Study of Circulating Adiponectin Level in Children with Type 1 Diabetes Mellitus in Najran-KSA
Mohammed Ayed Huneif1, Amar B Elhussein2, Omar E Fadlelseed3, Walid G Babikr4, Njood M Alwadei5 and Elhashimi Eltayb Hassan6*
1Department of Pediatrics, Najran University, Saudi Arabia
2 Department of Biochemistry, Nile College for Medicine and Medical Sciences, Sudan
3Department of Biochemistry, Najran University, Saudi Arabia
4Department of Medicine, Najran University, Saudi Arabia
5 Department of Pediatric, King Khalid university, Saudi Arabia
6Department of Clinical Laboratory Sciences, Najran University, Saudi Arabia
Submission: September 28, 2018; Published: October 25, 2018
*Corresponding author: Elhashimi Eltayb Hassan, Assistant Professor of Clinical Chemistry, Department of Clinical Laboratory Sciences, College of Applied Medical Sciences, Najran University, Najran, Saudi Arabia.
How to cite this article: Md Ayed H, Amar B E, Omar E F, Walid G B, Njood M A et al. Study of Circulating Adiponectin Level in Children with Type 1 Diabetes Mellitus in Najran-KSA. Curre Res Diabetes & Obes J. 2018; 9(1): 555755. DOI: 10.19080/CRDOJ.2018.09.555755.
Abstract
Background: Adiponectin is a polypeptide hormone produced exclusively by adipocytes and is believed to play a vital role in the pathophysiology of diabetes, especially Type 1 Diabetes. This study aimed to determine the relationship between total plasma adiponectin concentrations and body mass index (BMI), fasting glucose and glycated hemoglobin levels among Saudi children with type 1 diabetes mellitus in Najran city -Najran region Southern KSA.
Methods: Two hundred and four Saudi children were enrolled in the study, 132 with type 1 diabetes mellitus were selected as a test group compared with a control group which included 72 apparently healthy volunteers, ages and sex in both groups were matched.
Results: The adiponectin level was evaluated with special regard to the control of diabetes, gender, duration of disease, body mass index (BMI). Results showed that the plasma adiponectin concentrations were significantly higher in the diabetic group when compared to the controls, results also revealed a significant negative correlation between the serum levels of Adiponectin ng/ml and age/ year, Duration / Year, FBG mg/dl & BMI Kg ̸ m2 in the diabetic group, while it revealed a significant positive correlation with HbA1c%.
Conclusion: Serum level of adiponectin in uncontrolled type 1 diabetic children was significantly higher than in healthy and good controlled type 1 DM children, it indicate that Adiponectin may play roles in diabetic control.
Keywords: Type 1 Diabetes mellitus; Adiponectin; Glycated hemoglobin
Abbrevations: T1 DM: Type 1 Diabetes Mellitus; BMI: Body Mass Index; HbA1c: Glycated Hemoglobin
Introduction
Diabetes mellitus is one of the most serious and widespread metabolic diseases today, affecting 10%-15% of the United States population. According to a report by the Saudi Arabian Ministry of Health, approximately 0.9 million people were diagnosed with diabetes in 1992, but this figure rose to 2.5 million people in 2010, representing a 2.7 time increase in the incidence rates in less than two decades [1]. Type 1 diabetes mellitus (T1DM) accounts for 5% of cases, affecting 11-22 million people worldwide, with 78000 new cases diagnosed each year, it is characterized by absolute deficiency of insulin resulting in severe hyperglycemia, and it is fatal if untreated [2-4].
Adiponectin is a 30kDa polpeptide hormone produced only by adipose tissues cells with anti- atherogenic, anti-inflammatory and insulin-sensitizing properties and glucose homeostasis [5] Low levels have been reliably connected with obesity and foresee the advancement of insulin resistance and type 2 diabetes mellitus (T2DM) [6,7]. The exact role of adiponectin in pathogenesis of type 1 diabetes is not fully clear. Some studies in children and most studies in adults have shown adiponectin to be higher in type 1 diabetes than in nondiabetic people and in those with type 2 diabetes [8-13]. Nonetheless, the low levels of adiponectin may likewise be identified with insulin resistance in type 1 diabetes [10,12,14-17]. Adiponectin has been shown to have anti-inflammatory and anti-thermogenic effects [18] including upgraded secretion of nitric oxide and vasodilation and inversion of the proinflammatory impacts of tumor necrosis factor-alpha (TNF-𝛼) on endothelial function [15]. It has been hypothesized that a compensatory system may lead adiponectin levels to react to inflammation and oxidative stress [11,14,16,19]. Other factors that may affect adiponectin include peripheral hyperinsulinemia accompanying subcutaneous insulin administration or the chronic hyperglycemic state of type 1 diabetes [5-7]. A reduced clearance of adiponectin may contribute to higher levels found in individuals with advanced kidney disease [6-8].
According to several studies, plasma concentration of adiponectin is reduced in human obesity, particularly visceral, and negatively correlated with insulin resistance (IR) [9,10]. Additionally, hypoadiponectinemia is independently associated with the metabolic syndrome (MS), surrogate marker of IR, and significantly related to T2DM development [10,11]. MS represents an important risk factor for mortality and micro- and macrovascular complications development both in patients with type 1 diabetes mellitus (T1DM) and T2DM [12]. Adiponectin was found to accumulate in damaged vascular walls and beneficially modulate the endothelial inflammatory response to vascular injury as it possesses anti-inflammatory and antiatherogenic properties [5]. Consequently, normal adiponectin concentrations or even induction of elevated concentrations are considered to be beneficial. However, increased adiponectin concentrations have been found to be associated with an increased cardiovascular mortality in T1DM [5,13], but in cross sectional data, the elevation in adiponectin levels has been hypothesized as a compensatory response in T1DM patients who have microvascular complications [5]. In addition, there are a growing number of patients with T1DM and MS who appear to be at increased risk of cardiovascular mortality and development of diabetes related complications, a greater need for higher insulin doses, and multifactorial intervention, thus, more aggressive treatment [6,14]. Therefore, the purpose of this study was to determine the relationship between total plasma adiponectin concentrations and body mass index (BMI), waist circumference, fasting glucose levels, and glycated hemoglobin.
Materials and Methods
This was an analytic, hospital based- case control study that was conducted in Najran University Hospital & King Khalid Hospital in Najran City- Southern Saudi Arabia from March 2015 to August 2017. The study samples comprised 132 Saudi children (76Male & 56 female) clinically diagnosed as type 1 diabetes mellitus as defined by The American Diabetes Association [15]; in contrast, 72 healthy volunteers (40 males, 32 females) were involved as a control group. Both groups were age and sex matched and their ages ranged between 2 and 14 years.
Inclusion criteria
Those with other types of diabetes mellitus, thyroid disease, renal disease, liver disease, anaemia and any medication that may be affect the Parameters under study were excluded from this study. An interview with a questionnaire to obtain the clinical data was done for each participant in this study, clinical history and examination of the test group and the controls were done by physicians working in Najran University Hospital & King Khalid Hospital. Weight was measured using electronic digital scales. Height was measured using a wall-mounted stadiometer. BMI was subsequently calculated as weight (kg) per height (m2).
In sterile condition and using a local antiseptic for skin following an overnight fasting (8-12hrs), 5mls of venous blood was collected from each participant and separated into 3mls as serum and 2mls as plasma in EDTA tubes. For serum preparation the blood samples were separated after complete clotting by centrifugation at 4000rpm for 5 minutes and serum was obtained. The sera were used to measure the concentrations of adiponectin using ELISA (Enzyme-Linked Immunosorbent Assay) from abcam-UK, FBG was measured using spectrophotometric method., HbA1c was measured from sample of EDTA tubes using chromatography technique (boronate affinity chromatography). Results of this study were statistically analyzed using statistical package for social science (SPSS) program. Independent t-test, ANOVA and Persons correlation coefficients were used; significance levels were set at (P<0.05).
Results
One hundred and thirty two Type 1 diabetic patients (76 males and 56 females) and seventy two matched controls (40 males and 32 females) were participated in this study, their ages ranged between 4 and 14 years (10.45±3.92 for diabetic group and 10.53±3.43 for control group). There were no significant differences between the two groups concerning ages & gender. Adiponectin (ng/ml) was 13.5±3.0 (mean±S.D.) for diabetic children while it was 8.5±2.8 for control group (p=0.001) , Fasting plasma glucose, FPG (mg/dl) was 196.86±56.32 (mean±S.D.) for diabetic children while it was 82.78±8.39 for control group ( p = 0.001) , HbA1c % was significantly higher in the diabetic group than that of the healthy control group (9.90±1.79 vs. 4.53±0.46, p=0.001) and BMI Kg ̸m2 was significantly decreased in the diabetic group than that of the healthy controls (17.5±1.8vs. 18.1±1.4, p=0.009), which were demonstrated by (Table 1).
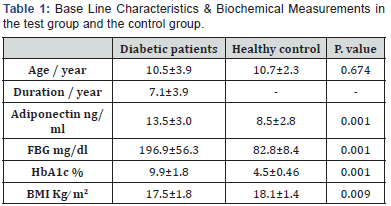
• The table shows the mean±SD.
• P value Less than 0.05 considered significant
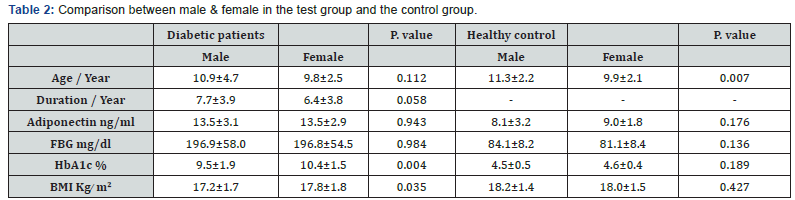
• The table shows the mean±SD.
• P value Less than 0.05 considered significant.
The study showed insignificant differences between male & female regarding the means of Duration of disease/Year, Adiponectin ng/ml & FBG mg/dl in the diabetic group when compared to the controls (p>0.05) while the means of HbA1c% and BMI Kg ̸ m2 shows significant increase in female compared to male in diabetic group (p<0.05) (Table 2).
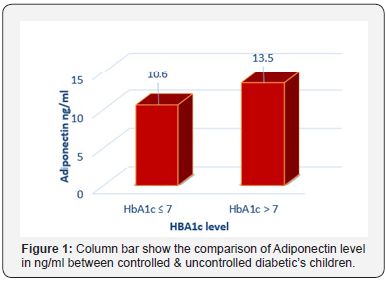
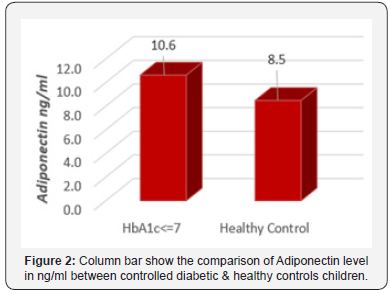
Our results showed significant increase in the mean of Adiponectin ng/ml in uncontrolled DM when compared with controlled DM (p<0.01) (Figure 1) and in controlled diabetic when compared to healthy controls children (p<0.01) (Figure 2).
Regarding Pearson correlation coefficients the results revealed a significant negative correlation between the serum levels of Adiponectin ng/ml and age/ year, Duration / Year, FBG mg/dl & BMI Kg ̸m2 in the diabetic group, while it revealed a significant positive correlation with HbA1c% (Table 3).
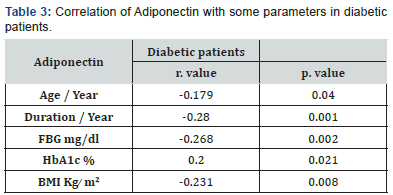
• P value Less than 0.05 considered significant.
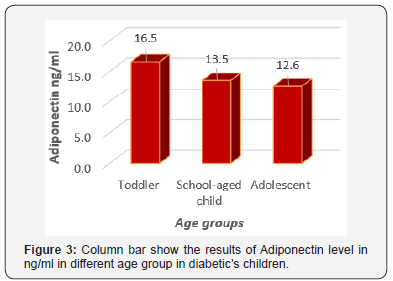
Concerning the age groups, results showed that the Adiponectin values were significantly different among the study groups (P=0.003) with the highest values were for Toddlers (16.54±2.92) while School –aged children were (13.52±3.13), and the lowest values were for Adolescent group (12.60±2.19) (Figure 3).
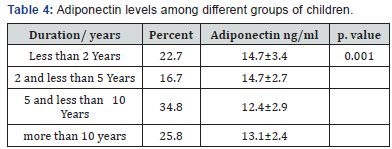
(Table 4) shows the mean and SD of Adiponectin among the four studied groups of children regarding to the duration of DM. The four groups differed significantly from each other (P = 0.001). The highest values were for children with duration Less than 2 Years while the lowest values were for children with duration between 5 years and less than 10 Years.
Discussion
Anti-inflammatory and anti-atherosclerotic effects of adiponectin have been suggested that had a role in protection against development of atherosclerosis [7]. Our study showed that, the mean adiponectin levels were considerably higher in the diabetic patients than the controls. The same results were obtained by Barnes et al. and Ljubicet et al. they found that adiponectin levels were considerably higher in diabetics than in controls [15,16]. Also Furuta et al. found that adiponectin levels exaggerated in conjunction with β-cells malfunction [17]. Researchers assumed that the increase in adiponectin levels is mediated by insulin, the accurate mechanism of insulin activate the gene of adiponectin expression, production and secretion is heretofore unknown [18]. In study that carried out in vitro showed that the adiponectin secretion was stimulated by insulin via phosphatidylinositol 3-kinase pathway [19] this signaling pathway stimulate both production and secretion of adiponectin hormone [20]. Several studies showed that higher serum adiponectin levels was reported in T1DM patients, specially patients surfing from diabetes complication like vascular complications or nephropathy [21]. The increased adiponectin concentration in patients with T1DM can interpreted as a response to posttranslational modifications (glycosylation) that exist in T1DM patients [22], decreased excretion due to renal failure [23], vascular injury [5], and impact of subcutaneous insulin therapy [24]. Also, possibly the fat distribution between diabetic patients and healthy control subjects and reduction of suppression effect by insulin [25]. Adiponectin perform a fundamental role in metabolism of carbohydrates and fats by its effect on insulin sensitivity by activating peroxisome proliferator-activated receptor- γ and the AMP-activated protein kinase [26].
The serum adiponectin level in poorly-controlled subgroup was much greater than in controlled diabetics subgroup and healthy control group, there was a significant difference regarding serum adiponectin level between controlled diabetics subgroup and healthy control children. Many previous studies found that the level of adiponectin was high in type 1 DM patients [27-29]. The collagenous domain of the adiponectin molecule has four conserved lysines. Glycosylation of these molecules is one of the major posttranslation modifications of adiponectin. In diabetic patients with constant hyperglycemia, the glycosylation process is altered, and this could lead to an altered adiponectin function. Consequently, a modified adiponectin molecule could lead to diminished negative feedback, and thus to increased adiponectin concentrations in diabetics [30].
There were no significant differences between male & female in diabetic group and healthy controls regarding adiponectin level and fasting blood glucose, this results was in harmonization with Diwan et al. which found that there was no significant difference between the two groups with respect to gender, but the level of serum adiponectin in males was lower compared diabetic females [31], this slight differences could be interpreted by the action of testosterone hormone [32]. Saltevo et al. found that the level of adiponectin in females was low compared to males in prediabetics and type 2 diabetic patients [33]. HbA1c % and BMI Kg ̸ m2 were significantly higher in females than in males in diabetic group, while there were no differences between male and female in healthy control group.
Our results showed negative correlation between the serum levels of Adiponectin ng/ml and duration of disease/ year, this finding disagrees with the finding of Lindström et al. who reported that the increase in circulating adiponectin concentrations in patients with type 1 diabetes appears to be strongly associated with long diabetes duration [34], however, in our study group the majorities of patients with long duration of diabetes are well- controlled patients. Our data showed that there was an inverse correlation between the level of serum adiponectin and BMI in type 1 diabetic children, many studies showed the same finding [31,35,36]. Decreased adiponectin levels was linked to Obesity after gender, age and diabetes were adjusted when compared to none obese patients [31,37], serum adiponectin level increases with weight reduction in both diabetic and healthy control individuals [38]. Researchers found that convers relation between the number of positive islet cell autoantibodies and body mass index, which explain that the obesity associated with insulin resistance lead to speed up the clinical presentation of TDM at an early phase of β-cell destruction [39].
Conclusion
The serum level of adiponectin in uncontrolled type 1 diabetic children was significantly higher than in healthy and good controlled type 1 DM children, indicates that the serum level of adiponectin hormone has a direct association with HbA1c in children with type 1 diabetes.
References
- Alotaibi A, Perry L, Gholizadeh L, Al-Ganmi A (2017) Incidence and prevalence rates of diabetes mellitus in Saudi Arabia: An overview. J Epidemiol Glob Health 7(4): 211-218.
- Friedman AL, Friedman EA (2002) Pancreas transplantation for type 2 diabetes at US transplant centers. Diabetes Care 25(10): 1896-1897.
- Gruessner RW, Gruessner AC (2013) The current state of pancreas transplantation. Nat Rev Endocrinol 9(9): 555-563.
- Margreiter C, Resch T, Oberhuber R, Aigner F, Maier H, et al. (2013) Combined pancreas-kidney transplantation for patients with endstage nephropathy caused by type-2 diabetes mellitus. Transplantation 95(8): 1030-1036.
- Frystyk J, Tarnow L, Hansen TK, Parving H-H, Flyvbjerg A (2005) Increased serum adiponectin levels in type 1 diabetic patients with microvascular complications. Diabetologia 48(9): 1911-1918.
- Ghosh S, Collier A, Hair M, Malik I, Elhadd T (2010) Metabolic syndrome in type 1 diabetes. International Journal of Diabetes Mellitus 2(1): 38- 42.
- Okamoto Y, Arita Y, Nishida M, Muraguchi M, Ouchi N, et al. (2000) An adipocyte-derived plasma protein, adiponectin, adheres to injured vascular walls. Horm Metab Res 32(02): 47-50.
- Report of the Expert Committee on the Diagnosis and Classification of Diabetes Mellitus (1997) Diabetes Care 20(7): 1183-1197.
- Arita Y, Kihara S, Ouchi N, Takahashi M, Maeda K, et al. (1999) Paradoxical decrease of an adipose-specific protein, adiponectin, in obesity. Biochem Biophys Res Commun 257(1): 79-83.
- Yatagai T, Nagasaka S, Taniguchi A, Fukushima M, Nakamura T, et al. (2003) Hypoadiponectinemia is associated with visceral fat accumulation and insulin resistance in Japanese men with type 2 diabetes mellitus. Metabolism 52(10): 1274-1278.
- Daimon M, Oizumi T, Saitoh T, Kameda W, Hirata A, et al. (2003) Decreased serum levels of adiponectin are a risk factor for the progression to type 2 diabetes in the Japanese Population: the Funagata study. Diabetes care 26(7): 2015-2020.
- Isomaa B, Almgren P, Tuomi T, Forsén B, Lahti K, et al. (2001) Cardiovascular morbidity and mortality associated with the metabolic syndrome. Diabetes care 24(4): 683-689.
- Kistorp C, Faber J, Galatius S, Gustafsson F, Frystyk J, et al. (2005) Plasma adiponectin, body mass index, and mortality in patients with chronic heart failure. Circulation 112(12): 1756-1762.
- Orchard TJ, Olson JC, Erbey JR, Williams K, Forrest KYZ, et al. (2003) Insulin resistance–related factors, but not glycemia, predict coronary artery disease in type 1 diabetes: 10-year follow-up data from the Pittsburgh Epidemiology of Diabetes Complications study. Diabetes care 26(5): 1374-1379.
- Barnes M, Curran‐Everett D, Hamman R, Maahs D, Mayer‐Davis E, et al. (2008) Determinants of adiponectin levels in young people with Type 1 diabetes. Diabetic Medicine 25(3): 365-369.
- Ljubic S, Boras J, Jazbec A, Lovrencic MV, Vidjak V, et al. (2009) Adiponectin has different mechanisms in type 1 and type 2 diabetes with C-peptide link. Clin Invest Med 32(4): 271-279.
- Furuta M, Tamai M, Hanabusa T, Yamamoto Y, Nanjo K, et al. (2006) Serum adiponectin is associated with fasting serum C-peptide in nonobese diabetic patients. Diabetes Res Clin Pract 72(3): 302-307.
- Hecht Baldauff NTH, Dong W, Arena VC, Gurtunca N, Pietropaolo M, et al. (2016) Relationship of adiponectin and leptin with autoimmunity in children with new-onset type 1 diabetes: a pilot study. Pediatr Diabetes 17(4): 249-256.
- Pereira RI, Draznin B (2005) Inhibition of the phosphatidylinositol 3’-kinase signaling pathway leads to decreased insulin-stimulated adiponectin secretion from 3 T3-L1 adipocytes. Metabolism 54(12): 1636-1643.
- Blumer RM, van Roomen CP, Meijer AJ, Houben Weerts JH, Sauerwein HP (2008) Regulation of adiponectin secretion by insulin and amino acids in 3 T3-L1 adipocytes. Metabolism 57(12): 1655-1662.
- Gokulakrishnan K, Aravindhan V, Amutha A, Abhijit S, Ranjani H, et al. (2013) Serum adiponectin helps to differentiate type 1 and type 2 diabetes among young Asian Indians. Diabetes Technol Ther 15(8): 695-701
- Richards AA, Stephens T, Charlton HK, Jones A, Macdonald GA, et al. (2006) Adiponectin multimerization is dependent on conserved lysines in the collagenous domain: evidence for regulation of multimerization by alterations in posttranslational modifications. Mol Endocrinol 20(7): 1673-1687
- Jorsal A, Tarnow L, Frystyk J, Lajer M, Flyvbjerg A, et al. (2008) Serum adiponectin predicts all-cause mortality and end stage renal disease in patients with type I diabetes and diabetic nephropathy. Kidney Int 74(5): 649-654.
- Havel PJ (2004) Update on adipocyte hormones: regulation of energy balance and carbohydrate/lipid metabolism. Diabetes 53(Suppl 1): S143-S151.
- Fasshauer M, Klein J, Neumann S, Eszlinger M, Paschke R (2002) Hormonal regulation of adiponectin gene expression in 3T3-L1 adipocytes. Biochem Biophys Res Commun 290(3): 1084-1089.
- Gable D, Hurel S, Humphries S (2006) Adiponectin and its gene variants as risk factors for insulin resistance, the metabolic syndrome and cardiovascular disease. Atherosclerosis 188(2): 231-244.
- Karamifar H, Habibian N, Amirhakimi G, Karamizadeh Z, Alipour A (2013) Adiponectin is a good marker for metabolic state among type 1 diabetes mellitus patients. Iran J Pediatr 23(3): 295.
- Maahs DM, Hamman RF, D’agostino R, Dolan LM, Imperatore G, et al. (2009) The association between adiponectin/leptin ratio and diabetes type: the SEARCH for Diabetes in Youth Study. J Pediatr 155(1):133- 135.
- Meral C, Tascilar E, Karademir F, Tanju IA, Cekmez F, et al. (2010) Elevated plasma levels of apelin in children with type 1 diabetes mellitus. J Pediatr Endocrinol Metab 23(5): 497-502.
- Habeeb NMM, Youssef OI, Saab AAR, El Hadidi ES (2012) Adiponectin as a marker of complications in type I diabetes. Indian Pediatr 49(4): 277-280.
- Diwan AG, Kuvalekar AA, Dharamsi S, Vora AM, Nikam VA, et al. (2018) Correlation of serum adiponectin and leptin levels in obesity and Type 2 diabetes mellitus. Indian J Endocrinol Metab 22(1): 93.
- Nishizawa H, Shimomura I, Kishida K, Maeda N, Kuriyama H, et al. (2002) Androgens decrease plasma adiponectin, an insulin-sensitizing adipocyte-derived protein. Diabetes 51(9): 2734-2741.
- Saltevo J, Kautiainen H, Vanhala M (2009) Gender differences in adiponectin and low-grade inflammation among individuals with normal glucose tolerance, prediabetes, and type 2 diabetes. Gend Med 6(3): 463-470.
- Lindström T, Frystyk J, Hedman CA, Flyvbjerg A, Arnqvist HJ (2006) Elevated circulating adiponectin in type 1 diabetes is associated with long diabetes duration. Clin Endocrinol 65(6): 776-782.
- Blaslov K, Bulum T, Zibar K, Duvnjak L (2013) Relationship between adiponectin level, insulin sensitivity, and metabolic syndrome in type 1 diabetic patients. International journal of endocrinology.
- LeCaire TJ, Palta M (2015) Longitudinal analysis of adiponectin through 20-year type 1 diabetes duration. Journal of diabetes research.
- Yang WS, Lee WJ, Funahashi T, Tanaka S, Matsuzawa Y, et al. (2002) Plasma adiponectin levels in overweight and obese Asians. Obes Res 10(11): 1104-1110. 38. Hotta K, Funahashi T,
- Hotta K, Funahashi T, Arita Y, Takahashi M, Matsuda M, et al. (2000) Plasma concentrations of a novel, adipose-specific protein, adiponectin, in type 2 diabetic patients. Arterioscler Thromb Vasc Biol 20(6): 1595- 1599.
- Libman IM, Pietropaolo M, Arslanian SA, LaPorte RE, Becker DJ (2003) Evidence for heterogeneous pathogenesis of insulin-treated diabetes in black and white children. Diabetes Care 26(10): 2876-2882






























