Modeling Schemes for Control of Bed-Temperature Gradients in Solid-State Fermentation
Ramón Larios-Cruz1, Gerardo Lara-Cisneros2, Julio C Moñtanez1and Cristóbal N Aguilar1*
1Universidad Autónoma de Coahuila, México
2División de Matemáticas Aplicadas, Instituto Potosino de Investigación Científicay Tecnológica, México
Submission: February 27, 2017; Published: April 21, 2017
*Corresponding author: Cristóbal N Aguilar, Group of Bioprocesses, School of Chemistry, Universidad Autónoma de Coahuila, Saltillo, Coahuila, 25280, México, Tel: +52-844-4161238; Fax: +52-844-4159534; Email : cristobal.aguilar@uadec.edu.mx
How to cite this article: Ramón L, Gerardo L, Julio C M, Cristóbal N A. Modeling Schemes for Control of Bed-Temperature Gradients in Solid-State Fermentation. Adv Biotech & Micro. 2017; 3(2): 555607. DOI: 10.19080/AIBM.2017.03.555607
Abstract
Mathematical modeling is a useful tool to represent true processes on solid-state fermentation, more employed on scale-up and for better automatic control strategy designs. In the case of temperature profiles during the growth of microorganisms, models reproduce the events well, given valuable information to prevent or know the maximum temperature along the process. The election of differential mathematical models over others are better to use because of the simplicity and further modifications on the models, according to different bioreactor design, they could improve solid fermentation processes. In the present work, a review on the mathematical models for heat transfer in packed bed bioreactor is showed. In order to evaluate the best models for control design effects, the main characteristics for the models reported in the literature are discussed. Also, the analysis of the models can be useful to define an optimal bed-temperature profile that improves the solid-state fermentation.
Keywords: Solid-state fermentation; Mathematical modeling; Bed temperature; Packed bed bioreactor; Control process
Introduction
The solid-state fermentation (SSF) is defined as the growth of the microorganism on a solid porous matrix which can be biodegradable or not, with spaces between particles where we can found a gas phase and a minimum of water, enough sources of carbohydrates and nitrogen and mineral salts that allow the appropriate develop of the organisms [1,2]. SSF has been used traditionally on Asian cultures as Japanese for the production of "koji" byAspergilusoryzae, [2], Chinese for "red rice" by Monascus [3], and European as French for "blue cheese" by PeniciHium roquefortii [2], among others. Also SSF has been employed in bioprocess, nutritional enrichment of crops, biopulping and production of secondary metabolites [4].
Comparing SSF with submerged fermentation (SmF) in some processes, the performance is larger in SSF and it is particularly attractive for industrial applications [5]. But in the real, the experiments are developed in laboratory scale based on different types of bioreactors design, most of them on petri dishes or Erlenmeyer flasks [6,7]. When packed-bed bioreactors are employed for SSF the performance is better, because they maintain stable the substrate during the fermentation, where microorganisms sensitive to agitation forces do not suffer damages that could reduce their growth [8,9]. However, when scale up strategies is followed by researches, the disadvantages of the processes appear. One of the most important aspects to control on the solid fermentation is bed temperature; it can reach levels so high that the microorganisms decrease their growth or died as fermentation time proceed [10].
In this way, the application of mathematical modeling is an important tool to describe these processes as we could expect in laboratory experiments [11] and its use is effective for large scale bioreactors to evaluate the design and the best operation approach during the experiments [12,13]. Then, the main variables in the SSF can be controlled via feedback schemes in order to increase the bioreactor productivity [10,14].
Operating a packed-bed bioreactor for SSF with feedback control schemes can improve the yield of the fermentation, but it is needed a correct model to describe the procedure and support the phenomenon; also, difficulties to reproduce the events due to the bed heterogeneity, gas occlusion in the bed and electromagnetic interference, could make difficult the design of control strategies [14]. For modeling heat transference in packed-bed bioreactors, axial temperature gradients are considered [12,15-17] due to the air flow direction, from the button to the top or backwards.
In these sense the study of models for heat transfer in packed- bed bioreactor became useful to different strategies to control the temperature during the fermentation. Where ordinary differential equations could be more appropriate to the analysis and design of control strategies; because of that, they result easy to solve by numerical simulation. However, the possible solution of reduce high temperatures during the microbial metabolism on SSF continue far to be controllable.
The aim of this paper is to show mathematical models for heat transfer in packed bed bioreactors for SSF and their potential application to the analysis and design of control strategies for regulating of axial bed temperature to lead at the optimal temperature during the fermentation. It is important to note that these models could be used for automatic control strategies in larger scales bioreactor designs to improve the productivity of the fermentation.
Technological advantages of SSF
The use of microorganisms on biotechnological processes is for the transformation of solid or liquid substrates into several products. Depending of the characteristics of the substrates it could be classified as solid-state or submerged fermentation [18]. Submerged fermentation (SmF) is defined as the fermentation in presence of excess water '[19] and the preference for industrial processes is due to the advantages on control the processes along the fermentation; besides, for larger production, the yields are almost the same compare with minor scales.
However, SSF has gain importance with years because of high productivity fields and numeral applications including utilization and treatment of agricultural-industrial wastes. Different studies on secondary metabolites or enzyme production are commonly compared between both fermentation processes, in majority of cases SSF is over SmF [20-22].
Secondary metabolites
Uses of secondary metabolites from fermentation processes in food, pest control and antibiotic, among others, have become reaching a particular interest to develop more efficient procedures to improve quality and quantity of the products. Table 1 shows some examples of secondary metabolites produced in SSF; microorganisms, substrate and bioreactor design are exposed too. It is important to note one of the most attractive characteristic of SSF which is the use of agriculture residues as carbon source and on the other hand, the lack of engineering aspects for massive production when use laboratory scale bioreactors.
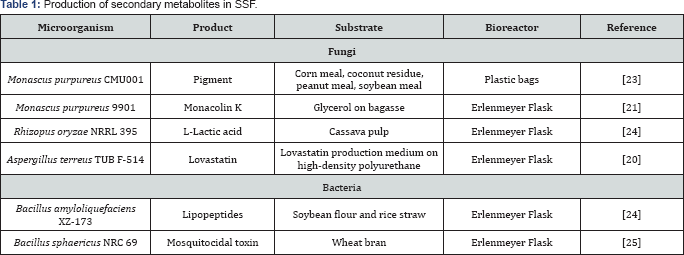
Common uses of that metabolites includes: monacolin K (lovastatin) for lowering blood cholesterol [20,21], lactic acid for use as organic acid in food, chemical and pharmaceutical industries or compostable plastic [23], pigment from Monascus applied on food industry [24] and biomass as bio-control of mosquitoes [25], among others.
Enzyme production in SSF
In case of enzymes, they are more studied for many applications including value added to industrial wastes [26] which could be used as feed for ruminant [27]. Food industries take enzymes for clarification purposes in fruit juices, like pectinases [28] andlipases for hydrolysis of fats and oils [29], other industries use cellulases for second generation ethanol production [30] and chitinases for application in fungal protoplast formation [22].
Table 2 shows the productions of enzymes under SSF by bio-conversion of wastes and their respectively bioreactor design employed. As it was expected, laboratory scale on SSF is developed. The lack of knowledge on larger scale bioreactors designs limits the potential industrial production; even if high productivity yields were obtained, in case of comparing results obtained with others alternatives.
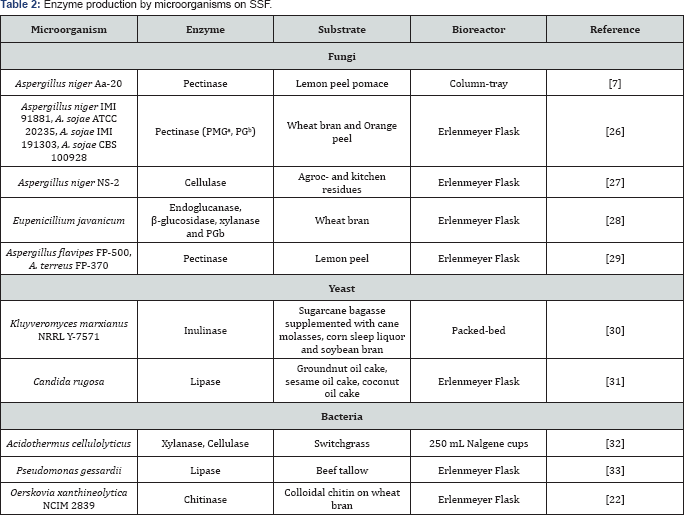
aPolymethylgalacturonase.
bPolygalacturonase.
cCorn cobs, carrot peelings, composite, grass, leaves, orange peelings, pineapple peelings, potato peelings, rice husk, sugarcane baggage, saw dust, wheat bran, wheat straw.
As it was showed in both, secondary metabolites and enzymes production, there was not common to use different bioreactor designs. But when SSF are developed for purposes of scale-up, different bioreactors designs are found in the literature including rotational drum, horizontal drum, zymotis, fluidized bed, evaporative cooling, tray fermenters and packed bed bioreactors, among others [6,7].
SSF bioreactors
In SSF bioreactors are used according to the quantity of substrate/biomass and the type of system employed on laboratory-scale, pilot-scale or industrial-scale. On laboratory- scale the dry solid medium used begin from a few grams reaching a few kilograms, and the pilot and industrial-scale from kilograms reaching several tons of dry medium [6]; wherever the scale, bioreactors could be on aerated or mixed system to improve quality of the process [31]. The first step on the studies in SSF over an specific metabolism or growth of the microorganism is on laboratory scale, using commonly Erlenmeyer flask because of the facilities to control the fermentation [6] or Petri dishes for the selection of strain and substrate [7]. But these kinds of methods are not developed using a specific bioreactor design; there is not air force or substrate mixing as a characteristic on the operation mode of bioreactors [6,9]. However, some important parameters as temperature of incubation, pH of the medium, moisture content and particle size of the substrate can be analyzed on this pathway (Erlenmeyer flask) [32,33]. Still on laboratory-scale, some studies on bioreactors are made in columns as packed bed bioreactors [8,34], rotating [8,9] or column-tray [9] where the operation conditions could be taking place to improve the productivity or evaluation between reactors and purposes of scale-up.
On pilot-scale, the studies are focused on going to industrial- scale. But it is hard to reproduce the events when volume of bioreactors increases. It is there where real troubles showing up because of the cooling system or operation system of the bioreactor do not offer enough capacity of controlling temperature. In this case, it is an important parameter to take into account, which increases during microorganism growth due to the metabolic heat [35,36]. Other factors involved the bioreactor design affecting normal growth of some microorganisms like filamentous fungi [36]; or properties of the substrate as bed density and porous of the bed impacting on the air flow direction and its diffusion, principally [36].
With respect to the nature of the microorganism on the production of a specific metabolite, for example filamentous fungi, maintain the bed without movement is critical because of possible ruptures of the mycelium. The high productivity obtain is the basis of the bioreactor choose [8] and the preference to use aerobic microorganisms on SSF [2] is taking into account to find the best bioreactor design to use [6,7,36]. In that case, packed bed bioreactors are good enough to be employed on SSF.
Packed bed bioreactor
The advantage of choose packed bed bioreactors is that it does not cause damaging to the microorganisms during the fermentation as done by a rotating drum or a fluidized bed bioreactor [8,37] specially on fungus which are more sensitive to changes in the bed orientation and with disturbs of the mycelium causing a reduction of natural growth of the microorganisms. But high temperatures caused by metabolic heat are difficult to remove just with forced aerated because the unidirectional way of air through the bed [12]. Besides, engineering problems limited the reproducibility of the fermentations like different substrate size, humidity and water disposition [38]. Water is a parameter to take a special care just because its participation in the direct organism develop in SSF process, the transfer of materials during fermentation is one of the primary works and its quantity determined the kind of microorganism appropriate for a particular experiment [39].
Biomass Growth
Biomass is probably the most important parameter to evaluate in SSF; it is the basis for kinetic studies. It determines the success or the failure of the fermentation processes, its estimation with growth models is a good tool for automatic control. Secondary metabolites are correlated with microorganism growth like production of CO2 and cellular components as ergosterol or gulucosamine, but a direct measure method is not employed [2].
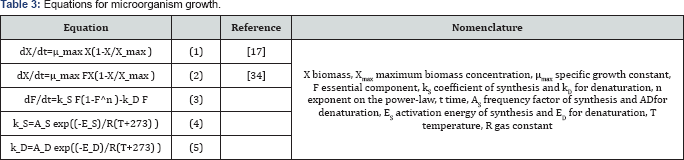
Table 3 shows equations (1-5) for biomass estimation. As suggested by Saucedo-Castaneda et al. [17] in the kinetic model for solid culture, the biomass in the bioreactor grows according to equation (1). Where there is an independent specific growth over substrate concentration. And changes on CO2 and substrate are not affecting the maximum biomass concentration.
Others authors like Dalsenter et al. [40] [equations [2-5]] and Fanaei & Vaziri [13] consider an essential factor (F) on the model to describe the biomass growth. This factor is depending of the temperature; corresponding to the coefficients of synthesis and denaturation (equations (4-5)). Where a healthy cell represents a value of 1 of this component [F] [40]. Those expressions are essential for heat transfer models.
Mathematical modeling
Various phenomena on SSF bioreactor could be described in a set of equations [models]. In order to predict temperatures in SSF, equations that describe heat transfer are written. With models, the simulation of operation conditions and productivity can be emulated for the purpose of depict the events for optimization or prediction of undesirable measures in case of automatic control applications [1]. Table 4 shows energy balance equations used in SSF bioreactors. From conservative relation for mass and energy, taking into account heat and mass transfer phenomena, as well as the growth biomass kinetic model, Sahir et al. [16] proposed an N-tanks model (eq. (7)). In this model the energy balance for a packed-beb bioreactor for SSF considers nth tank. It receives air from the bottom of [n - 1)th tank and provide it to the [n + 1)th tank. Equations [8-10] estimate the humidity of air, depending on the vapor pressure, and total dry solids in the substrate bed.
The important assumptions of the model are the well-mixed tank, the same concentration of biomass and temperature within the same tank, no pressure drop of air flows, the total biomass and the heat transfer area are divided in N-tanks, the water temperature of the jacket does not vary over the time, negligible radial gradients, and the basis of the model, the solids and air within the packed bed bioreactor exist as a pseudo homogeneous single phase. The profile of temperature showed by the authors [16] demonstrates that as the height of bed increased, the maximum temperature of the fermentation increased as well. This model became useful to different bioreactor design and its use on control strategies for larger scales could be considered; this is for prediction of higher temperatures at different bed locations. That is why little modifications including other assumptions or cooling strategies as air flow direction could be employed on new bioreactor designs. Because of lack of changes on the bioreactor function, the model only reproduces the profile of temperature over the time across of the SSF and there is not any improve on the temperature control. But the height of the bed could be choosing accurately and the fermentation processes might be enhanced.
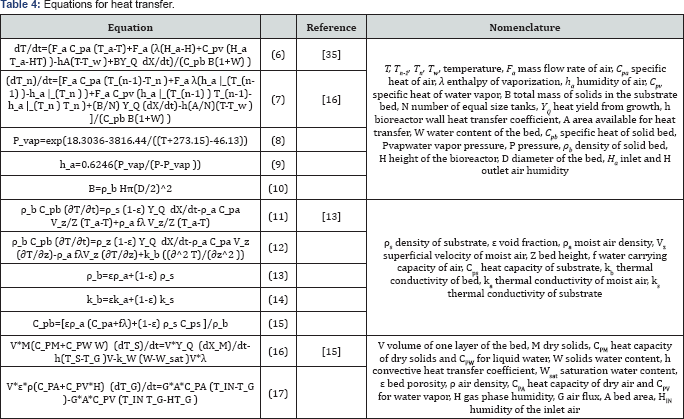
It is normal to find models which consider the whole bioreactor to depict temperature during the fermentation process and the profile only shows the performance on the extremes of the bioreactor. As done by Santos et al. [41] the description of the bioreactor and mathematical model on heat transfer is for the temperature reach at the top of the bioreactor. The energy balance for a well-mixed bioreactor is on eq. (6). The contribution of Sahir et al. [16] over this equation for heat transfer is the consideration of N-tanks visualized on the bed. This became useful for further studies on SSF, specially on larger scale strategies to temperature control. In the case of Dalsenter et al. [40], only changes of the sing [minus instead of a plus] in the term that describe the heat removal by the water vapor differ from eq. (6), of course the bioreactor design is for a tray bioreactor.
Taking others mathematical models, a comparison of two dynamic models for heat transfer on a packed bed bioreactor for SSF was use to evaluate the best model selection and applications on the height of the bed [13]. Where metabolic heat generation, convective and evaporative heat removal was considered into the lumped dynamic model [eq. [11]]. In contrast axial conduction term is also considered at the distributed model [eq. [12]]. For the bed properties as density, thermal conductivity and heat capacity, values were computed as weighted averages of the air and substrate [eqs. [13-15]].According to the authors, the distributed model predictions are better adjustable to the experimental data. Most important is that this model could be used to determine the temperature bed at different heights and fermentation time. The same experimental data were taken by Sahir et al. [16] to validate the predictions and it seems that the distributed model adjust in better way to them.
This kind of model [eq. [12]] is applicable to the developed of new strategies for temperature control in SSF on larger scales with options of automatic control designs. But the solution of the model is harder, the advantage is the precision that offer to the experimental data; important for control strategies and better designs of control algorithms. In another way, an equation based in two phase model of a well-mixed bioreactor with differential equation for individual layer is developed for a solid and gas phase. On this every layer is a complete bioreactor and different operation design was described for a multi-layer of a packed bed bioreactor for SSF (eqs. (16-17)).?
As it was expected by the authors, the model predicted the temperature at different heights of packed bed bioreactor during a SSF process. In this work, differing from others, the authors finished the paper with two different configurations of the bioreactors for multi-layer operation in case of experimental reproducibility of the processes. Talking to the model, it became easy to solve because the inclusion of differential equations. It is more practical for control strategies on larger scales of the fermentation events.
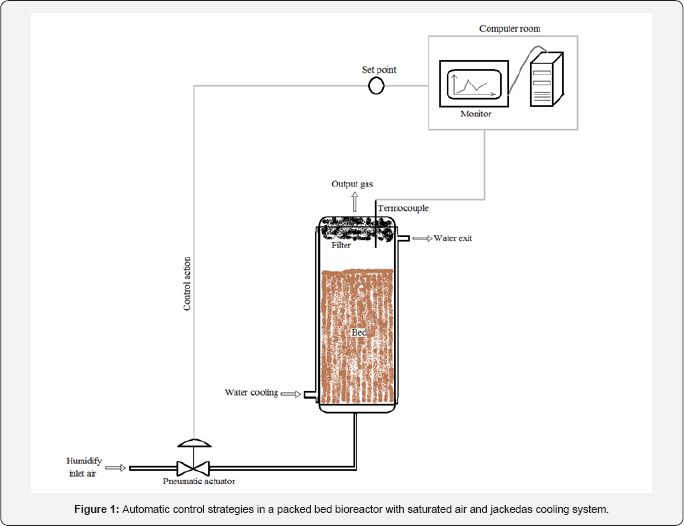
The purposes of these models [Table 4] are to describe the effect of temperature profile during SSF. As an example, Figure 1 shows a packed bed bioreactor employing automatic control to improve productivity. Thermocouples are used to temperature measure as fermentation process proceed and from equations for heat transfer, the prediction of high heat values could be anticipate increasing the air flux, gaining time to reduce high temperatures of the bed. This of course, respecting normal operation system corresponding to the design of bioreactor exposed. Which leads to problems describe above as rupture of mycelium by high flux, drying of the bed, high temperatures on the top and low mass transfer, among others. It is not express in Figure 1, but output gas could be taken to evaluation of humidity by the use of an appropriate sensor and biomass estimation by measure of the CO2 content; increasing considerably the productivity of the bioreactor when more instruments for control processes are employed.
With respect to the numerical solution, lumped models could be solved by the application of an appropriate method; e.g. for equations exposed, MATLAB® for Runge-Kutt-Fehlberg method [16] and fourth-order Runge-Kutta method [13] or FORTRAN subroutine DASSL [15]. In case of the distributed model, it must be discretized to obtain a non-linear ordinary differential equation; which is approximated by finite difference method (Fanaei and Vaziri, 2009). Then, the resulting equation is solved by the chosen method as a lumped model.
After the modeling simulation, with the use of partial differential equations data nearest to the experimental are expected; the reason could be achieve, in case of the evaluation of temperature in SSF, to the dependence of more than just one variable like time and bed height [13]. Even if the solution of the lumped models is easier that distributed models, they are better in automatic control strategies due to the advantage on creating algorithms.
Final Comments
The correct choose of bioreactor design for SSF is important to define the success of the microorganism growth into the fermenter. Mathematical modeling is a useful tool to reproduce de events to choose with more severity and advantage the best operation conditions of the bioreactor for the SSF. The lack of new designs of bioreactor for SSF makes difficult the solution of the principal trouble, maintaining optimal temperature during fermentation processes. That is way models only describe the events of actual bioreactor designs, but on a positive way it is useful to avoid costs on reproduce fail experiments. Another application is on control strategies for SSF on larger scales, and on this point easier models to compute and solve are more possible to use because they need less knowledge on mathematical modeling.
The lumped model proposed by Sahir et al. [16] presents best attributes in order to design control strategies for regulates bed-temperature gradients. Unlike control strategies could be studied and apply for SSF, maintain optimum temperature is a little harder. Works that adjust mathematical models on different control strategies to optimize the temperature during the fermentation process are expected to be developed in the near future. This could improve the temperature controlling in SSF with application of new bioreactor designs [42-49].
Acknowledgment
First author want to acknowledge to the Mexican Council of Science and Technology CONACyT for the financial support in the form of scholarship for postgraduate studies.
References
- Mitchell DA, Krieger N, Berovic M (2006) Solid-State Fermentation Bioreactors: Fundamentals of Design and Operation. [1 edn], Germany: Springer-Verlag, Germany, ISBN 978-3-540-31286-4.
- Raimbault M [1998] General and microbiological aspects of soid substrate fermentation. Electronic J Biotechnol 1[3]: 174-188.
- Carvalho JCd, Pandey A, Oishi BO, Brand D, Rodri'guez-Leon JA, et al. (2006) Relation between growth, respirometric analysis and biopigments production from Monascus by solid-state fermentation. Biochem Eng J 29: 262-269.
- Pandey A [2003] Solid-state fermentation. Biochem Eng J 13: 81-84.
- Subramaniyam R, Vimala R [2012] Solid state and submerged fermentation for the production of bioactive substances: A comparative study. International Journal of Science and Nature IJSN 3[3] 2012: 480486.
- Durand A [2003] Bioreactor designs for solid state fermentation. Biochem Eng J 13: 113-125.
- Ruiz-Leza HA, Rodri'guez-Jasso RM, Rodri'guez-Herrera R, Contreras- Esquivel JC, Aguilar CN [2007] Diseno de biorreactores para fermentacion en medio solido. Rev Mex Ing Quim 6(1): 33-40.
- Breukelen FRv, Haemers S, Wijffels RH, Rinzema A (2011) Bioreactor and substrate selection for solid-state cultivation of the malaria mosquito control agent Metarhizium anisopliae. Process Biochem 46: 751-757.
- Rodri'guez-Fernandez DE, Rodri'guez-Leon JA, Carvalho JCd, Karp SG, Sturm W, et al. (2012) Influence of airflow intensity on phytase production by solid-state fermentation. Bioresour Technol 118: 603606.
- Meien OFV, LFLL, Mitchell DA, Perez-Correa JR, Agosin E, et al. (2004) Control strategies for intermittently mixed, forcefully aerated solid- state fermentation bioreactors based on the analysis of a distributed parameter model. Chem Eng Sci 59: 4493-4504.
- Mitchell DA, Meien OFV, Krieger N [2003] Recent developments in modeling of solid-state fermentation: heat and mass transfer in bioreactors. Biochem Eng J 13: 137-147.
- Ashley VM, Mitchell DA, Howes T (1999) Evaluating strategies for overcoming overheating problems during solid-state fermentation in packed bed bioreactors. Biochem Eng J 3: 141-50.
- Fanaei MA, Vaziri BM (2009) Modeling of temperature gradients in packed-bed solid-state bioreactors. Chem Eng Process Process Intensif 48: 446-451.
- Fernandez-Fernandez M, Perez-Correa JR (2007) Realistic model of a solid substrate fermentation packed-bed pilot bioreactor. Process Biochem 42: 224-234.
- Mitchell DA, Cunha LEN, Machado AVL, Luz LFL, Krieger N (2010) A model-based investigation of the potential advantages of multi-layer packed beds in solid-state fermentation. Biochem Eng J 48: 195-203.
- Sahir AH, Kumar S, Kumar S (2007) Modelling of a packed bed solid- state fermentation bioreactor using the N-tanks in series apporach. Biochem Eng J 35: 20-28.
- Saucedo-Castaneda G, Gutierrez-Rojas M, Bacquet G, Raimbault M, Viniegra-Gonzalez G (1990) Heat transfer simulation in solid substrate fermentation. Biotechnol Bioeng 35[8]: 802-808.
- Chisti Y [1999] Fermentation [industrial]: Basic considerations. In: Robinson R, et al. [Eds.], Encyclopedia of Food Microbiology. Academic Press, London, pp. 663-674.
- Singhania RR, Sukumaran RK, Patel AK, Larroche C, Pandey A [2010] Advancement and comparative profiles in the production technologies using solid-state and submerged fermentation for microbial cellulases. Enzyme Microb Technol 46: 541-549.
- Barrios-Gonzalez J, Banos JG, Covarrubias AA, Garay-Arroyo A (2008) Lovastatin biosynthetic genes of Aspergillus terreus are expressed differentially in solid-state and in liquid submerged fermentation. Appl Microbiol Biotechnol 79(2): 179-186.
- Lu LP, Zhang BB, Xu GR (2013) Efficient conversion of high concentration of glycerol to Monacolin K by solid-state fermentation of Monascus purpureus using bagasse as carrier. Bioprocess Biosyst Eng 36: 293-269.
- Waghmare SR, Kulkarni SS, Ghosh JS (2011) Chitinase production in solid-state fermentation from Oerskovia xanthineolytica NCIM 2839 and its application in fungal protoplasts formation. Curr Microbiol 63: 295-299.
- Nimnoi P, Lumyong S (2011) Improving solid-state fermentation of Monascus purpureus on agricultural products for pigment production. Food Bioprocess Technol 4: 1384-1390.
- Phrueksawan P, Kulpreecha S, Sooksai S, Thongchul N (2012) Direct fermentation of L[+]-lactic acid from cassava pulp by solid state culture of Rhizopus oryzae. Bioprocess Biosyst Eng 35(8): 1429-1436.
- Tao NG, Shi WQ, Lui YJ, Huang SR (2011) Production of feed enzymes from citrus processing waste by solid-state fermentation with Eupenicillium javanicum. Int J Food Sci Technol 46: 1073-1079.
- Santos MMd, Rosa ASd, Dal'Boit S, Mitchell DA, Krieger N [2004] Thermal denaturation: is solid-state fermentation really a good technology for the production of enzymes?. Bioresour Technol 93: 261-268.
- Gervais P, Molin P (2003) The role of water in solid-state fermentation. Biochem Eng J 13: 85-101.
- Ramani K, Kennedy LJ, Ramakrishnan M, Sekaran G (2010) Purification, characterization and application of acidic lipase from Pseudomonas gessardii using beef tallow as a substrate for fats and oil hydrolisis. Process Biochem 45: 1683-1691.
- Dalsenter FDH, Viccini G, Barga MC, Mitchell DA, Krieger N (2005) A mathematical model describing the effect of temperature variations on the kinetics of microbial growth in solid-state culture. Process Biochem 40: 801-807.
- Kapoor M, Gupta MN [2012] Lipase promiscuity and its biochemical applications. Process Biochem 47: 555-569.
- Bansal N, Tewari R, Soni R, Soni SK (2012) Production of cellulases from Aspergillus niger NS-2 in solid-state fermentation on agicultural and kitchen wate residues. Waste Manage 32(7): 1341-1346.
- Heerd D, Yegin S, Tari C, Fernandez-Lahore M (2012) Pectinase enzyme- complex production by Aspergillus spp. in solid-state fermentation: A compartive study. Food Bioprod Process 90: 102-110.
- Holker U, Lenz J (2005) Solid-state fermentation - are there any biotechnological advantages?. Curr Opin Microbiol 8: 301-306.
- Singhania RR, Patel AK, Soccol CR, Pandey A [2009] Recent advances in solid-state fermentation. Biochem Eng J 44: 8-13.
- Jahromi MF, Liang JB, Ho YW, Mohamad R, Goh YM, et al. (2012) Lovastatin production by Aspergillus terreus using agro-biomass as substrate in solid state fermentation. BioMed Research International 2012: 196264.
- El-Bendary MA [2010] Production of mosquitocidal Bacillus sphaericus by solid state fermentation using agricultural wastes. World J Microbiol Biotechnol 26: 153-159.
- Chen HZ, He Q (2012) Value-added bioconversion of biomass by solid- state fermentation. J Chem Technol Biotechnol 87: 1619-1625.
- Graminha EBN, Goncalves AZL, Pirota RDPB, Balsalobre MAA, Silva RD, et al. (2008) Enzyme production by solid-state fermetation: Application to animal nutrition. Anim Feed Sci Technol 144: 1-22.
- Esawy MA, Gamal AA, Kamel Z, Ismail AMS, Abdel-Fattah AF (2013) Evaluation of free and immobilized Aspergillus niger NRC1ami pectinase applicable in industrial processes. Carbohydr Polym 92[2]: 1463-1469.
- Maeda RN, Barcelos CA, Anna LMMS, Pereira N [2013] Cellulase production by PeniciHium funiculosum and its application in the hydrolysis of sugar cane bagasse for second generation ethanol production by fed batch operation. J Biotechnol 163: 38-44.
- Velmurugan P, Hur H, Balachandar V, Kamala-Kannan S, Lee KJ, et al. [2011] Monascus pigment production by solid-state fermentation with corn cob substrate. J Biosci Bioeng 112: 590-594.
- Chavez-Gonzalez ML, Rodri'guez-Duran LV, Cruz-Hernandez MA, Hernandez R, Prado-Barragan LA, et al. (2011) Effect of packing density and aeration rate on tannase production by Aspergillus niger GH1 in solid state fermentation. In: Sabu A, et al. (Eds.), Chemistry and Biotechnology of Poliphenols. India: CiBET Publishers 21[9]: 101-111.
- Mazutti MA, Zabot G, Boni G, Skovronski A, Oliveira DD, et al. (2010) Kinetics of inulinase production by solid-state fermentation in a packed-bed bioreactor. Food Chem 120: 163-173.
- Raghavarao KSMS, Ranganathan TV, Karanth NG [2003] Some engineering aspects of solid-state fermentation. Biochem Eng J 13: 127-135.
- Foong CW, Janaun J, Krishnaiah K, Prabhakar A (2009) Effect of superficial air velocity on solid state fermentation of palm kernel cake in a lab scale fermenter using locally isolated fungal strain. Ind Crops Prod 30: 114-118.
- Marti'nez-Trujillo A, Arregui'n-Rangel L, Garci'a-Rivero M, Aguilar- Osorio G [2011] Use of fruit residues for pectinase production by Aspergillus favipes FP-500 and Aspergillus terreus FP-370. Lett Appl Microbiol 53: 202-209.
- Rajendran A, Thangavelu V (2013) Utilizing agricultural wastes as substrates for lipase production by Candida rugosa NCIM 3462 in solid- state fermentation: response surface optimization of fermentation parameters. Waste Biomass Valor 4(2): 347-357.
- Rezaei F, Joh LD, Kashima H, Reddy AP, VanderGheynst JS (2011) Selection of conditions for cellulase and xylanase extraction from switchgrass colonized by Acidothermus cellulolyticus. Appl Biochem Biotechnol 164(6): 793-803.
- Zhu Z, Zhang G, Luo Y, Ran W, Shen Q (2012) Production of lipopeptides by Bacillus amyloliquefaciens XZ-173 in solid state fermentation using soybean flour and rice straw as the substrate. Bioresour Technol 112: 254-260.






























