Plant Growth Promoting Bacteria: Biodiversity and Multifunctional Attributes for Sustainable Agriculture
Ajar Nath Yadav1*, Priyanka Verma2, Bhanumati Singh3, Vinay Singh Chauahan3, Archna Suman4 and Anil Kumar Saxena5
1Department of Biotechnology, Akal College of Agriculture, Eternal University, India
2Department of Microbiology, Akal College of Basic Science, Eternal University, India
3Department of Biotechnology, Bundelkhand University, India
4Division of Microbiology, Indian Agricultural Research Institute, India
5ICAR-National Bureau of Agriculturally Important Microorganisms, India
Submission: May 24, 2017; Published: August 24, 2017
*Corresponding author: Ajar Nath Yadav, Department of Biotechnology, Akal College of Agriculture, Eternal University, India,Tel: +91-9882545085; Email: ajarbiotech@gmail.com
How to cite this article: Ajar N Y, Priyanka V, Bhanumati S, Vinay S C, Archna S, Anil K S. Plant Growth Promoting Bacteria: Biodiversity and Multifunctional Attributes for Sustainable Agriculture. Adv Biotech & Micro. 2017; 5(5): 555671. DOI: 10.19080/AIBM.2017.05.5556671
Abstract
The use of plant growth promoting bacteria may prove useful in developing strategies to facilitate plant growth under normal as well as diverse abiotic stress conditions. The application of microbes with the aim of improving nutrients availability for plants is an important practice and necessary for sustainable agriculture. During the past couple of decades, the use of microbial inoculants for sustainable agriculture has increased tremendously in various parts of the world. Significant increases in growth and yield of agronomically important crops in response to inoculation with plant growth promoting (PGP) microbes have been repeatedly reported. The actual biodiversity of PGP microbes belong to different groups including Actinobacteria, Bacteroidetes, Balneolaeota Firmicutes, Proteobacteria and Spirochaetes. PGP bacteria are naturally occurring soil bacteria that aggressively colonize plant roots and benefit plants by providing growth promotion either directly by solubilization of phosphorus, potassium and zinc; production of indole acetic acids, gibberellic acid, cytokinin; biological nitrogen fixation or in-directly by production of ammonia, hydrogen cyanide, siderophore and biocontrol against different plant pathogens. In this review, we have discussed method of isolation, characterization, identification and biodiversity of bacteria associated with crops and further mechanisms of plant growth promotion under the normal as well as diverse abiotic stress conditions.
Keywords: Abiotic stress; Biodiversity; Crop improvement; Plant growth promotion; Sustainable agriculture
Introduction
To provide food security to the ever increasing population greater agriculture production is a pressing need in 21st century. The increasing demand for a steady and healthy food supply by a burgeoning human population will require efficient management practices along with controlling disease that reduce crop yield. During last few decades, agricultural production has increased due to the use of high yielding varieties and enhanced consumption of chemicals, which are used both as fertilizers to provide nutrition and as protection agents to control the damage caused by phytopathogens. Excessive use of chemicals and change in traditional cultivation practices has resulted in the deterioration of physical, chemical and biological health of the cultivable soil.
Globally need to increase agricultural production from a steadily decreasing and degrading land resource base has placed significant strain on the delicate agro-ecosystems. Current strategies to maintains and enhanced agricultural productivity via high input practices places considerable emphasis on 'failsafe' techniques for each component of the production sequence with little consideration to the integration of these components in a holistic, systems approach. The use of chemical fertilizers is considered the quickest and surest way of boosting crop production, their cost and other constraints deter farmers from using them in recommended quantities. Various studies report the projections of impact of climate change on different crops production under different simulation models in different continents [1,2]. Under various climate change scenarios the major influencing factor is availability and management of soil water reserves of winter wheat, especially in frequently occurring extreme events such as heat waves and droughts with decrease in annual soil and groundwater recharge [3-6]. However, it is expected that future climate change will affect wheat yields. This is due to the opposing effects of temperature, salt, pH and drought.
Microbial diversity in soil is considered important for maintaining for the sustainability of agriculture production systems. However, the links between microbial diversity and ecosystem processes is not well understood. Region of contact between root and soil where soil is affected by roots is designated as "rhizosphere". The rhizosphere is the zone of soil influenced by roots through the release of substrates that affect microbial activity. The rhizoplane is the surface, including the strongly adhering soil particles. Root colonizing bacteria that exert beneficial effect on plant development via direct or indirect mechanisms have been defined as plant growth promoting bacteria [7-10]. The concept of plant growth promoting bacteria is now well established both for growth promotion and biocontrol. Plant growth promoting bacteria were first defined by Kloepper & Schroth [11] to describe soil bacteria that colonize the roots of plants following inoculation onto seed and they enhance plant growth. The ineffectiveness of PGP bacteria in the field has often attributed to their inability to colonize plant roots [12-14]. The crops associated bacterial has been extensively investigated in the past few years with a focus on culture dependent techniques and PGP bacteria have been reported e.g. Agrobacterium, Allorhizobium, Arthrobacter, Azospirillum, Azotobacter, Bacillus, Bradyrhizobium, Burkholderia, Caulobacter, Chromobacterium, Erwinia, Exiguobacterium, Flavobacterium, Mesorhizobium, Micrococcous, Providencia, Pseudomonas, Rhizobium andSerratia [6,15-17]. In addition to understanding the mechanisms of the interaction between plants and microorganisms, colonization mechanisms and strategies represent an important aspect of the interaction. Successful colonization of a PGPB inoculants strain is a requirement to promote plant growth or soil health. This review focuses on bacterial biodiversity, its characterization and mechanisms of PGP under normal as well as abiotic stress conditions.
Isolation and characterization of plant growth promoting bacteria
The microbial communities associated with crops growing in diverse abiotic stress condition have been received much attention, because the food security to the ever increasing population greater agriculture production is a pressing need in 21st century. The microbes associated the crops can be characterized using culture-dependent techniques. The microbes could be isolated using enrichment and serial dilution methods followed by spread or pour plates technique [18]. The rhizospheric, endophytic and phyllospheric microbes can be isolated using standard method of serial dilution, surface sterilization and leaf imprinting techniques [19]. The different specific and selective growth mediums should be used to isolate the maximum possible culturable morphotypes different genera such as Arthrobacter (trypticase soy agar), BBDG (T3 agar), Methylobacterium (Ammonium mineral salt), N2 -fixing bacteria (Jensen N2-free agar), Pseudomonas spp. (Kings' B agar), Rhizobium (Congo red yeast mannitol) and soil-specific actinobacteria (soil extract agar) [20]. The isolated microbes should be screened for tolerances to different abiotic stresses of salinity, pH, temperature and drought [21].
To know the PGP ability and other potential application of bacteria should be screened qualitatively for direct-PGP attributes which included solubilization of phosphorus [22], potassium [23] and zinc [24]; production of phytohormones indole-3-acetic acid [25], gibberellic acid [26]; biological N2- fixation [27] and 1-aminocyclopropane-1-carboxylate (ACC) deaminase [28]. The bacteria should be also screened for qualitatively in-direct PGP attributes which included production of ammonia [29], HCN [30], siderophore [31], lytic enzyme [32] and biocontrol against different fungal pathogens [33]. After qualitatively screening the selected bacteria with PGP attributes should be quantitatively screened for N2-fixing attributes by using the acetylene reduction assay (ARA) [34], P-solubilization [35] K-solubilization [9] and IAA production [36].
For the molecular characterization, the genomic DNA should be isolated from purified pelleted microbial cells of 1.5mL broth should be washed 2-3 times in 1.0mL of TE Buffer (10mM Tris HCl and 1mM EDTA pH 8.0). Microbial lysis should be performed using 0.5mL SET buffer (75mM NaCl, 25mM EDTA and 20mM Tris) with 10μL of lysozyme (10mgmL-1) for 30min at 37 °C and 10% SDS with 20mgmL-1 proteinase K for 1h at 55 °C. DNA should be extracted using phenol/chloroform/isoamyl alcohol and aqueous phase can be transferred to a fresh tube. Finally, the washed DNA pellet should be incubated at 37 °C for 25-30min to completely remove ethanol and then resuspended in 50μL of TE buffer. The amount of DNA extracted should be electrophoresed on 0.8% agarose gel [37]. The primers universal 16S rRNA should be used for the amplification of conserved genes. The PCR amplified 16S rRNA should be purified with a Quiaquick purification kit (Qiagen). The partial 16S rRNA gene sequences should be compared with those available in the NCBI databases. Identification at the species level was determined using a 16S rRNA gene sequence similarity of ≥97% with that of a prototype strain sequence in the GenBank. Sequence alignment and comparison can perform, using the program CLUSTAL W. The phylogenetic tree can be constructed on the aligned datasets using the neighbor-joining method implemented in the program MEGA 4.0.2 [38]. A schematic representation of the isolation, characterization and identification of plant growth promoting bacteria have presented in Figure 1.
Plant-microbe interactions
Plant-microbe interactions may be beneficial or harmful, depending on the characteristics of the bacteria involved and the ways in which they interact with plants. Among such microbes, PGP bacteria are distributed on plant roots or in the surrounding soil and have beneficial effects on plants [13]. PGP microbes may promote plant growth, thus providing high crop yields, and they also function as biocontrol agents against plant diseases caused by phytopathogenic microbes. Moreover, recent studies indicate that PGP microbes are able to boost plant tolerance to abiotic stresses such as salinity, temperature, pH and drought [5,9,39]. The PGP bacteria with multifunctional attributes can be used to replace chemical fertilizers and pesticides that are agents of pollution.


PGP bacteria produce a wide range of metabolites that regulate cell content according to ambient biotic and abiotic stresses e.g. some produce hormones such as indole acetic acids (IAAs), ethylene, and gibberellins that enhance plant growth, seed germination, and root growth. Rhizosphere bacteria are also able to fix nitrogen symbiotically and to solubilize mineral phosphates, potassium and zinc. Plant growth promotion by PGP bacteria requires a close relationship between the bacteria and their host plants. This interaction may be recognized as rhizospheric or endophytic [40,41]. During the PGP microbial colonization process, bacteria first occupy the rhizosphere. Endophytes are then able to enter plant tissues through the root zone, after which they penetrate plant cells, often conferring beneficial effects on hosts [41]. Biological control is achieved through mechanisms such as parasitism, competition and antibiosis which adversely affect the fitness, survival and reproduction of nematodes (Figure 2).
Biodiversity of plant growth promoting bacteria
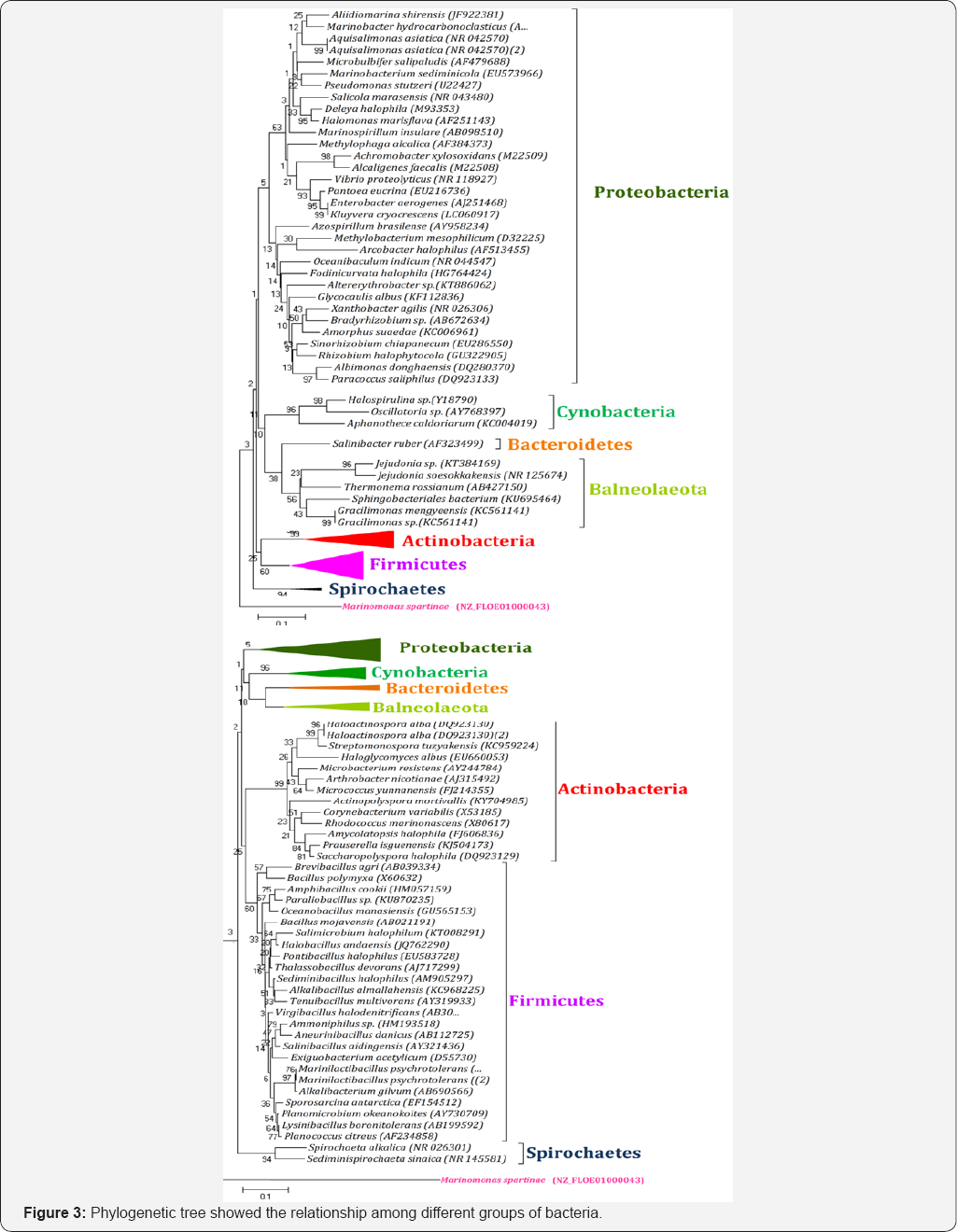
Biodiversity is defined as the variety of living organisms at species, inter-species and intra-species level in ecology. Microbes are quite beneficial and exploited in gain of quite lot revenue for sustainable agriculture and human health. Microbial diversity in rhizospheric soil ecosystem exceeds more than eukaryotic organisms. One gram of soil may harbor up to 10billion microbiomes of possible thousands of different species, and thus the rhizosphere of any plant is hot spot of biodiversity of microbes (archaea, bacteria and fungi). On review of different research on biodiversity of microbes associated with crops, it was found that microbes belonged to different phylum mainly: Actinobacteria, Bacteroidetes, Balneolaeota, Basidiomycota, Cyanobacteria, Firmicutes, Proteobacteria and Spirochaetes (Figure 3). Among all reported phylum the members of phylum Firmicutes were most dominant followed by Proteobacteria (Figure 4). The Proteobacteria were further grouped as α, β, and γ-proteobacteria. These microbes been reported from cereal crops (Wheat, rice, maize, sugarcane, chick pea etc.) [17,27,42-45]. The microbes have beneficial ecological significance as they have been reported as common as well as niche or host-specific. The niche of host-specific microbes may play important role in vegetation of specific plants in different abiotic stress regions.
Actinobacteria
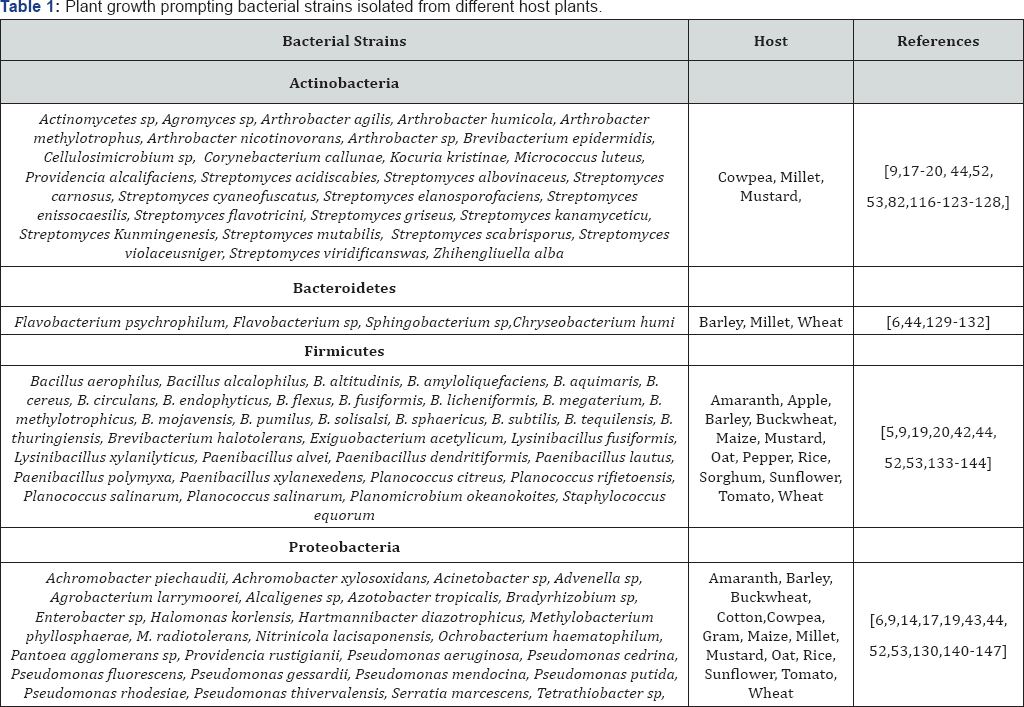
Actinobacteria are of agriculturally important as they can enhance plant growth; improve plant nutrition through direct plant growth promoting. Several studies have indicated that Actinobacteria colonization can also result in increased plant vigor, and it confers tolerance to biotic and abiotic stresses, enhanced drought tolerance, and improved nutrients utilization [36,46]. On review diversity of actinobacteria associated with different crops all 6-classes have been reported, whose phylogenetic investigations have been carried out mainly by using 16S rRNA sequences. On review of different crops, it was found that microbes were most predominant and studied belong to all six classes in which many bacteria like Arthrobacter, Brevibacterium, Cellulomonas, Corynebacterium, Kocuria, Microbacterium, Micrococcus, Mycobacterium, Rhodococcus and Streptomyces have been reported from all crops reviewed.
In rhizospheric soil, Actinobacteria represents a high proportion of the microbial biomass. Their populations are found at 106-109 bacteria g-1 and they represent more than 30% of total population of soil microbiomes. Among different genera of actinobacteria two genera namely Streptomyces and Nocardia have been well represented in rhizospheric soil. It has been reported that Streptomyces may be represent about >95% of the whole soil Actinobacterial microbiomes. The members of Actinobacteria play a major role in agricultural soil quality and soil fertility. Actinobacteria represents a large fraction of microbiomes in the root systems and is well established that they are dominant fraction of the microbial community in soils of wild and agricultural plant species [47-51]. Together with other phyla, the members of actinobacteria account for a large proportion in the rhizosphere of numerous plants including wheat (Triticum aestivum) [9,17,44,52,53]; rice (Oryza sativa) [48,54,55]; maize (Zea mays) [50,56,57]; Sugarcane (Saccharum officinarum) [58-60]; soybean (Glycine max) [51,61-63]; pea (Pisum sativum) [51,64,65]; sunflower (Helianthus annuus) [49,66,67] and chickpea (Cicer arietinum) [68,69].
Firmicutes
The member Bacillus and Bacillus derived genera (BBDG) belonged to phylum Firmicutes are associated with different plant, show different plant growth promoting attributes. Among bacilli, Bacillus and Paenibacillus are the most dominant genera followed by Alicyclobacillus, Aneurinibacillus, Virgibacillus, Salibacillus, and Gracilibacillus reported from different crops plants [32,33]. Garbeva et al. [70] showed that the majority (95%) of Gram-positive bacteria in soils under different types of management regimes (permanent grassland, grassland turned into arable land, and arable land), were putative Bacillus species; B. mycoides, B. pumilus, B. megaterium, B. thuringiensis, and B. firmus, as well as related taxa such as Paenibacillus, were frequently identified by sequencing the DNA bands obtained on DGGE gels. Bacteria of the genus Bacillus Cohn are widely dispersed in nature, easy to multiply, have a long shelf life when sporulated and are nonpathogenic. B. subtilis, B. mycoides, B. pumilus, B. megaterium, B. thuringiensis and B. firmus are wide range of Bacillus present in rhizosphere soil [9,17,52,53].
Verma et al. [52], investigated forty one endophytic bacteria were isolated from surface-sterilized roots and culms of wheat var. HS507, growing in NW Indian Himalayas. These bacteria were screened in vitro for multifarious plant growth promoting attributes such as solubilization of phosphorus, potassium, zinc; production of indole acetic acids, hydrogen cyanide, gibberellic acid, siderophore and activities of nitrogen fixation, ACC deaminase and biocontrol against Rhizoctonia solani and Macrophomina phaseolina at low temperature (4 °C). One isolate IARI-HHS2-30, showed appreciable level of potassium solubilization was further characterized in vivo at control condition of low temperature. Based on 16S rDNA sequence analysis, this isolate was identified as Bacillus amyloliquefaciens assigned accession number KF054757. Analysis of the phylogenetic characterization showed close homology with typical psychrotolerant bacteria Bacillus amyloliquefaciens, B. methylotrophicus, B. polyfermenticus, B. siamensis, B. subtilis, and B. vallismortis. Endophytic nature and plant growth promoting ability of IARI-HHS2-30 was tested by qualitatively and followed by inoculation onto wheat seedlings in low temperature conditions. At 30 days after inoculation, Bacillus amyloliquefaciens IARI-HHS2-30 to wheat plants resulted in significant increase in root/shoot length, fresh weight, and chlorophyll a content. Plant growth promoting features coupled with psychrophilic ability suggest that this endophytic bacterium may be exploited as bio-inoculants for various crops in low temperature and high altitude condition.
Yadav et al. [33], reported and characterized psychrotrophic Bacilli from different sites in north western Indian Himalayas. A total of 247 morphotypes were obtained from different soil and water samples and were grouped into 43 clusters based on 16S rDNA-RFLP analysis. Sequencing of representative isolates from each cluster led to their identification and 43 Bacilli belonged to different species ofeleven genera viz. Desemzia, Exiguobacterium, Jeotgalicoccus, Lysinibacillus, Paenibacillus, Planococcus, Pontibacillus, Sinobaca, Sporosarcina, Staphylococcus and Virgibacillus. With an aim to develop microbial inoculants that can perform efficiently at low temperatures, all representative isolates were screened for different plant growth promoting traits at low temperatures (5-15 °C). Among the strains, variations were observed for production of ammonia (22%), indole-3-acetic acid (20%), siderophores (8%), gibberellic acid (4%) and hydrogen cyanide (3%); solubilization of phosphate (12%), zinc (16%) and potassium (8%); 1-aminocyclopropane-1-carboxylate deaminase activity (5%) and biocontrol activity (19%) against Rhizoctonia solani and Macrophomina phaseolina. Among all the strains Bacillus licheniformis, Bacillus muralis, Desemzia incerta, Paenibacillus tylopili and Sporosarcina globispora were found to be potent candidates to be developed as inoculants as they exhibited multiple PGP traits at low temperature.
Verma et al. [9] investigated culturable Bacilli in six wheat cultivating agro-ecological zones of India viz: northern hills, north western plains, north eastern plains, central, peninsular and southern hills. These agro-ecological regions are based on the climatic conditions such as pH, salinity, drought and temperature. A total of 395 Bacilli were isolated by heat enrichment and different growth media. Phylogenetic analysis based on 16S rRNA gene sequencing led to the identification of 55 distinct Bacilli that could be grouped in five families, Bacillaceae (68%), Paenibacillaceae (15%), Planococcaceae (8%), Staphylococcaceae (7%) and Bacillales incertae sedis (2%), which included eight genera namely Bacillus, Exiguobacterium, Lysinibacillus, Paenibacillus, Planococcus, Planomicrobium, Sporosarcina and Staphylococcus. All 395 isolated Bacilli were screened for their plant growth promoting attributes, which included direct-plant growth promoting (solubilization of phosphorus, potassium and zinc; production of phytohormones; 1-aminocyclopropane-1-carboxylate deaminase activity and nitrogen fixation), and indirect-plant growth promotion (antagonistic, production oflytic enzymes, siderophore, hydrogen cyanide and ammonia). To our knowledge, this is the first report for the presence of Bacillus endophyticus, Paenibacillus xylanexedens, Planococcus citreus, Planomicrobium okeanokoites, Sporosarcina sp. and Staphylococcus succinus in wheat rhizosphere and exhibit multifunctional PGP attributes. These niche-specific and multifarious PGP Bacilli may serve as inoculants for crops growing in respective climatic conditions.
Proteobacteria
The phylum proteobacteria are a major group of Gram- negative bacteria which included α/β/γproteobacteria. The γ-proteobacteria is the largest class in terms of species Pseudomonas and Azotobacter. Azotobactor is a gram-negative, aerobic, heterotrophic, rod shaped nitrogen fixing bacteria present in alkaline and neutral soils. They are free living organism present in soil, water and also in association with some plants [71-74]. Various species of Azotobacter such as A. agilis, A. chrococcum, A. beijerinckii, A. vinelandii, A. ingrinis has been reported from different crops. Among proteobacteria most dominant and plant growth promoting genera belong to Pseudomonas. Pseudomonas is a genus of Gram-negative, aerobic γ-proteobacteria, belonging to the family Pseudomonadaceae and containing 191 validly described species. The certain members of the Pseudomonas genus have been applied to cereal seeds or applied directly to soils as a way of preventing the growth or establishment of crop pathogens. The biocontrol properties of P. fluorescens and P. protegens are currently best-understood, although it is not clear exactly how the plant growth-promoting properties of P. fluorescens are achieved. Other notable Pseudomonas species with biocontrol properties include P. chlororaphis, which produces a phenazine-type antibiotic active agent against certain fungal plant pathogens [75].
Verma et al. [20], reported 135 wheat associated plant growth promoting bacteria from acidic soil, among all isolates Pseudomonas chlororaphis IARI-THD-13, Pseudomonas fluorescens IARI-THD-21, Pseudomonas rhodesiae IARI-THD-11 and Pseudomonas rhodesiae IARI-THD-28 exhibited direct and indirect plant growth promoting attributes. Verma et al. [44], investigated thermotolerant wheat associated plant growth promoting bacteria which has been identified using 16S rRNA gene sequencing. Bacillus and Pseudomonas were dominant in rhizosphere while Methylobacterium were in phyllosphere. Different species of Pseudomonas fuscovaginae IARI-IIWP-29, Pseudomonas lini IARI-IIWP-33, Pseudomonas monteilii IARI-IIWP-27, Pseudomonas stutzeri IARI-IHD-4 and Pseudomonas thivervalensis IARI-IHD-3 have been sort out from wheat as endophytic, rhizospheric as well as epiphytic. Verma et al. [19], reported psychrotolerant wheat associated bacteria from northern hills zone of India. A total 247 bacteria were isolated from five different sites. 16S rRNA gene based phylogenetic analysis, revealed that 65, 26, 8 and 1% bacteria belonged to four phyla namely Proteobacteria, Firmicutes, Actinobacteria and Bacteroidetes respectively. Overall 28% of the total morphotypes belonged to Pseudomonas followed by Bacillus (20%), Stenotrophomonas (9%), Methylobacterium (8%), Arthrobacter (7%), Pantoea (4%), Achromobacter, Acinetobacter, Exiguobacterium and Staphylococcus (3%), Enterobacter, Providencia, Klebsiella and Leclercia (2%), Brevundimonas, Flavobacterium, Kocuria, Kluyvera and Planococcus (1%).
Mechanisms of plant growth promotion
Plants play an important role in selecting and enriching the types of bacteria by the constituents of their root exudates. Thus, depending on the nature and concentrations of organic constituents of exudates, and the corresponding ability of the microbes to utilize these as sources of energy, the microbial community have been developed. Microbes associated with crops are of agriculturally important as they can enhance plant growth; improve plant nutrition through biological N2-fixation and other mechanisms. The growth stimulation by microbes can be a consequence of biological N2-fixation, production of phytohormones, such as IAA and cytokines; biocontrol of phytopathogens through the production of antifungal or antibacterial agents, siderophore production, nutrient competition and induction of acquired host resistance or enhancing the bioavailability of minerals (Table 1).
Indole-3-acetic acid production
Plant hormones are chemical messengers that affect a plant's ability to respond to its environment. Hormones are organic compounds that are effective at very low concentration; they are usually synthesized in one part of the plant and are transported to another location. They interact with specific target tissues to cause physiological responses, such as growth or fruit ripening. Each response is often the result of two or more hormones acting together. Because hormones stimulate or inhibit plant growth, many botanists also refer to them as plant growth regulators. Researchers recognize five major groups of hormones: auxins, gibberellins, ethylene, cytokinins, and abscisic acid [14,76].
IAA (indole-3-acetic acid) is the member of the group of phytohormones and is generally considered the most important native auxin. It functions as an important signal molecule in the regulation of plant development including organogenesis, tropicresponses, cellular responses such as cell expansion, division, and differentiation, and gene regulation. Diverse bacterial species possess the ability to produce the auxin phytohormone IAA [77-79]. Different biosynthesis pathways have been identified and redundancy for IAA biosynthesis is widespread among plant-associated bacteria. Interactions between IAA-producing bacteria and plants lead to diverse outcomes on the plant side, varying from pathogenesis to phytostimulation. Verma et al. [19], biodiversity of wheat- associated bacteria from the northern hills zone of India was deciphered. A total of 247 bacteria were isolated from five different sites. Among all isolated bacteria 14% showed IAA production in which strain IARI-HHS1-3 showed highest IAA production (70.8±1.5μg mg-1 protein day-1) followed by IARI- HHS1-8 (69.1±0.5μg mg-1 protein day-1)
Tabatabaei et al. [76], have reported Pseudomonas isolated from wheat. An in vitro experiment was conducted to observe the effect of the inoculation of four indole-3-acetic acid (IAA)- producing Pseudomonas isolates and exogenous IAA on seed germination traits and α-amylase activity of durum wheat. The results showed inoculation with all bacterial isolates led to a decrease in the germination percent, although the extent of the depression varied with the isolate. A significant relationship between concentrations of bacterial IAA and the germination inhibition percent in durum wheat seeds by different bacteria strains was observed. Several plant growth promoting rhizobacteria Azotobacter sp., Rhizobium sp., Pantoea agglomerans, Rhodospirillum rubrum, Pseudomonas fluorescens, Bacillus subtilis and Paenibacillus polymyxa can produce cytokinins or gibberellins or both can produce either cytokinins or gibberellins or both for plant growth promotion. Some strains of phytopathogens can also synthesize cytokinins. However, it appears that plant growth promoting bacteria produce lower cytokinin levels compared to phytopathogens so that the effect of the plant growth promoting rhizobacteria on plant growth is stimulatory while the effect of the cytokinins from pathogens is inhibitory
Phosphate and potassium solubilization
Phosphorus (P) is major essential macronutrients for biological growth and development. Bacteria offer a biological rescue system capable of solubilizing the insoluble inorganic P of soil and make it available to the plants. The ability of some bacteria to convert insoluble phosphorus (P) to an accessible form, like orthophosphate, is an important trait in PGP bacterium for increasing plant yields. The rhizospheric phosphate utilizing bacteria could be a promising source for plant growth promoting agent in agriculture. The use of phosphate solubilizing bacteria as inoculants increases the P uptake by plants. Phosphorus (P) is major essential macronutrients for biological growth and development. P in soils is immobilized or becomes less soluble either by absorption, chemical precipitation, or both. Under such conditions, microorganisms offer a biological rescue system capable of solubilizing the insoluble inorganic P of soil and make it available to the plants. Phosphate solubilizing bacteria include largely bacteria and fungi, which can grow in media containing tricalcium, iron and aluminium phosphate, hydroxyapatite, bonemeal, rock phosphate and similar insoluble phosphate compounds as the sole phosphate source. Such microbes not only assimilate P but a large portion of soluble phosphate is released in quantities in excess of their own requirement.
Phosphate solubilization is a common trait among microbes associated with different crops. For instance, the majority of microbial populations from wheat, rice, maize, and legumes were able to solubilize mineral phosphates in plate assays and A vast number of PGP microbes with phosphate solubilizing property have been reported which include members belonging to Burkholderia, Enterobacter, Halolamina, Pantoea, Pseudomonas, Citrobacter and Azotobacter [14,52,80-82]. Possible mechanisms for solubilization from organic bound phosphate involve either enzymes namely C-P lyase, non- specific phosphatases and phytases. However, most of the bacterial genera solubilize phosphate through the production of organic acids such as gluconate, ketogluconate, acetate, lactate, oxalate, tartarate, succinate, citrate and glycolate.
Verma et al. [9], have reported 395 Bacilli from wheat and these bacteria have been screened for direct and indirect PGP traits. Of 55 representatives, 39, 18, and 40 strains exhibited solubilization of phosphorus, potassium, and zinc respectively. Among P, K, and Zn solubilizers, Paenibacillus polymyxa BNW6 solublized highest amount of phosphorus 95.6±1.0mg L-1 followed by Sporosarcina sp. BNW4 75.6±1.0mg L-1. Planococcus salinarum BSH13 (46.9±1.2mg L-1) and Bacillus pumilus BCZ15 (7.5±0.5mg L-1) solubilized highest amount of potassium and zinc respectively.
Potassium (K) is the third major essential macronutrient for plant growth. The concentrations of soluble potassium in the soil are usually very low and more than 90% of potassium in the soil exists in the form of insoluble rocks and silicate minerals. Moreover, due to imbalanced fertilizer application, potassium deficiency is becoming one of the major constraints in crop production. Without adequate potassium, the plants will have poorly developed roots, grow slowly, produce small seeds and have lower yields. This emphasized the search to find an alternative indigenous source of potassium for plant uptake and to maintain potassium status in soils for sustaining crop production [52]. Plant growth promoting bacteria are able to solubilize potassium rock through production and secretion of organic acids. Potassium solubilizing plant growth promoting rhizobacteria such as Acidothiobacillus ferrooxidans, Bacillus edaphicus, Bacillus mucilaginosus, Burkholderia, Paenibacillus sp. and Pseudomonas has been reported to release potassium in accessible form from potassium bearing minerals in soils.Thus, application of potassium solubilizing plant growth promoting bacteria as biofertilizer for agriculture improvement can reduce the use of agrochemicals and support ecofriendly crop production. K-solubilizing bacteria (KSB) were found to resolve potassium, silicon and aluminium from insoluble minerals. Among different groups of microbes, BBDG were best characterized for K-solubilization [52,83]. The K-solubilizing bacteria may have use in the amelioration of K-deficient soil in agriculture. There are only few reports on K-solubilization by endophytic bacteria isolated from wheat [9,20,53].
Biological nitrogen fixation
Nitrogen is the major limiting factor for plant growth, the application of N2-fixing microbes as biofertilizers has emerged as one of the most efficient and environmentally sustainable methods for increasing the growth and yield of crop plants. Biological nitrogen fixation (BNF) is one of the possible biological alternatives to N-fertilizers and could lead to more productive and sustainable agriculture without harming the environment. Many associative bacteria are now known to fix atmospheric nitrogen and supply it to the associated host plants. A variety of nitrogen fixing microbes like Arthrobacter, Azoarcus, Azospirillum, Azotobacter, Bacillus, Enterobacter, Gluconoacetobacter, Herbaspirillum, Klebsiella, Pseudomonas, and Serratia have been isolated from the rhizosphere of various crops, which contribute fixed nitrogen to the associated plants [84-88].
Choudhury et al. [89], reported that that the Azolla and cyanobacteria can supplement the nitrogen requirements of plants, replacing 30-50% of the required urea-N in rice production. BNF by Azotobacter, Clostridium, Azospirillum, Herbaspirillum and Burkholderia can substitute for urea-N, while Rhizobium can promote the growth physiology or improve the root morphology of the rice plant. Green manure crops can also fix substantial amounts of atmospheric N. Among the green manure crops, Sesbania rostrata has the highest atmospheric N2-fixing potential, and it has the potential to completely substitute for urea-N in rice cultivation. Gtari et al. [90], reported that non-Frankia actinobacteria has dramatically increased and has opened investigation on the origin and emergence of diazotrophy among actinobacteria. During the last decade, Mycobacterium flavum, Corynebacterium autotrophicum and a fluorescent Arthrobacter sp. have been reported to have nitrogenase activity. Verma et al. [44], isolated and characterized drought tolerant nitrogen fixing rhizospheric actinobacteria Arthrobacter humicola IARI-IIWP-42 associated with wheat growing in in central zone of India. Verma et al. [19], reported the psychrotrophic microbes from wheat rhizosphere growing in northern hills zone of India. Arthrobacter nicotinovorans IARI- HHS1-1 and Arthrobacter methylotrophus IARI-HHS1-25 fixed atmospheric nitrogen 18.25±1.2 and 9.65±1.5nmol ethylene h-1 mg-1 protein respectively. Among isolated actinobacteria, three strains were identified as Arthrobacter methylotrophus,Arthrobacter nicotinovorans and Kocuria kristinae exhibited more than six different plant growth promoting activities at low temperature.
ACC-deaminase activity
Ethylene (C2H4) an important plant-signaling molecule involves in many plant functions including seed germination, root hair development, root nodulation, flower senescence, leaf abscission and fruit ripening. Ethylene is produced by a two-step process that consists of enzymatic conversion of S-adenosyle methionine (SAM) to ACC followed by the conversion of ACC to ethylene, which is catalyzed by ACC-oxidase. Ethylene production in plants is increased upon exposure to biotic and abiotic stresses, including extreme salinity, temperature, drought, and infection by viral, bacterial and fungal pathogens. It has been investigated that certain microbes contain an enzyme ACC-deaminase which hydrolyses ACC, the precursor of ethylene into ammonia and a-ketobutyrate, thereby reducing the levels of ethylene which can inhibit plant growth. Currently, bacterial strains exhibiting ACC deaminase activity have been identified in a wide range of genera such as Acinetobacter, Achromobacter, Agrobacterium, Alcaligenes, Azospirillum, Bacillus, Burkholderia, Enterobacter, Pseudomonas, Ralstonia, Serratia and Rhizobium [19,20,33,44,52,53,91]. Ethylene is a key regulator of the colonization of plant tissue by bacteria which in turn suggests that the ethylene inhibiting effects of ACC- deaminase may be a bacterial colonization strategy. Regardless of why plant-associated bacteria produce ACC-deaminase, their application can clearly is a very useful strategy to mitigate the effects of various stressors on cultivated plants. Generally, ethylene is an essential metabolite for the normal growth and development of plants [92,93]. Under stress conditions like those generated by salinity, drought, water logging, heavy metals and pathogenicity, the endogenous level of ethylene is significantly increased which negatively affects the overall plant growth. Plant growth promoting bacteria which possess the enzyme, 1-aminocyclopropane-1-carboxylate (ACC) deaminase, facilitate plant growth and development by decreasing ethylene levels, inducing salt tolerance and reducing drought stress in plants [20,44,93,94].
Biocontrol
Phytopathogenic microbes are a major and chronic threat to sustainable agriculture and ecosystem stability worldwide subverts the soil ecology, disrupt environment, degrade soil fertility and consequently show harmful effects on human health, along with contaminating ground water. Plant growth promoting bacteria is a promising sustainable and environmentally friendly approach to obtain sustainable fertility of the soil and plant growth indirectly. This approach takes inspire a wide range of exploitation of plant growth promoting bacteria led to reducing the need for agrochemicals (fertilizers and pesticides) for improve soil fertility by a variety of mechanisms that via production of antibiotics, siderophores,HCN, hydrolytic enzymes. Phytopathogenic microorganism can control by releasing siderophores, B-1, 3-glucanase, chitinases, antibiotics, fluorescent pigment or by cyanide production. World agriculture faces a great loss every year incurred from infection by pathogenic organisms. Application of microorganism for the control of diseases seems to be one of the most promising ways. Biocontrol systems are ecofriendly, cost-efficient and involved in improving the soil consistency and maintenance of natural soil flora. To act efficiently, the biocontrol agent should remain active under large range of conditions viz., varying pH, temperature and concentrations of different ions. Biocontrol agents limit growth of pathogen as well as few nematodes and insects. Recent studies have indicated that biological control of bacterial wilt disease could be achieved using antagonistic bacteria. Different bacterial species, namely, Alcaligenes sp., Bacillus pumilus, B. subtilis, B. megaterium, Clavibacter michiganensis, Curtobacterium sp., Flavobacterium sp., Kluyvera sp., Microbacterium sp., Pseudomonas alcaligenes, P. putida, P. fluorescens have been reported as endophytes and were inhibitory to plant pathogens [19,95-100].
Iron is an essential growth element for all living organisms. The scarcity of bioavailable iron in soil habitats and on plant surfaces foments a furious competition. Under iron-limiting conditions PGPB produce low-molecular-weight compounds called siderophores to competitively acquire ferric ion. Siderophores (Greek: «iron carrier») are small, high-affinity iron chelating compounds secreted by microorganisms such as bacteria, fungi and grasses [101-104]. Microbes release Siderophores to scavenge iron from these mineral phases by formation of soluble Fe3+ complexes that can be taken up by active transport mechanisms. Many siderophores are non-ribosomal peptides, although several are biosynthesised independently. Siderophores are low molecular weight bio-molecules secreted by micro-organisms in response to iron starvation for acquisition of iron from insoluble forms by mineralization and sequestration. Although some siderophores are known to chelate other ions, their specificity and avidity for iron is the most consistent feature. Many plants can use various bacterial siderophores as iron sources, although the total concentrations are probably too low to contribute substantially to plant iron uptake. Siderophores have been implicated for both direct and indirect enhancement of plant growth by plant growth promoting bacteria. The direct benefits of bacterial siderophores on the growth of plants have been demonstrated by using radio labeled ferric siderophores as a sole source of iron showed that plants are able to take up the labeled iron by a large number of plant growth promoting bacteria including Aeromonas, Azadirachta, Azotobacter, Bacillus, Burkholderia, Pseudomonas, Rhizobium, Serratia and Streptomyces sp. [99,105-107].
Certain bacteria synthesize a wide spectrum of multifunctional polysaccharides including intracellular polysaccharides, structural polysaccharides, and extracellular polysaccharides. Production of exo polysaccharides is generally important in biofilm formation; root colonization can affect the interaction of microbes with roots appendages. Effective colonization of plant roots by EPS-producing microbes helps to hold the free phosphorous from the insoluble one in soils and circulating essential nutrient to the plant for proper growth and development and protecting it from the attack of foreign pathogens. Other innumerable functions performed by EPS producing microbes constitute shielding from desiccation, protection against stress. PGP bacteria improve plant growth by preventing the proliferation of phytopathogens and thereby support plant growth. Some PGP bacteria synthesize antifungal antibiotics, e.g. Pseudomonas fluorescens produces 2,4-diacetyl phloroglucinol which inhibits growth of phytopathogenic fungi. In recent years, fluorescent Pseudomonas has been suggested as potential biological control agent due to its ability to colonize rhizosphere and protect plants against a wide range of important agronomic fungal diseases such as black root-rot of tobacco, root-rot of pea, root-rot of wheat, damping-off of sugar beet and as the prospects of genetically manipulating the producer organisms to improve the efficacy of these biocontrol agents [108-112].
Growth enhancement through enzymatic activity is another mechanism used by plant growth promoting bacteria. Plant growth promoting bacterial strains can produce certain enzymes such as chitinases, dehydrogenase, p-glucanase, lipases, phosphatases, proteases etc. exhibit hyperparasitic activity, attacking pathogens by excreting cell wall hydrolases. Through the activity of these enzymes, plant growth promoting bacteria play a very significant role in plant growth promotion particularly to protect them from biotic and abiotic stresses by suppression of pathogenic fungi including Botrytis cinerea, Sclerotium rolfsii, Fusarium oxysporum, Phytophthora sp., Rhizoctonia solani, and Pythium ultimum [113-147].
Effects of soil bacteria on the soil ecosystem
Soil bacteria including different groups along with fungi are able to enhance the availability of different nutrients by utilizing different mechanisms. PGP abcteria are able to enhance the availability of different nutrients including N, P and micronutrients e.g. Rhizobium sp., in symbiosis with their legume host plant, and Azospirillum in non-symbiotic association with their host plant, can fix atmospheric N2 [148-153]. PGP bacteria including Arthrobacter, Bacillus and Pseudomonas are able to enhance P availability, by production of organic acids and phosphatase enzymes through producing siderophores, PGP bacteria can also increase Fe solubility and hence uptake by plant [46]. Soil particles are bound by organic chemicals including compounds produced by soil bacteria. A wide range of biochemical is produced by soil bacteria among which polysaccharides are the ones with the highest impact on binding soil particles. The mineralizing effects of soil microbes on organic matter can also influence soil structure. The enhanced growth of plant growth by soil microbes and hence the increased amount of root exudates and rhizo deposition can also affect soil structure directly or by increasing the microbial population and activities, indirectly [154-156].
Soil microbes produce a wide range of biochemical, affecting soil environment. Among which there are the products, adversely affecting the growth and activities of soil pathogens including soil bacteria and soil fungi e.g. PGP bacteria produce hydrogen cyanide (HCN), which can have unfavorable effects on the growth of soil pathogens. In addition through stimulating plant systemic resistance, soil microbes can enhance plant resistance to pathogens. The presence of soil microbes in the rhizosphere and production of different compounds can stimulate plant growth and systemic resistance. Generally, the adverse effects of PGP bacteria, which is mostly related to Pseudomonas and Bacillus on soil pathogens is through the production of antibiotic compounds (HCN, pyrrolnitrin, phloroglucinols, phenazines, pyoluteorin, and cyclic lipopeptides); plant induced systemic resistance and interfering with pathogens ability to suppress plant growth.
In the recent years there have been some interesting research work regarding the use of PGP bacteria under stress. There are many soil stressors affecting plant growth and yield production including soil pH, suboptimal root zone temperature, and heavy metals. Interestingly, researchers have found that the adverse effects of different stressors on the Rhizobium-legume N2 fixation can be alleviated by the use of signal molecule genistein. Genistein is produced by the specific legume host plant like soybean (Glycine max L.) activating the nodulation (nod) genes in Bradyrhizobium japonicum [157,158]. Under humid or dry climate conditions soil pH can fluctuate much. The high amounts of rain in the humid area reduce soil pH significantly. PGPR bacteria can also alleviate salinity stress on plant growth. They are able to produce the important bacterial enzyme ACC- deaminase under different conditions including stress, which can effectively control the stress. This is because, as previously mentioned, ACC-deaminase can catalyze the ACC, which is the prerequisite for the production of the stress hormone, ethylene. Increased level of ethylene in the plant can adversely affect plant growth and yield production. Plant physiological and morphological characters are also important in the alleviation of stress in symbiosis of non symbiosis association with their associative soil microbes. The more resistant plant species can perform more efficiently under stress and their symbiosis with their associative soil microbes also can intensify such abilities.
Conclusion and Future Prospect
Sustainable agriculture requires the use of strategies to increase or maintain the current rate of food production while reducing damage to the environment and human health. The use of microbial plant growth promoters is an alternative to conventional agricultural technologies. Plant growth-promoting microbe can affect plant growth directly or indirectly. The direct promotion of plant growth by PGP microbes, for the most part, entails providing the plant with a compound that is synthesized by the bacterium or facilitating the uptake of certain nutrients from the environment. The indirect promotion of plant growth occurs when PGP microbes decrease or prevent the deleterious effects of one or more phytopathogenic organisms. The need of today's world is high output yield and enhanced production of the crop as well as fertility of soil to get in an ecofriendly manner. Hence, the research has to be focused on the new concept of rhizo engineering based on favorably partitioning of the exotic biomolecules, which create a unique setting for the interaction between plant and microbes. Future research in rhizosphere biology will rely on the development of molecular and biotechnological approaches to increase our knowledge of rhizosphere biology and to achieve an integrated management of soil microbial populations. The application of multi strain bacterial consortium over single inoculation could be an effective approach for reducing the harmful impact of stress on plant growth.
Acknowledgement
The authors duly acknowledge the Department of Biotechnology and Vice Chancellor, Eternal University for providing the motivation and research infrastructure.
References
- Altenbach SB (2012) New insights into the effects of high temperature, drought and post-anthesis fertilizer on wheat grain development. J Cereal Sci 56 (1): 39-50.
- Kersebaum K, Nendel C (2014) Site-specific impacts of climate change on wheat production across regions of Germany using different CO2 response functions. Eur J Agron 52: 22-32.
- Barnett A, Hajat S, Gasparrini A, Rocklov J (2012) Cold and heat waves in the United States. Environ Res 112: 218-224.
- Elliott L, Lynch J (1984) Pseudomonads as a factor in the growth of winter wheat (Triticum aestivum L.). Soil Biol Biochem 16 (1): 69-71.
- Saxena AK, Yadav AN, Rajawat M, Kaushik R, Kumar R, et al. (2016) Microbial diversity of extreme regions: An unseen heritage and wealth. Indian J Plant Genet Resour 29(3): 246-248.
- Yadav AN, Verma P, Kumar V, Sachan SG, Saxena AK (2017) Extreme Cold Environments: A Suitable Niche for Selection of Novel Psychrotrophic Microbes for Biotechnological Applications. Adv Biotechnol Microbiol 2(2): 1-4.
- Hill G, Mitkowski N, Aldrich-Wolfe L, Emele L, Jurkonie D, Ficke A, et al. (2000) Methods for assessing the composition and diversity of soil microbial communities. Appl Soil Ecol 15 (1): 25-36.
- Yadav AN, Sharma D, Gulati S, Singh S, Kaushik R, et al. (2015) Haloarchaea endowed with phosphorus solubilization attribute implicated in phosphorus cycle. Sci Rep Doi:10.1038/srep12293
- Verma P, Yadav AN, Khannam KS, Kumar S, Saxena AK, et al. (2016) Molecular diversity and multifarious plant growth promoting attributes of Bacilli associated with wheat (Triticum aestivum L.) rhizosphere from six diverse agro-ecological zones of India. J Basic Microbiol 56(1): 44-58.
- Kumar V, Yadav AN, Verema P, Sangwan P, Abhishake S, et al. (2017) β-Propeller phytases: Diversity, catalytic attributes, current developments and potential biotechnological applications. Int J Biol Macromolec 98: 595-609.
- Kloepper J, Schroth M (1978) Plant growth-promoting rhizobacteria on radishes. In: Proceedings of the 4th international conference on plant pathogenic bacteria, pp. 879-882.
- Benizri E, Baudoin E, Guckert A (2001) Root colonization by inoculated plant growth-promoting rhizobacteria. Biocon Sci Technol 11(5): 557-574.
- Lugtenberg B, Kamilova F (2009) Plant-growth-promoting rhizobacteria. Ann Rev Microbiol 63: 541-556.
- Kaur R, Saxena A, Sangwan P, Yadav AN, Kumar V, et al. (2017) Production and characterization of a neutral phytase of Penicillium oxalicum EUFR-3 isolated from Himalayan region. Nus Biosci 9(1): 68-76.
- Suman A, Verma P, Yadav AN, Saxena AK (2015) Bioprospecting for extracellular hydrolytic enzymes from culturable thermotolerant bacteria isolated from Manikaran thermal springs. Res J Biotechnol 10: 33-42.
- Suman A, Verma P, Yadav AN, Srinivasamurthy R, Singh A, et al. (2016) Development of hydrogel based bio-inoculant formulations and their impact on plant biometric parameters of wheat (Triticum aestivum L.). Int J Curr Microbiol Appl Sci 5(3): 890-901.
- Verma P, Yadav AN, Shukla L, Saxena AK, Suman A (2015) Hydrolytic enzymes production by thermotolerant Bacillus altitudinis IARI- MB-9 and Gulbenkiania mobilis IARI-MB-18 isolated from Manikaran hot springs. Int J Adv Res 3(9): 1241-1250.
- Yadav AN, Verma P, Kumar M, Pal KK, Dey R, et al. (2015) Diversity and phylogenetic profiling of niche-specific Bacilli from extreme environments of India. Ann Microbiol 65(2): 611-629.
- Verma P, Yadav AN, Khannam KS, Panjiar N, Kumar S, et al. (2015) Assessment of genetic diversity and plant growth promoting attributes of psychrotolerant bacteria allied with wheat (Triticum aestivum) from the northern hills zone of India. Ann Microbiol 65: 1885-1899.
- Verma P, Yadav AN, Kazy SK, Saxena AK, Suman A (2013) Elucidating the diversity and plant growth promoting attributes of wheat (Triticum aestivum) associated acidotolerant bacteria from southern hills zone of India. Natl J Life Sci 10(2): 219-227.
- Yadav AN, Verma P, Sachan SG, Kaushik R, Saxena AK (2016) Microbiome of Indian Himalayan regions: Molecular diversity, phylogenetic profiling and biotechnological applications. In: Proceeding of 86th Annual Session of NASI & Symposium on "Science, Technology and Entrepreneurship for Human Welfare in The Himalayan Region”, p. 58.
- Pikovskaya R (1948) Mobilization of phosphorus in soil in connection with vital activity of some microbial species. Mikrobiologiya 17: 362370.
- Hu X, Chen J, Guo J (2006) Two Phosphate- and Potassium-solubilizing Bacteria Isolated from Tianmu Mountain, Zhejiang, China. World J Microbiol Biotechnol 22(9): 983-990.
- Fasim F, Ahmed N, Parsons R, Gadd GM (2002) Solubilization of zinc salts by a bacterium isolated from the air environment of a tannery. FEMS Microbiol Lett 213 (1): 1-6.
- Bric JM, Bostock RM, Silverstone SE (1991) Rapid in situ assay for indoleacetic acid production by bacteria immobilized on a nitrocellulose membrane. Appl Environ Microbiol 57(2): 535-538.
- Brown ME, Burlingham SK (1968) Production of plant growth substances by Azotobacter chroococcum. J Gen Microbiol 53 (1): 135-144.
- Boddey R, De Oliveira O, Urquiaga S, Reis V, De Olivares F, et al. (1995) Biological nitrogen fixation associated with sugar cane and rice: contributions and prospects for improvement. Plant Soil 174(1-2): 195-209.
- Jacobson CB, Pasternak J, Glick BR (1994) Partial purification and characterization of 1-aminocyclopropane-1-carboxylate deaminase from the plant growth promoting rhizobacterium Pseudomonas putida GR12-2. Can J Microbiol 40(12): 1019-1025.
- Cappucino JC, Sherman N (1992) Nitrogen Cycle. In: Microbiology: A Laboratory Manual. (4th edn), Benjamin/Cumming Pub Co, New York, USA, pp. 311-312.
- Bakker AW, Schippers B (1987) Microbial cyanide production in the rhizosphere in relation to potato yield reduction and Pseudomonas SPP-mediated plant growth-stimulation. Soil Biol Biochem 19(4): 451-457.
- Schwyn B, Neilands J (1987) Universal chemical assay for the detection and determination of siderophores. Anal Biochem 160(1): 47-56.
- Yadav AN, Sachan SG, Verma P, Kaushik R, Saxena AK (2016) Cold active hydrolytic enzymes production by psychrotrophic Bacilli isolated from three sub-glacial lakes of NW Indian Himalayas. J Basic Microbiol 56(3): 294-307.
- Yadav AN, Sachan SG, Verma P, Saxena AK (2016) Bioprospecting of plant growth promoting psychrotrophic Bacilli from cold desert of north western Indian Himalayas. Indian J Exp Biol 54(2): 142-150.
- Han SO, New P (1998) Variation in nitrogen fixing ability among natural isolates of Azospirillum. Microb Ecol 36(2): 193-201.
- Mehta S, Nautiyal CS (2001) An efficient method for qualitative screening of phosphate-solubilizing bacteria. Curr Microbiol 43(1): 51-56.
- Patten CL, Glick BR (2002) Role of Pseudomonas putida indoleacetic acid in development of the host plant root system. Appl Environ Microbiol 68(8): 3795-3801.
- Yadav AN, Sachan SG, Verma P, Tyagi SP, Kaushik R, et al. (2015) Culturable diversity and functional annotation of psychrotrophic bacteria from cold desert of Leh Ladakh (India). World J Microbiol Biotechnol 31(1): 95-108.
- Yadav AN (2015) Bacterial diversity of cold deserts and mining of genes for low temperature tolerance. IARI, New Delhi/ BIT, Ranchi, India, p. 234.
- Yang J, Kloepper JW, Ryu C-M (2009) Rhizosphere bacteria help plants tolerate abiotic stress. Trends Plant Sci 14(1): 1-4.
- Barea J-M, Pozo MJ, Azcon R, Azcon-Aguilar C (2005) Microbial co-operation in the rhizosphere. J Exp Bot 56(417): 1761-1778.
- Suman A, Yadav AN, Verma P (2016) Endophytic Microbes in Crops: Diversity and Beneficial impact for Sustainable Agriculture. In: Singh DP, Abhilash PC, Prabha R (Eds.), Microbial Inoculants in Sustainable Agricultural Productivity, Research Perspectives. Springer-Verlag, India, pp. 117-143.
- Hassen AI, Labuschagne N (2010) Root colonization and growth enhancement in wheat and tomato by rhizobacteria isolated from the rhizoplane of grasses. World J Microbiol Biotechnol 26(10): 1837-1846.
- Suarez C, Cardinale M, Ratering S, Steffens D, Jung S, et al. (2015) Plant growth-promoting effects of Hartmannibacter diazotrophicus on summer barley (Hordeum vulgare L.) under salt stress. Appl Soil Ecol 95: 23-30.
- Verma P, Yadav AN, Kazy SK, Saxena AK, Suman A (2014) Evaluating the diversity and phylogeny of plant growth promoting bacteria associated with wheat (Triticum aestivum) growing in central zone of India. Int J Curr Microbiol Appl Sci 3(5): 432-447.
- Gaba S, Singh RN, Abrol S, Yadav AN, Saxena AK, et al. (2017) Draft Genome Sequence of Halolamina pelagica CDK2 Isolated from Natural Salterns from Rann of Kutch, Gujarat, India. Genome Announc 5(6): 1-2.
- Glick BR (1995) The enhancement of plant growth by free-living bacteria. Can J Microbiol 41(2): 109-117.
- Ikunaga Y, Sato I, Grond S, Numaziri N, Yoshida S, et al. (2011) Nocardioides sp. strain WSN05-2, isolated from a wheat field, degrades deoxynivalenol, producing the novel intermediate 3-epi- deoxynivalenol. Appl Microbiol Biotechnol 89(2): 419-427.
- Bal HB, Das S, Dangar TK, Adhya TK (2013) ACC deaminase and IAA producing growth promoting bacteria from the rhizosphere soil of tropical rice plants. J Basic Microbiology 53(12): 972-984.
- Cavalca L, Corsini A, Bachate SP, Andreoni V (2013) Rhizosphere colonization and arsenic translocation in sunflower (Helianthus annuus L.) by arsenate reducing Alcaligenes sp. strain Dhal-L. World J Microbiol Biotechnol 29(10): 1931-1940.
- Li X, Rui J, Xiong J, Li J, He Z, et al. (2014) Functional potential of soil microbial communities in the maize rhizosphere. PloS one 9(11): e112609.
- Ali A, Bashir U, Akhtar N, Haider MS (2016) Characterization of growth promoting rhizobacteria of leguminous plants. Pak J Phytopathol 28(1): 57-60.
- Verma P, Yadav AN, Shukla L, Saxena AK, Suman A (2015) Alleviation of cold stress in wheat seedlings by Bacillus amyloliquefaciens IARI- HHS2-30, an endophytic psychrotolerant K-solubilizing bacterium from NW Indian Himalayas. Natl J Life Sci 12(2): 105-110.
- Verma P, Yadav AN, Khannam KS, Mishra S, Kumar S, et al. (2016) Appraisal of diversity and functional attributes of thermotolerant wheat associated bacteria from the peninsular zone of India. Saudi J Biol Sci DOI: https://doi.org/10.1016/j.sjbs.2016.01.042.
- Prasanna R, Nain L, Pandey AK, Saxena AK (2012) Microbial diversity and multidimensional interactions in the rice ecosystem. Arch Agron Soil Sci 58(7): 723-744.
- Thawai C, Kittiwongwattana C, Thanaboripat D, Laosinwattana C, Koohakan P, et al. (2016) Micromonospora soli sp. nov., isolated from rice rhizosphere soil. Antonie van Leeuwenhoek 109(3): 449-456.
- Martin-Laurent F, Barres B, Wagschal I, Piutti S, Devers M, et al.(2006) Impact of the maize rhizosphere on the genetic structure, the diversity and the atrazine-degrading gene composition of cultivable atrazine-degrading communities. Plant Soil 282(1-2): 99-115.
- WEN X-y, Dubinsky E, Yao W, Rong Y, Fu C (2016) Wheat, maize and sunflower cropping systems selectively influence bacteria community structure and diversity in their and succeeding crop's rhizosphere. J Integ Agric 15(8): 1892-1902.
- Mayilraj S, Krishnamurthi S, Saha P, Saini H (2006) Kitasatospora sampliensis sp. nov., a novel actinobacterium isolated from soil of a sugar-cane field in India. Int J Syst Evol Microbiol 56(Pt3): 519-522.
- de Oliveira ZM, Floh EI, Ferrara FI, Barbosa HR (2011) Diazotrophyc rhizobacteria isolated from sugarcane can release amino acids in a synthetic culture medium. Biol Fert Soils 47(8): 957-962.
- Wei F, Lin YH, YE BY, Zhang JS, Chen YQ (2011) Screening and identifying of xylanase producing strain from sugarcane rhizosphere soil. Subtropical Agric Res 3: 014.
- Cattelan A, Hartel P, Fuhrmann J (1997) Bacterial composition in the rhizosphere of nodulating and non-nodulating soybean. Soil Sci Soc Am J 62(6): 1549-1555.
- El-Tarabily KA, Nassar AH, Sivasithamparam K (2008) Promotion of growth of bean (Phaseolus vulgaris L.) in a calcareous soil by a phosphate-solubilizing, rhizosphere-competent isolate of Micromonospora endolithica. Appl Soil Ecol 39(2): 161-171.
- Shen Y, Liu C, Wang X, Zhao J, Jia F, et al. (2013) Actinoplanes hulinensis sp. nov., a novel actinomycete isolated from soybean root (Glycine max (L.) Merr). Antonie van Leeuwenhoek 103(2): 293-298.
- Carro L, Sproer C, Alonso P, Trujillo ME (2012) Diversity of Micromonospora strains isolated from nitrogen fixing nodules and rhizosphere of Pisum sativum analyzed by multilocus sequence analysis. Syst Appl Microbiol 35(2): 73-80.
- Tariq M, Hameed S, Yasmeen T, Zahid M, Zafar M (2014) Molecular characterization and identification of plant growth promoting endophytic bacteria isolated from the root nodules of pea (Pisum sativum L.). World J Microbiol Biotechnol 30(2): 719-725.
- Ambrosini A, Beneduzi A, Stefanski T, Pinheiro FG, Vargas LK, et al. (2012) Screening of plant growth promoting rhizobacteria isolated from sunflower (Helianthus annuus L.). Plant Soil 356(1-2): 245-264.
- Wang W, Qiu Z, Tan H, Cao L (2014) Siderophore production by actinobacteria. Biometals 27(4): 623-631.
- Yadav AK, Srivastava AK, Yandigeri MS, Kashyap SK, Modi DR, et al. (2010) Characterization of indigenous copper-resistant Streptomycetes from chickpea (Cicer arietinum L.) fields. Ann Microbiol 60(4): 605-614.
- Khan MA, Gangopadhyay S (2012) Effect of Soil Inhabiting Antagonistic Microflora Against f. sp. Incitant of Wilt in Chickpea. J Mycol Plant Pathol 42(3): 341.
- Garbeva P, Van Veen J, Van Elsas J (2003) Predominant Bacillus spp. in agricultural soil under different management regimes detected via PCR-DGGE. Microb Ecol 45(3): 302-316.
- Martyniuk S, Martyniuk M (2003) Occurrence of Azotobacter spp. in some Polish soils. Polish J Environm Stud 12(3): 371-374.
- Jimenez DJ, Montana JS, Martinez MM (2011) Characterization of free nitrogen fixing bacteria of the genus Azotobacter in organic vegetable-grown Colombian soils. Brazi J Microbiol 42(3): 846-858.
- Lenart A (2012) Occurrence characteristics and genetic diversity of Azotobacter chroococcum in various soils of Southern Poland. Pol J Environ Stud 21(2): 415-424.
- Czaban J, Wroblewska B (2017) The Effect of Bentonite on the Survival of Azotobacter chroococcum in Sandy Soil in a Long-Term Plot Experiment. Polish J Environ Stud 26(1): 1-8.
- Chin-A-Woeng TF, Bloemberg GV, Mulders IH, Dekkers LC, Lugtenberg BJ (2000) Root colonization by phenazine-1-carboxamide-producing bacterium Pseudomonas chlororaphis PCL1391 is essential for biocontrol of tomato foot and root rot. Mol Plant-Microbe IN 13(12): 1340-1345.
- Tabatabaei S, Ehsanzadeh P, Etesami H, Alikhani HA, Glick BR (2016) Indole-3-acetic acid (IAA) producing Pseudomonas isolates inhibit seed germination and a-amylase activity in durum wheat (Triticum turgidum L.). Spanish J Agric Res 14(1): 0802.
- Ivanova E, Doronina N, Trotsenko YA (2001) Aerobic methylobacteria are capable of synthesizing auxins. Microbiology 70(4): 392-397.
- Ivanova E, Pirttila A, Fedorov D, Doronina N, Trotsenko Y (2008) Association of methylotrophic bacteria with plants: metabolic aspects. Prospects and applications for plant associated microbes A laboratory manual, part A: bacteria Biobien Innovations, Turku, Finland, pp. 225-231.
- Shukla L, Suman A, Yadav AN, Verma P, Saxena AK (2016) Syntrophic microbial system for ex-situ degradation of paddy straw at low temperature under controlled and natural environment. J App Biol Biotech 4(2): 030-037.
- Forchetti G, Masciarelli O, Alemano S, Alvarez D, Abdala G (2007) Endophytic bacteria in sunflower (Helianthus annuus L.): isolation, characterization, and production of jasmonates and abscisic acid in culture medium. Appl Microbiol Biotechnol 76(5): 1145-1152.
- Kumar V, Yadav AN, Saxena A, Sangwan P, Dhaliwal HS (2016) Unravelling rhizospheric diversity and potential of phytase producing microbes. SM J Biol 2(1): 1009.
- Singh RN, Gaba S, Yadav AN, Gaur P, Gulati S, et al. (2016) First high quality draft genome sequence of a plant growth promoting and Cold Active Enzymes producing psychrotrophic Arthrobacter agilis strain L77. Stand Genomic Sci 11(1): 54.
- Sheng XF, Xia JJ, Jiang CY, He LY, Qian M (2008) Characterization of heavy metal-resistant endophytic bacteria from rape (Brassica napus) roots and their potential in promoting the growth and lead accumulation of rape. Environ Poll 156(3): 1164-1170.
- Giller KE (2001) Nitrogen fixation in tropical cropping systems. CABI.
- Elbeltagy A, Nishioka K, Sato T, Suzuki H, Ye B, et al. (2001) Endophytic colonization and in planta nitrogen fixation by a Herbaspirillum sp. isolated from wild rice species. Appl Environ Microbiol 67(11): 5285-5293.
- Boddey RM, Urquiaga S, Alves BJ, Reis V (2003) Endophytic nitrogen fixation in sugarcane: present knowledge and future applications. Plant Soil 252(1): 139-149.
- Wei CY, Lin L, Luo LJ, Xing YX, Hu CJ, et al. (2014) Endophytic nitrogen- fixing Klebsiella variicola strain DX120E promotes sugarcane growth. Biol Fert Soils 50(4): 657-666.
- Reis VM, Teixeira KRdS (2015) Nitrogen fixing bacteria in the family Acetobacteraceae and their role in agriculture. J Basic Microbiol 55(8): 931-949.
- Choudhury A, Kennedy I (2004) Prospects and potentials for systems of biological nitrogen fixation in sustainable rice production. Biol Fert Soils 39(4): 219-227.
- Gtari M, Ghodhbane-Gtari F, Nouioui I, Beauchemin N, Tisa LS (2012) Phylogenetic perspectives of nitrogen-fixing actinobacteria. Arch Microbiol 194(1): 3-11.
- Yadav AN, Rana KL, Kumar V, Dhaliwal HS (2016) Phosphorus solubilizing endophytic microbes: potential application for sustainable agriculture. In: EU Voice 2(1): 21-22.
- Khalid A, Arshad M, Zahir Z (2004) Screening plant growth- promoting rhizobacteria for improving growth and yield of wheat. J Appl Microbiol 96(3): 473-480.
- Khalid A, Akhtar M, Mahmood M, Arshad M (2006) Effect of substrate-dependent microbial ethylene production on plant growth. Microbiology 75(2): 231-236.
- Xu M, Sheng J, Chen L, Men Y, Gan L, et al. (2014) Bacterial community compositions of tomato (Lycopersicum esculentum Mill.) seeds and plant growth promoting activity of ACC deaminase producing Bacillus subtilis (HYT-12-1) on tomato seedlings. World J Microbiol Biotechnol 30(3): 835-845.
- Gholami M, Khakvar R, Niknam G (2013) Introduction of some new endophytic bacteria from Bacillus and Streptomyces genera as successful biocontrol agents against Sclerotium rolfsii. Arch Phytopathol Plant Prot 47(1): 122-130.
- Inderiati S, Franco CM (2008) Isolation and identification of endophytic actinomycetes and their antifungal activity 1: 1-6.
- Nagendran K, Karthikeyan G, Peeran MF, Raveendran M, Prabakar K, et al. (2013) Management of Bacterial Leaf Blight Disease in Rice with Endophytic Bacteria. World Appl Sci J 28(12): 2229-2241.
- Purnawati A, Sastrahidayat IR, Abadi AL, Hadiastono T (2014) Endophytic Bacteria as Biocontrol Agents of Tomato Bacterial Wilt Disease. J Trop Life Sci 4(1): 33-36.
- Gulati A, Vyas P, Rahi P, Kasana RC (2009) Plant growth-promoting and rhizosphere-competent Acinetobacter rhizosphaerae strain BIHB 723 from the cold deserts of the Himalayas. Curr Microbiol 58(4): 371-377.
- Verma P, Yadav AN, Kumar V, Khan MA, Saxena AK (2017) Microbes in Termite Management: Potential Role and Strategies. In: Sustainable Termite Management.
- West SA, Buckling A (2003) Cooperation, virulence and siderophore production in bacterial parasites. Proc Biol Sci 270(1510): 37-44.
- Takase H, Nitanai H, Hoshino K, Otani T (2000) Impact of Siderophore Production on Pseudomonas aeruginosa Infections in Immunosuppressed Mice. Infect Immun 68(4): 1834-1839.
- Dale SE, Doherty-Kirby A, Lajoie G, Heinrichs DE (2004) Role of siderophore biosynthesis in virulence of Staphylococcus aureus: identification and characterization of genes involved in production of a siderophore. Infect Immun 72(1): 29-37.
- Niehus R, Picot A, Oliveira NM, Mitri S, Foster KR (2017) The evolution of siderophore production as a competitive trait. Evolution International journal of organic evolution 71(6): 1443-1455.
- Mishra PK, Bisht SC, Ruwari P, Selvakumar G, Joshi GK, et al. (2011) Alleviation of cold stress in inoculated wheat (Triticum aestivum L.) seedlings with psychrotolerant Pseudomonads from NW Himalayas. Arch Microbiol 193(7): 497-513.
- Vyas P, Rahi P, Gulati A (2009) Stress tolerance and genetic variability of phosphate-solubilizing fluorescent Pseudomonas from the cold deserts of the trans-Himalayas. Microb Ecol 58(2): 425-434.
- Vyas P, Joshi R, Sharma K, Rahi P, Gulati A (2010) Cold-adapted and rhizosphere-competent strain of Rahnella sp. with broad-spectrum plant growth-promotion potential. J Microbiol Biotechnol 20(12): 1724-1734.
- Raaijmakers JM, Weller DM, Thomashow LS (1997) Frequency of antibiotic-producing Pseudomonas spp. in natural environments. Appl Environ Microbiol 63(3): 881-887.
- Maurhofer M, Keel C, Schnider U, Voisard C, Haas D, et al. (1992) Influence of enhanced antibiotic production in Pseudomonas fluorescens strain CHA0 on its disease suppressive capacity. Phytopathology 82(2): 190-195.
- De Bruijn I, De Kock MJ, Yang M, De Waard P, van Beek TA, et al.(2007) Genome-based discovery, structure prediction and functional analysis of cyclic lipopeptide antibiotics in Pseudomonas species. Mol Microbiol 63(2): 417-428.
- Davis R, Brown PD (2016) Multiple antibiotic resistance index, fitness and virulence potential in respiratory Pseudomonas aeruginosa from Jamaica. J Medical Microbiol 65(4): 261-271.
- Van der Meij A, Worsley SF, Hutchings MI, Van Wezel GP (2017) Chemical ecology of antibiotic production by actinomycetes. FEMS Microbiol Rev 41(3): 392-416.
- Arora NK (2013) Plant microbe symbiosis: Fundamentals and advances. Springer Science & Business Media, India.
- Kim IS, Yang SY, Park SK, Kim YC (2017) Quorum sensing is a key regulator for the antifungal and biocontrol activity of chitinase- producing Chromobacterium sp. C61. Mol Plant Pathol 18(1): 134-140.
- Wang K, Yan PS, Cao LX (2014) Chitinase from a novel strain of Serratia marcescens JPP1 for biocontrol of aflatoxin: molecular characterization and production optimization using response surface methodology. BioMed Res Int. Doi.org/10.1155/2014/482623.
- Agbessi S, Beausejour J, Dery C, Beaulieu C (2003) Antagonistic properties of two recombinant strains of Streptomyces melanosporofaciens obtained by intraspecific protoplast fusion. Appl Microbiol Biotechnol 62 (2-3): 233-238.
- Anwar S, Ali B, Sajid I (2016) Screening of rhizospheric actinomycetes for various in-vitro and in-vivo plant growth promoting (PGP) traits and for agroactive compounds. Front Microbiol 7:1334.
- Dimkpa C, Svatos A, Merten D, Buchel G, Kothe E (2008) Hydroxamate siderophores produced by Streptomyces acidiscabies E13 bind nickel and promote growth in cowpea (Vigna unguiculata L.) under nickel stress. Can J Microbiol 54(3): 163-172.
- Gupta R, Saxena R, Chaturvedi P, Virdi J (1995) Chitinase production by Streptomyces viridificans: its potential in fungal cell wall lysis. J Appl Bacteriol 78 (4): 378-383.
- Kawase T, Saito A, Sato T, Kanai R, Fujii T, et al. (2004) Distribution and phylogenetic analysis of family 19 chitinases in Actinobacteria. Appl Environ Microbiol 70(2): 1135-1144.
- Macagnan D, Romeiro RdS, Pomella AW (2008) Production of lytic enzymes and siderophores, and inhibition of germination of basidiospores of Moniliophthora (ex Crinipellis) perniciosa by phylloplane actinomycetes. Biol Con 47(3): 309-314.
- Peoples MB, Craswell ET (1992) Biological nitrogen fixation: investments, expectations and actual contributions to agriculture. Plant Soil 141(1-2): 13-39.
- Suman A, Shasany AK, Singh M, Shahi HN, Gaur A, et al. (2001) Molecular assessment of diversity among endophytic diazotrophs isolated from subtropical Indian sugarcane. World J Microbiol Biotechnol 17(1): 39-45.
- Tiwari S, Singh P, Tiwari R, Meena KK, Yandigeri M, et al. (2011) Salt-tolerant rhizobacteria-mediated induced tolerance in wheat (Triticum aestivum) and chemical diversity in rhizosphere enhance plant growth. Biol Fert Soils 47(8): 907.
- Trejo Estrada S, Paszczynski A, Crawford D (1998) Antibiotics and enzymes produced by the biocontrol agent Streptomyces violaceusniger YCED-9. J Industrial Microbiol Biotechnol 21(1): 81-90.
- Wang Q, Duan B, Duan B, Yang R, Zhao Y, Zhang L (2015) Screening and Identification of Chitinolytic Actinomycetes and Study on the Inhibitory Activity against Turfgrass Root Rot Disease Fungi. J Biosci and Med 3(3): 56-65.
- Xue L, Xue Q, Chen Q, Lin C, Shen G, et al. (2013) Isolation and evaluation of rhizosphere actinomycetes with potential application for biocontrol of Verticillium wilt of cotton. Crop Prot 43: 231-240.
- Zhao J, Xue QH, Niu GG, Xue L, Shen GH, (2013) Extracellular enzyme production and fungal mycelia degradation of antagonistic Streptomyces induced by fungal mycelia preparation of cucurbit plant pathogens. Ann Microbiol 63(2): 809-812.
- Antaya CL (2008) Current Eco-Economical Impacts of Flavobacterium psychrophilum. Basic Biotechno J 4(1): 16-21.
- Pereira SI, Moreira H, Argyras K, Castro PM, Marques AP, et al. (2016) Promotion of sunflower growth under saline water irrigation by the inoculation of beneficial microorganisms. Appl Soil Ecol 105: 36-47.
- Rana KL, Kour D, Yadav AN, Kumar V, Dhaliwal HS (2016) Endophytic microbes from wheat: Diversity and biotechnological applications for sustainable agriculture. In: Proceeding of 57th Association of Microbiologist of India & International symposium on "Microbes and Biosphere: What's New What's Next”, p. 453.
- Yadav AN, Sachan SG, Verma P, Saxena AK (2015) Prospecting cold deserts of north western Himalayas for microbial diversity and plant growth promoting attributes. J Biosci Bioeng 119(6): 683-693.
- Ambrosini A, Beneduzi A, Stefanski T, Pinheiro F, Vargas L, et al. (2012) Screening of plant growth promoting rhizobacteria isolated from sunflower (Helianthus annuus L.). Plant Soil 356(1-2): 245-264.
- Beneduzi A, Peres D, da Costa PB, Bodanese Zanettini MH, Passaglia LMP (2008) Genetic and phenotypic diversity of plant-growth- promoting bacilli isolated from wheat fields in southern Brazil. Res Microbiol 159(4): 244-250.
- Beneduzi A, Peres D, Vargas LK, Bodanese-Zanettini MH, Passaglia LMP (2008) Evaluation of genetic diversity and plant growth promoting activities of nitrogen-fixing bacilli isolated from rice fields in South Brazil. Appl Soil Ecol 39(3): 311-320.
- Hariprasad P, Niranjana S (2009) Isolation and characterization of phosphate solubilizing rhizobacteria to improve plant health of tomato. Plant Soil 316(1-2): 13-24.
- Karlidag H, Esitken A, Turan M, Sahin F (2007) Effects of root inoculation of plant growth promoting rhizobacteria (PGPR) on yield, growth and nutrient element contents of leaves of apple. Sci hort 114(1): 16-20.
- Qadri M, Rajput R, Abdin MZ, Vishwakarma RA, Riyaz-Ul-Hassan S (2014) Diversity, molecular phylogeny, and bioactive potential of fungal endophytes associated with the Himalayan blue pine (Pinus wallichiana). Microb Ecol 67(4): 877-887.
- Rajput L, Imran A, Mubeen F, Hafeez FY (2013) Salt-tolerant PGPR strain Planococcus rifietoensis promotes the growth and yield of wheat (Triticum aestivum L.) cultivated in saline soil. Pak J Bot 45(6): 1955-1962.
- Sgroy V, Cassan F, Masciarelli O, Del Papa MF, Lagares A, et al. (2009) Isolation and characterization of endophytic plant growth-promoting (PGPB) or stress homeostasis-regulating (PSHB) bacteria associated to the halophyte Prosopis strombulifera. Appl Microbiol Biotechnol 85(2): 371-381.
- Yu X, Ai C, Xin L, Zhou G (2011) The siderophore-producing bacterium, Bacillus subtilis CAS15, has a biocontrol effect on Fusarium wilt and promotes the growth of pepper. Eup J Soil Biol 47(2): 138-145.
- Deepa C, Dastager SG, Pandey A (2010) Isolation and characterization of plant growth promoting bacteria from non-rhizospheric soil and their effect on cowpea (Vigna unguiculata (L.) Walp.) seedling growth. World J Microbiol Biotechnol 26(7): 1233-1240.
- Joe MM, Islam MR, Karthikeyan B, Bradeepa K, Sivakumaar PK, et al. (2012) Resistance responses of rice to rice blast fungus after seed treatment with the endophytic Achromobacter xylosoxidans AUM54 strains. Crop Prot 42: 141-148.
- Forchetti G, Masciarelli O, Alemano S, Alvarez D, Abdala G (2007) Endophytic bacteria in sunflower (Helianthus annuus L.): isolation, characterization, and production of jasmonates and abscisic acid in culture medium. Appl Microbiol Biotechnol 76(5): 1145-1152.
- Forchetti G, Masciarelli O, Izaguirre MJ, Alemano S, Alvarez D, et al. (2010) Endophytic bacteria improve seedling growth of sunflower under water stress, produce salicylic acid, and inhibit growth of pathogenic fungi. Curr Microbiol 61(6): 485-493.
- Siddikee MA, Chauhan P, Anandham R, Han GH, Sa T (2010) Isolation, characterization, and use for plant growth promotion under salt stress, of ACC deaminase-producing halotolerant bacteria derived from coastal soil. J Microbiol Biotechnol 20 (11): 1577-1584.
- Villarraga DMV, Tibambre MEM, Romero IAG, ia, Suarez-Moreno ZR, Moreno-Sarmiento N (2017) Evaluation of biocontrol properties of Streptomyces spp. isolates against phytopathogenic fungi Colletotrichum gloeosporioides and Microcyclus ulei. Afr J Microbiol Res 11(5): 141-154.
- Niste M, Vidican R, Pop R, Rotar I (2013) Stress Factors Affecting Symbiosis Activity and Nitrogen Fixation by Rhizobium Cultured in vitro. Environ Prom 6(13): 42-45.
- Oldroyd GE, Dixon R (2014) Biotechnological solutions to the nitrogen problem. Curr Opin Biotechnol 26: 19-24.
- Olivares J, Bedmar EJ, Sanjuan J (2013) Biological nitrogen fixation in the context of global change. Mol Plant-Microbe IN 26 (5): 486-494.
- Hungria M, Nogueira MA, Araujo RS (2015) Soybean seed coinoculation with Bradyrhizobium spp. and Azospirillum brasilense: a new biotechnological tool to improve yield and sustainability. Am J Plant Sci 6(6): 811.
- Alves GC, Videira SS, Urquiaga S, Reis VM (2015) Differential plant growth promotion and nitrogen fixation in two genotypes of maize by several Herbaspirillum inoculants. Plant Soil 387(1-2): 307-321.
- Brusamarello-Santos LC, Gilard F, Brule L, Quillere I, Gourion B, et al. (2017) Metabolic profiling of two maize (Zea mays L.) inbred lines inoculated with the nitrogen fixing plant-interacting bacteria Herbaspirillum seropedicae and Azospirillum brasilense. PloS one 12(3): e0174576.
- Lehmann J, Rillig MC, Thies J, Masiello CA, Hockaday WC, et al. (2011) Biochar effects on soil biota-a review. Soil Biol Biochem 43(9): 18121836.
- Burns RG, DeForest JL, Marxsen J, Sinsabaugh RL, Stromberger ME, et al. (2013) Soil enzymes in a changing environment: current knowledge and future directions. Soil Biol Biochem 58: 216-234.
- Rillig MC, Mummey DL (2006) Mycorrhizas and soil structure. New Phytol 171(1): 41-53.
- Miransari M, Smith D (2007) Overcoming the stressful effects of salinity and acidity on soybean nodulation and yields using signal molecule genistein under field conditions. J Plant Nutr 30(12): 19671992.
- Miransari M, Smith D (2009) Alleviating salt stress on soybean (Glycine max (L.) Merr.)-Bradyrhizobium japonicum symbiosis, using signal molecule genistein. Eur J Soil Biol 45(2): 146-152.






























