In Vitro Evaluation of Porcupine Bezoar Extracts as Anticancer Agent on A549 -A Preliminary Study
Firus Khan AY1, Mohd Nasir MH2, Tara Jalal1, Razali S1, Hassan H1, Asuhaimi FA1 and Wahab RA1*
1Department of Biomedical Science, International Islamic University, Malaysia
2Department of Biotechnology, International Islamic University Malaysia
Submission: June 15, 2017; Published: July 28, 2017
*Corresponding author: Ridhwan Abdul Wahab, Department of Biomedical Science, Kuliyyah of Allied Health Sciences, International Islamic University Malaysia, Malaysia, Tel: +06-570-5262; Email: ridhwan@iium.edu.my
How to cite this article: Firus K AY, Mohd N MH, Tara J, Razali S, Hassan H, Asuhaimi FA, Wahab RA. In Vitro Evaluation of Porcupine Bezoar Extracts as Anticancer Agent on A549 -A Preliminary Study. Adv 007 Biotech & Micro. 2017; 5(1): 555651. DOI:10.19080/AIBM.2017.05.555651
Abstract
Porcupine bezoar (PB) was reported to possess medicinal properties in old medical manuscript. However, its potential as anticancer agent on human lung cancer cells (A549) is not yet studied. In present study, porcupine bezoar was tested to observe its ability in inhibiting cell growth of cancer cell (A549) and its cytotoxicity on Normal Human Gingival Fibroblast Cell (HGF-1). A549 cells morphology was observed after treated with bezoars for 72 hours. The ability of bezoars to induce DNA damage and apoptosis was analyzed by staining cells with Hoechst 33428(nucleus) and Rhodamine Phalloidin (f-actin). The A549 IC50 is 13.6±1.58^g/ml A549 growths was inhibited in dose-dependent pattern, but no inhibition found on normal HGF-1 cells. Treated A549 morphology shows sign of apoptosis such as DNA fragmentation, cytoplasm shrunk and vacuolation. The finding in this study suggests PB extracts able to inhibit cell growth, induce DNA damage and apoptosis, further analysis need to be done to verify the mechanism.
Keywords: A549; Porcupine bezoar; Proliferation; Apoptosis; DNA damage
Introduction
Lung cancer is one of the leading cause for mortality in most developing and developed country. Incidence in 2015 reported lung cancer as the second leading in male and female by 14% and 13% respectively [1]. Additionally, estimated death reported in 2016 shows that lung cancer had the highest mortality in male and female by 27% and 26% respectively [2]. Apart from so many research done and developing new cancer treatments to cure cancer, a recent study in 2016 reported that less than ~1.0% mortality rate due to cancer had decreased in which indicating the effort is still not enough [3]. Furthermore, common treatment for non-small lung cancer is surgery, chemotherapy and radiation therapy depending on stages [4]. One of the long term side effects from the treatments is impaired lung function. Hence, studying new anticancer agent with less side effects and target specifically becomes urgent to control increasing cancer cases every year.
Anticancer study is known as tertiary prevention, which focuses on the malignant cell. It is known that tumor cells replicate rapidly and differentiate into malignant cells, hence the study of anticancer is targeting on specific malignant cells which able to metastasize [5]. Current anticancer drugs are targeting to block molecular signals which promote cancer cell proliferation, obstruct cell death, cell differentiation and angiogenesis [6].
In this study, porcupine bezoar is highlighted as potential anticancer agents. Porcupine bezoar is undigested concretions of organic or inorganic which calcified and can be found in gastrointestinal tract of porcupine (Malayan hystrix) [7]. The bezoars found can be either round or elongated with peel like an onion, the colour range from green grass of blackish to green hue[8] . Bezoar was well known as the prince of antidotes due to its medicinal properties and was used as earliest 8th century [8,9]. However, the understanding of porcupine bezoar on anticancer activities is poorly understood. Hence, the principle objective of this study was to investigate how porcupine bezoar extracts affecting A549 cells on cell growth and morphology.
Materials and Methods
Maintaining cell culture
Human lung cancer cells (A549) and Normal Human Gingival Fibroblast (HGF-1) were obtained from American Type Culture Collection (ATCC), USA. Cells were grown in complete growth media of Dulbecco's modified eagle medium (DMEM) (Gibco, USA) with 10% Fetal Bovine Serum (Gibco, USA) & 1% of Penicillin-Streptomycin (Gibco, USA). Cells were incubated in 37 °C of 5% CO2 humidified atmosphere.
Porcupine bezoar extracts (PB extract) preparation
This study is granted permission from Malaysian Government Wild Life and National Parks Department as porcupine bezoar is strictly controlled material to protect porcupine which are an endangered species animals (JPHL&TN (IP): 100-34/1.24 Jld 8). The study conducted is for academic and research purposes. The PB extract was prepared by adapting traditional method[9] . Porcupine bezoar powder was prepared by soaking extract in DH20 for 3 days before filtered. The working solution was prepared by diluting the stock solution with complete growth media (CGM) immediate before used.
IC50 determination
The viability of the A549 was determined using MTT assay, cells of 5.0 X 103 were seeded in 24-well plate with 2ml of CGM. Before treatment with serial concentration (5.0-160.0μg/ml) of PB extract, cells were washed with Phosphate buffer saline (PBS) (Gibco, USA) and fresh media added. After 72h of exposure with PB extract, medium was discarded subsequently cells were exposed to MTT solution (Sigma Al-drich, USA) that was prepared at 5mg/mL in sterile PBS prior incubated at 37 °C for 3h. The water insoluble for mazan salts was solubilised with 1ml of DSMO/well. Absorbance was read using infinite Pro200 TECAN, the primary wavelength of 570nm. Each plate consists of negative control, samples, and blank. IC50 was determined using Graph pad prism version 6.0.
Cell proliferation assay
Cells with a density of 5 X 104cells/ml were plated in 6 well plates. After incubated for 24 hours cells were washed and PB extract at IC50 added for treated cells. Untreated cells served as negative control. Cells were grown in 5% CO2 at 37 °C for 24, 48 and 72 hours. Cells proliferation rate was determined using trypan blue dye (Sigma Al-drich, USA) exclusion method. At 24, 48 and 72 hours cells were counted to investigate the pattern of treated and untreated cells growth rate.
Cytotoxicity assay on normal cells
Approximately 5 X 104 of normal Human Gingival Fibroblast (HGF-1) were seeded in 6 well plates. Once cells attached, old media were discard, cells were washed with PBS before new media with a serial concentration of PB extracts (10, 25, 50, 75, 100 and 160μg/ml) were added and incubated for next 24,48 and 72 hours. Viable cells were counted using Tiypan blue dye exclusion method. The viability percentage of treated and untreated cells was compared.
Cell morphological analyses
Cells morphology was analyzed at the same time with cells proliferation assay. The cells morphology was observed after 72 hours of incubation before the cells were trypsinized for cell count. Cells were examined under an inverted microscope at low (10X) and high (40X) magnification.
Cytoskeleton analysis
The A549 treated and untreated cells which served as negative control were grown on cover slip in 6-well plates for 72 hours. Cells on cover slips were fixed with 4% Paraformaldehde (Sigma Al-drich, USA) for half hour and washed with PBS thrice before permeabilized with 0.1% of Triton X-100(Sigma Al-drich, USA) for one minute. Subsequently washed thrice, cells were stained with Rhodamine-Phalloidin (Molecular Probe, USA) and counter stained with Hoechst 33428(Molecular Probe, USA) for 10 minutes prior mounted with 50% glycerol (Sigma Al-drich, USA). Cells were observed under fluorescence for the cytoskeleton analysis.
DNA fragmentation assay
Further analysis was carried out to observe DNA damage. The assay aim was to observe for DNA fragmentation using Hoechst 33428 staining. PB extracts treated and untreated cells were grown on cover slip in 6-well plate and incubate for 72 hours. Cells were fixed with 4% Paraformaldehde before washed with PBS. Cells then permeabilized with 0.1% Triton-X prior stained with Hoechst 33428 and mounted with glycerol. Cell's DNA fragmentation was observed using fluorescence microscope.
Statistical analysis
Statistical analysis of all data was performed using a one way analysis variance (ANOVA) followed by Bonferroni correction. (Graph-Pad Prism 6.0; Graph Pad Software, Inc., CA, USA). P*, 0.01 denoted significance in all cases.
Results
50% Inhibitory concentration
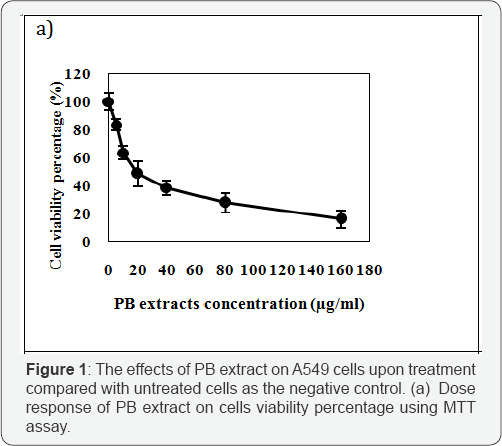

Anticancer agents have the common ability in inhibiting cell progression. As far as our literature review could ascertain, no specific information was found on specific dosages of porcupine bezoar used previously. As shown in (Figure 1a), the percentages of viable cells upon treatment with serial concentration of 5.0- 160.0|ig/ml were decreased corresponded with the increasing dosage of the PB extracts used. Results indicate that the effect of the extracts on A549 CF7 was in a concentration-dependent manner. From the graph, the IC50 was found to be at 13.6 ± 1.58μg/ml for A549 cells.
Proliferation assay
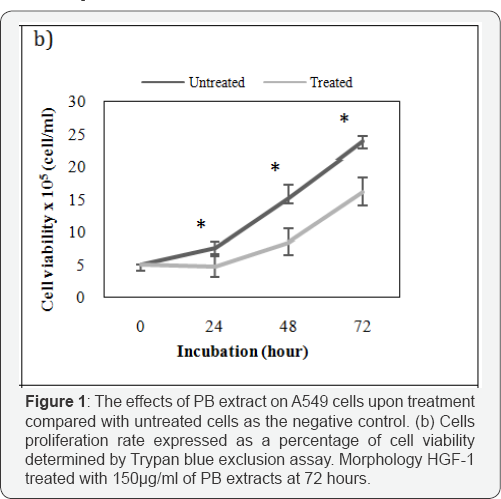
Results from IC50 determination for A549 shown that PB extracts exhibit a concentration-dependent pattern Hence, to verify the effect of the PB extracts on cell growth pattern with respect to time, the cell proliferation assay was conducted. PB extracts exposure on A549 in (Figure 1b) shown cytotoxic effect and cause reduction cell growth activity of A549 cells by difference by 2.8 X 105, 6.8 X 105, 7.7 X 105 cells/ml at 24,48 and 72 hours respectively. The result demonstrated PB extracts induced cytotoxic effect in concentration-dependent pattern when compared to untreated.
Cytotoxicity assay
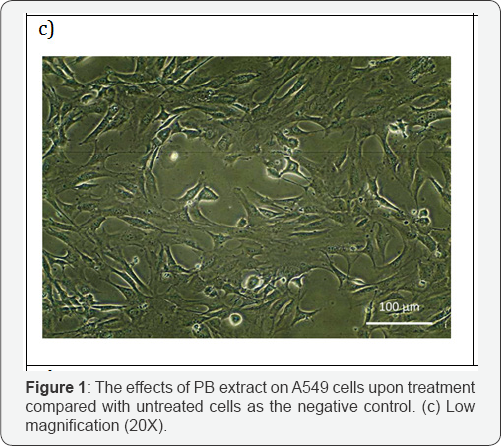
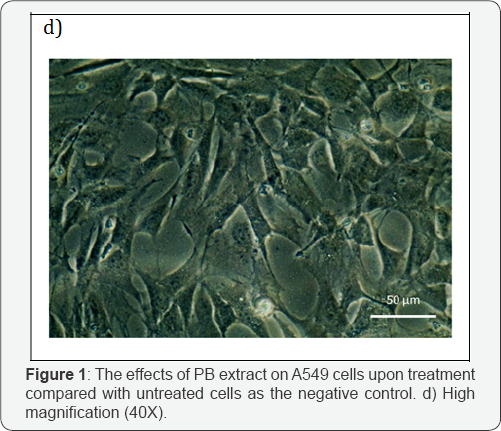
This part of test was done to investigate the effect of PB extracts on non-cancerous human cells as reported earlier; the extracts have effects on cancer cell. Morphology of treated HGF-1 at 150μg/ml in 72 hours demonstrated neither visible sign of apoptosis nor necrosis. Cells in Figure 1c-1d appeared in high density, with intact nucleus and cytoplasm membrane as it attaching to its neighboring cells. Additionally, Figure 1e shown that the HGF-1 viable cell percentage of PB extracts treated was higher in all concentration and incubation hours compared to untreated. PB extracts exposure on normal cells HGF-1 demonstrated neither cytostatic nor cytotoxic.
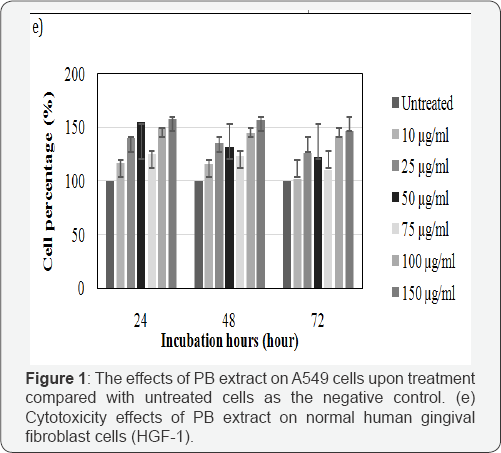
Morphology assay
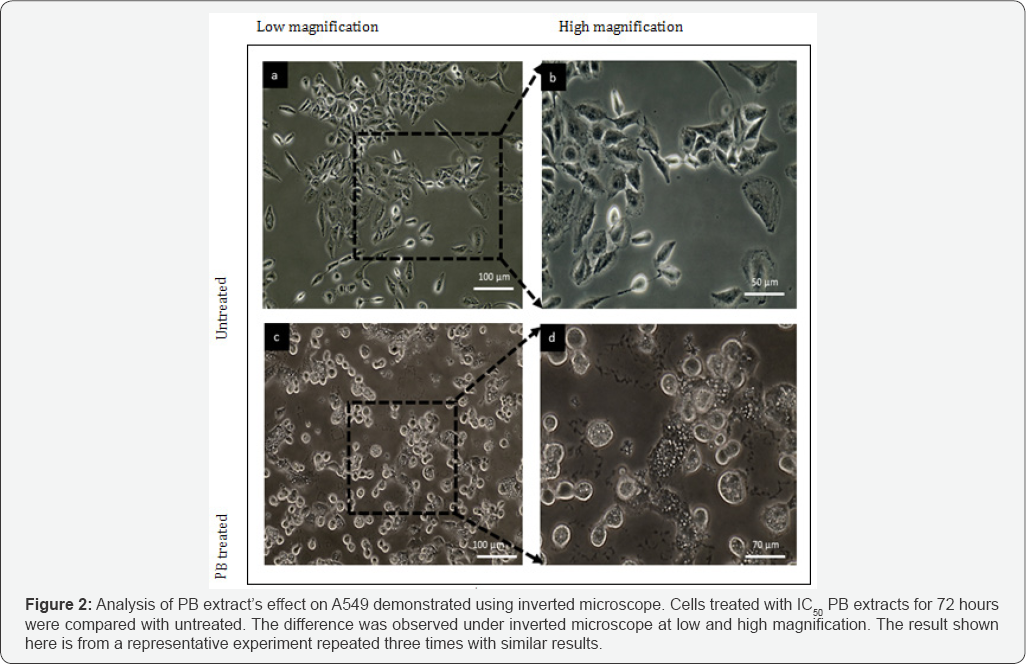
To determine the morphological changes of the A549, the cells were treated at what concentration for 72 hours. Untreated cells were used as a negative control for comparison. Untreated cells demonstrated grown attaching to neighboring cells in monolayer and the morphology of the cells shown no abnormalities. However, A549 cells Figure 2 clearly shown the infamous effect of PB extract. Treated A549 displayed unhealthy and apoptotic features. Figure 2demonstrated the difference between untreated cells of A549 with PB extracts treated at low (10X) and high (40X) magnification. Treated cells exhibit sign of apoptosis, cells appeared to be in rounded shape, nucleus condensed and the cytoplasm shrunk whilst still attached to plate. Furthermore, the cells also exhibit vacuolation which was one of the signs of apoptosis and paraptosis. Additionally, it can be seen that the untreated cells were shown to reach out other cells and expanding its cell membrane, but for treated cells, the cytoplasm rounded up and detached from its neighbour cells.
Cytoskeleton analysis
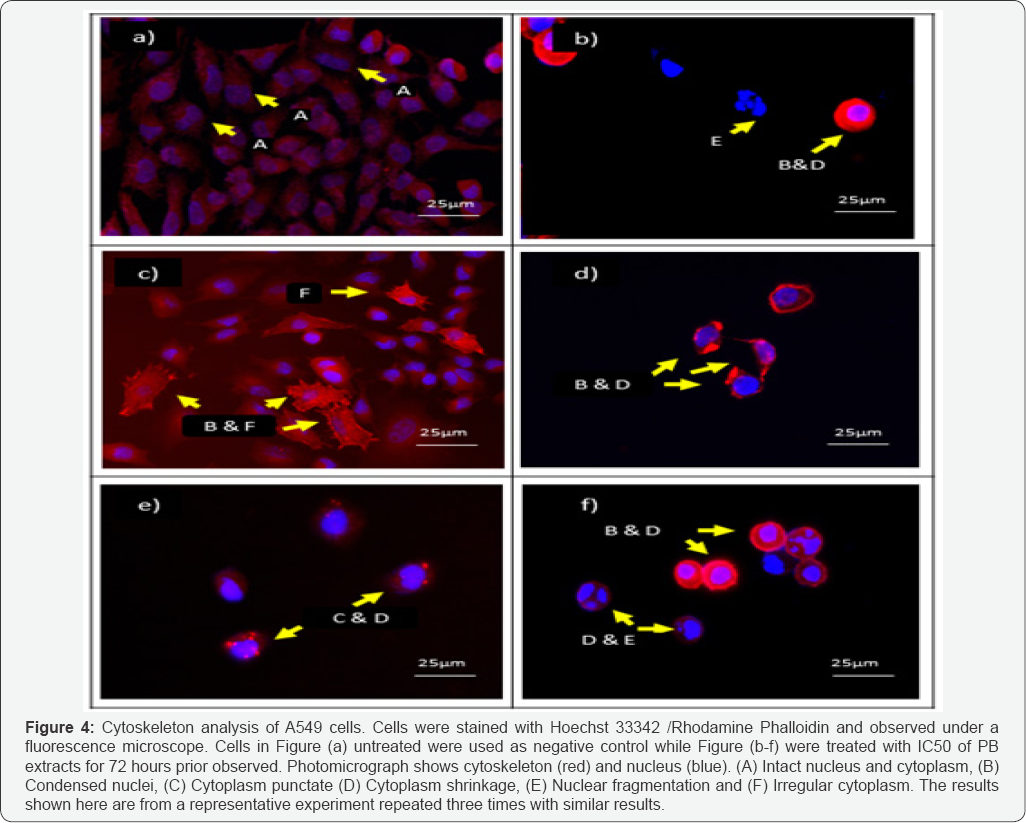
Cytoskeleton analysis done to investigate morphological changes of f-actin on A549 treated with PB extract. Cells were counter stained with Hoechst 33428 for nucleus and Rhodamine- Phalloidin for the cytoskeleton (f-actin). The cytoskeleton structure of cells stained blue for nucleus and bright red for filament actin. Figure 3a demonstrated nucleus of the untreated appeared intact and the cytoplasm of the cells displayed attaching to its neighboring cells with absence abnormalities. Untreated cells demonstrated well-developed actin cytoskeleton with sub cortical actin filaments was observed in the cells Differently, in Figure 3b-3f, A549 cells treated with PB extracts exhibited features of apoptosis where the cytoplasm appeared punctate, irregular and shrunk. Furthermore, the shrunk cytoplasm emitted brighter red colour compared to untreated suggesting the actin aggregate and volume of cytoplasm decreased. Interestingly, the f-actin of the PB extract treated cells for 72 hour appeared densely at the perimeter of the cells which opposite to the appearance observed in untreated cells.
DNA damage assay
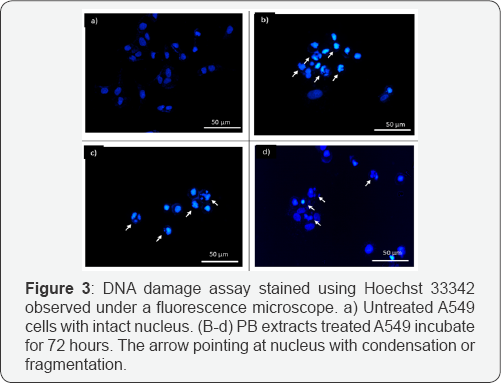
Earlier result pointing cell inhibition and morphological changes with apoptosis feature occurred. Hence further test was done to investigate whether cells experience DNA damage post PB extracts exposure. Cells were stained with Hoechst 33428 which binds specifically at adenine-thymine (A-T) regions of DNA and stained the minor groove of DNA. Figure 4a displayed intact morphology of stained nucleus. On the other hand, arrows in Figure 4b-4c pointing at nucleus which indicates condensation and fragmentation which is a hallmark feature of late stages apoptosis.
Discussion
To the best available knowledge we had, no report that evaluates PB extracts in details neither in-vivo nor in-vitro study. Bezoar was remembered as a medical relic which possesses variety medicinal properties which mostly mention in medical manuscripts only. Therefore, in the present study, several observations have been made concerning the inhibitory concentration, cell proliferation, morphology, cytoskeleton analysis and cell DNA damage.
In present study anti proliferation analysis, PB extracts demonstrated to exhibit cytotoxic effect and cause cell growth activity reduction in dose-dependent only. A study reported similar result where tamoxifen worked only at specific concentration without having any time dependant effect on different type of breast cancer cells and demonstrated their ability to survive at a higher dose and longer duration of exposure drugs [10]. Cytotoxicity study on human noncancerous cells suggesting PB extracts not toxic towards tested cells.
The biochemical hallmarks and distinct morphology of apoptosis has been characterized by cell shrinkage, membrane blebbing, or deformation of the cells and loose its contact with the neighboring cells resulting in the appearance of apoptotic bodies containing cytosol, broken organelles and condensed chromatin that will eventually be engulfed and digested by macrophages [11-15].
Morphology finding of PB treated A549 in this study, suggest the cells undergone apoptosis as cellular shrinkage, rounding, poor adherence, vacuolation and nucleus condensed were observed by phase-contrasted microscopy. Similar finding supported where chloroquine induces apoptosis feature with vacuolation on A549 [16]. Treated A549 shown presence of vacuolation, even though the formation of vacuoles was not considered among the hallmarks of apoptosis, but study in 2000 explained the formation of vacuoles sometimes happens in apoptotic cells [17]. This condition was supported [18], where the A375 cells with vacuolation undergone apoptosis through mitochondrial pathway and review study [15] that explain vacuolation could happen in some cells in early apoptosis together with the other morphology of apoptosis. Moreover, Elmore reported that cells in apoptosis, the cells will start to lose the cell-to-cell adhesions by rounding up its cytoplasm, hence separated from neighboring cells. The same author also explained that in late phase of apoptosis, the nucleus would fragment and lead to the development of apoptotic bodies with intact cell membranes that contain cytoplasm organelles.
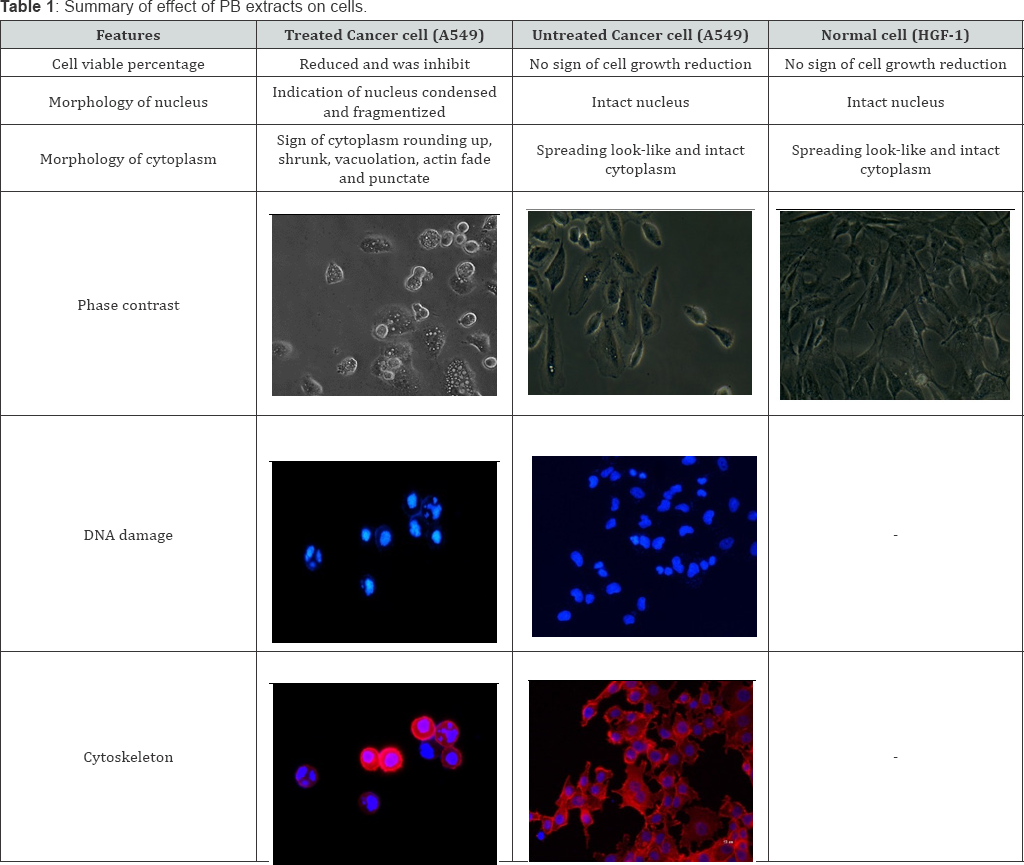
DNA damage assay in this study suggests A549 treated with PB extracts experienced nuclear condensation and fragmentation. DNA damage indicate the nucleus fragmentize into smaller size where the DNA chemical structure altered and breaking (pyknosis) [19]. When cells undergo prominent feature of DNA damage, this marked A549 cells undergone late stage of apoptosis [15,20,21]. Cytoskeleton analysis supported this finding where stained f-actin (red) indicates to exhibit other hall mark of apoptosis which are cell shrunken. According to the literature, actin cytoskeleton plays an important role in apoptosis execution as it influences cell blebbing and karyorrhexis [22]. Additionally, actin of cytoskeleton is crucial in maintaining cellular homeostasis and cell morphology [23]. When the cytoplasm shrunk, it's indicated the volume of the cells decrease, hence cytoplasm rounding up and will detached from flask. This is related to another finding in this study which the actin staining appeared to be fading compared to untreated [24]. Actin depolymerization as White reported in his study suggested actin depolymerization later leads to DNA degradation [25]. The same study also reported actin degradation makes the cells ready from detachment which explains treated A549 that have faded cytoplasm exhibit rounding up morphology instead of spreading lookalike compared to untreated. Additionally, Figure 3 (b-f), treated cells exerted punctate in the cytoplasm, the punctate indicating the actin cytoskeleton rearranged there to hold vacuoles. This appearance similarly reported in where the punctate appears due to rearranging of the cytoskeleton in apoptosis cells [26]. These findings will aids in understanding PB extract ability on anticancer, but also help to development a potential anticancer agents targeting on inducing apoptosis by disrupting actin cytoskeleton and fragmentize DNA. Table 1 summarized the effects of PB extracts on HGF-1, treated and untreated A549 [26].
Conclusion
In summary, based on the result from this study supported from literature review, it is evidenced that PB extracts might induce cell growth inhibition in concentration dependent pattern on A549 with no sign of toxicity on normal human fibroblast cells (HGF-1) The morphology analysis suggested PB extract promote destabilize of actin and induced DNA damage hence lead to apoptosis on A549 cells. However, further study is needed to investigate the mechanism behind it.
Acknowledgement
This work is funded by Fundamental Research Grant Scheme (FRGS) by High education ministry. Special thanks to staff of Integrated Centre for Research Animal Care & Use (ICRACU), Microbiology laboratory, Kuliyyah of Medicine and Kuliyyah of Allied Health Science IIUM, Kuantan Malaysia for assistance during this research.
Conflict of Interest
Authors declared there is no conflict of interest.
References
- Siegel R, Miller K, Jemal A (2015) Cancer statistics. CA Cancer J Clin 65(1): 5-29.
- Siegel R, Miller KD, Jemal A (2016) Cancer statistics. A Cancer J Clin 66(1): 7-30.
- Hashim D, Boffetta P, La Vecchia C, Rota M, Bertuccio P, et al. (2016) The global decrease in cancer mortality: trends and disparities. Ann Oncol 27(5): 926-933.
- DeSantis CE, Lin CC, Mariotto AB, Siegel R, Smith T, et al. (2016) Cancer Treatment and Survivorship Statistics, 2012. Cancer J Clin 62(4): 220241.
- Xu J, Mao W (2016) Overview of Research and Development for Anticancer Drugs. J Cancer Ther 10(7): 762-772.
- Pan JG, Mak TW (2007) Metabolic targeting as an anticancer strategy: dawn of a new era?. Sci STKE 381: 14.
- Sanders MK (2004) Bezoars: From Mystical Charms Management. Nutr Issues Gastro enterol, pp. 37-50.
- Barroso MDS (2014) The Bezoar Stone: A Princely Antidote, The Tavora Sequeira Pinto Collection-Oporto. Acta Med Hist Adriat 12(1): 77-98.
- Duffin CJ (2013) Porcupine Stones. Pharm Hist (Lond) 43(1): 13-22.
- Karami-Tehrani F, Salami S (2003) Cell Kinetic Study of Tamoxifen Treated MCF-7 and MDA-MB 468 Breast Cancer Cell Lines. Iran Biomed J 7(2): 51-56.
- Majno G, Joris I (1995) Apoptosis, oncosis, and necrosis. An overview of cell death. Am J Pathol 146(1): 3-15.
- Trump BE, Berezesky IK, Seung H, Phelps PC (1997) Pathways Oncosis, Apoptosis, and Necrosis. Toxicol Pathol. 25(1): 82-88.
- Cruchten SV, Broeck WVD (2002) Morphological and biochemical aspects of apoptosis, oncosis and necrosis. Anat Histol Embryol 31(4): 214-223.
- Fink SL, Cookson BT (2005) Eukaryotic Cells Minireview Apoptosis , Pyroptosis , and Necrosis : Mechanistic Description of Dead and Dying Eukaryotic Cells. Infect Immun 73(4): 1907-1916.
- Elmore S (2007) Apoptosis: A Review of Programmed Cell Death. Toxicol Pathol 35(4): 495-516.
- Fan C, Wang W, Zhao B, Zhang S, Miao J, et al. (2006) Chloroquine inhibits cell growth and induces cell death in A549 lung cancer cells. Bioorg Med Chem 14(9): 3218-3222.
- Häcker G (2000) The morphology of apoptosis. Cell Tissue Res 301(1): 5-17.
- Looi CY, Moharram B, Paydar M, et al. (2013) Induction of apoptosis in melanoma A375 cells by a chloroform fraction of Centratherum anthelminticum (L) seeds involves NF-kappaB, p53 and Bcl-2- controlled mitochondrial signaling pathways. BMC Complement Altern Med 13(1): 166.
- Ioannou YA, Chen FW (1996) Quantitation of DNA fragmentation in apoptosis. Nucleic Acids Res 24(5): 992-993.
- Collins JA, Schandl CA, Young KK, Vesely J, Willingham MC, et al. (1997) Major DNA Fragmentation Is a Late Event in Apoptosis. J Histochem Cytochem 45(7): 923-934.
- Plesca D, Mazumder S, Almasan A (2008) DNA damage response and apoptosis. Methods Enzymol 446(8): 107-122.
- Craig DH, Zhang J, Basson MD (2007) Cytoskeletal signaling by way of α-actinin-1 mediates ERK1/2 activation by repetitive deformation in human Caco2 intestinal epithelial cells. Am J Surg 194(5): 618-622.
- Ahn YT, Shin IJ, Kim JM, Kim YS, Lee C, et al. (2013) Counteracting the activation of pAkt by inhibition of MEK/Erk inhibition reduces actin disruption-mediated apoptosis in PTEN-null PC3M prostate cancer cell lines. Oncol Lett 6(5): 1383-1389.
- Taatjes DJ, Sobel BE, Budd RC (2008) Morphological and cytochemical determination of cell death by apoptosis. Histochem Cell Biol 129(1): 33-43.
- White SR, Williams P, Wojcik KR (2001) Initiation of apoptosis by actin cytoskeletal derangement in human airway epithelial cells. Am J Respir Cell Mol Biol 24(3): 282-294.
- Dong QM, Ling C, Zhao L (2015) Immuno fluorescence analysis of cytokeratin 8/18 staining is a sensitive assay for the detection of cell apoptosis. Oncol Lett 9(3): 1227-1230.






























