Binding Activity of Mosquitocidal Protein of Pseudomonas Fluorescens Migulab426 to the Midgut of Larvae and Pupae of Culex Quinquefasciatus (DIPTERA: CULICIDAE)
Usharani Brammacharry1 and Muthuraj Muthaiah2*
1Department of Biomedical Genetics, University of Madras, India
2State TB Training and Demonstration Centre, Government Hospital for Chest Diseases, India
Submission: November 24, 2016; Published: December 02, 2016
*Corresponding author: Muthuraj Muthaiah, State TB Training and Demonstration Centre, Intermediate Reference Laboratory, Government Hospital for Chest Diseases, Puducherry, India,Email:muthuraj1970@gmail.com
How to cite this article: Usharani B, Muthuraj M.Binding Activity of Mosquitocidal Protein of Pseudomonas Fluorescens Migulab426 to the Midgut of 003 Larvae and Pupae of Culex Quinquefasciatus (DIPTERA: CULICIDAE). Adv Biotech & Micro. 2016; 1(5): 555572. DOI: 10.19080/AIBM.2016.01.555572
Abstract
To study the mode of action of mosquitocidal protein of Pseudomonas fluorescens Migula B426 on larvae and pupae of Culex quinquefasciatus, the presence of specific receptors for the mosquitocidal protein of P. fluorescens Migula B426 on Brush-Border Membrane Vesicles (BBMF) from Cx. quinquefasciatus larvae and pupae midgut cells was demonstrated by an in-vitro binding assay, immunofluorescence localization and Immunohistochemical localization. The presence of 55kDa and 35kDa bands in western blot assay revealed that the binding of mosquitocidal protein to the midgut of treated larvae and pupae of Cx.quinquefascistus.
The immunofluorescence localization study showed that the mosquitocidal protein binds preferentially to midgut of larvae and pupae of mosquito species Cx. quinquefasciatus. The present investigation confirmed that the binding of mosquitocidal protein to the gut regions of the pupae (non-feeding stage) by degrading the cuticle and overcome the peritrophic membrane and thus found to be effective pupicidal activity. However, in the larvae of Cx.quinquefascistus, the mosquitocidal protein is ingested orally and binds to the midgut epithelium causes the swelling of mitochondrial, endoplasmic reticulum and enlargement of vacuoles, followed by lysis of epithelial cells, midgut perforation and the death of larvae. It is concluded that the endogenous chitinases of P. fluorescens Migula B426 is to weaken the insect’s peritrophic membranes allowing the more ready access of the bacterial proteins to the gut epithelium of the larvae and pupae of Cx. quinquefasciatus.
Keywords: Pseudomonas fluorescens; Culex quinquefasciatus; Midgut; Mosquitocidal protein; Larvae; Pupae
Introduction
Mosquitoes are vectors of tropical diseases such as malaria, yellow fever, and dengue. Control of mosquito vectors has been accomplished with bio insecticides developed from the bacteria Bacillus thuringiensis sub sp. israelensis and Bacillus sphaericus. The B. thuringiensissubsp. israelensis produces Cry4Aa, Cry4Ba, Cry10Aa, Cry11Aa, and Cyt1Aa proteins, while B.sphaericus produces a binary protein. These insecticidal proteins are synthesized as inactive proproteins in the form of cytoplasmic crystalline inclusions that are solubilized and subsequently processed to active proteins by gut proteases in the larval midgut lumen.
The activated proteins bind to specific receptors on the microvilli of larval midgut epithelial cells and possibly undergo conformational changes followed by membrane insertion and pore formation, eventually resulting in the death of the larvae [1]. The Brush Border Membrane Vesicles (BBMV) isolated from lepidopteron midgut epithelium have been successfully used for the functional characterization of amino acid transport proteins [2] to analyze in vitro the activity of the insecticide fenoxycarb and, far more extensively, to study the mode of action of Cry1 proteins of B.thuringiensis [3,4]. The preparation of purified BBMV from the midgut of insects of interest is therefore fundamental to investigate several membrane phenomena.
The analysis of the in-vivo and in-vitro binding of the Cry4Aa, Cry4Ba, and Cry11Aa B. thuringiensis subsp. israelensis proteins in the midgut of the An. gambiae larvae have shown that these proteins are located in the apical microvilli of the posterior midgut with a mild binding being observed in the anterior midgut, suggesting that in the posterior midgut are receptors present with higher affinity or at higher concentration [5]. The binding of the protein to the apical microvilli of the midgut or to the midgut vesicles has generally been correlated with insect’s susceptibility to the particular protein; however, binding does not necessarily mean that the Cry protein is toxic [6].
The mosquitocidal protein(s) of a novel P fluorescens Migula B-426 strain and tested against 4th instar larvae and pupae of three species of vector mosquitoes, Anopheles stephensi Liston, Cx. quinquefasciatus Say and Aedesaegypti. The larvae and pupae of An. stephensi were the most susceptible to the formulation, followed by those of Cx. quinquefasciatus and Ae. aegypti in that order, and the dosage requirement for pupal mortality was less than that required for larval mortality. The LC50 dosage requirements for larvae of these mosquito species were, respectively, 70.4, 511.5 and 757.3mg protein ml_1, whereas for pupae they were, respectively, 2.0, 9.4 and 19.2mg protein ml_1. The lethal vesicles were purified from the culture broth and its molecular masses were determined by high performance liquid chromatography [7].
In this study, we described the preparation of purified midgut BBMV using whole larvae and pupae of Cx. Quinquefasciatus as a starting material and investigated the in-vivo binding of the mosquitocidal protein of P.fluorescens Migula B426 to the mosquito larvae midgut, through immunohistochemical methods in order to determine the primary site of action of this protein.
Materials and Methods
Production of mosquitocidal protein from P.fluorescens Migula B426
The culture supernatant of Pseudomonas fluorescens was collected after 72h of growth in GPS medium and the proteins were precipitated with 30 - 80% ammonium sulphate. The precipitated protein was dialyzed and fractionated by gel filtration using sephacryl S300 columns (Amersham-Pharmacia, Sweden) in an FPLC system. The column was eluted with Tris- HCl, containing sodium chloride buffer at a rate of 0.5ml/m.
The thoroughly prewashed fractions displayed at 280nm a peak with a retention time of 17m. The fraction gave a single peak when checked by HPLC analysis, indicating homogeneity of the fraction.Pure protein was subjected to 10% Native page and single band was observed with a molecular weight of 90kDa. Thus the FPLC fractionated pure protein was found to be a monomer. Two prominent bands were observed on 10% SDS PAGE and their molecular weight was found to be 55kDa and 35kDa [8].
Binding of mosquitocidal protein to the midgut of larvae and pupae of Cx. quinquefasciatus
Mosquitocidal protein treatment and rising of polyclonal antibodies Polyclonal antibodies were raised against the mosquitocidal protein by inoculating each of 200μg protein subcutaneously to 6-8 weeks old male BALB/C mice as per standard immunization protocol as indicated by Harlow et al. [9]. Blood from the challenged animals were collected after 20 days by tail vein puncture, serum was separated and the specificity were evaluated by ELISA and Western blot with 1:100, 1:1000dilutions respectively [10].
Preparation of midguts from the larvae and pupae of Cx.quinquefasciatus
The mosquitocidal protein (1mg/ml) was exposed to the 4th instars larvae and pupae of Cx.quinquefasciatus and mortality was observed after 16 hours .The 4th instars larvae and pupae of Cx.quinquefasciatus were placed in sterile petridish. The paddles were removed from the larvae, thorax and the last abdominal segment was pulled in the opposite direction with the help of dissection needle to get the midgut. The isolated midguts from the treated untreated larvae and pupae of Cx.quinquefasciatus were washed in 0.5ml of PBS and resuspended in 0.2ml of PBS.
The guts were homogenized in ice-cold gut extracting (GET) buffer (10mM Tris-HCl, pH 8.0, 1mM EGTA, 300mM mannitol, 1mM PMSF). BBMVs were prepared according to the method of Wolfersberger et al [11] with some modifications. An equal volume of ice-cold 24mM MgCl2 was added to the gut homogenate and the mixture was kept on ice for 15m. After centrifugation at 2,500rpm in 4 °C for 5 min, the supernatant was transferred to another centrifuge tube. The pellet was suspended in icecold GET buffer with 0.5 volume of the gut homogenate. The mosquitocidal protein concentration was estimated by a modification of the Lowry et al. [12] method using bovine serum albumin as a standard.
Immunoblot assay
The midgut proteins of untreated larvae and pupae were suspended in 4x Lammeli sample loading buffer. The samples were boiled for 5 minutes and separated by SDS-PAGE in Trisglycine buffer (192mM glycine, 25mM Tris-HCl, pH 8.3) were electrophoretically transferred onto a nitrocellulose membrane in the MiniTrans-Blot electrophoretic transfer cell (Bio-Rad) using transferring buffer (190mM glycine, 20% methanol and 25mM Tris-HCl, pH 8.3) at constant voltage for overnight [13,14].
The membranes were washed thrice in the phosphate buffered saline containing 0.05% Tween 20 and blocked with BSA for 2 hours to block the non-specific binding sites. After blocking, the proteins immobilized on the membrane were probed with 1: 500 dilution of the polyclonal antibodies developed against the mosquitocidal protein for 5hours. The nitrocellulose membrane was washed thrice with PBS-0.05% Tween 20 and the immunocomplexes were incubated with horseradish peroxidase-conjugated rabbit anti-mouse IgG (whole molecule) as the secondary antibody for 2 h. The binding was visualized using TMB/H2O2 as the substrate.
In-vivo binding of mosquitocidal protein to the midgut of larvae and pupae of Cx.quinquefasciatus
The midguts of untreated treated 4th larvae and pupae of mosquito species Cx.quinquefasciatus were dissected at 4oC and the guts were fixed with neutral formaldehyde. Mosquito larvae and pupae guts were dehydrated in increasing isopropyl alcohol concentrations, rinsed in xylol and hydrated in decreasing concentrations of ethanol, washed in distilled water and balanced in Tris buffer saline -TBS (66mM NaCl, 1.6mM KCl, 25mM Tris base, pH 7.4). Antigen unmasking was carried out with 1mg/ml of trypsin in 0.2N HCl for 5 min at room temperature.
The midguts were blocked with 3% BSA in TBS for 1h at a room temperature, rinsed with TTBS (0.5% Triton-X100 TBS), and incubated 1h at 37 °C in a humid chamber with 1: 500 of the polyclonal antibody raised in mouse against the mosquitocidal protein diluted in TBS with 1% BSA, incubated for 40 min a 37 °C in humid chamber with goat anti-mouse IgG fluorescence conjugated. The slides were washed three times in washing solution and mounted in D.P.X and observed under Fluorescence microscope [15].
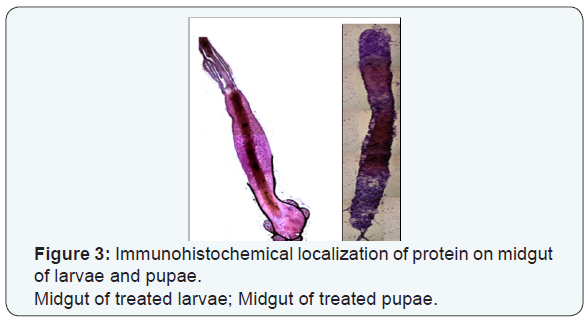
Immunohistochemical localization of the mosquitocidal protein
Mosquitocidal protein treatments: The 4th instar larvae of Cx.quinquefasciatus mosquitoes under starvation for 20hours were treated with 1mg/ml, which corresponded to approximately 500-fold the half lethal concentration for the mosquitoes for 2h, this concentration had been previously used by Orduz, et al. [16]. When intoxication and mortality were observed, larvae were removed to a petri dish with distilled water to wash the protein excess. The control larvae were treated with 100μl of 50mM PBS.
Preparation and sectioning of insect tissues: After exposure to the protein treatments, mosquito larvae were placed in neutral formaldehyde. After feeding Cx.quinquefasciatus larvae with the protein, guts were dissected at 4 °C and the midgut was fixed in neutral formaldehyde. Mosquito larvae and their midguts were dehydrated in increasing isopropyl alcohol concentrations, rinsed in xylol and included in paraffin. 5μm sections were obtained and placed in carriers loaded with 2% 3-aminopropyltriethoxy-silane [4].
Antibodies: Mice polyclonal antibody against protein (90kDa) was prepared as indicated by Harlow et al. [9]. Sensitivity and specificity were evaluated by ELISA and Western blot with 1:100, 1:1000 dilutions, respectively, of the mice sera using standard techniques [10]. The immunodetection was performed in combination with a peroxidase-conjugated goat IgG fraction to mouse IgG (whole molecule) (Sigma Chemicals co).
Detection of mosquitocidal protein was performed through modifications of the technique developed by Bravo et al. [4]. The sections were de-paraffinated in 100% xylol and hydrated in decreasing concentrations of ethanol, washed in distilled water and balanced in Tris buffer saline (TBS) (66mM NaCl, 1.6mM KCl, 25mM Tris base, pH 7.4). Antigen unmasking was carried out with 1 mg/ml of trypsin in 0.2N HCl for 5 min at room temperature. Trypsin was inactivated by addition of 0.5mM PMSF with incubation for 5 min and then the endogen peroxidase activity was blocked by incubating the tissue sections with 0.5% H2O2 for 30m and 6% H2O2 for 15m in methanol, at room temperature.
The tissue section boundaries were marked with hydrophobic pencil in order to achieve uniformity in the incubations. The tissue samples were blocked with 3% BSA in TBS for 1h at a room temperature, rinsed with TTBS (0.5% Triton-X100 TBS ), and incubated 1h at 37 °C in a humid chamber with 1:1000 of the polyclonal antibody raised in mouse against the mosquitocidal protein diluted in TBS with 1% BSA, incubated for 40 min a 37 °C in humid chamber with 1:200 of peroxidase-conjugated goat IgG fraction to mouse IgG, and washed again with TBS.
Colour development was achieved with 3,3-diaminobenzidine (DAB) in chromogen solution in imidazole-HCl buffer pH 7.5 containing hydrogen peroxide and an antimicrobial agent (Liquid DAB large volume substrate-chromogen) for 3 min at room temperature. The counter staining of the tissue sections was performed with Harris hematoxilin for 10s and ammoniacal water for 10s; the tissues were dehydrated in increasing concentrations of ethanol and clarified in xylol; finally, the sections were covered with DPX (Sigma Chemicals co) mounting resin and analyzed by light microscopy.
Binding of mosquitocidal protein to brush-border membrane vehicle (BBMV)
Brush border membrane vehicle isolation: Toxicity assays were performed on early 4th instar larvae of Cx.quinquefasciatus, in 5cm diameter Petri dishes containing 10 ml of 100mg per ml mosquitocidal protein dilutions in demineralised water. Dead larvae were counted after 24 h, and lethal concentrations killing 50% of the population (LC50) evaluated by probit analysis [17].
Brush Border Membrane Vesicles were prepared from midguts of Cx.quinquefasciatuslarvae following methods modified from those of Biber et al [18], Wolfersberger et al [11], Houk et al. [19]. The 4th instar larvae were chilled on ice, midguts extruded by dissection under a binocular microscope, peritrophic membranes and Malpighian tubules removed, and midguts rinsed in ice-cold buffer A (0.3M mannitol/5mM EGTA/20mM Tris/HCl, pH 7.4).
Midguts were then placed in 1ml cryogenic vials, frozen by immersing the vials in liquid nitrogen and stored at -70 °C until required. BBMF were prepared freshly before each series of experiments. Midguts were thawed, pooled in ice-cold buffer A and ground with a 25-75μm clearance homogenizer (40 strokes). The homogenate was adjusted to 3ml and made upto 12mM MgC12. The samples were allowed to stand on ice for 20m.
The mixture was then centrifuged at 3000xg in 1.5ml capacity polyethy1ene microfuge for 15m at 2 °C.
The supernatant was collected and kept on ice. The pellets were suspended in buffer A and treated as above for the first midgut homogenization. The resulting supernatant was pooled with the first and centrifuged in the same rotor at 14100xg for 60rn at 2 °C. The resulting pellet was then suspended in the proportion of 1 midgut/ μl buffer A and kept on ice. Protein content was determined by the Bio-Rad protein assay with bovine serum albumin as standard. The protein yield was approximately 0.5-1pg membrane protein/midgut. Each step of BBMF preparation was checked for aminopeptidase activity as a marker; a typical enrichment between the crude homogenate and BBMF was approximately 3-5/U protein [20].
Immunoblotting assay
Binding was performed in 100μl binding buffer [PBS (pH 7.6), 0.1% (w/v) BSA, 0.1% (v/v) Tween 20]. BBMV protein (10μg) was incubated with biotinylated protein (10nM) in the presence of a several-fold excess of unlabelled protein for 1 h. The unbound protein was removed by centrifugation for 10m at 14000g. BBMV were washed three times in binding buffer and suspended in 15μl of PBS, 5μl of Laemmli sample loading buffer 4X [0.125M Tris/HCl (pH 6.8), 4% (w/v) SDS, 20% (v/v) glycerol, 10% (v/v) 2-mercaptoethanol, 0.01% (w/v) Bromophenol Blue. The samples were boiled for 5m, separated by SDS/10% PAGE, and electro blotted on to Hybond nitrocellulose membranes (Amersham Biosciences).The biotinylated protein was visualized by incubating membranes with streptavidin-peroxidase conjugate (1:6000 dilution; Amersham Biosciences) for 1h followed by incubation with Super Signal chemiluminescence substrate (Pierce) as described in the manufacturer’s protocol [21].
Protein overlay assay
Analysis of BBMV proteins recognized by mosquitocidal protein was as follows. BBMV proteins (10μg) were separated through SDS/10% PAGE gels and electro blotted on to Hybond nitrocellulose membranes as described above. After blocking, the membranes were incubated for 1 h with 30nM protein in blocking buffer [0.1M NaHCO3 (pH 9.6), 5mg/ml BSA, 0.02% (w/v) NaN3] at 25oC. Unbound proteins were removed by washing six times for 10m in washing buffer [PBS, 0.1% (v/v) Tween 20] at 25 °C. The bound protein was identified by incubation with anti-M13 mouse antibody and anti-mouse peroxidase conjugate (1:2000 dilutions), for 1h at 25 °C. Binding was visualized using Super Signal chemiluminescence substrate as described above [22].
Analysis of GPI anchored proteins that interact with proteins
BBMV were prepared from dissected midguts of 4th instar larvae of Cx.quinquefasciatus by differential precipitation using MgCl2, as described above in this chapter and stored at −70 °C until used. Cx.quinquefasciatus BBMV (300μg) were suspended in 500μl of PBS2 [137mM NaCl, 2.7mM KCl, 10mM Na2HPO4 (pH 7.2)].PIPLC (phospholipase-C) (3 units) of from Bacillus cereus was added to the BBMV suspension and incubated for 90m at 30 °C, as previously reported by Lawrence, et al. [23]. Membranes were recovered by centrifugation at 11000g for 20 m, and the supernatant was analysed for the presence of GPI-anchored proteins by SDS/10% PAGE and protein overlay as described above.
Then mosquitocidal protein was biotinylated using biotinyl- N-hydroxysuccinimide ester (catalogue number RPN28; Amersham Biosciences) according to the manufacturer’s directions. A matrix of agarose-streptavidin (200μl; Sigma- Aldrich) was incubated with 7μg of biotinylated- protein overnight at 4 ºC. After incubation, the matrix was washed ten times in 1X PBS2 to remove any unbound protein. The solution containing GPI-anchored proteins (42μg) was incubated with the 200μl of protein-agarose matrix. The matrix was washed four times with 1X PBS2. The binding proteins were then eluted using 200μl of 1X PBS2, pH 9.5. The pH was adjusted to 7.2 and the samples were stored at 4 ºC in the presence of protease inhibitors [24].
ALP and APN specific activities
Specific ALP and APN enzymatic activities were measured using 1 mg/ml of p-nitro phenyl phosphate and leucine-pnitroanilide (Sigma-Aldrich) as substrates respectively. BBMV proteins (5μg) were mixed with ALP buffer [0.5mM MgCl2, 100mM Tris/HCl (pH 9.5)] containing 1.25mM of p-nitro phenyl phosphate and APN buffer [0.2M Tris/HCl (pH 8), 0.25M NaCl] containing 1mM of leucine-p-nitroanilide. Enzyme activities were monitored as the change in the absorbance at 450 nm for 3 min at 25 ºC [23].
Results
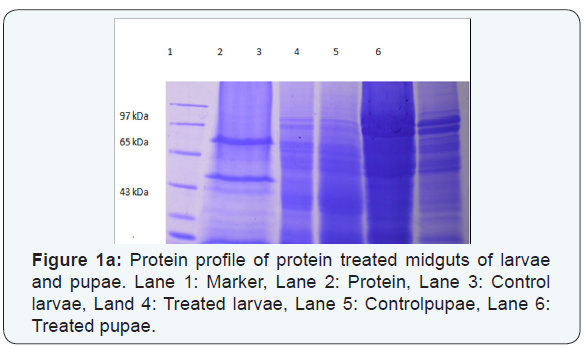
The 55kDa protein on the membrane was detected with the pooled antiserum against 55kDa protein (1:1000 dilutions) as primary antibodies and peroxidase-conjugated rabbit antimouse IgG as the secondary antibody. Thus the presence of 55kDa alone in the membrane confirmed that the protein is not degraded to 35kDa protein(Figure 1a).The presence of 55kDa and 35kDa bands in western blot revealed that the binding of mosquitocidal protein to the midgut of treated larvae and pupae of Cx.quinquefascitus (Figure 1b,1c). It has been reported that the binding of the protein to the midgut epithelium causes swelling of mitochondrial and endoplasmic reticulum and enlargement of vacuoles, followed by lysis of epithelial cells, midgut perforation and the death of the larvae.

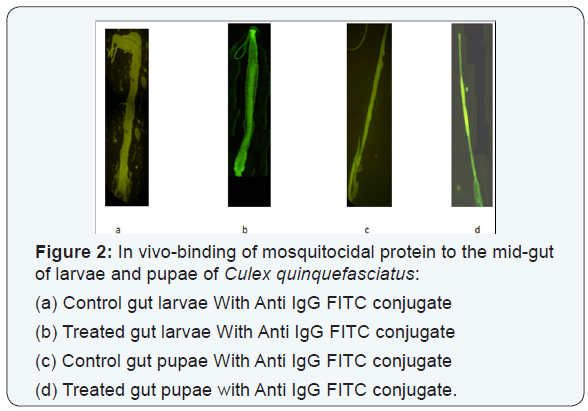
The binding activity of the mosquitocidal protein to the midgut of larvae and pupae of Cx.quinquefasciatus were analysed by immunofluorescence localization in which regions of the midgut this protein binds. In addition, we analyzed the binding of mosquitocidal protein to the midgut epithelium of Cx.quinquefasciatus larvae and also determined the different regions of midgut epithelium of Cx.quinquefasciatus pupae. Figure 2 showed that the mosquitocidal protein binds preferentially to midgut of larvae and pupae of mosquito species Cx.quinquefasciatus.
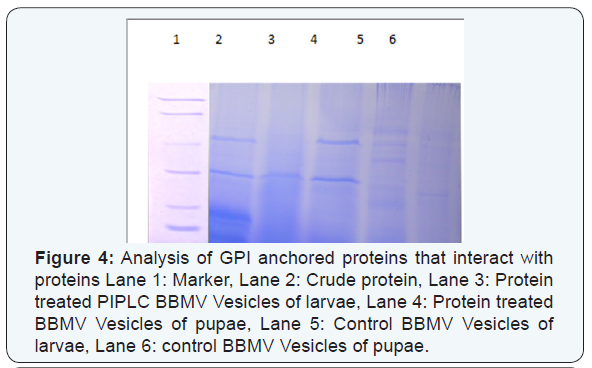

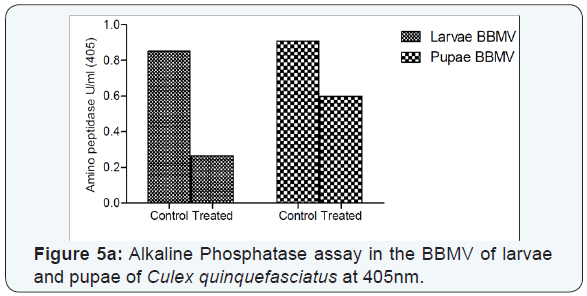
The alkaline phosphatase and amino peptidase activity was found to be decreased in the treated larvae and pupae than the control larvae and pupae (Figure 5a & 5b). The results revealed that the binding of protein to the receptor molecules of Brush Border Membrane Vesicles of Cx.quinquefasciatus leading to the impairment of the enzymatic machinery of midgut cells and reduced the specific activity of the enzymes. It has also been reported that the GPI-anchored proteins like alkaline phosphatase and amino peptidase are important receptor molecules that mediates the toxicity in Ae.aegypti larvae and reducing the activity of enzymes.
Discussion
The present study revealed that the chitin is the target substrate for the mosquitocidal protein and it overcomes peritrophic membrane of the mosquito species and thereby supports the proposed mode of action for mosquitocidal metabolites. Thus, the present investigation confirmed that the mosquitocidal protein efficiently hydrolysis the cuticular protein chitin and overcome the peritrophic membrane which is a protective sleeve for the midgut epithelium mosquito species and binds to the gut regions of larvae and pupae of Cx. quinquefasciatus. The immunofluorescence studies further confirmed that the binding of mosquitocidal protein to the gut regions of the pupae (non-feeding stage) by degrading the cuticle and overcome the peritrophic membrane and thus found to be effective pupicidal activity.
However, in the larvae of Cx.quinquefascistus, the mosquitocidal protein is ingested orally and binds to the midgut epithelium causes the swelling of mitochondrial, endoplasmic reticulum and enlargement of vacuoles, followed by lysis of epithelial cells, midgut perforation and the death of larvae. The results marker enzymes revealed that the binding of protein to the receptor molecules of brush border membrane vesicles of Cx.quinquefasciatus leading to the impairment of the enzymatic machinery of midgut cells and reduced the specific activity of the enzymes.
The immunohistochemical localization of the Cry11Bb protein observed in the basal membrane of Ae. aegypti and An. albimanus larvae was possibly due to the protein’s leakage or at least part of it from the midgut lumen to the basal area of the epithelium after disruption of the cellular integrity of the midgut tissue. The deterioration of the intercellular junctions could be a consequence of the damage caused by the Cry11Bb protein at the cellular membrane level, as this situation has also been observed in Heliothis virescens midgut after 3h exposure to the Cry1Ac [25].
Additionally, the Cry11Bb protein was observed in Cx.quinquefasciatus larval midgut 15m after treatment and 1h after exposure to the protein on the apical microvilli of the Malpighian tubules. It is unknown if the binding of the Cry11Bb protein to the Malpighian tubules contributes to mortality of mosquito larvae. The binding sites of several Cry proteins have been located on the Malpighian tubule epithelium of some insect species; however, their role in toxicity has not been fully characterized. Although Maddrell, et al. [26] reported important changes in the trans-epithelial potential difference of Rhodnius prolixus Malpighian tubules after treatment with B.thuringiensis protein and Reisner, et al. [27] located B.thuringiensis kurstaki δ-endoproteins on the Malpighian tubules of Calpodes ethlius larvae and described its effect as inhibitory of fluid secretion causing cytological alterations, which ended-up in cellular lysis and epithelial damage, Denolf, et al. [28] reported binding of Cry1Ab1, Cry1Ac1, and Cry1Ba1 proteins to the Malpighian tubules of Ostrinia nubilalis, but did not suggest a relevant role in the mortality of this lepidopteran. It is possible that the damage caused by the Cry11Bb protein in the mosquito larvae midgut intercellular junctions could have permitted protein leakage into the haemolymph to reach the Malpighian tubules, producing shrinking of their epithelium, and perhaps impairing their osmoregulatory functions before larval death.
The alkaline phosphatase and amino peptidase activity was found to be decreased in the treated larvae and pupae than the control larvae and pupae. The results revealed that the binding of protein to the receptor molecules of Brush Border Membrane Vesicles of Cx.quinquefasciatus leading to the impairment of the enzymatic machinery of midgut cells and reduced the specific activity of the enzymes. It has also been reported that the GPIanchored proteins like alkaline phosphatase and amino peptidase are important receptor molecules that mediates the toxicity in Ae.aegypti larvae and reducing the activity of enzymes [14]. It has also been reported that after exposure of BBMV to cry1A proteins, these proteins were found to associate with the lipid rafts micro domains and the integrity of these micro domains was essential for in-vitro cry1 A pore forming activity [29].
Conclusion
The mosquitocidal protein of P.fluorescens Migula B426 showed active hydrolysis of chitin polymers and the enhancement of chitinase activity revealed that the invasion of mosquitocidal protein through the cuticular region of larvae and pupae. Thus, the present investigation confirmed that the mosquitocidal protein efficiently hydrolysis the cuticular protein chitin and overcome the peritrophic membrane which is a protective sleeve for the midgut epithelium mosquito species and binds to the gut regions of larvae and pupae of Cx.quinquefasciatus. The immunofluorescence studies further confirmed that the binding of mosquitocidal protein to the gut regions of the pupae (non-feeding stage) by degrading the cuticle and overcome the peritrophic membrane and thus found to be effective pupicidal activity.
This supports the hypothesis that the mosquitocidal protein alters the structural integrity of peritrophic membrane and binds to the gut regions of larvae and pupae of Cx.quinquefasciatus. The immunofluorescence studies and western blot analysis revealed the specific binding of mosquitocidal proteins to the epithelial cells of midgut regions of larvae and pupae of Cx.quinquefasciatus.
The alkaline phosphatase and amino peptidase activity was found to be decreased in the treated larvae and pupae than the control larvae and pupae revealed that the binding of protein to the receptor molecules of Brush Border Membrane Vesicles of Cx.quinquefasciatus leading to the impairment of the enzymatic machinery of midgut cells and reduced the specific activity of the enzymes. It is concluded thattheendogenous chitinases of P.fluorescens Migula B426 is to weaken the insect’s peritrophic membranes allowing the more ready access of the bacterial proteins to the gut epithelium of the larvae and pupae of Cx.quinquefasciatus.
Acknowledgement
Authors thank Dr. P. Jambulingam, Director, Vector Control Research Centre, Indian Council of Medical Research, Puducherry for his support and critical suggestions.
References
- Knowles BH (1994) Mechanism of action of Bacillus thuringiensis insecticidal d-endoproteins. Adv Insect Physiol 24: 275-308
- Wolfersberger M, Liithy P, Maurer A, Parenti P, Sacchi, VF, et al. (1987) Preparation and partial characterization of amino acid transporting brush border membrane vesicles from the larval midgut of the cabbage butterfly (Pieris brassicae). Comp Biochem Physiol MA 301-308.
- Sacchi VF, Parenti P, Giordana B, Hanozet GM, Liithy P (1986) Bacillus thuringiensis protein inhibits K+ -gradient-dependent amino acid transport across the brush border membrane of Pieris brassicae midgut cells. FEBS Lett 204: 213-218.
- Bravo A, Hendricks K, Jansens S, Peferoen M (1992) Immunocytochemical analysis of specific binding of Bacillus thuringiensis insecticidal crystal proteins to lepidopteron and coleopteran midgut membranes. J Invertebr Pathol 60(3): 247-253.
- Charles JF (1987) Ultra structural midgut events in culicidal larvae fed with Bacillus sphaericus spore/crystal complex. Ann Inst Pasteur Microbiol 138(4): 471-484.
- Fonsfeldmann, Annette Dullemans, Cess waalwijk (1995) Binding of the Cry IVD Protein of Bacillus thuringiensis subsp. israelensis to Larval Dipteran Midgut Protein. App Environ Microbial 61(7): 2601- 2605.
- Prabakaran G, Paily KP, Padmanabhan, V, Holti SL, Balaraman K (2002) Isolation of a Pseudomonas fluorescensmetabolite/exoprotein active against both larvae and pupae of vector mosquitoes. Pest Manag Sci 59(1): 21-24.
- Usharani Brammacharry, Kummankottil Paily (2014) Gelatinase activity of metabolites of Pseudomonas fluorescens migula on larvae and pupae of Culexquinquefasciatus (Diptera: Culicidae). Int J Pharm Bio Sci 3(11): 234-245.
- Harlow E, Lane D (1999) Using Antibodies: A Laboratory Manual, Cold Spring Harbor Laboratory Press, New York, USA, pp. 495.
- Voller A, Bid well D, Burek C (1980) An enzyme-linked immunosorbent assay (ELISA) for antibodies to thyroglobulin.Proc Soc Exp Biol Med 163(3): 402-405.
- Wolfersberger MG (1989) Neither barium nor calcium prevents the inhibition by Bacillus thuringiensis 8-endoprotein of sodium- or potassium gradient dependent amino acid accumulation by tobacco hornworm midgut brush border membrane vesicle. Arch Insect Biochem Phys 12: 267-277.
- Lowry OH, Rosen rough NJ, Randall AJ (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193(1): 265-275.
- Towbin H, Staehelin T, Gordon J (1979) Electrophoretic transfer of proteins from polyacrilamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A 76(9): 4350-4354.
- Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227(5259): 680- 685.
- Luisa EF, Karlygash GA, Sarjeet SG, Alejandra Bravo, Mario Soberon A (2006) GPI-anchored alkaline phosphatase is a functional midgut receptor of cry 11Aa protein in Aedes aegypti larvae. Biochem J 394(Pt 1): 77-84.
- Orduz S, Diaz T, Thiery I, Charles JF, Rojas W (1994). Crystal proteins from Bacillus thuringiensis serovar. Medellin. Appl Microbial Biotechnol 40(6): 794-799.
- Finney DJ (1971) Probit analysis, 3rd edn, S Chand & Co Ltd, New Delhi, India, p 50-80.
- Biber J, Stieger B, Haase W, Murer H (1981) A high yield preparation for rat kidney brush border membranes. Different behaviour of lysosomal markers. Biochim Biophys Acta 647(2): 169-176.
- Houk EJ, Arcus YM, Hardy JL (1986) Isolation and characterization of brush border fragments from mesenterons. Arch Insect Biochem Physiol 3: 135-146.
- Erlanger B, Kokowsky N, Cohen W (1961) The preparation and properties of two new chromogenic substrates of trypsin. Arch Biochem Biophys 95: 271-278.
- Xue Jun Chen, April Curtiss,Edwin Alcantara, Donald H, Dean (1995) Mutations in Domain I of Bacillus thuringiensis δ -endoprotein Cry IAb Reduce the Irreversible binding to Manduca sexta Brush Border membrane Vehicles. J Bio Chem 270(11): 6412-6419.
- Christina Nielsen-Leroux, Jean-François Charles (1992) Binding of Bacillus sphaericus binary protein to a specific receptor on midgut brush-border membranes from mosquito larvae. Eur J Biochem 210(2): 585-590.
- Lawrence A, Darszon A, Bravo A (1997) Amino peptidase dependent pore formation of Bacillus thuringiensis Cry1Ac protein on Trichoplusiani membranes. FEBS Lett 414(2): 303-307.
- Laurence Abrami, Marc Fivaz, Pierre-Etienne Glauser, Robert GP, Gisou vander Goot F (1998) A Pore-forming Protein Interacts with a GPI-anchored Protein and Causes Vacuolation of the Endoplasmic Reticulum. J Cell Bio 140(3): 525-540.
- Forcada C, Alcácer E, Garcerá MD, Tato, A, Martínez, R (1999) Resistance to Bacillus thuringiensis Cry1Ac protein in three strains of Heliothis virescens: Proteolytic and SEM study of the larval midgut. Arch Insect Biochem Physiol 42(1): 51-63.
- Maddrell SH, Overton JA, Ellar DJ, Knowles BH (1989) Action of activated protein from Bacillus thuringiensis var. israelensis on Malpighian tubules of the insect, Rhodnius prolixus. J Cell Sci 94(pt 3): 601-608.
- Reisner W, Feir D, Lavrik P, Ryerse J (1989) Effect of Bacillus thuringiensis kurstaki d-endoprotein on insect Malpighian tubule structure and function. J Invertebr Pathol 54: 175-190.
- Denolf P, Jansens S, Peferoen M, Degheele D, Van Rie J (1993) Two different Bacillus thuringiensis delta-endoprotein receptors in the midgut brush border membrane of the European corn borer, Ostrinia nubilalis (Hübner) (Lepidoptera: Pyralidae). Appl Microbiol Biotechnol 59(6): 1828-1837.
- Zhuang M, Oltean DI, Gomez I, Pullikith AK, Soberon M, et al. (2002) Heliothis virescens, Manduca Sexta, Lipid rafts are involved in Cry1A toxin binding to the midgut epithelium and subsequent pore formation. J Biol chem 277(16): 13863-13872.






























