of organic acids. W0/2016/194003.
- Wee Y, Kim J, Ryu H (2006) Biotechnological production of lactic acid and its recent applications. Food Technol Biotechnol 44(2): 163-172.






























