Impediments in the Tissue Culture of High Altitude Medicinal Plants
Harsh Kumar Chauhan1,2, Anil Kumar Bisht1 and Indra Dutt Bhatt2*
1GB pant National Institute of Himalayan Environment and Sustainable Development, India
2GB Pant National Institute of Himalayan Environment & Sustainable Development, India
Submission: June 06, 2017; Published: July 17, 2017
*Corresponding author: Indra Dutt Bhatt, G.B. pant National Institute of Himalayan Environment and Sustainable Development, Kosi-Katarmal, Almora, India, Tel: +91 5962241041; Fax: +915962241014; Email: idbhatt@yahoo.com; idbhatt@gbpihed.nic.in
How to cite this article: Harsh K C, Anil K B, Indra D B.Impediments in the Tissue Culture of High Altitude Medicinal Plants. Adv Biotech & Micro. 2017; 4(2): 555631. DOI: 10.19080/AIBM.2017.04.555631
Abstract
High altitude medicinal plants (HAMPs) are facing tremendous pressure of over-exploitation. Most of them have narrow distribution range, unique for their medicinal properties and sensitive to environmental perturbation. In addition, poor seed germination, low seed viability, lesser survival percentage and slow growth further puts pressure on the overall growth of HAMPs. In this context, tissue culture offers a sustainable way for conservation and sustainable utilization. The production and extraction of the active constituents through tissue culture approaches are increasingly growing. Large numbers of protocols are available for propagating and utilizing HAMPs through tissue culture, still the commercial exploitation is meager. The present article highlights various impediments that a researcher might face during the tissue culture of HAMPs. The sustainable utilization of these plants through tissue culture needs collaboration with the various experts from the field of life, engineering and management sciences so that suitable conditions can be optimized for their commercialization.
Keywords: Active constituents; Conservation; High altitude medicinal plants (HAMPs); In vitro propagation; Optimization
Abbreviations: WHO: World Health Organization; HAMPs: High Altitude Medicinal Plants
Introduction
The world health organization (WHO) estimated that more than 80% population of the developing countries depends on the medicinal plants for primary health care [1]. About two third of the medicinal plants in use are collected from wild [2]. As such, the demand of the medicinal plant based raw drugs is increasing at the rate of 15 to 25% annually and may reach more than US$ 5 trillion in 2050 [3]. The HAMPs are being over-exploited and threatening their existence particularly due to the increasing herbal medicine demand, deforestation, overgrazing, rapid agricultural expansion, habitat destruction, destructive harvesting and by the climate change. It has been reported that nearly 4000 to 10000 medicinal plants might now be endangered [2]. The major target of plant tissue culture includes sustainability, food security, and the production of novel biomaterial [4]. Advancement in plant tissue culture provides new means for conservation and rapidly propagating the valuable, rare and endangered medicinal plants [5]. In addition, the plant tissue culture offers integrated approaches for the mass production of bioactive constituents via callus or suspension cultures. Plant tissue culture thus promises efficient and reliable production system for phyto chemicals as well plants as a whole [6].
High altitude medicinal plants (HAMPs) are the choice for pharmaceutical industries as they hold greater possibilities of having novel molecules and even larger quantities of active compounds due to their exposure to extreme climatic and geophysical conditions [7,8]. The indiscriminate exploitation coupled with unregulated harvesting and lack of attention to strategic cultivation practices have led to the endangered or threatened status of several HAMPs [9]. Considering to this, several researchers are working for the development of in vitro propagation and production of active constituents from these species. Several HAMPs are successfully propagated and used as the source of active constituents by tissue culture methods [5,10-20]. Unfortunately, most of the research remains up to the laboratory and commercial propagation of these plants is meager. The researchers face several impediments in development of the successful and replicable tissue culture protocols for such species. In the present article we tried to summarize the impediments that a researcher might face during the tissue culture of HAMPs (Figure 1).
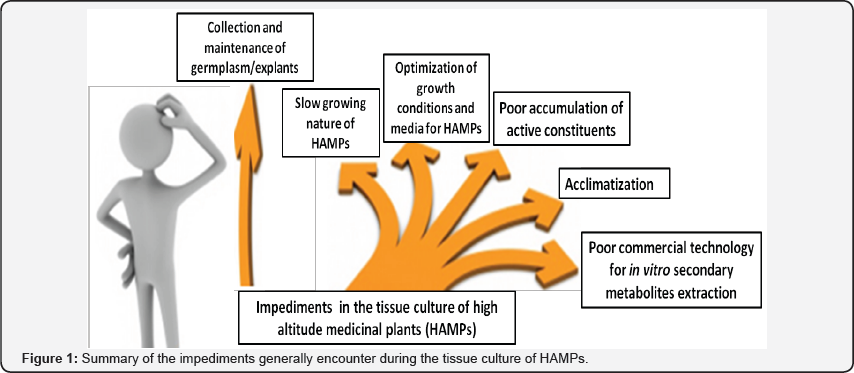
Collection and maintenance of germ plasm/explants
High altitude regions are characterized by extreme weather conditions such as cold and arid climate, scanty rainfall, heavy snow fall, high wind velocity, avalanches, landslides etc. The angle of slope is generally high. Environmental conditions in alpine areas change sharply within pockets [21]. The process of initiation of plant material into culture usually occurs in the laboratory after collection from the field [22]. The collection of the plant material from the high altitude region is not an easy task and it needs specialized collection techniques. Locations of the availability are generally remote. The transport facilities are very poor, and the plant collectors might need to walk several kilometers to reach the exact locations. Further, there is either lack of geo-coordinated data regarding locations (as high altitude regions are less explored) or sometimes when the reports available, one does not find them at all on reported locations due to exploitation and destruction of habitats by anthropogenic and natural factors (flash flood and landslides). Thus, eventually the plant collector has to look for alternate locations which might accommodate limited number of plants. Under such conditions the replicates of experiments have to be reduced accordingly. The researcher has to carry the plants to the laboratory/arboretum from a far flung region. Transporting plant/explants to laboratory/arboretum is yet another tedious job. The maintenance of these plants outside their natural habitat (at low altitude) is not easy and their survival is usually very low. Thus, it is difficult to maintain the continuous supply of explants for the running experiments. It is also evidenced that the limited amount of plant material available from rare species poses major challenges in the application of in vitro methods to endangered species [22].
Slow growing nature of HAMPs
Alpine plants are generally slow growing. Seeds of alpine plants may remain dormant for long period at low temperature since they require temperature well above the freezing for germination [23]. Threatened alpine plant species are often slow to reach maturity (7-8 years) and to produce the tradable component [24]. It is also known that the seedling establishment in alpine plants is rare and very slow; it may take several years before safe establishment. HAMPs often show similar growth performance in the in-vitro conditions. HAMPs cells generally have low biomass yields in callus and suspension cultures as compared to the other plants. The slow growth of HAMPs in in- vitro conditions incorporates the risk of contamination by fast growing bacteria/fungi. Contamination is of great concern for the large scale propagation and cultivation of HAMPs.
Growth conditions and media for HAMPs
The HAMPs prefer highly specific habitat [25]. Most of the alpine plants grow in association with other plants as these associations normally result in increased biomass, growth and reproduction of sub-alpine and alpine species [26]. The cultivation of HAMPs generally requires a thorough knowledge on the biology, otherwise ex situ replication of these conditions is tedious. As such, there is lack of published methods for in vitro propagation of medicinal plants and this makes the choice and development of the initiation culture medium arbitrary [27]. Even if the cultures are initiated, the growth and multiplication of in vitro cultures can remain problematic requiring specific novel treatments [22]. The growth conditions of large number of medicinal plants are yet to be optimized [28]. There are number of chemical and physical factors like media components, phyto hormones, pH, temperature, aeration, agitation, light, etc. that are to be optimized for the better growth of the plants in in vitro conditions. This needs long time and huge efforts which may hinder the interest of the researchers and one of the major impediments in the large scale quality plant production.
Accumulation of active constituents
The accumulation of active constituents varied greatly within and among the species, habitat and altitude [29]. These observations are equally true in in vitro conditions where variations in the physical and chemical environment may lead to the variation in accumulation of active constituents. HAMPs generally accumulate more bioactive constituents in natural conditions in response to extreme climatic and geophysical conditions [8]. Although, reports are available for the accumulation of more bioactive constituents in in vitro cultures and non native habitat as compared to natural habitat [18,30] but, this require a perfect optimization of plant growth regulators and media type which is being used. In most cases, plant cells often produce reduced quantities of active constituents in vitro and their qualitative features may also be changed. Better results can be obtained only after manipulating the environmental parameters and medium, selecting high yielding cell clones, precursors feeding and elicitation [31]. Obtaining these results is very laborious, costly and time consuming affair, however, at the same time if it get optimized and can stable the production will help in reducing pressure on the natural sites.
Acclimatization
The use of in vitro propagated HAMPs for reintroduction is one of the critical step; however, it relies on the successful methods for acclimatization. Acclimatization of the tissue culture raised plants has never been an easy task. A substantial number of tissue culture propagated plants do not survive from in vitro conditions to green house or field environment [32]. The condition becomes more adverse for HAMPs as the natural fields are generally far apart and the greenhouse or nonnative field may have changed humidity, light level and other environmental conditions as compared to native environment of the species. The transplantation continues the major bottleneck in the conservation of HAMPs as most of them require specific protocols for the successful acclimatization.
Commercial consideration
Despite the fact that, lots of protocols have been developed for the in vitro propagation of HAMPs and the active constituent extraction from in vitro cultures has also been done successfully very few workers focused on the cost-benefit analysis of these protocol [4]. Due to this the commercial exploitation of these protocols are still lacking [33]. It is often observed that most of the pharmaceutical or medicinal plants based industries are still collecting and extracting the active constituents from the wild plants as there is no commercial technology for in vitro secondary metabolites extraction.
Conclusion
In spite of several impediments in the propagation of the HAMPs, the importance of tissue culture in the conservation and production of secondary metabolites from in vitro raised culture are gaining popularity. A plenty of literature is available wherein researchers have significantly overcome with field and laboratory problems and implemented new ideas and innovations [5-10,20]. In addition, methods of seed banking, development of living collections, cryopreservation and molecular genetics may provide additional backup for propagation and conservation of HAMPs. Greater progress can be expected if the plant culturists are given more opportunities to closely collaborate with the chemists, biochemist, biotechnologist, molecular biologist, geneticists and engineers. The commercialization of tissue culture techniques for HAMPs needs more time and patient. Researchers working in this area need motivation and facilitation by special recognition of their contribution.
Conflict of Interest
Authors declare no conflict of interest.
References
- Canter PH, Thomas H, Ernst E (2005) Bringing medicinal plants into cultivation: opportunities and challenges for biotechnology. Trends Biotechnol 23(4): 180-185.
- Edwards R (2004) No remedy in sight for herbal ransack. New Sci 181: 10-11.
- Kala CP, Dhyani PP, Sajwan BS (2006) Developing the medicinal plants sector in northern India: challenges and opportunities. J Ethnobiol Ethnomed 2(1): 32.
- Moshelion M, Altman A (2015) Current challenges and future perspectives of plant and agricultural biotechnology. Trends Biotechnol 33(6): 337-342.
- Nalawade SM, Tsay HS (2004) In vitro propagation of some important Chinese medicinal plants and their sustainable usage. In Vitro Cell Dev- Pl 40(2): 143-154.
- Davies KM, Deroles SC (2014) Prospects for the use of plant cell cultures in food biotechnology. Curr Opin Biotech 26: 133-140.
- Hazlett DL, Sawyer NW (1998) Distribution of Alkaloid-Rich Plant Species in Shortgrass Steppe Vegetation. Conserv Biol 12(6): 1260-1268.
- Kaul MK (2010) High altitude botanicals in integrative medicine-Case studies from Northwest Himalaya. Indian J Tradit Know 9(1): 18-25.
- Verma PC, UR Rahman L, Negi AS, Jain DC, Khanuja SPS, et al. (2007) Agrobacterium rhizogenes-mediated transformation of Picrorhiza kurroa Royle ex Benth: establishment and selection of superior hairy root clone. Plant Biotechnol Rep 1(3): 169-174.
- Bhatt ID, Dhar U (2000) Micropropagation of Indian wild strawberry (Fragaria indica Andr.) Plant Cell Tiss Org 60: 83-88.
- Bhatt ID, Dhar U (2000) Combined effect of cytokinins on multiple shoot production from cotyledonary node explants of Bauhinia vahlii. Plant Cell Tiss Org 62: 79-83.
- Bhatt ID, Dhar U (2004) Factors controlling micropropagation of Myrica esculenta: a high value wild edible of kumaon Himalaya. Afr J Biotechnol 3(10): 534-540.
- Dhar U, Upreti J, Bhatt ID (2000) Micropropagation of Pittosporum napaulensis (DC.) Rehder & Wilson - a rare and endemic Himalayan medicinal tree. Plant Cell Tiss Org 63: 231-235.
- Joshi M, Dhar U (2003) In vitro propagation of Saussurea obvallata (DC.) Edgew.-an endangered ethnoreligious medicinal herb of Himalaya. Plant Cell Rep 21(10): 933-939.
- Manjkhola S, Dhar U, Joshi M (2005) Organogenesis, embryogenesis, and synthetic seed production in Arnebia euchroma-a critically endangered medicinal plant of the Himalaya. In Vitro Cell Dev-Pl 41(3): 244-248.
- Joshi SK, Dhar U (2009) In vitro propagation from axenic explants of Lilium oxypetalum (D. Don) Baker, an endemic bulbous plant of high altitude Himalaya. Acta Physiol Plant 31(4): 833.
- Pandey M, Dhar U, Bhatt ID, Thengane SR (2010) In vitro propagation of Selinum wallichianum (DC), A high value medicinal plant of the Himalaya. Asian Journal of Experimental Science 24(1): 179-183.
- Giri L, Dhyani P, Rawat S, Bhatt ID, Nandi SK, et al. (2012) In vitro production of phenolic compounds and antioxidant activity in callus suspension cultures of Habenaria edge worthii: a rare Himalayan medicinal orchid. Ind Crop Prod 39: 1-6.
- Giri L, Jugran A, Rawat S, Dhyani P, Andola H, et al. (2012) In vitro propagation, genetic and phytochemical assessment of Habenaria edgeworthii: an important Astavarga plant. Acta Physiol Plant 34: 869-875.
- Purohit S, Jugran AK, Bhatt ID, Palni LMS, Bhatt A, et al. (2016) In vitro approaches for conservation and reducing juvenility of Zanthoxylum armatum DC: an endangered medicinal plant of Himalayan region. Trees 31(3): 1101-1108.
- Shimono Y, Kudo G (2005) Comparisons of germination traits of alpine plants between fellfield and snowbed habitats. Ecol Res 20(2): 189-197.
- Sarasan V, Cripps R, Ramsay MM, Atherton C, McMichen M, et al. (2006) Conservation In vitro of threatened plants-progress in the past decade. In Vitro Cell Dev-Pl 42(3): 206-214.
- Billings WD, Mooney HA (1968) The ecology of arctic and alpine plants. Biol Rev 43(4): 481-529.
- Nautiyal MC (1995) Agro-techniques of some high altitude medicinal herbs. In: Sudriyal RC & Sharma E (Eds), Cultivation of Medicinal Plants and Orchids in Sikkim Himalaya. PGR Newsletter, Dehradun, India, pp. 53-64.
- Airi S, Rawal RS, Dhar U, Purohit AN (2000) Assessment of availability and habitat prefernce of Jatamansi-a critically endangered medicinal plant of west Himalaya. Curr Sci 79(10): 1467-1470.
- Callaway RM, Brooker RW, Choler P, Kikvidze Z, Lortie CJ, et al. (2002) Positive interactions among alpine plants increase with stress. Nature 417(6891): 844-848.
- Krogstrup P, Find JI, Gurskov DJ, Kristensen MMH (2005) Micropropagation of socotran fig, Dorstenia gigas Schweinf. Ex Balf. F.-A Threatened Species, endemic to the island of Socotra, Yemen. In Vitro Cell Dev-Pl 41(1): 81-86.
- Briskin DP (2000) Medicinal plants and phytomedicines. Linking plant biochemistry and physiology to human health. Plant Physiol 124(2): 507-514.
- Jugran AK, Bahukhandi A, Dhyani P, Bhatt ID, Rawal RS, et al. (2016) Impact of altitudes and habitats on valerenic acid, total phenolics, flavonoids, tannins, and antioxidant activity of Valeriana jatamansi. Appl Biochem Biotech 179(6): 911-926.
- Bhatt ID, Dauthal P, Rawat S, Gaira KS, Jugran A, et al. (2012) Characterization of essential oil composition, phenolic content, and antioxidant properties in wild and planted individuals of Valeriana jatamansi Jones. Sci Hortic-Amsterdam 136: 61-68.
- Mulabagal V, Tsay HS (2004) Plant cell cultures-an alternative and efficient source for the production of biologically important secondary metabolites. Int J Appl Sci Eng 2(1): 29-48.
- Hazarika BN (2003) Acclimatization of tissue-cultured plants. Curr Sci 85(12): 1704-1712.
- Chaturvedi HC, Jain M, Kidwai NR (2007) Cloning of medicinal plants through tissue culture-a review. Indian J Exp Biol 45: 937-948.






























