The Gut: A Key to the Pathogenesis of Type 2-Diabetes?
Holst JJ*, Pedersen J, Wewer Albrechtsen NJ and Knop Fk
Department of Biomedical Sciences, University of Copenhagen, Denmark
Submission: February 6, 2017; Published: March 23, 2017
*Corresponding author: Holst JJ, Department of Biomedical Sciences, Center for Diabetes Research, Gentofte Hospital, University of Copenhagen NNF Center for Basal Metabolic Research, Denmark, Tel: +45-28757518; Email: jjholst@sund.ku.dk
How to cite this article: Holst J, Pedersen J, Wewer A N , Knop F. The Gut: A Key to the Pathogenesis of Type 2-Diabetes?. Curre Res Diabetes & Obes J 2017; 1(4): 555570. DOI: 10.19080/CRDOJ.2017.1.555570
Mini Review
The gastrointestinal tract plays a predominant role in the regulation of the postprandial plasma glucose levels [1]. The first regulating factor is the gastric antral motility which, by incompletely elucidated neuroendocrine mechanisms, regulates the rate of emptying and hence exposure of chyme to the small intestine. Thus, it is a main function of the stomach to receive and retain the incoming meal, and eventually to expel the triturated, emulsified and partly digested contents, the chyme, at a slow and surprisingly constant at a rate of maximally 4Kcal per minute. The duration of gastric emptying thereby becomes regulated by the energy content of the ingested meal [2]. The exposure rate of the small intestine is similarly limited and relatively constant. If the regulatory mechanisms are interfered with [for instance with surgical procedures like pyloroplasty or gastric bypass) and the normal emptying rate is exceeded, a so-called "dumping syndrome" may be elicited.
This syndrome which includes nausea, malaise, desire to lie down, maybe fainting, is caused by the increased osmotic load presented to the small intestine which shifts fluid from the circulation to the intestinal lumen via the leaky epithelium in the proximal gut. The more or less constant emptying rate also means that the secretion and thereby the plasma concentration of some of the gut hormones, e.g. the insulin-stimulating hormone, glucose-dependent insulin tropic polypeptide [GIPJ which is dependent on the intestinal absorption rate of nutrients, quickly rises to a certain elevated level which is then maintained as long as there is emptying from the stomach, depending on the total amount of stimulatory nutrients that were present in the meal [and are being retained in the stomach) [2].
It is possible that hydrogen ions in the duodenum [released from the stomach) play a role in the regulation of the emptying rate, but nutrients in the duodenum may also inhibit the emptying rate by neuroendocrine mechanisms. The hormone, GIP, has little effect on gastric emptying, whereas the other incretin hormone, glucagon-like peptide-1 [GLP-1), the secretion of which follows a similar pattern, powerfully inhibits gastric emptying. There is no doubt that the regulation of gastric emptying plays a major role for the postprandial glucose responses, again as illustrated in conditions of accelerated emptying rates, where particularly the early postprandial glucose excursions may be dramatically increased [3]. The increased secretion of GIP and GLP-1 powerfully amplifies the glucose-induced insulin secretion, and thereby also the ability of the organism to limit and restore to normal the increased glucose concentrations-indeed postprandial reactive hypoglycemia [sometimes designated "late dumping") may ensue, and is relatively often observed in individuals with accelerated gastric emptying [4].
The amplifying effect of the gut hormones on the glucose- induced insulin secretion is designated the incretin effect, and this is responsible for the proportional increase in insulin secretion according to the ingested amount of glucose [and other meal components). The effects is small after small meals [small amount of glucose), but increases so that it may be responsible for up to 80% of the elimination of glucose from the circulation after larger meals [corresponding to 100g of glucose) [2,5]. By means of the regulation of gastric emptying and the incretin effect, it is ensured that post prandial glucose excursions are normally moderate and in fact independent of the ingested amount of calories [glucose) [1].
It is likely that these mechanisms are of major importance to prevent complications elicited by hyperglycemia. As mentioned these powerful mechanisms may also lead to severe postprandial reactive hypoglycemia [6]. The question then arises whether the same mechanisms play a role in the development of type 2diabetes mellitus [T2DM)? The gastric emptying rates in patients with T2DM show great variability and a consistent abnormality do not seem to prevail (although abnormalities may very well exist in selected groups).
The early response (0-60min) to ingestion of a mixed meal of the two hormones, GIP and GLP-1, does not seem to be importantly affected in patients with T2D, but GLP-1 secretion is more consistently impaired in the later phase (from 60 minutes and onwards) [7] and this may play a significant role. A similar impairment (and a similar impairment of the incretin effect, see below) is seen in obesity [8,9]. Probably it is of greater importance that the insulin tropic action of both hormones is also impaired. During the progressive development of glucose in tolerance, a gradual loss of the incretin effect, as well as the insulin tropic action of the two hormones are observed [10] and their effect is almost completely lost in full-blown diabetes as far as physiological amounts of the hormones are concerned [11].
However, in contrast to the almost complete loss of insulin tropic action of GIP, regardless of dose, suprahysiological concentrations of GLP-1 may still have considerable effect [12]. Evidently, the loss of the incretin effect is of major importance for the postprandial hyperglycemia of the patients with T2D and restoration of the incretin effect with GLP-1-receptoragonists provides part of the explanation for the antidiabetic effects of these agents. The GLP-1receptoragonistsalso powerfully inhibits the gastric emptying rate, at least acutely and this explains part of the effect on postprandial glycaemia of the short-acting GLP- 1-receptor agonists (eventide and lixisenatide).
Regarding the long-acting GLP-1-receptoragonists, tachyphylaxis rapidly develops regarding their effects on gastric emptying, while their incretin effect is preserved and when during meal intake the plasma glucose concentration rises, they will exert their incretin action, which at the cellular level in essence consists of potentiating of the glucose-induced insulin secretion [13,14]. There is, however, an additional factor which plays a rather important role: patients with T2DM typically have increased plasma concentrations of glucagon, both in the fasting state and postprandial [7].
There is a general misconception that meal intake should lead to inhibition of glucagon secretion; on the contrary, most meals, in particular protein rich meals, will stimulate glucagon secretion, but for patients with T2DM the increase is even larger Because glucagon stimulates the hepatic glucose production, this rise has considerable consequences for the postprandial glucose excursions, as illustrated in experiments with glucagon receptor antagonists, which effective lower both fasting and postprandial glucose levels [15]. But what is the explanation for the elevated postprandial levels of glucagon in the patients with T2DM? Several mechanisms have been proposed and they may all contribute. First of all, there are probably signals from the GI-tract.
Several hormones might be of importance, including GIP and GLP-2, which stimulate glucagon secretion and GLP-1, which inhibits secretion. Their combined influence was investigated in a systematic study where T2D patients received intravenous infusions of each of these hormones (mimicking their postprandial plasma profiles) superimposed on an intravenous glucose infusion mimicking the concentration curves resulting from an OGTT [16]. Individually, it turned out, each of the hormones had the mentioned, expected effects, but infused together the resulting glucagon secretion profile was very similar to that observed after oral glucose alone. Particularly GIP stimulated glucagon secretion. An increased activity of GIP might therefore contribute to the postprandial hyperglucagonemia in T2DM. This together with the loss of its insulin tropic effect in T2DM might therefore suggest that GIP actually has diabetogenic effects [17].
However, it is also possible that the postprandial hyperglucagonemia is derived not from the pancreas but from the GI-tract where some of the endocrine cells, under certain circumstances, which may include T2DM, may be able to produce glucagon. This is clearly seen in individuals after total pancreatectomies, who exhibit a large postprandial glucagon response after oral glucose [18]. It can actually be demonstrated that the response has important effects for hepatic glucose production.
Extra pancreatic glucagon may therefore contribute to the diabetic hyperglucagonemia. Furthermore, there is strong evidence from recent research that circulating amino acids play a predominant role in the regulation of glucagon secretion, and in view of this, it should always be suspected that altered amino acid levels underlie clinical hyperglucagonemia. The reverse also seems to be true: that the plasma level of glucagon plays a predominant role in the regulation of the plasma levels of the amino acids; thus conditions with hyper- and hypoglucagonaemiaare associated with low and high levels of plasma amino acids, respectively [19]. Indeed, amino acid metabolism and the glucagon-producing alpha cells in the pancreas seem to be coupled in a close, negative feed-back loop. Most recently we examined patients with varying degrees of nonalcoholic fatty lived disease and found a correlation between on one hand liver function and amino acid levels and on the other hand the plasma glucagon levels [20].
The hypothesis is that the steatosis impairs liver function which also comprises an impairment of the effects of glucagon on amino acid metabolism and urea genesis. As a consequence, plasma levels of amino acids rise, which in turn results in hyperglucagonemia. Because glucagon also influences glucose metabolism and hepatic glucose production, this will also result in glucose intolerance and increased postprandial glucose excursions. A lot of work still has to be done with respect to unravelling these associations but the recent application of glucagon receptor antagonists and the use of transgenic animals have greatly facilitated these studies in both people and experimental animals Figure 1 [21].
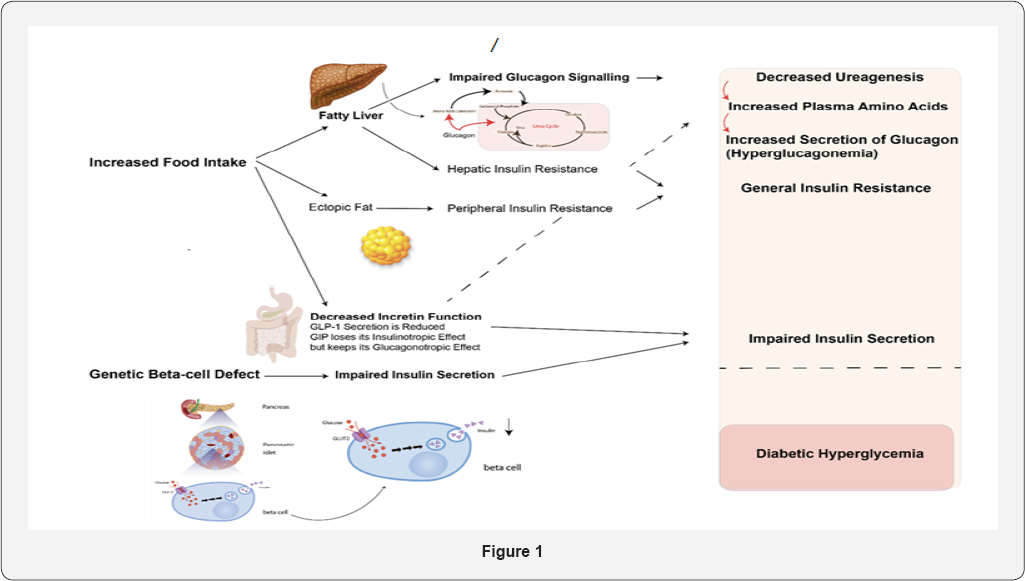
One final factor deserves discussion: there is actually a fasting level of the hormoneGLP-1, and it can be demonstrated, also in humans, that blocking of the GLP-1 receptor with a GLP- 1 receptor antagonist [extending 9-39) results in increased glucagon secretion, decreased insulin secretion and presumably as a consequence of this, glucose intolerance and hyperglycemia in the fasting state [22]. As already mentioned, the GLP-1 secretion is often impaired in T2DM, perhaps mainly related to simultaneously occurring obesity [8]. Because the pathogenesis of T2DM also is associated with the development of obesity, a picture emerges where decreased GLP-1secretion leads to both an impaired incretin effect and increased glucagon secretion, both with diabetogenic actions. As hepatic steatosis develops because of increased calorie intake, the liver's sensitivity to both insulin and to the actions of glucagon on amino acid and glucose? metabolism will be impaired. Furthermore, parallel development of insulin resistance [because of ectopic fat) in the skeletal muscles results in impaired peripheral glucose uptake. Together, all of these result in a vicious diabetogenic cycle.
References
- Holst JJ, Gribble F, Horowitz M, Rayner CK [2016) Roles of the Gut in Glucose Homeostasis. Diabetes Care 39[6): 884-892.
- Bagger JI, Knop FK, Lund A, Vestergaard H, Holst JJ, et al. [2011) Impaired regulation of the incretin effect in patients with type 2 diabetes. J Clin Endocrinol Metab 96[3): 737-745.
- Jacobsen SH, Bojsen MKN, Dirksen C, Jorgenson NB, Clousen TR, et al. [2013) Effects of gastric bypass surgery on glucose absorption and metabolism during a mixed meal in glucose-tolerant individuals. Diabetologia 56[10): 2250-2254.
- Holst JJ, Knop FK, Vilsboll T, Krarup T, Madsbad S [2011) Loss of incretin effect is a specific, important, and early characteristic of type 2 diabetes. Diabetes Care 34[Suppl 2): S251-S257.
- Nauck MA, Homberger E, Siegel, Allen RC, Eaton RP, et al. [1986] Incretin effects of increasing glucose loads in man calculated from venous insulin and C-peptide responses. J Clin Endocrinol Metab 63[2): 492-498.
- Craig CM, Liu LF, Deacon CF, Holst JJ, McLaughlin TL [2016) Critical role for GLP-1 in symptomatic post-bariatric hypoglycaemia. Diabetologia 60[3): 531-540.
- Toft-Nielsen MB, Damholt MB, Madsbad S, Hilsted LM, Michelsen BK, et al. Determinants of the impaired secretion of glucagon-like peptide-1 [GLP-1) in type 2 diabetic patients. J Clin Endocrinol Metab 86[8): 3717-3723.
- Matikainen N, Bogl LH, Hakkarainen A, Lundbom J, Lundbom N, et al. [2014) GLP-1 responses are heritable and blunted in acquired obesity with high liver fat and insulin resistance. Diabetes Care 37[1): 242251.
- Faerch K, Torekov SS, Vistisen D, Jhonsen NB, Witte DR, et al. [2015) GLP-1 Response to Oral Glucose Is Reduced in Prediabetes, Screen- Detected Type 2 Diabetes, and Obesity and Influenced by Sex: The ADDITION-PRO Study. Diabetes 64[7): 2513-2525.
- Eriksen M, Jensen DH, Tribler S, Holst JJ, Madsbad S, et al. [2015) Reduction of insulinotropic properties of GLP-1 and GIP after glucocorticoid-induced insulin resistance. Diabetologia 58[5): 920928.
- Hojberg PV, Zander M, Vilsboll T, Knop k, karup T, et al. [2008) Near normalisation of blood glucose improves the potentiating effect of GLP-1 on glucose-induced insulin secretion in patients with type 2 diabetes. Diabetologia 51[4): 632-640.
- Vilsboll T, Krarup T, Madsbad S, Holst JJ [2002) Defective amplification of the late phase insulin response to glucose by GIP in obese Type II diabetic patients. Diabetologia 45[8): 1111-1119.?
- Holst JJ (2007) The Physiology of Glucagon-like Peptide 1. Physiol Rev 87(4): 1409-1439.
- Holst JJ, Deacon CF (2013) Is there a place for incretin therapies in obesity and prediabetes? Trends Endocrinol Metab 24(3): 145-152.
- Kazda CM, Ding Y, Kelly RP, Garyan P, Shi C, et al. (2016) Evaluation of Efficacy and Safety of the Glucagon Receptor Antagonist LY2409021 in Patients with Type 2 Diabetes: 12- and 24-Week Phase 2 Studies. Diabetes Care 39(7): 1241-1249.
- Lund A, Vilsboll T, Bagger JI, Holst JJ, Knop FK (2011) The separate and combined impact of the intestinal hormones, GIP, GLP-1 and GLP-2, on glucagon secretion in type 2 diabetes. Am J Physiol Endocrinol Metab 300(6): E1038-E1046.
- Chia CW, Carlson OD, Kim W, Shin Yk, Charles CP, et al. (2009) Exogenous glucose-dependent insulinotropic polypeptide worsens post prandial hyperglycemia in type 2 diabetes. Diabetes 58(6): 1342-1349.
- Lund A, Bagger JI, Wewer Albrechtsen NJ, Christensen M, Grondahl M, et al. (2016) Evidence of Extrapancreatic Glucagon Secretion in Man. Diabetes 65(3): 585-597.
- Holst JJ, Pedersen J, Wewer Albrechtsen NJ, Knop FK (2017) Glucagon and amino acids are linked in a mutual feed-back cycle: the liver-alpha cell axis. Diabetes 66(2): 235-240.
- Junker AE, Gluud L, Holst JJ, Knop FK, Vilsboll T (2016) Diabetic and nondiabetic patients with nonalcoholic fatty liver disease have an impaired incretin effect and fasting hyperglucagonaemia. J Intern Med 279(5): 485-493.
- Bagger JI, Knop FK, Holst JJ, Vilsboll T (2011) Glucagon receptor antagonism for the treatment of type 2 diabetes. Diabetes Obes Metab 13(11): 965-971.
- Edwards CM, Todd JF, Mahmoudi M, Wang Z, Wang RM, et al. (1999) Glucagon-like peptide 1 has a physiological role in the control of postprandial glucose in humans: studies with the antagonist exendin 9- 39 [In Process Citation]. Diabetes 48(1): 86-93.






























