Activated Charcoal Increases the Viability of Ectomycorrhizal fungi During the Stages of Cultivation Aiming the Production of Large-Scale Inocula via Submerged Cultivation
Rossi MJ1*, Nascimento FX1, Brandão PMFDR1, Camelini CM2 and Giachini AJ1
1Departamento de Microbiologia, Universidade Federal de Santa Catarina, Brazil
2Departamento de Engenharia Química e Engenharia de Alimentos, Universidade Federal de Santa Catarina, Brazil
Submission: August 03, 2017; Published: October 03, 2017
*Corresponding author: Rossi MJ, Departamento de Microbiologia, Imunologia e Parasitologia, Centro de Ciências BiolɃgicas, Universidade Federal de Santa Catarina, CP 476, 88040-900 Florianópolis, SC, Brazil, Tel: +55-48-3721-5204; Fax: +55-48-3721-2011; Email: marcio.rossi@ufsc.br
How to cite this article: Rossi MJ, Nascimento FX, Brandao PMFDR, Camelini CM, Giachini AJ. Activated Charcoal Increases the Viability of Ectomycorrhizal fungi During the Stages of Cultivation Aiming the Production of Large-Scale Inocula via Submerged Cultivation. Adv Biotech & Micro. 2017; 6(4): 555692.DOI:10.19080/AIBM.2017.06.555692
Abstract
Ectomycorrhizal fungi play an important role in promoting the growth and development of several tree species indispensable in worldwide forestation programs and, also, industrial processes. Yet, the large-scale production of ectomycorrhizal fungi necessary for these applications is still difficult and incipient. One alternative consists on the submerged cultivation of these fungi in high capacity bioreactors and the consequent production of inoculants by encapsulation. However, ectomycorhizal fungi present some limitations when growing under these conditions, and maintaining fungal viability is essential.
In this work we studied the growth and viability of several of ectomycorhizal fungi such as, Rhizopogon, Pisolithus, Chondrogaster and Scleroderma spp. when subjected to submerged cultivation aiming for the production of large scale inocula, and tested the effect of activated charcoal in promoting fungal growth and viability. The results obtained showed that after fragmentation of mycelia, aiming for both inocula production for the application directly in the bioreactor or to the encapsulation process, most fungal isolates were unable to grow and maintain there viability when cultivated without charcoal. On the other hand, after fragmentation, activated charcoal increased the viability of the fragmented fungal mycelium, and also increased the fungal long term viability when encapsulated and stored under refrigerated conditions. Moreover, encapsulated ectomycorhizal fungi were able to efficiently colonize Eucalyptus plants. A model for the submerged cultivation and large scale commercial production of ectomycorrhizal fungi is therefore presented. Maximizing the growth and production of ectomycorrizal fungi may be the key for a wide range of agricultural and biotechnological applications worldwide.
Keywords: Ectomycorrhiza; Fungi; Activated charcoal; Submerged cultivation; Pinus; Eucalyptus
Introduction
Ectomycorrhizal fungi (ECM) are essential for the establishment ofplants in forestation programs around the world. The effects promoted by ECM fungi are more pronounced when the association occurs early in plant development, preferably at the nursery, with the inoculation of selected effective fungi [1,2]. Several countries around the world, including Brazil, vast in reforestation areas, do not present any commercial formulations for ECM. Also, the commercial production of fungal inoculum to attend the ever-growing needs of important sectors, such as the pulp and paper industry is still incipient, even after decades of studies, indicating the difficulties behind this technology.
Currently, mycorrhizal inoculants are mainly produced via solid substrate fermentation (SSF) [3]. Aside from fungal slower growth characteristics under these conditions, there is also a potential increase in contamination and more associated costs of production [4]. One promising alternative to produce ECM-based inoculants consists of cultivating the fungi in liquid media in bioreactors and then encapsulate the produced mycelium in calcium alginate beads [5,6]. Studies developed at nurseries have shown that this method is more effective than those employing solid substrates probably because it reduces contamination and provides a much higher survival rate for the fungi while encapsulated [5,6]. Furthermore, encapsulated fungi may maintain viability for months if properly stored under refrigeration [2]. Yet, the production of commercial ECM inoculum based on submerged cultivation of the fungi is dependent on the development of more adequate bioreactors [7,8]. These have to minimize the mycelium shearing damages caused by the oxygen transfer system. One additional concern is related to the viability of the fungi during the scaling up and storing [9]. Due to the filamentous growing characteristics, ECM fungi need necessarily to be fragmented into smaller propagule pieces in every step of the scaling process, from the inoculum production to apply in the bioreactor (pre-production) to the inoculant bead production. These smaller propagules are important so there will be enough surface area to allow for mass transfer and to disperse the mycelium [10,11]. Still, fragmentation results in viability losses. Some studies have shown differences in fungal viability (beads) according to the species of fungi tested. Kuek et al. [6] observed fungal viability losses of up to 60% for Elaphomyces sp. within 30 days of storage. On the other hand, fungi such as Hebeloma westraliensi and Laccaria laccata presented viability above 80%, even after stored for up to 7 months. Viabilities above 90% were obtained for fungi such as Hebeloma crustuliniforme (after 5 months of storage) [12], Paxillus involutus, (after 2 months of storage) [13], and Rhizopogon nigrescens (after 18 months of storage) [2]. Factors frequently involved in the viability losses of fungi over time include the different fungal species employed, isolates, age, form and degree of mycelium fragmentation, temperature of storage, humidity of the beads, and the accumulation of toxic compounds resulting from the fragmentation processes. One possible way to decrease the accumulation of toxic compounds resulting from the fragmentation process may reside on the use of activated charcoal (AC), which possesses significant adsorption properties. Curiously, AC has been shown to promote an earlier and more intensive ECM formation by Piloderma croceum [14].
The aim of this study was to determine the factors related to biological viability of ECM fungi during all the stages of cultivation necessary for the production of commercial, large- scale inocula via submerged cultivation.
Materials and Methods
Microorganisms
All isolates used in the study were obtained from pine and eucalypt plantations located in the states of Santa Catarina (SC) and Minas Gerais (MG), Brazil. They belong to the Federal University of Santa Catarina Microbial Diversity Laboratory and have been deposited at CBMAI - Coleção Brasileira de Microrganismos de Ambiente e Industria [15]. Six isolates were employed, as indicated in Table 1. Isolates were chosen based on its growth characteristics and from previous studies developed with seedlings of the cited plant species (Table 1) [16,17]. The isolates were monthly sub cultured onto solid MMN [18] and kept at 25±1 °C.
Culture media and inoculum preparation
Variations of the MMN medium were used in the assays aiming for increased biomass production. The original MMN 0.25g/L (C/N ratio; 50/1) nitrogen concentration was increased to 0.62g/L [19]. The medium contained (g/L): glucose 10.0; yeast extract 3.33; malt extract 3.0; CaCl2 0.050; NaCl 0.025; (NH4)2HPO4 0.75; KH2PO4 0.5; and MgSO4.7H2O 0.15. In addition, the medium received 1.2mL of a 1% FeCl3 solution and 100 ^ g of thiamine-HCl. The initial inoculations were based on mycelial suspensions obtained from liquid culturing in flasks. These cultures were prepared using 5 mycelial-agar disks transferred to 25mL liquid media in 250mL recipients, and incubated statically for a period of 14 days at 25 °C. The suspensions were prepared by fragmenting the fungal mycelium in saline solution (0.85% NaCl) using a blender (LAR-15 Metvisa, Brazil) set at 3600rpm for 20s.
Effects of activated charcoal on the viability of fragmented mycelium suspensions
Assays were performed in order to determine the effects of ACon the viability of mycelial suspensions in relation to time of storage. Mycelial suspensions were prepared from liquid medium by fragmenting the fungi for 20s in saline solution (0.85% NaCl) using a blender (same settings described earlier). The suspensions were further separated into two portions. One was treated with 0.25% neutralized activated charcoal [20]. Part of the suspension with charcoal was employed for the production of beads (described below in the Biomass encapsulation and viability tests section). The second part of the suspension containing charcoal, together with the fraction without charcoal, was kept refrigerated and used for viability tests, by periodically adding 2mL of the suspension onto solid MMN medium in Petri dishes incubated at 25 °C. Both suspensions were tested in solid medium with and without 0.25% AC. Solutions were considered viable when hyphal growth was observed (after incubation).
Determination of mycelium time of fragmentation
To determine the effect of fragmentation time in the mycelium produced in liquid media two fragmentation conditions were tested: 3600rpm for 10 and 60s. The suspensions received 0.25% of neutralized active charcoal and were stored refrigerated at 8 °C for viability tests, performed periodically by adding 2mL of solution onto solid medium in Petri dishes incubated at 25 0 C. The suspensions were tested only in solid MMN medium containing 0.25% active charcoal. The suspensions were considered viable when hyphal growth was observed (after incubation).
Biomass encapsulation and viability tests
The fungal biomass was encapsulated in calcium alginate beads following the methodology described by Mauperin et al. [12]. Each mycelial suspension, composed of about 0.5g of biomass (dry weight), received 0.1 to 0.4% of activated charcoal (this amount varied according to the fungal isolate). Part of the suspension (100mL) was separated and used to perform the viability tests throughout the entire period of storage. Using a magnetic stirring, an aliquot of 150mL of fungal suspension was mixed with a 2% solution of sodium alginate. The mix was then dripped, under constant stirring, to a 0.7mol/L solution of CaCl2 for polymerization, for bead formation (≈4mm diameter). After full polymerization, obtained in about 30 minutes, beads were gently washed and left standing in 400mL of sterile distilled water for about 2 minutes for the removal of any residual chlorides. Finally, they were stored in distilled water at 8 0c for different periods of time.
All encapsulated fungal isolates (inoculant beads) were periodically tested for their viability for a minimum period of six months. The viability tests were performed by placing 25 beads onto solid MMN medium in Petri dishes, and hyphal development was evaluated. Every other day, for a period of two weeks, dishes were inspected under a stereomicroscope to verify the emanation of hyphae from the beads. Bead hyphal growth corresponded to germination, and therefore, viability. These tests were performed on intervals of 20 days. The response obtained was the percentage of viable beads as a function of the period of storage.
Production of inoculants via solid-state fermentation
In order to compare fungal viability for inoculants produced in submerged culture, all fungal isolates were also grown under the traditional SSF method, described elsewhere [21,22]. For this, mycelia from two different Erlenmeyer flasks were used to prepare fungal suspensions. These were performed using 100mL MMN medium. From these, 60mL were withdrawn and used to inoculate 250mL of a vermiculite-peat mixture (1:4; v:v) in 600mL glass flasks. Peat and vermiculite particles smaller than 1mm and 5mm, respectively, were removed by sieving.The mix was moisturized with 40mL of MMN medium, the pH stabilized at 5.8, and then sterilized for 20min at 121 o c. 100mL of MMN medium were added per flask (40%; v:v), allowing for an estimated biomass production of 0.35g/L, considering a yield of 42%, as described by Rossi et al. [23].
This arrangement was adopted to obtain equivalence in relation to biomass encapsulated into alginate gel beads, to allow for a more trustful comparison between the values. The lids of the flasks were perforated with two 1cm 0 holes to improve gas exchange, and covered with surgical Micropore® tape (3M do Brasil) prior to sterilization. The lids rims were then sealed with parafilm, and the flasks incubated for 2 months under the same conditions for the cultivations described earlier. After this period, the colonized substrate was washed with distilled sterile water to remove any residual metabolites. The washing consisted of adding 300mL of water to the substrate and carefully stirring the mix using a glass wand. Washing was performed twice. All heavy peat particles sank, so it was relatively easy to separate the colonized vermiculite particles from the rest of the mix. While moist, these were separated and immediately transferred to new recipients and stored at 8 oc. Mycelium viability was periodically tested in solid MMN medium by adding portions of the inoculant similar in size to that of a bead.
Inoculant viability in the presence of plants
Fungal growth out of the inoculant beads is an important preliminary information for controlled inoculation studies with plants, since this in vitro scenario mimics the conditions in the environment. Therefore, inoculant beads were tested for its viability when applied in Eucalyptus seedlings growing inpeat: vermiculite substrate.
Seeds of Eucalyptus dunnii were disinfected with 70% alcohol for 30s and rinsed in distilled sterile water. They were then placed in 1% sodium hypochlorite for 20min under agitation, rinsed three consecutive times in sterile water, and dried in sterile filter paper. The seeds were then placed in 25mL of germination solid medium (glucose 2g/L, CaSO4. 2H2O 0.068g/L, H3BO3 0.186mg/L, agar 9g/L at pH 5.7) [24], and incubated at 25+1 °C in the dark for five days. Seven randomly taken germinated plants were inserted into a peat: vermiculite (1:3; v:v) substrate and placed into a 9cm 0 Petri dish. The substrate was arranged in order that only half of the dish was occupied, leaving enough room for plants to develop. The substrate was previously sterilized and moisturized to 25% (v/v) humidity. A single bead was inserted immediately below and adjacent to each plant.
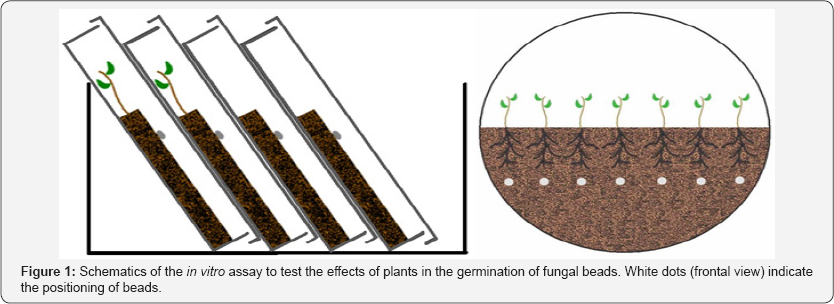
In order to get a proper comparison, three plates containing plants and beads and three plates containing only beads (control) were prepared to each of the fungal isolates. The lower half of all plates was covered with aluminum foil to protect roots and fungal mycelium from direct light exposure. The assay was conducted under laboratory conditions (average room temperature of 25 °C). Periodically, for a period of 90 days, all plates were checked under a stereomicroscope for hyphal growth from the beads. This experiment was carried out only with inoculation beads of fungal isolates that associate with eucalypts (UFSC-Pt116, UFSC- Ch163 e UFSC-Pt188) (Figure 1).
Results
Effects of activated charcoal on the viability of fragmented mycelium
The use of AC was crucial for the viability maintenance of the fragmented mycelium through the long periods of storage (at least 4 months) in saline solution under refrigeration (Table 2). When no AC was applied to the fragmented mycelium, no hyphal growth was observed in most of the tested isolates. Only some hyphal growth under these conditions. Nevertheless, when AC was added to the fragmented mycelium, all fungal isolates, even those not prone to prompt viability losses presented an increased hyphal growth (Table 2). For instance, the growth of the fungal isolate Chondrogaster angustisporus UFSC-Ch163 was much more vigorous in the presence of AC during the first week of trials (Figure 2A).
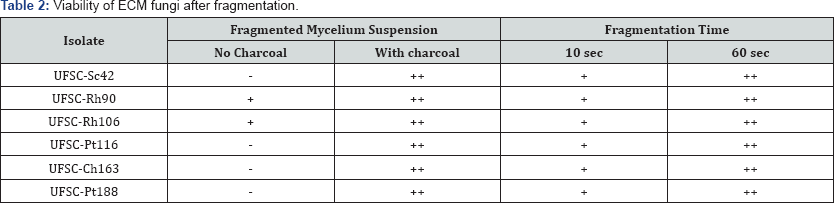
(-) no hyphal growth, (+) intermediate hyphal growth (++), abundant hyphal growth
It is also important to mention that even the suspensions containing AC, after some time, had their viability affected when grown in solid, charcoal-free medium. For example, the 30 day- old fragmented mycelial suspension (containing AC) of Pisolithus microcarpus UFSC-Pt116 showed a poor hyphal growth when inoculated in solid medium without AC (Figure 2B). However, when samples from the same suspensions were placed in solid medium containing AC, the fungal development and, therefore viability, was expressive (Figure 2C).
Determination of mycelium fragmentation time
Tests performed during this study have revealed that long fragmentation times did not affected mycelium viability over time (Table 2). Prior to this result, fragmentation was done in very short periods, of a few seconds, since it was believed that longer periods were detrimental to the fungal viability [25]. Curiously, an increased in viability was detected when fragmentation time was increased to 60s, when compared to 10s. More fragmented mycelium created smaller fungal propagules (Figure 2D-2G).
Viability of encapsulated ECM fungi
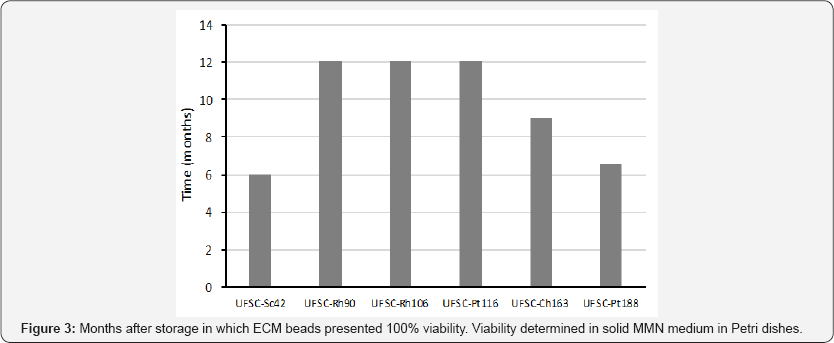
The presence of AC was crucial for the viability ofthe inoculant beads (Table 2). For example, 15 day-old C. angustisporus UFSC- Ch163 inoculation beads (containing AC) cultivated in solid media containing AC showed an increased hyphal growth (Figure 2) when compared to the same beads grown in solid medium without AC (Figure 2). Based on previous studies, encapsulated inoculants were produced and tested in conditions to obtain the best fungal viability possible (20s fragmentation and with charcoal). With the exception of UFSC-Sc42 and UFSC-Pt88, all other fungal isolates maintained 100%viability for more than six months (Figure 3). One particular batch of inoculants produced with fungal isolate UFSC-Rh90 preserved its viability for more than 24 months stored in a fridge. It is important to note that as storage time increases, most fungi tend to germinate slowly detected. In some cases, depending on the fungi, it may take out of the beads, and therefore viability may take longer to be more than a week for germination to take place.
SSF inoculant viability
The viability of inoculants produced by the traditional SSF method was determined monthly for six months. During this period, all isolates presented viability of more than 95% in peat vermiculite particles. The longer the storage time, the longer it took for hyphae to be noticed growing out of the beads.
Inoculant viability in the presence of plants
It was observed that the presence of plants stimulated the germination of the fungi from the beads for all tested isolates. Bead germination was detected after 30 (isolate UFSC-Ch163) and 40 days (UFSC-Pt116 and UFSC-Pt188) after incubation. No germination was observed for the control (beads in peat: vermiculite), even after 60 days of incubation. Pellet diameter reductions of up to 35% (more than 70% in volume) caused by water losses, did not affect fungal germination. The percentage of germinated beads reached more than 90% in the presence of seedlings. Hyphal growth was more prominent near the roots, indicating a typical rhizospheric effect of the association. The inoculation of beads in Eucalyptus seedlings growing in tubes resulted in an augmented plant growth. The fungi successfully colonized the roots of the seedlings (Figure 4).

Discussion
During inoculum preparation for the cultivation in the bioreactor, it was possible to detect and determine difficulties maintaining fungal viability. The required two to three-day wait to determine the mycelial suspensions viability in Petri dishes (prior to bioreactor inoculation), creates unavoidable uncertainties regarding the quality of the mycelium. The mycelium can undergo changes during this period, which can cause viability losses. Fragmentation may release constituents in the medium, such as phenolic compounds, lipids and membrane constitutive proteins, along with intracellular enzymes [26]. Oxidation of these substances, or still yet other unknown reactions, can create a toxic environment in the suspension, which generally gets more pronounced over time since these tend to accumulate in the medium. For suspensions not treated with charcoal, successive washes of the centrifuged solid phase, conferred viability to the mycelium (data not shown). Many times, due to operational reasons, the inoculum preparation may not coincide with the time of inoculation. These events are strong contributors to increase the levels of these substances in solution and, therefore, reduce viability, with subsequent failures in the cultivation in the bioreactor.
Mycelial suspensions and beads of most of the fungal isolates tested (except for Rhizopogon spp.) and kept in solution without active charcoal, lost viability in less than 24h. Viability losses are irreversible, indicating that the use of active charcoal is vital for the process. Furthermore, charcoal needs to be added right after fragmentation. It is also recommended to use charcoal in the biomass for better preservation of the commercial formulations of the inoculants. Charcoal seems to be important for not only a full expression of the fungal viability, but also to indicate the significance of physiological and biochemical studies of the inhibitory products that maybe formed following fragmentation. Pre-determined amounts of charcoal have been established for the inoculum, the inoculant and the solid culture media. Charcoal is currently used also in all culture media employed in the maintenance of fungal cultures in our lab, as the most suitable way to preserve viability of fungal isolates over time. Although beneficial, charcoal is not intended for all situations. In liquid media, for instance, it is not recommended since its unselective chemical attraction pattern may extract nutrients from the medium, which can limit fungal growth in the bioreactor. High dosages also interfere with fungal development. This study has shown that the best concentration for solid media is between 0.2 and 0.3%.
Even though preserving fungal biomass in the form of mycelial suspension may not be the most adequate practice, with the addition of charcoal, viability of these fungal suspensions can be maintained for months. Nevertheless, ideally, the mycelium should be kept in a solid matrix, such as calcium alginate. Besides the typical protection advantages, the gel matrix will also yield a homogeneous distribution of charcoal, culminating with longer fungal viability. As for the mycelium time of fragmentation, the literature indicates values in the range of 1 to 60 s for ECM fungi [11,12,23,25,27]. Besides the objectives of each particular situation, this time span is related to the equipment used and the mycelium resistance to fragmentation. In many of the published literature, fragmentation is an issue when dealing with mycelium viability losses. Furthermore, adequate fragmentation periods are very important for the growing kinetics and for biomass encapsulation. Large fragments may obstruct the encapsulating dispositive and cause uneven distribution of mycelium in the beads. Even though fragmentation may destroy a portion of the active mycelium, its negative effects are far lower from those caused by the absence of charcoal in the medium. Furthermore, fragmentation is important because it breaks hyphal chunks apart, minimizing, consequently, the limitation of nutrients by diffusion. In this study, we found that fragmentations of 20s in a blender set at 3600rpm were sufficient to produce very fine suspensions, ideal for all isolates tested, both for the bioreactor inoculum as for the inoculants formulations. Other fragmentation times should be tested for other fungal isolates. Nevertheless, every time the mycelium is fragmented, charcoal should be used to minimize the potential negative effects of fragmentation.
One important observation in relation to the encapsulation is that the concentration and volume of calcium chlorite used in the process should be consistently checked in order to avoid or reduce the accumulation of residues in the final product. In their seminal work, Maupérin et al. [12] did not clearly discuss the effects caused by CaCl2 in ECM fungi. However, other studies, such as that of Kernaghan et al. [28], clearly show the toxic effect that CaCl2 has on ECM fungi. These authors have shown that a concentration of 30%, lower than the one proposed by Mauperin et al. [12], and a rinsing period of 2 minutes for beads, were sufficient to propitiate starting viability of 100% for all fungi tested. Such procedure did not cause de-polymerization of beads as observed when other salts were tested.
Aside from being languid, potentially increasing chances for contamination, and costlier, SSF presented much higher practical cultivation difficulties when compared to submerged cultivation. Washing the mycelium produced via SSF was difficult, considerably increasing the risks for contamination. Another negative point related to SSF was the separation and consequent loss of peat particles, removing, together with them, considerable quantities of biomass. All difficulties put aside, mycelium washing was very important for the maintenance of viability, propitiating a more homogeneous humidity distribution. Non-washed inoculants presented large humidity variations, culminating with viability losses.
In conclusion, even though washing is important for the humidity of the inoculant, it can also cause biomass losses due to the physical treatment suffered by the samples, varying in severity according to the morphology of each fungal isolate and the level of hyphal adhesion to the substrate. The biological viability of inoculants obtained via SSF is favored by the absorption of metabolites and the stabilization of pH promoted by the peat and vermiculite, aside from the physical protection of the mycelium promoted by the vermiculite layers [29,30]. Thus, inoculants obtained under such conditions may fit well for commercialization since they will attend the market specifications.
Alves et al. [21] and Souza et al. [22] have obtained encouraging results when testing the infectivity and efficiency of mycorrhizal inoculants produced via SSF in the colonization and growth of plants. The inoculant tested by the former authors has been stored under refrigeration for more than six months prior to use. Although viability for inoculants obtained via SSF was high, it was even higher for those produced in submerged cultivation. One possible explanation for this higher viability lays on the fact that in submersion, while in the bioreactor, the shorter period to produce the mycelium maintains the levels of ATP high [25], a phenomenon responsible for the potentially stronger capability of colonizing the plant roots [31,32]. Studies have shown that cultivation periods above 20 days cause a considerable reduction in the ATP levels in the mycelium and, as a consequence, in viability. Lapeyrie & Bruchet [25] have shown that aside from the time of cultivation, the levels of ATP are also related to the fungal growth phase, in which higher levels of energy correspond to higher growth rates. Maximum viability was obtained for cultivation periods inferior to 20 days. This demonstrates that any attempt to produce commercial ECM inoculant must observe this three-week time frame. Observing this condition will guarantee high ATP levels and, therefore, inoculant viability.
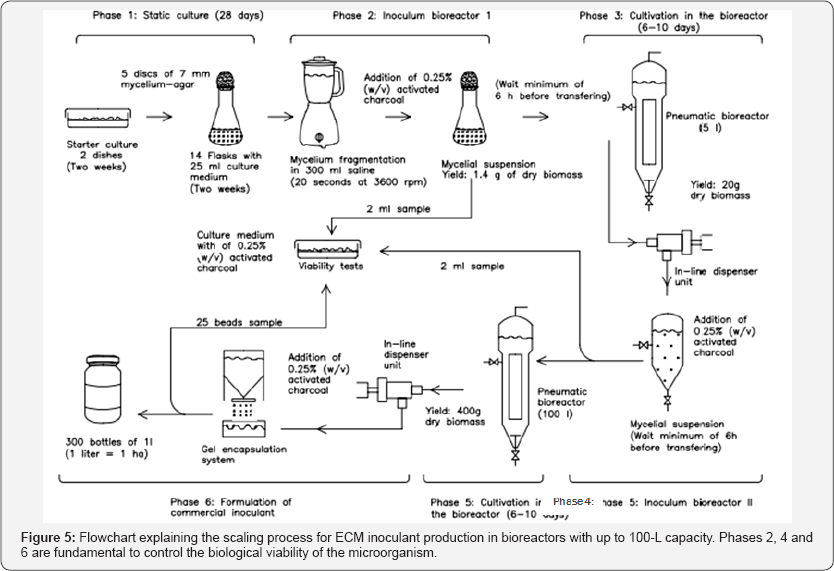
Figure 5 shows a flowchart for scheduling the production of ECM fungi inoculants to bioreactors with 100-L capacity, capable of producing a batch inoculant in sufficient amount to inoculate 300 hectares of land (≈480 thousand trees). Cultivation for 6 to 10 days in the bioreactor meet the different growth rates observed for ECM fungi (unpublished data), considering an initial inoculum concentration above 0.25g/L and the culture medium with 10g/L glucose. The use of pneumatic bioreactors and charcoal in the steps indicated previously is critical to the success of the process [33]. Fragmentation is crucial for scaling up, and it should be done using in line dispersing units, such as the Z42 model, manufactured by Ystral GmbH, which has a 2mm stator slot and a X40/38 drive. Low growth rates of ECM fungi, the need for axenic conditions, associated to large volumes, creates inherent risks for contamination, and, therefore, scaling to higher volumes is not recommended [34].
Acknowledgement
The authors gratefully acknowledge the Fundaêão de Amparo à Pesquisa e Inovaêão do Estado de Santa Catarina (FAPESC), and the Brazilian Conselho Nacional de Ciência e Tecnologia(CNPq) for supporting this work. Francisco Nascimento acknowleges a Ph.D fellowship (SFRH/BD/ 86954/2012) from Fundaêão de Ciencia e Tecnologia, Portugal.
References
- Marx DH (1969) The influence of ectotrophic mycorrhizal fungi on the resistence of pine roots to pathogenic fungi and soil bacteria. Phytopatol 59: 153-163.
- Oliveira LP, Rossi MJ, Furigo JA, Silva Filho GN, Oliveira VL (2006) Viability and infectivity of an ectomycorrhizal inoculum produced in an airlift bioreactor and immobilized in calcium alginate. Braz J Microbiol 37: 251-255.
- Siddiqui ZA, Kataoka R (2011) Mycorrhizal inoculants: progress in inoculant production technology. In: Microbes and Microbial Technology. In: Ahmad I, Ahmad F, Pichtel J (Eds.), Agricultural and Environmental Applications, Springer, New York, USA, pp. 489-506.
- Rossi MJ, Furigo A, Oliveira VL (2007) Inoculant production of ectomycorrhizal fungi by solid and submerged fermentations. Food Technol Biotechnol 45: 277-286.
- Le Tacon F, Jung G, Mugnier J, Michelot P, Mauperin C (1985) Efficiency in a forest nursery of an ectomycorrhizal fungus inoculum produced in a fermenter and entrapped in polymeric gels. Can J Bot 63: 1664-1668.
- Kuek C, Tommerup IC, Malajczuck N (1992) Hydrogel bead inocula for the production of ectomycorrhizal eucalyptus for plantations. Mycol Res 96: 273-277.
- Rossi MJ, Cardoso Cortez DH, Giachini AJ, Oliveira VL, Furigo JA (2015) Evaluation of different pre-setting conditions in airlift bioreactor to determine the respiratory kinetics of fungi. J Adv Biotechnol 5: 625-633.
- Rossi MJ, Nascimento FX, Giachini AJ, Oliveira VL, Furigo JA (2016) Airlift bioreactor fluid-dynamic characterization for the cultivation of shear stress sensitive microorganisms. J Adv Biotechnol 5: 640-651.
- Duarte Filho PFM (2009) Estudos sobre o crescimento e a viabilidade de fungos ectomicorrizicos em cultivo submerso. Dissertation. Universidade Federal de Santa Catarina, Brazil.
- Rossi MJ, Nascimento FX, Giachini AJ, Oliveira VL, Furigo JA (2016) Transfer and consumption of oxyfen during the cultivation of the ectomycorrhizal fungus Rhizopogon nigrescens in an airlift bioreactor. App Microbiol Biotechnol 101(3): 1013-1024.
- Kuek C (1996) Shake-flask culture of Laccaria laccata, an ectomycorrhizal basiodiomycete. Appl Microbiol Biotechnol 45: 319-326.
- Mauperin CH, Mortier F, Garbaye J, Le Tacon F, Carr G (1987) Viability of an ectomycorrhizal inoculum produced in a liquid medium and entrapped in a calcium alginate gel. Can J Bot 65: 2326-2329.
- Rodrigues LS, Kasuya MC, Borges AC (1999) Viability of ectomycorrhizal fungus mycelium entrapped in calcium alginate gel. Mycorrhiza 8(5): 263-266.
- Herrmann S, Oelmuller R, Buscot F (2004) Manipulation of the onset of ectomycorrhiza formation by indole-3-acetic acid, activated charcoal or relative humidity in the association between oak microcuttings and Piloderma croceum : influence on plant development and photosynthesis. J Plant Physiol 161: 509-517.
- http://webdrm.cpqba.unicamp.br/cbmai/
- Souza LAB (2003) Seleçâo de fungos ectomicorrizicos eficientes para promoçâo do crescimento de Eucalyptus dunnii Maiden. Dissertation. Universidade Federal de Santa Catarina. Florianopolis, Brazil.
- Oliveira LP (2004) Produçâo de inoculante, seleçâo e aplicaçâo de fungos ectomicorrizicos em mudas de Pinus taeda L. Dissertation. Universidade Federal de Santa Catarina, Florianopolis, Brasil.?
- Marx DH, Cordell CE (1989) The use of specific ectomycorrhizas to improve artificial forestation practices. In: Whipps JM, Lumsden RD (Eds.), Biotechnology of fungi improving plant growth, New York, USA, pp. 1-25.
- Rossi MJ, Oliveira VL (2011) Growth of the ectomycorrhizal fungus Pisolithus microcarpus in different nutritional conditions. Braz J Microbiol 42(2): 624-632.
- Teng WL (1997) Activated charcoal affects morphogenesis and enhances sporophyte regeneration during leaf cell suspension culture of Platycerium bifurcatum. Plant Cell Reports 17: 77-83.
- Alves J, Souza O, Podlech P, Giachini AJ, Oliveira Vl (2001) Efeito de inoculante ectomicorrízico produzido por fermentado semi-sólida sobre o crescimento de Eucalyptus dunnii Maiden. Pesq Agrop Bras 36: 307-313.
- Souza LAB, Silva Filho GN, Oliveira VL (2004) Eficiencia de fungos ectomicorrízicos na absorfäo de fósforo e na promofäo do crescimento de eucalipto. Pesq Agrop Bras 39: 349-355.
- Rossi MJ, Souza JAR, Oliveira VL (2002) Inoculum production of the ectomycorrhyzal fungus Pisolithus microcarpus in an airlift bioreactor. App Microbiol Biotechnol 59(2-3): 175-181.
- Thomson BD, Grove TS, Malajczuk N (1994) The effectiveness of ectomycorrhizal fungi in increasing the growth of Eucalyptus globulus Labill. in relation to root colonization and hyphal development in soil. New Phytol 126(3): 517-524.
- Lapeyrie FF, Bruchet G (1985) Some factors influencing viability of ectomycorrhizal fungal inoculum. New Phytol 100: 585-593.
- Iwase K (1992) Gluconic acid synthesis by the ectomycorrhizal fungus Tricholoma robustum. Can J Bot 70(1): 84-88.
- Pradella JC, Zuccolo M, Lopes SAR, Oliveira MS (1991) Pisolithus tinctorius vegetative mycelia production: effects of nitrogen sources and cultivation in stirred tank fermenter. Rev Microbiol 22: 7-11.
- Kernaghan G, Hambling B, Fung M, Khasa D (2002) In vitro selection of borel ectomycorrhizal fungi for use in reclamation of saline alkaline habitats. Restor Ecol 10: 43-51.
- Marx DH, Zak B (1965) Effect of pH on mycorrhizal formation of slash pine in aseptic culture. For Sci 11: 66-75.
- Hacskaylo E (1973) Carbohydrate physiology of ectomycorrhizae. In: Marks AC, Kozlowsky TT (Eds.), Ectomycorrhizae: Their ecology and physiology. Academic Press, London, UK, pp. 207-230.
- Littke WR, Bledsoe CS, Nadkarni M, Cordell CE (1980) Survival and growth of pine seedlings with Pisolithus ectomycorrhizae after two years on reforestation sites in North Carolina and Florida. For Sci 23: 363-373.
- Le Tacon F, Jung G, Mugnier J, Michelot P (1983) Efficacité en pépinière forestière d'un inoculum de champignon ectomycorhizien produit en fermenteur et inclus dans une matrice de polymères. Ann Sci For 40: 165176.
- Rossi MJ (2006) Tecnologia para produçao de inoculantes de fungos ectomicorrizicos utilizando cultivo submerso em biorreator airlift. Dissertation. Universidade Federal de Santa Catarina, Brazil.
- Oliveira VL, Oliveira, LP, Rossi MJ (2010) Ectomicorrizas no Brasil: biologia e nutriçao de plantas. In: Micorrizas: 30 anos de pesquisas no Brasil. In: Siqueira JO, Souza FA, Cardoso, EJBN, Tsai SM (Eds.), Lavras- MG: Editora UFLA.






























