An Investigation of the Effect of Copper Oxide and Silver Nanoparticles on E. Coli Genome by Rapd Molecular Markers
Amjady Fariba1, Golestani Emani Bahram2*, Karimi Farrokh3, Sevda Tabrizy4 and Seyed Sayyah Pooneh5
1,2,4,5Department of Biology, Islamic Azad University, Iran
3Department of Biotechnology, University of Maragheh, Iran
Submission: July 25, 2016; Published: August 12, 2016
*Corresponding author: Golestani Emani Bahram, Department of Biology, Urmia Branch, Islamic Azad University, Urmia, Iran.
How to cite this article: Amjady F, Golestani EB, Karimi F, Sevda T, An Investigation of the Effect of Copper Oxide and Silver Nanoparticles on E. Coli Genome by Rapd Molecular Markers. Adv Biotech & Micro. 2016; 1(2): 555559. DOI: 10.19080/AIBM.2016.01.555559
Abstract
Regarding the bacterial resistance of current antibiotics, many studies have been conducted to evaluate the antimicrobial properties of metal nanoparticles such as silver and copper oxide. This study was carried out to compare the effects of these nanoparticles on the genome of Escherichia coli strain O157: H7 as a model for gram-negative bacteria.
For this purpose, the bacteria were first treated by 30 and 60 μg/ml the nanoparticles and the growth of bacteria was controlled at certain time intervals by spectrophotometry to evaluate the antimicrobial activity. Then the DNA was extracted to investigate the effects of the nanoparticles on the genomes 4 hours after treatment using RAPD-PCR. The bands obtained from electrophoresis of PCR products on agarose gel 2% were analyzed.
The results of the study revealed that silver and copper oxide nanoparticles not only inhibit the growth of bacteria, but also change the genomic DNA sequences and cause genetic differences between control and treated samples. Metal nanoparticles are antibacterial compounds, and copper oxide was more effective than silver on E. coli genome as a model for gram-negative bacteria.
Keywords: Silver and copper oxide nanoparticles; Changes in DNA sequences; Escherichia coli; RAPD polymerase chain reaction (RAPD-PCR)
Abbreviations: EMB: Eosin Methylene Blue Agar; BHI: Brain Heart Infusion; OD: Optical Density; NDM-1: New Delhimetallo-Beta-Lactamase-1; RAPD-PCR: RAPD Polymerase Chain Reaction
Background
In general, antibacterial agents are classified into two categories: bactericidal and bacteriostatic (growth inhibitor). However, the widespread and indiscriminate use of anti-bacterial agents has been led to bacterial resistance, which is a serious challenge in this area. Resistance often affects different stages of development such as inheritance to new strains, which requires therapeutic strategies for diseases caused by bacteria [1]. Metal nanoparticles including copper and silver have antimicrobial effect against bacteria, viruses and other microorganisms [2]. Copper nanoparticles, especially inexpensive nanoparticles at the level of micro-electrical applications, have received more attention in recent years and are likely to be the latest discovered antimicrobial agents [3].
Unlike the usual chemical disinfections, antimicrobial nanomaterials are not expected to manufacture harmful disinfections [4]. Antibacterial effects of metal nanoparticles are because of very small size and high surface area to volume ratio, which allow being in contact directly with microbial membranes and releasing metal ions [5]. Targeted uses of silver and copper oxide nanoparticles with antimicrobial properties require more attention in terms of compatibility with the environment. Considering the fact that copper nanoparticles are lethal to other organisms, for example against crustaceans such as Daphnia magna and Thamnocephalusplatyurus and also Pseudo- kirchneriella subcapitata algae at low concentrations (3.2, 0.18 and <1 mg/l, respectively) reported in 2008 and 2009 [6,7] and thereby a degree of bactericidal of nanoparticles have been shown to aquatic organisms.
It has been also reported that low concentrations of silver ions released from silver nanoparticles are able to haemolysis of red blood cells, in vitro [8,9]. In addition, silver nanoparticles can directly affect the normal functioning of cells in the body, causing dysfunction of the organs [10]. As well, numerous reports and laboratory studies have shown that silver nanoparticles can induce immune responses [11]. Several experimental researches have proved that silver nanoparticles cause damages to DNA and cells as well as result in cancer, oxidative stress and detoxification of metal [12,13].
Therefore, further investigations are required on nanoparticles to determine the effective concentrations of antibacterial and their genotoxicity effects as a suitable replacement for antibiotics and disinfectants. Since the antibacterial effects of silver and copper oxide nanoparticles have not been yet studied on the genomes; so this study was conducted to evaluate the effect of silver and copper oxide nanoparticles on the genome of Escherichia coli as a model for gram-negative bacteria and compare the effect of nanoparticles on the genome using random amplified polymorphic DNA- PCR (RAPD-PCR) technique.
Materials and Methods
Bacterial culture and the conditions
Characteristics of Escherichia coli 0157: H7 (ATCC 25922) bacteria was identified on eosin methylene blue agar (EMB) medium. The bacteria were cultured in 5 ml of the Brain Heart Infusion (BHI) broth, and were left overnight in a shaking incubator at 37°C at rpm 200. The growth rate of bacteria was controlled by measuring optical density (OD) of medium at a wavelength of 600 nm [14].
Evaluate the antimicrobial properties of copper oxide and silver nanoparticles
Copper oxide and silver nanoparticles less than 20nm in diameter were synthesized by the Nanotechnology South Korea. Characterization and analysis of the nanoparticles by electron microscopy have been presented in (Figure 1). Phosphate buffer saline (pH 7.4) was used as a solvent for preparing 30 and 60μg/ ml concentrations of the nanoparticles, separately in tubes. The tubes were placed in a shaking incubator at 37°C at 200rpm and their OD was measured at 600nm with intervals of 2, 4 and 24 hours for the bacteria treated by nanoparticles to measure growth.

DNA extraction and RAPD-PCR
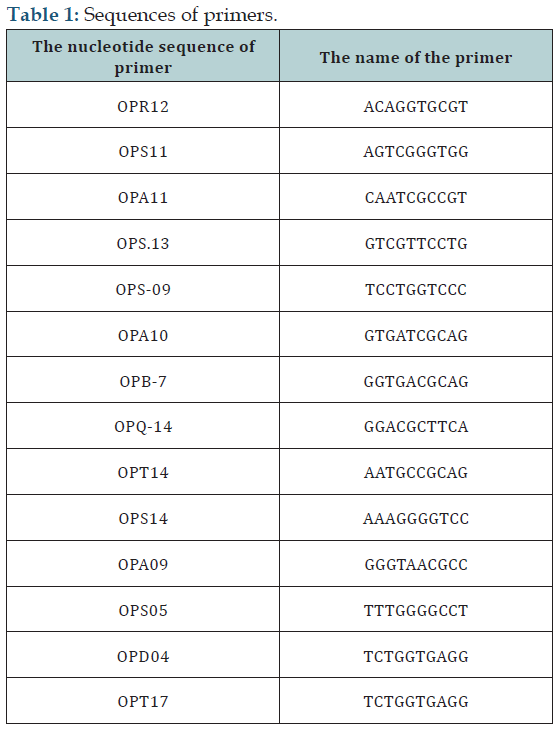
DNAs of treated and control bacteria were extracted using DNA extraction kit (ExirAzma Co.) in accordance with the kit instructions, and then the quantity and quality were analyzed by spectrophotometry and electrophoresis on agarose gel 1%. RAPD- PCR molecular marker method was used to examine the effect of nanoparticles on bacterial genome. Initially, RAPD 10 bases-primers (Cinnagene Co.) were prepared to perform RAPDPCR method. Sequences and characterization of primers have been shown in (Table 1).
Ingredients listed below at concentrations of 25ml volume were prepared for PCR reaction to amplify samples; 1μl primer, 2.5μl (10 x) PCR buffer, 3μl MgCl2, 1μl dNTP mix, 1μl of extracted DNA samples and 0.3μl of DNA Taq polymerase that was reached to the volume of 25ml by 16.2μl of deionized distilled water.
The mixture was placed in a thermocycler (Corbett research, Australia) with the following schedule: the initial template DNA denaturation at 95°C for 5 minutes, followed by 40 cycles of PCR reactions, so that 95°C for 35 seconds for denaturation of template DNA strands, 30°C for 45 seconds to attach the primers to the template strand, 72°C for 45 seconds for the polymerization of a new strand from template strand. 7 minutes were required to complete the polymerization of incomplete strands. These compositions and the temperature profiles after optimizing the PCR conditions were used for all 14 primers.
Evaluation the results of RAPD-PCR
After completion of the PCR reaction, electrophoresis was done for 10μl of PCR products on agarose gel 2% (the size of 14×26cm) containing red safe in TBE buffer (1x) for 4 hours with a voltage of 120 volt to detect appeared bands; DNA ladder marker with 100bp was used to determine the product size and the images of gel were taken using imaging system (Uvitec, France).
Analysis of data obtained from electrophoresis
The bands resulting from analyzed RAPD were scored based on the presence or absence, respectively, as one and zero. The data was then entered into the software based on molecular weight; the similarity matrix was calculated by Dic and dendrogram were derived by UPGMA method in NTSYS-PC software.
Results
SEM electron microscopes were used for study of the copper oxide and silver nanoparticles (Figure 1).
Evaluate the antimicrobial properties of copper oxide and silver nanoparticles
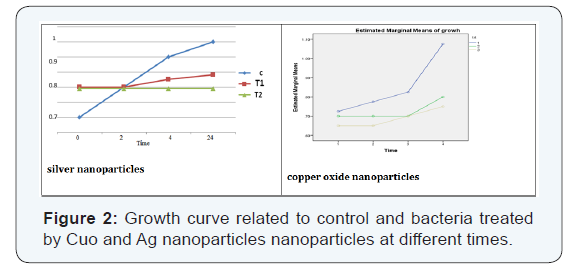
The results of experiments on antimicrobial activities of copper oxide and silver nanoparticles against Escherichia coli bacteria have been presented in (Figure 2), demonstrating that the growth of bacteria was clearly stopped after treatment by nanoparticles in intervals of 2 and 4 hours and they had only minor growth after 24 hours.
RAPD- PCR product analysis

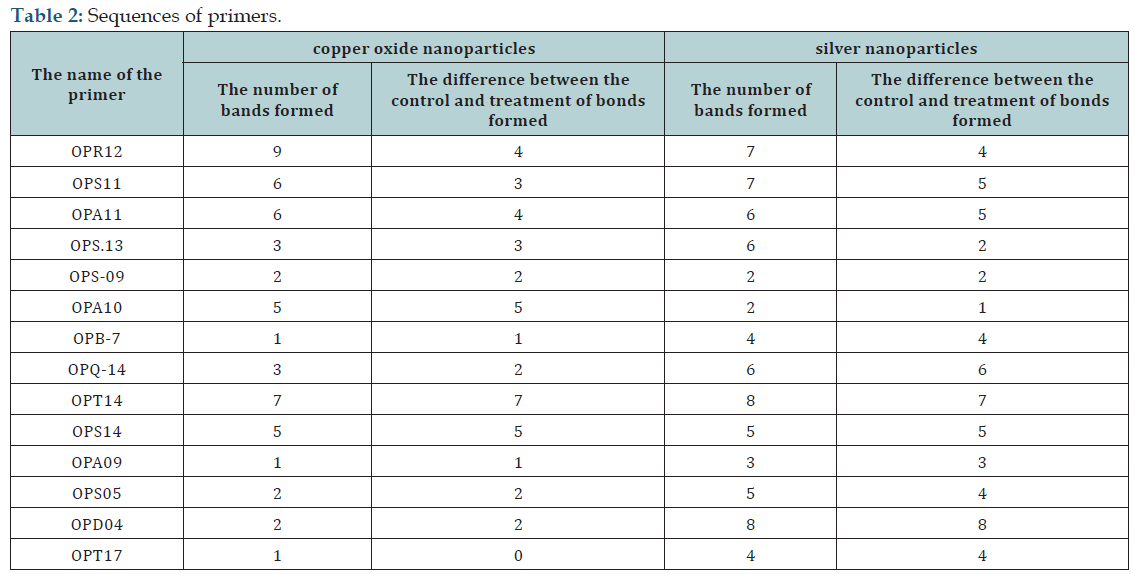
Electrophoretic bands obtained from amplification of 14 primers by the RAPD- PCR have been shown in (Figure 3) for bacteria treated by copper oxide and silver nanoparticles. The bands obtained from analyzed RAPD were scored based on the presence or absence, respectively, as one and zero. The conclusion was based on the difference in the bands formed by each primer, for control and treated samples (Table 2) as can be seen in (Table 2), totally 53 bands were produced for bacteria treated with copper oxide nanoparticles from 14 primers that 41 bands were different between the control and treated samples. Collectively 73 bands were also appeared for bacteria treated with silver nanoparticles which 60 bands were different between the control and treated samples.
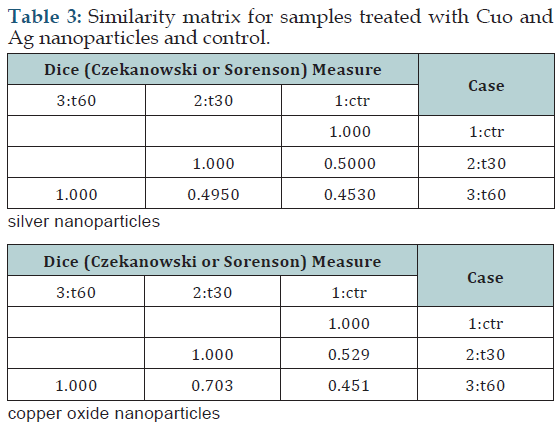
(Table 3) shows the results of NTSYS-PC software in order to compare genetic variations between control and treated samples.
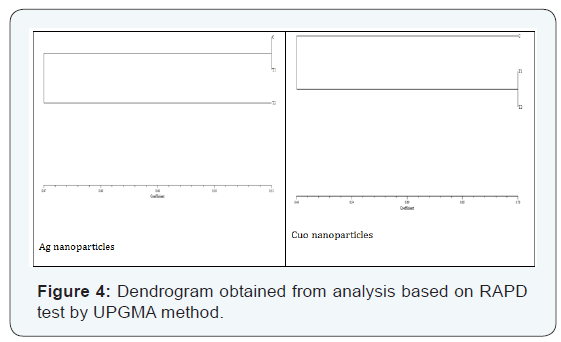
The dendrogram was drawn by UPGMA in NTSYS-PC software to compare genetic variations between control and samples treated with nanoparticles. As seen in (Figure 4), control and samples treated with copper oxide nanoparticles have been located in two separate main branches, suggesting a genetic difference; but the control and first treatment of silver nanoparticles in a branch, and the second treatment in a separate main branch, reflecting the impact of nanoparticles at high concentrations.
Discussion
The development of severe bacterial resistance to antibiotics is a major health problem. In this regard, the nanoparticles are considered new antimicrobial agents [15]. Various industries and human daily life have been changed with the advent of nanotechnology. Antimicrobial potential of nanoparticles has attracted the attention of researchers and industrialists, which nanoparticles can be used as an alternative to antibacterial agents and antibiotics, as it is predicted that bacteria cannot become resistant to nanoparticles because nanoparticles can be effective on different parts and various enzymes, which this issue is confirmed by research of Sondi et al. [16] in 2004 on the antimicrobial activity of silver nanoparticles. They argued that antimicrobial activity of silver is carried out by blocking the electron transport system, changing the bacterial membrane function and inhibit the DNA replication. Silver ions are known to particularly inhibit thiol group-containing enzymes and proteins and thereby this mechanism plays an essential role in the antimicrobial activity of silver ions, although other cellular factors such as hydrogen bonds may also be involved [16].
New Delhi metallo-beta-lactamase-1 (NDM-1) is an enzyme that makes bacteria resistant to a broad range of beta-lactam antibiotics.Most isolates with NDM-1 enzyme are resistant to all standard intravenous antibiotics for treatment of severe infections [17]. DNA gyrase enzyme in E.coli participates in several important processes, and thus is physiological target for a series of antibiotics. E.coli mutants resistant to the two classes of drugs have provided important evidences about subunit structure of the enzyme.
Drug resistance is controlled by two groups of genes (gyr A and gyr B) that are structural genes for subunits of the enzyme [18,19]. Therefore, the nanoparticles can be used for antimicrobial activities.But both advantages and disadvantages need to be considered in the host cell. Nanoparticles arenot toxic to cells in the body at low concentrations.In this study, certain concentrations of silver and copper oxide nanoparticles were used for bacterial treatment to find out antimicrobial properties. The results of the present study (Figure 1) showed that the nanoparticles with diameters less than 20nm in doses of 30 and 60μg/ml had relatively good antimicrobial effects so that were able to almost inhibit the growth of all bacteria in the samples.
According to numerous studies that have demonstrated the effect of silver and copper oxide nanoparticles as antimicrobial agents [16,20-25], the main purpose of this research was to evaluate and compare the effect of nanoparticles on bacterial genome at the lowest effective dose.Reports have been also presented based on the nanoparticles effects on bacterial genome which can induce DNA single-strand breakage and affect gene expression [26].
Li et al. [26] in 2012 during a study stated that silver nanoparticles are imported into bacterial cells and influence on the DNA twisting, thus inhibit the replication and cell proliferation. The silver nanoparticles are combined with the thiol groups in respiratory enzymes and inhibit respiration process in bacterial cells [27,28] or as expressed in previous studies, metal oxide nanoparticles may interfere with the transcription and translation [29].
This study was also conducted to investigate the effects of silver and copper oxide nanoparticles on the genome of Escherichia coli strain O157: H7 as a model for gram-negative bacteria. In this regard, based on the RAPD-PCR reaction with 14 primers, the presence or absence of bands in the gel images (Figure 3) suggest changing the DNA sequence by silver and copper oxide nanoparticles. A large number of primers failed to detect target sequences, and therefore the related segments were unable to replicate and we have seen the absence of bands on the agarose gel.
The difference among the bands observed in the treated and control groups of bacteria suggests that the target sequences of primers have been changes in the treated bacteria that make a difference in binding of primers and PCR amplification. The genomic sequence variations could possibly be in the process of replication.
It can be concluded that a change in the base pairing properties could be one of the possible causes of DNA sequence variations due to treatments of bacteria by silver and copper oxide nanoparticles which during the replication can lead to change the sequences in daughter strands. Also, silver nanoparticles could possibly cause dysfunction in DNA pol enzyme and are able to target the molecular mechanisms of replication accuracy which is involved in the synthesis of new strands based on the structure of Watson and Crick, changing the sequence of daughter strands [30].
Variations observed in DNA sequence in this study could also be a factor for growth inhibition and cell cycle through the occurrence of mutations, followed by gene expression changes associated with growth and cell cycle control [31]. Copper oxide and silver nanoparticles inside the cells can release ions of copper oxide [30,31] and silver, which react with DNA phosphorus and then disable the replication.
Silver ions increase the level of ROS, react with the sulfurcontaining proteins and inhibit the respiratory enzymes, resulting in cell death [31,32]. The energetic ions of copper cations with moving easily among the lipid layers are trapped by the cells which produce a specific reaction of oxygen, penetration of lipid peroxidation and protein oxidation.
Cell wall components are responsible for binding with copper nanoparticles. Amine and carboxyl groups of peptidoglycan participate in copper process and cell wall damages [33]. Accordingly, given that growing and replicating bacteria in the present research have been treated with silver and copper oxide nanoparticles, it could be argued that these nanoparticles most likely can create disruption in replication as well as in repair mechanisms to cause multiple mutations in DNA sequences.
In accordance with the results of (Figure 1) there are significant differences between treatment and control samples and in fact bacteria were separated into two distinct strains in terms of genome. Based on dendrogram, samples of control and treated with copper oxide nanoparticles by being in separate categories demonstrate great genetic distances. But the control and first treatment of silver nanoparticle were placed in single branch, and the second treatment was subjected to a separate main branch, reflecting the impact of nanoparticles on bacterial genome at high concentrations. Therefore, according to previous studies and the current research, it can be concluded that the nanoparticles can reduce the expression of genes involved in cell cycle control by creating mutations in their sequences, and thus reduce the growth of bacteria.
Conclusion
The results of this study and similar findings indicate the proper efficacy of nanoparticles as antibacterial compounds, but copper oxide nanoparticles compared with silver nanoparticles was more effective on E. coli genome as the model for gramnegative bacteria and since the copper nanoparticles are less expensive, thus is cost-effective as antibacterial agents. But because the nanoparticles can bind with DNA, so in the long term can make hard mutants and have adverse effects on eukaryotic host cells and it is impossible to be tested in a short time. But it is suggested to be further investigated in future works on the effects of nanoparticles in eukaryotic cells, which the nanoparticles can be used in various industries with greater certainty.
Acknowledgment
The authors hereby express their appreciation and gratitude to Biotechnology Department, University of Maragheh, Iran, due to sincere cooperation in providing primers and nanoparticles used in this study.
References
- Baker-Austin C, Wright MS, StepanauskasR, McArthurJV (2006) Coselection of antibiotic and metal Resistance. Trends Microbiol 14(4): 176-182.
- Gong P, Li H, He X, Wang K, Hu J, et al. (2007) Preparation and antibacterial activity of Fe3O4@Ag nanoparticles. J of Nanotechnology 18(28): 604-611.
- Yoon KY, Byeon JH, PARK JH, Hwang J (2007) Constants of Escherichia coli and Bacillus subtilis to silver and copper nanoparticles. Sci Total Environ 373(2-3): 572-575.
- Fahmy B, Cormier SA (2009) Copper oxide nanoparticles induce oxidative stress and cytotoxiy in airway epithelial cells. Toxicol In Vitro 23(7): 1365-1371.
- Morones JR, Elechiguerra JL, Camacho A, Holt K, Kouri JB, et al. (2005) The bactericidal effect of silver nanoparticles. Nanotechnology 16(10): 2346-2353.
- Aruoja V, Dubourguier HC, KasemetsK, Kahru A (2009) Toxicity of nano particles of CuO, ZnO and TiO2 to microalgae Pseudokirchneriella subcapitata. Sci Total Environ 407(4): 1461-1468.
- Heinlaan H, Ivask A, Blinova I, Dubourguier HC, Kahru A (2008) Toxicity of nano sized and bulk ZnO, CuO and TiO2 to bacteria Vibrio fischeri and crustaceans Daphnia magna and Thamnocephalus paltyurus. Chemosphere 71(7): 1308-1316.
- Ballinger PM, Brown BS, Griffin MM, Steven FS (1982) Evidence for carriage of silver by sulphadimidine: haemolysis of human erythrocytes. Br J Pharmacol 77(1): 141-145.
- Loke CN, Ho CM, Chen R, He QY, Yu WY, et al. (2007) Silver nanoparticles: partial oxidation and antibacterial activities. J Biol Inorg Chem 12(4): 527-534.
- Ahmadi F, Kordestany AH (2011) Investigation on silver retention in different organs and oxidative stress enzymes in male broiler fed diet supplemented with powder of nano silver. Amer Eurasian J Toxicol Sci 3(1): 28-35.
- Yoshimaru T, Suzuki Y, Inoue T, Niide O, Ra C (2006) Silver activates mast cells through reactive oxygen species production and a thiolsensitive store-independent Cainflux. Free Radic Biol Med 40(11): 1949-1959.
- Asharani PV, Wu YL, Gong Z, Valiyaveettil S (2008) Toxicity of silver nanoparticles in zebrafish models. Nanotechnology 19(25): 255102.
- Kim S, Choi JE, Choi J, Chung KH, Park K, et al. (2009) Oxidative stressdependent toxicity of silver nanoparticles in human hepatoma cells. Toxicol In vitro 23(6): 1076-1084.
- Brown AN, Smith K, Samuels TA, Lu J, Obare SO, et al. (2012) Nanoparticles functionalized with ampicillin destroy multipleantibiotic- resistant isolates of Pseudomonas aeruginosa and Enterobacteraerogenes and methicillin-resistant Staphylococcus aureus. Appl Environ Microbiol 78(8): 2768-2774.
- Huh AJ, Kwon YJ (2011) Nanoantibiotics: a new paradigm for treating infectious diseases using nanomaterials in the antibiotics resistant era. J Control Release 156(2): 128-145.
- Sondi I, Sondi SB (2004) Silver nanoparticles as antimicrobial agent: a case study on E. coli as a model for gram-negative bacteria. Colloid Interf Sci 275(1): 177-182.
- Rakow NA, Suslick KS (2000) A colorimetric sensor array for odour visualization. Nature 406(6797): 710-713.
- Pal S, Tak YK , Song JM (2007) Does the antibacterial activity of nanoparticles depend on the shape of the nanoparticles A study of the gram negative bacterium Escherichia coli. J of Appl Environ Microbiol 73(6): 1712-1720.
- Blecher K, Nasir A, Friedman A (2011) The growing role of nanotechnology in combating infectious disease. J of Virulence 2(5): 395-401.
- Hindi KM, DittoAJ, Panzner MJ, MedvetzDA, Han DS, et al. (2009) The antimicrobial efficacy of sustained release silver-carbene complexloaded L-tyrosine polyphosphate nanoparticles: characterization, in vitro and in vivo studies. Biomaterials 30(22): 3771-3779.
- Knetsch ML, Koole LH (2011) New strategies in the development of antimicrobial coat-ings: the example of increasing usage of silver and silver nanoparticles. Polymers 3(1): 340-366.
- Lara HH, Ayala Nunez NV, Turrent LDI, Padilla CR (2010) Bactericidal effect of silver nanoparticles against multidrug-resistant bacteria. World J Microbiol Biotechnol 26(4): 615-621.
- Ruparelia JP, Chatterjee AK, Duttagupta SP, Mukherji S (2008) Strain specificity in antimicrobial activity of silver and copper nanoparticles. Acta Biomater 4(3): 707-716.
- Chang Y, Zhang M, XiaL, ZhangJ, Xing G (2012) The Toxic Effects and Mechanisms of CuO and ZnO Nanoparticles. Materials 5(12): 2850- 2871.
- Shankar S, Jong Whan R (2014) Effect of copper salts and reducing agents on characteristics and antimicrobial activity of copper nanoparticles. Materials Letters 132: 307-311.
- Li WR, Xie XB, Shi QS, Duan SS, Ou-Yang YS, et al. (2011) Antibacterial effect of silver nanoparticles on Staphylo- coccusaureus. Biometals 24(1): 135-141.
- Klasen HJ (2000) A historical review of the use of silver in the treatment of burns. I. Early issues Burns 26(2): 117-130.
- Soenen SJ, Himmelreich U, Nuytten N, Pisanic TR, Ferrari A, et al. (2010) Intracellular nanoparticle coating stability determines nanoparticle diagnostics efficacy and cell functionality. Small 6(19): 2136-2145.
- Ruparelia JP, Chatterjee AK, Duttagupta SP, Mukherji S (2008) Strain specificity in antimicrobial activity of silver and copper nanoparticles. Acta Biomater 4(3): 707-716.
- Hoshino N, Kimura T, Yamaji A, Ando T (1999) Damage to the cytoplasmic embrane of Escherichia coli by catechin-copper (II) complexes. Free Radic Biol Med 27(11-12): 1245-1250.
- Macomber L, Rensing C, Imlay JA (2007) Intracellular copper does not catalyze the formation of oxidative DNA damage in Escherichia coli. J Bacteriol 189(5): 1616-1626.
- Matsumura Y, Yoshikata K, Kunisaki Si, Tsuchido T (2003) Mode of bactericidal action of silver zeolite and its comparison with that of silver nitrate. Appl Environ Microbiol 69(7): 4278-4281.
- Bogdanovića U, Lazićb V, Vodnik V (2014) Copper nanoparticles with high antimicrobial activity . J of Materials Letter 219: 558-567.






























