Enrichment Blood Culture Isolation of Bartonella henselae from Horses with Chronic Circulatory, Musculoskeletal and/or Neurologic Deficits
Brenda L Bishop1, Natalie A Cherry2, Barbara C Hegarty3 and Edward B Breitschwerdt3*
1Sport Horse Associate, USA
2Galaxy Diagnostics Inc, Research Triangle Park, USA
3Department of Clinical Sciences and the Intracellular Pathogens Research Laboratory, North Carolina State
Submission: April 13, 2017; Published: July 27, 2017
*Corresponding author: Edward Breitschwerdt, Department of Clinical Sciences and the Intracellular Pathogens Research Laboratory, Comparative Medicine Institute, College of Veterinary Medicine, North Carolina State University, 1060 William Moore Dr., Raleigh, North Carolina, USA, Tel: 919-513-8277; Fax: 919-513-6464 ; Email: ed_breitschwerdt@ncsu.edu
How to cite this article: Bishop LB, Cherry NA, Hegarty CB and Breitschwerdt EB. Enrichment Blood Culture Isolation of Bartonella henselae from Horses with Chronic Circulatory, Musculoskeletal and/or Neurologic Deficits. Adv Biotech & Micro. 2017; 4(5): 555646. DOI: 10.19080/AIBM.2017.04.555646
Abstract
Background: Because published case-based evidence is limited and only one experimental equine infection study has been reported to date, bartonellosis, a recently recognized equine disease, is rarely considered as a differential diagnosis by equine clinicians. Microbiological documentation of Bartonella spp. infections has been difficult to achieve in the laboratory; therefore, elucidation of the potential clinical and pathological aspects of chronic bartonellosis in horses has not been thoroughly investigated.
Objective: To summarize the historical, clinical and laboratory findings in horses with microbiologically confirmed Bartonella spp. bacteremia.
Study Design: During medical consultations, testing for Bartonella spp. bacteremia was recommended for horses with histories of chronic circulatory, musculoskeletal and/or neurologic deficits.
Methods: Bartonella ePCR™ (an adaptation of the Bartonella alpha Proteobacteria growth medium [BAPGM] enrichment blood culture platform) was performed by Galaxy Diagnostics, Inc. to establish intravascular infection (bacteremia) in each horse. Bartonella henselae indirect fluorescent antibody testing was performed for horses with documented bacteremia.
Results: Within a 14 month time frame, the authors identified nine B. henselae bacteremic horses out of the total number (n=22) tested. Historical abnormalities included exercise intolerance, shifting leg lameness, circulatory deficits, cranial nerve dysfunction, endocrine imbalances and neurologic impairment. Serum samples were available from five of the nine bacteremic horses, and one horse was sero- reactive with antibody titer of 1:128 to Bartonella henselae antigens.
Main limitations: Variability of disease manifestations in these horses may decrease the clinical applicability of these findings. All horses had received prior therapeutic efforts while this study focused upon Bartonellosis, therefore, the presence and effect of other infectious or non-infectious diseases was not evaluated in the context of this study.
Conclusion: Prospective controlled studies are needed to define the duration of bacteremia, diagnostic testing criteria, and treatment outcomes for horses infected with B.henselae.
Keywords: Bartonellosis; Horses; BAPGM; Bartonella henselae
Supplementary Material
Indirect fluorescent antibody testing using a panel of Bartonella species antigens and the BAPGM enrichment blood culture/PCR platform, with DNA sequence confirmation of the infecting Bartonella species is available for clinical diagnostic and research study purposes through Galaxy Diagnostics, Inc.
Introduction
Genus Bartonella is currently comprised of at least 34 species or subspecies of fastidious, intracellular, Gram-negative aerobic bacteria [1-3]. Bartonella spp. has been increasingly recognized as pathogens of substantial medical importance in cats, dogs and humans [1-3]. However, equine bartonellosis remains largely undefined as a disease of horses. These highly adaptive, intravascular and endotheliotropic bacteria are transmitted among domestic and wild animals throughout the world, primarily by blood-feeding arthropod vectors, including bedbugs, biting flies, fleas, lice, mites, sand flies, ticks and potentially spiders [4-6]. In addition, bites, scratches, needle sticks, and for some species, transplacental, have all been implicated as modes of disease transmission [1-4]. Persistent intravascular infection with different Bartonella spp. has been reported in numerous pet, production and wildlife animals [13]. Following transmission in nature, duration of bacteremia in horses is unknown, whereas following experimental intra dermal inoculation of four horses with culture-grown Bartonella henselae, three sero-converted and developed bacteremia and mild cutaneous disease manifestations [7]. It was not until 2008 that B. henselae was isolated or detected for the first time by polymerase chain reaction (PCR) amplification in the blood of two adult horses from North Carolina (USA), one with vasculitis and the other with chronic intermittent shifting leg lameness [8]. In 2009, B. henselae infection was documented in an aborted equine fetus in Indiana (USA) [9], and in 2011, B. henselae DNA was amplified from the bone marrow and spleen of a horse that succumbed to hemolytic anemia in Germany [10]. More recently, B. henselae was isolated using the BAPGM enrichment blood culture approach from a Thoroughbred foal with granulomatous hepatitis, pathology that had been previously reported in dogs and humans [11].
Serology, PCR amplification of organism-specific DNA sequences from blood, and conventional blood culture isolation techniques have not proven useful for confirming a diagnosis of canine, equine or human Bartonella bacteremia [12-15]. Relatively recent advances in microbiological culture technology have enabled more sensitive documentation of B. henselae bacteremia in foals and in horses with presumptive vasculitis and chronic arthropathy [13]. The purpose of this study was to summarize historical, clinical and laboratory findings in horses with laboratory confirmation of Bartonella spp. bacteremia using Bartonella ePCR™ (Galaxy Diagnostics, Inc. Morrisville, NC USA) platform.
Materials and Methods
Horses included in this study had a history of chronic symptoms in conjunction with poor skeletal muscle exercise tolerance, microcirculatory deficits, and/or neurologic dysfunction. Chronicity was based upon owner reported symptoms extending over a period of at least two years. Poor exercise tolerance was defined as delayed forward phase of stride and/or muscle weakness (with or without muscle spasms) and/or shifting leg lameness. Microcirculatory involvement was defined in terms of sequelae indicative of poor arterial oxygenation of soft tissue(s) and/or chronic inflammation in a wide range of tissues and/or evidence of impaired venous and lymphatic drainage. Neurologic dysfunction was defined as unexplained cranial nerve deficits and/or symptoms consistent with neurotransmitter imbalances. Additional historical abnormalities involving endocrine system impairment, a finding reported in other mammalian species with confirmed bartonellosis, were considered as adjunct criteria for Bartonella bacteremia testing.
Historical, Physical Examination and Blood Specimen Collection
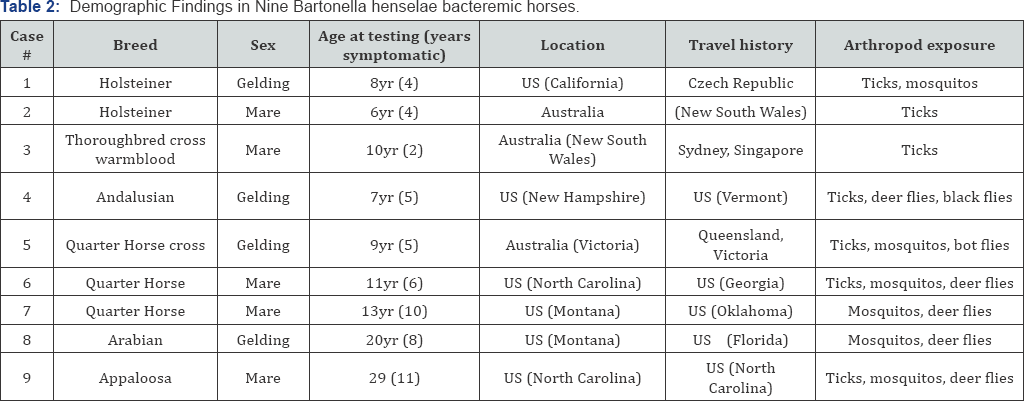
Study population: Each owner provided historical and demographic data for their horses' travel and arthropod exposure history. Physical examinations were concurrently performed by the respective primary care veterinarians in conjunction with blood specimen collection from each horse on three alternate days. Ethylene diamine tetraacetic acid (EDTA)- anti-coagulated blood and serum samples from horses were shipped to Galaxy Diagnostics, Inc. (Research Triangle Park, NC, USA) from Australia and the United States for Bartonella ePCR™ testing.
Bartonella IFA serology
As previously described Cherry NA et al. [14], IFA was performed to document detection of Bartonella antibodies using B. henselae (San Antonio-2, and Houston-1 strains), B.vinsonii subsp. berkhoffii (genotypes I and II), B. koehlerae and B. quintana organisms grown in mammalian cells as antigens. Serum was available from 10 of the 22 horses described in this study. Sera (kindly provided by Dr. Bruno Chomel, University of California, Davis) from three horses experimentally-infected with B. henselae (SA2 strain, isolate designation NCSU 2008- EO-1) were used as IFA positive controls. In our assay, endpoint titers for these three horse sera were 1:512, 1:512, and 1:2048. The starting dilution was 1:16, with endpoint titers defined as the last dilution at which brightly-stained organisms could be detected by fluorescence. A cut-off titer defining sero-reactivity was arbitrarily defined as 1:64.
Bartonella ePCRTM
Bartonella ePCRTM, an adaptation of a previously-described approach that combines PCR amplification of Bartonella spp. DNA from blood before and after inoculation into BAPGM, was performed [12,13,15,16]. For each blood sample, 2mL of EDTA blood was spiked into 10mL BAPGM and incubated at 35-37 °C with 5% CO2. DNA was extracted directly from 200µl of EDTA blood and from 200µl of BAPGM enrichment blood cultures after three incubation time points (8, 14 or 15, and 21 or 22 days) using an automated QIA Symphony Workstation and DSP DNA blood mini kit (Qiagen, Valencia, CA). As extraction controls, a positive culture control consisting of BAPGM spiked with B. henselae and a negative culture control containing only BAPGM were processed simultaneously with each sample set to ensure extraction was completed successfully and that extraction buffers and reagents were not contaminated with Bartonella DNA. Bartonella DNA was amplified using conventional Bartonella genus primers targeting the 16S-23S intergenic spacer region (ITS) as previously described [14,17].
In addition, Bartonella genus qualitative real-time PCR (qPCR) was performed targeting the Bartonella 16S-23S ITS gene using oligonucleotides 325s (5' CTTCAGATGATGATCCCAAGCCTTYTGGC3') and 438as (5' GCCCTCCGGGRTAAAYCGGAAACC 3’) as forward and reverse primers, respectively. Amplification was performed in a 25µl final volume reaction containing 12.5µL of MyTaq Premix (Bioline USA, Inc., Taunton, MA) for conventional PCR or SensiFAST™ SYBR No-ROX kit (Bioline USA, Inc., Taunton, MA) for qPCR, 0.3µL of 100|imol/L of each forward and reverse primer (IDT®DNA Technology, Coralville, IA), 6.9µiL molecular grade water, and 5µL DNA from each sample tested. Amplification was performed using an Eppendorf Mastercycler EPgradient® (Eppendorf, Hauppauge, NY) for conventional PCR Time PCR Detection System (Bio-Rad, Inc., Hercules, CA) for qPCR with 0.01 and 0.001 pg/µiL B. henselae SA2 genomic DNA as PCR positive controls. Negative controls included BAPGM and molecular-grade water. For conventional PCR, thermocycling conditions consisted of initial denaturation at 95 °C for 2 minutes, followed by 55 cycles with denaturation at 94 °C for 15 seconds (s), annealing at 68 °C for 15s, and extension at 72 °C for 18s. PCR products were analyzed by 2% agarose gel electrophoresis. For qPCR, thermo cycling conditions consisted of initial denaturation at 95 °C for 2 minutes, followed by 45 cycles with denaturation at 94 °C for 10s, annealing at 66 °C for 10s, and extension at 72 °C for 10s. Positive amplicons were sequenced to identify Bartonella sp. and ITS genotype. Bacterial species and genotype were defined by comparing similarities with other sequences deposited in the GenBank database using the Basic Local Alignment Search Tool (BLAST version 2.0).
Results
Bartonella pre and post-enrichment PCR and IFA serology results for the nine bacteremic horses are summarized in Table 1. Demographic findings for the B. henselae bacteremic horses are summarized in Table 2. Each of these horses had histories of poor exercise tolerance, microcirculation deficits, neurological abnormalities, neuroses, and/or evidence of endocrine system impairment. All nine horses had histories indicative of varying degrees of poor exercise tolerance. Delayed forward phase of stride was manifested as a tendency to land toe first (cases 1, 2, 3 and 9), toes worn flat as a result of toe dragging (cases 1, 3, 5 and 9), reluctance to move forward under saddle (cases 1, 3, 5 and 6), refusal to move at all with or without rearing (cases 5 and 6), subluxating patellas (locking stifles) (cases 1 and 4) and pastern and fetlock subluxation (case 2). Four horses (cases 4, 7, 8 and 9) were prone to stumbling on level as well as uneven surfaces. Skeletal muscle abnormalities included a wide range of symptoms: total body soreness (cases 1, 2, 5, 6, 7, 8 and 9), back soreness with top line muscle atrophy (cases 1, 4, 5, 6, 7 and 9), rigid muscles (cording) between hips and stifles (cases 1, 3, 4, 5 and 6), cross firing (disunited cantering, skipping) at the canter (cases 1, 3, 4, 6 and 7), chronic inter costal muscle soreness (cases 1, 4, 6 and 8), tendency to travel hollow backed and high headed (cases 5 and 8), grunting under saddle (cases 6 and 7), shivers (case 2), flaring nostrils at rest (case 2), esophageal muscle spasms (choke, swallowing problems) (case 9) and dropped head syndrome (case 1). Smooth muscle abnormalities included dribbling urine (with odd urination habits) (cases 1, 4 and 6), constipation (scant dry manure) (cases 4 and 9) and bloated abdomen (cases 2 and 5). Two horses (cases 2 and 4) exhibited shifting leg lameness (front to back and/or side to side) that would wax and wane over time.
Microcirculation deficits were reported in terms of hypersensitivity to cold weather, particularly cold wet weather (cases 3, 5, 6 and 8), absence of dapples (cases 1, 5, 6, 7 and 8) , pale mucous membranes including the tongue (cases 3 and 5) and increased central arterial and venous pressures (case 1). Complications associated with poor tissue oxygenation included recurring scratch-like lesions (cases 1, 2, 7 and 9), fungal skin conditions resistant to treatment (streptothricosis) (cases 1, 2 and 4), yeast-like secretions in the sheath of geldings (cases 1 and 4), swollen joints (knees, hocks, stifles) (cases 2, 5 and 6), cording of lymphatic vessels (cases 1 and 5), chronic exertional rhabdomyolysis (case 4), recurring cellulitis (case 1), chronic thrush in and around the frogs (cases 1 and 9), chronic bacterial foot abscesses (case 1), chronically swollen eyelids ( cases 5 and 9), chronically swollen parotid salivary glands (case 3) and chronic bacterial corneal infections with blepharospasm (cases 1, 4 and 9).
Neurologic abnormalities involved both central and peripheral nervous systems. Head and neck findings included tongue laxity (case 7) and odd compulsive habits involving the tongue (case 4) (hypoglossal nerve), mouthiness (nipping, biting, licking) (cases 4 and 6) and Bell's Palsy (case 7) (facial nerve), hypersensitivity to sound (cases 2 and 6) and balance problems in a moving horse trailer (cases 4 and 7) (vestibulocochlear nerve), intense cranial pruritis during and after exercise (case 6) (trigeminal nerve), repetitive flehmen response (case 3) (olfactory nerve), and in 2 horses (cases 2 and 6) optic nerve related issues: recurrent uveitis (ERU), depth perception deficits (altered spatial behavior), photic headshaking and preference for shade vs. bright light. Three horses (cases 4, 7 and 8) had generalized proprioceptive deficits. Peripheral integument hyper-reactivity was recorded as hypersensitivity to a variety of triggers: rain or bathing (cases 4 and 6), blankets (cases 2, 4 and 6), girthing or cinching (cases 1, 4, 5 and 6), grooming tools (cases 2, 4 and 6), insects or insect bites (cases 4 and 7), strong sunlight or sunburn (case 5), and fly spray (case 9). Additional dermal abnormalities included chronic capped elbows (shoe boils) (case 1), callouses on one or both elbows (cases 1 and 9), and sweet-itch of the mane and/or tail with intense pruritis of ventral midline and sheath area (case 4).
All nine horses displayed neuroses, consistent with neurotransmitter imbalances. Most common were depression (horses 1, 2, 5 and 7), moodiness, grumpiness, an acquired argumentative attitude (cases 5, 6, 7 and 9), and changes in temperament from mild mannered to belligerent (cases 1, 2, 5 and 6). Three horses (cases 5, 6 and 7) displayed anger, aggression and "temper tantrums". Similar behavior patterns included explosive outbursts such as bolting (cases 5 and 6), panic attacks (cases 2 and 3), running backwards during panic attack (cases 2 and 6), violently pulling back while tied (cases 2 and 6), violent kicking episodes (cases 7 and 9), and bizarre fearful hallucinogenic-like behavior (cases 2 and 6). Two horses (cases 4 and 7) displayed frequent yawning. Narcolepsy (falling asleep randomly and/or collapsing) (case 8), random seizures (case 7) and combined attention or hyperactivity deficits (case 6) were reported in 1 horse each.
Evidence of endocrine system impairment occurred in individual horses with goiter (case 5), anhydrosis (case 5), Cushing's syndrome (case 9), irregular estrus cycles (case 6) and chronic intermittent lactation (over a period of five years) (case 9) respectively. Aberrations with immune system components were recorded in terms of allergic reactions following routine vaccination (cases 3, 4 and 6), equine sarcoid lesions (cases 2 and 5) and alopecia aereata (cases 4 and 5). An abundance of miscellaneous performance limiting issues included shortened stride length going downhill (cases 2, 3, 4, 5, 6 and 9), inconsistent behavior from day to day (cases 2, 3, 5, 6 and 7), and extreme separation anxiety (cases 5 and 6). Inability to relax (muscle twitching, trembling, prancing), repetitive behavior (pacing), obsessive compulsive behavior patterns and inability to tolerate changes in routines were common to all nine horses.
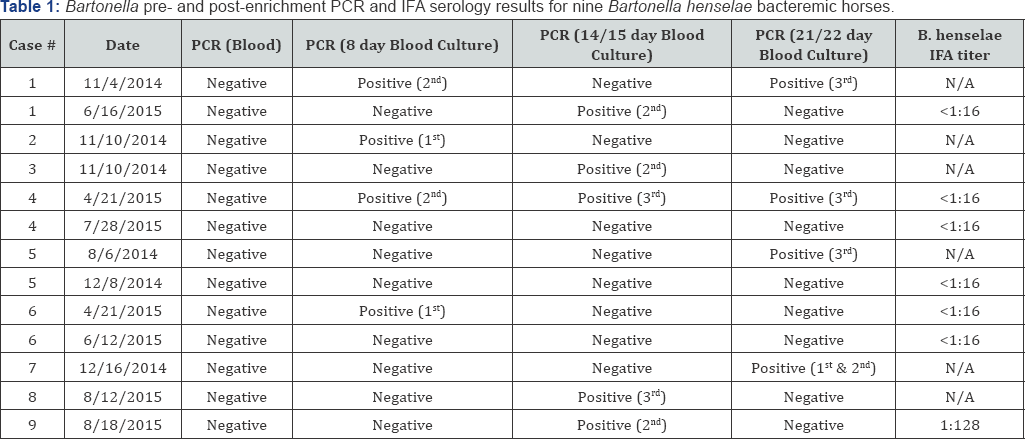
N/A: Serum not available for testing; "1st", "2nd" or "3rd" indicates which blood draw was B.henselae PCR positive.
Discussion
Bartonella henselae bacteremia was documented by BAPGM enrichment blood culture in nine horses with chronic, intermittent, relapsing, and at times progressive symptoms that spanned a several year time frame. Importantly, all of these horses had overlapping symptoms involving the musculoskeletal, nervous, and endocrine and circulatory systems. The extent to which B. henselae bacteremia contributed to individual or the collective symptoms reported in this case series could not be determined. Previously, Merrell and Falkow described the challenges associated with establishing disease causation for stealth pathogens, including members of the genus Bartonella [18]. As vector-transmitted, intracellular, organisms, the evolutionary lifestyle of this genus is characterized by erythrocyte and endothelial cell tropisms. Based upon in vitro studies, B. henselae can also invade monocytes, dendritic cells, CD34+ progenitor cells, pericytes, and synoviocytes (unpublished data) [19-22]. Thus, once bacteremic infection is established, the organism is able to circulate throughout the microvasculature of the host, and presumably localize in microcapillaries within various tissues, thereby involving organs throughout the body. Chronic micro vascular infection, in conjunction with interactions associated with the host immune response, presumably results in a diverse and fluctuating spectrum of symptoms that may span years in duration. Satisfying Koch's postulates has represented the historical benchmark for establishing infectious disease causation; however, the original postulates have substantial limitations when attempting to support disease causation associated with stealth pathogens. Because infections with various Bartonella species induce similar or identical pathology in different animal hosts, including humans, the Postulate of Comparative Infectious Disease Causation was proposed as an additional fifth Koch’s postulate [23].
This postulate states that microbiological documentation of the same infectious agent(s) in three different animal species with identical pathology supports stealth pathogen infectious disease causation. This postulate has medical utility when assessments are based upon common histopathological and microbiological findings in three different animal hosts. It is also possible that the comparative documentation of similar, subtle and non-specific symptoms among different companion animal species may further facilitate our understanding of the biological consequences of long standing B. henselae bacteremia in horses and other non-reservoir-adapted animals. Because animals share the same environment and because their behaviors are generally carefully monitored by their owners, symptoms and behavioral patterns observed in pets may have comparative medical implications for their human counterparts.
Diagnostic confirmation of B. henselae bacteremia in horses remains challenging. For reasons that remain unclear, serology in naturally-infected horses is insensitive. Similar to previous reports [13], only one horse in this study was B. henselae seroreactive, despite a history of chronic symptoms and concurrent diagnostic documentation of bacteremia. When using BAPGM enrichment blood culture to test dogs, cats and humans, shorter incubation periods are often sufficient for growth and detection of Bartonella in bacteremic individuals, whereas incubation for 21 or 22 days was necessary to document bacteremia in all but two of these nine horses. This observation could reflect the possibility that bacteremic horses maintain lower levels of bacteremia compared to most infected cats, dogs or humans, as comparable amounts of blood are inoculated into the liquid medium, regardless of the animal species being tested. When testing humans for evidence of Bartonella spp. bacteremia using BAPGM enrichment blood culture, obtaining specimens from three time points within a seven day period increased diagnostic sensitivity [24]. This approach also appears to have microbiological utility when testing horses, as no horse was PCR positive in all three blood culture specimens. Thus, until a more sensitive microbiological method becomes available, the Bartonella ePCR™ triple draw approach is recommended when assessing Bartonella spp. bacteremia in horses.
There are several limitations inherent in this study. The duration and types of symptoms varied widely among horses that were tested, thereby decreasing the clinical utility of the reported findings. Case controlled studies will be necessary to define specific symptoms or symptom patterns that would support the need for Bartonella spp. diagnostic testing. Clinical evaluations were performed by different equine clinicians, in different countries, all dealing with a broad range of economic, logistical and technical limitations related to specimen collection and the provided medical care. For all horses in this study, there had been extended, prior diagnostic and therapeutic efforts to manage their symptoms. Depending upon geographic location and clinician preference, some horses were tested for other infections contributing evidence of exposure to Borrelia and Toxoplasma spp. Testing performed by the authors was directed solely at determining if a horse was Bartonella spp. bacteremic. Thus, other infectious or non-infectious diseases were not systematically evaluated. Historically, use of the BAPGM blood culture platform has documented bacteremic coinfections with B. henselae and other bacteria in dogs [25] and humans [26]. Thus, there appears to be a complex pathobiome of blood that potentially changes with age, breed (genetics), nutrition, toxin exposure and interactions with a spectrum of bacteria, viruses and protozoa in the environment. Subcultures were not performed on samples from horses in this study to assess co-infections. Based upon evolving comparative clinical, microbiological and pathological data, tick borne pathogens, including Bartonella species, may constitute an important component of the pathobiome of blood in horses and other animals [27].
Within the context of differential diagnoses, the parameters described within this group of bacteremic horses could be incorporated by equine practitioners into a wide range of routine clinical evaluations such as lameness exams and pre-purchase exams. As part of a complete physical examination the examiner might review any and all presenting complaints and if necessary engage the horse owner in a series of specific questions for the express purpose of collecting complete historical data including history of exposure to biting insect or arthropod vectors and travel history. For horses presenting with history of chronic and/or sporadic exercise intolerance, fluctuating degrees of unsoundness, deficits in neurotransmission and/or chronic circulation deficits and/or relapsing symptoms such as those reported herein, testing for bartonellosis should be considered. The extent to which Bartonella bacteremia might contribute to behavioral abnormalities is beyond the scope of this report, however histories of neurotic, argumentative, unruly and/ or aggressive behavior patterns as seen in other species with chronic bartonellosis [28] lend support to a higher index of suspicion when considering testing options.
We conclude that the extent to which bartonellosis causes chronic disease manifestations in horses deserves additional research consideration. In-depth, sequential "lifetime" studies that span years in duration may be required to establish whether some or all of the symptoms described in the horses in this case series are causally associated with bartonellosis. It is possible that persistent intravascular infection with Bartonella spp. contributes to complex disease expression in horses, as seemingly occurs in humans and other animal species.
Competing Interest
In conjunction with Dr. Sushama Sontakke and North Carolina State University, Dr. Breitschwerdt holds U.S. Patent No. 7,115,385; Media and Methods for cultivation of microorganisms, which was issued October 3, 2006. He is the chief scientific officer for Galaxy Diagnostics, Inc. a company that provides diagnostic testing for the detection of Bartonella species infection in animals and human patients. Dr. Natalie Cherry is the Laboratory Supervisor and Research Specialist for Galaxy Diagnostics Inc. The remaining authors have no potential conflicts.
Source of Funding
This research received no specific grant or funding from any agency in the public, commercial, or not-for-profit sectors. This research was supported in part by Galaxy Diagnostics, Inc. and donations to the North Carolina State Veterinary Medical Foundation Vector Borne Diseases Research Fund.
References
- Breitschwerdt EB (2014) Bartonellosis: One health perspectives for an emerging infectious disease. ILAR J 55(1): 46-58.
- Chomel BB, Boulouis HJ, Breitschwerdt EB, Kasten RW, Vayssier- Taussat M, et al. (2009) Ecological fitness and strategies of adaptation of Bartonella species to their hosts and vectors. Vet Res 40(2): 29.
- Pulliainen AT, Dehio C (2012) Persistence of Bartonella spp. stealth pathogens: from subclinical infections to vasoproliferative tumor formation. FEMS Microbiol Rev 36(3): 563-599.
- Billeter SA, Levy MG, Chomel BB, Breitschwerdt EB (2008) Vector transmission of Bartonella species with emphasis on the potential for tick transmission. Med Vet Entomol 22(1): 1-15.?
- Bradley JM, Mascarelli PE, Trull CL, Maggi RG, Breitschwerdt EB (2014) Bartonella henselae infections in an owner and two Papillion Dogs exposed to Tropical Rat Mites (Ornithonyssus bacoti). Vector Borne Zoonotic Dis 14(10): 703-709.
- Mascarelli PE, Maggi RG, Hopkins S, Mozayeni BR, Trull CL, et al. (2013) Bartonella henselae infection in a family experiencing neurological and neurocognitive abnormalities after woodlouse hunter spider bites. Parasit Vectors 6: 98.
- Palmero J, Pusterla N, Kasten RW, Mapes S, Cherry NA, et al. (2012) Experimental Infection of horses with Bartonella henselae and Bartonella bovis. J Vet Intern Med 26(2): 377-383.
- Jones SL, Maggi RG, Shuler J, Alward A, Breitschwerdt EB (2008) Detection of Bartonella henselae in the blood of 2 adult horses. J Vet Intern Med 22(2): 495-498.
- Johnson R, Ramos-Vara J, Vemulapalli R (2009) Identification of Bartonella henselae in an aborted equine fetus. Vet Pathol 46(2): 277281.
- Cherry NA, Liebisch G, Liebisch A, Breitschwerdt EB, Jones SL, et al. (2011) Identification of Bartonella henselae in a horse from Germany Vet Microbiol 150(3-4): 414-415.
- Setlakwe EL, Sweeney R, Engiles JB, Johnson AL (2014) Identification of Bartonella henselae in the liver of a thoroughbred foal with severe suppurative cholangiohepatitis. J Vet Intern Med 28(4): 1341-1345.
- Duncan AW, Maggi RG, Breitschwerdt EB (2007) A combined approach for the enhanced detection and isolation of Bartonella species in dog blood samples: pre-enrichment liquid culture followed by PCR and subculture onto agar plates. J Microbiol Methods 69|(2): 273-281.
- Cherry NA, Jones SL, Maggi RG, Davis JL, Breitschwerdt EB (2012) Bartonella spp. infection in healthy and sick horses and foals from the southeastern United States. J Vet Intern Med 26(6): 1408-1412.
- Cherry NA, Maggi RG, Rossmeisl JH, Hegarty BC, Breitschwerdt EB (2011) Ecological diversity of Bartonella species infection among dogs and their owner in Virginia. Vector Borne Zoonotic Dis 11(11): 14251432.
- Maggi RG, Mascarelli PE, Pultorak EL, Hegarty BC, Bradley JM, et al.(2011) Bartonella spp bacteremia in high-risk immune competent patients. Diagn Microbiol Infect Dis 71(4): 430-437.
- Maggi RG, Mozayeni BR, Pultorak, EL, Hegarty BC, Bradley JM et al.(2012) Bartonella spp bacteremia and rheumatic symptoms in patients from Lyme Disease-endemic region. Emerg Infect Dis 18(5): 783-791.
- Maggi RG, Balakrishnan N, Bradley JM, Breitschwerdt EB (2015) Infection with Bartonella henselae in a Danish family. J Clin. Microbiol 53(5): 1556-1561.This work is licensed under Creative Commons Attribution 4.0 Licens DOI: 10.19080/AIBM.2017.04.555646
- Merrell DS, Falkow S (2004) Frontal and stealth attack strategies in microbial pathogenesis. Nature 430: 250-256.
- Schülein R, Seubert A, Gille C, Lanz C, Hansmann Y, et al. (2001) Invasion and persistent intracellular colonization of erythrocytes. A unique parasitic strategy of the emerging pathogen Bartonella. J Exp Med 193(9): 1077-1086.
- Vermi W, Facchetti F, Riboldi E, Heine H, Scutera S, et al. (2006) Role of dendritic cell-derived CXCL13 in the pathogenesis of Bartonella henselae B-rich granuloma. Blood 107(2): 454-462.
- Mändle T, Einsele H, Schaller M, Neumann D, Vogel W, et al. (2005) Infection of human CD34+ progenitor cells with Bartonella henselae results in intraerythrocytic presence of B. henselae. Blood 106(4): 1215-1222.
- Varanat M, Maggi RG, Linder KE, Breitschwerdt EB (2013) Infection of human brain vascular pericytes (HCVPs) by Bartonella henselae. Med Microbiol Immunol 202(2): 143-151.
- Breitschwerdt EB, Linder KL, Day MJ, Maggi RG, Chomel BB (2013) Koch's postulates and the pathogenesis of comparative infectious disease causation associated with Bartonella species. J Comp Pathol 148(2-3): 115-125.
- Pultorak EL, Maggi RG, Mascarelli PE Breitschwerdt EB (2013) Serial testing from a 3-day collection period by use of the Bartonella Alpha proteobacteria growth medium platform may enhance the sensitivity of Bartonella species detection in bacteremic human patients. J Clin Microbiol 51(6): 1673-1677.
- Davenport AC, Mascarelli PE, Maggi RG, Breitschwerdt EB (2013) Phylogenetic diversity of bacteria isolated from sick dogs using the BAPGM enrichment culture platform. J Vet Intern Med 27(4): 858-861.
- Cadenas MB, Maggi RG, Diniz PP, Breitschwerdt KT, Sontakke S, et al. (2007) Identification of bacteria from clinical samples using Bartonella alpha-Proteobacteria growth medium. J Microbiol Methods 71(2): 147-155.
- Vayssier-Taussat M, Kazimirova M, Hubalek Z, Hornok S, Farkas R, et al. (2015) Emerging horizons for tick-borne pathogens: from the 'one pathogen-one disease' vision to the pathobiome paradigm. Future Microbiol 10(12): 2033-2043.
- Breitschwerdt EB, Sontakke S, Hopkins S (2012) Neurological manifestations of bartonellosis in immuno competent patients: a composite of reports from 2005-2012. J Neuroparasit 3: 1-15.






























