The Endophytism of Bacillus thuringiensis in Cotton Plants at Acquisition and Oviposition by Bemisia tabaci
Flávia Santana S da Costa1*, Marcelo Tavares de Castro2 and Rose Monnerat3*
1 University of Brasília, Brazil
2 University Center ICESP, Brazil
3 Empresa Brasileira de Pesquisa Agropecuária (Embrapa) – Embrapa Genetic Resources and Biotechnology (Embrapa Cenargen), Brazil
Submission: November 12, 2021; Published: November 19, 2021
*Corresponding author: Flávia Santana S. da Costa, University of Brasília, University Campus Darcy Ribeiro - CEP 70910-900 Brasília, Brazil - E-mail: flaviasantana_agr@hotmail.com
Rose Monnerat, Empresa Brasileira de Pesquisa Agropecuária (Embrapa) - Embrapa Genetic Resources and Biotechnology (Embrapa Cenargen), Parque Estação Biológica, PqEB, Av. W5 Norte (final). Caixa Postal 03372, CEP 70770-917 Brasília, Brazil - E-mail: rose.monnerat@embrapa.br
How to cite this article: Flávia S S d C, Marcelo T de C, Rose M. The Endophytism of Bacillus thuringiensis in Cotton Plants at Acquisition and Oviposition by Bemisia tabaci. Agri Res & Tech: Open Access J. 2021; 26 (3): 556340. DOI: 10.19080/ARTOAJ.2021.26.556340
Abstract
Beneficial microorganisms can be applied on the soil or seeds and can protect plants against insect pests or pathogens. It is known which Bacillus thuringiensis (Bt) strains that, when introduced into the soil can be absorbed by the plant and translocate throughout this with insecticidal activity against insect pests that feed on them. For this reason, the ability of Bemisia tabaci ‘B biotype’ (Hemiptera: Aleyrodidae) to ingest Bt sap was evaluated in cotton plants inoculated with the Bt strain, which encodes the GFP gene, Btk-gfp (B. thuringiensis subsp. kurstaki - green fluorescent protein), and tested for oviposition, comparing untreated plants to those treated with Bt strains. The recovery of Btk-gfp was observed in the plant along with its ability to be acquired by nymphs and adults of B. tabaci. Plants treated with Bt strain S1806 had a lower number of B. tabaci eggs than plants that received only sterile water.
Keywords: Gossypium hirsutum; Antixenosis; Endophytic bacteria; Biological control; Agricultural entomology; Bioassay
Abbreviations: Embrapa: Empresa Brasileira de Pesquisa Agropecuária; Embrapa Cenargen – Embrapa Genetic Resources and Biotechnology; Bt: Bacillus thuringiensis; Btk-gfp: B. thuringiensis subsp. kurstaki: green fluorescent protein; GFP: Green Fluorescent Protein; B. tabaci: Bemisia tabaci; AbMV: Abutilon Mosaic Virus; P. xylostella: Plutella xylostella; UV: ultraviolet; ISLA: Brazil seed production company; BRS 8H: variety of cotton plants developed at Embrapa; rpm: revolution por minute; M: meters; μL: microliter; μg: microgram; nm: nanometer; h: hours; mL: milliliter; °C: degrees Celsius; ANOVA: Analysis of variance; F: test in ANOVA; p: value statistical significance
Introduction
The cotton plant (Gossypium hirsutum L., Malvaceae) production in Brazil attracts and hosts a significant complex of pests that attack the roots, stem, leaves, flower buds, fruits, and bolls [1]. Pests can cause significant yield losses in addition to raising concerns about sustainability of the productive system. Pest control in cotton is expensive and primarily done with broad-spectrum synthetic chemical insecticides [2]. To meet the growing demand for fibers, the crop must be protected from pests while still preserving natural resources, maintaining environmental quality [3] and ensuring human health. Thus, research continues to look for new measures to control these problems.
The whitefly [Bemisia tabaci (Gennadius) ‘B biotype’ (Hemiptera: Aleyrodidae)] is an important pest of cotton crops [4,5]. The damage caused by B. tabaci ‘B biotype’ includes,primarily, the large extraction of sap and, indirectly, the transmission of viruses, including the “common mosaic” AbMV (Abutilon Mosaic Virus) [6]. This biotype had shown to have a great capacity to develop resistance to most of the active ingredients on the market, including neonicotinoids and growth regulators [7-9] that also directly affect the natural enemies [10]. Through the excessive feeding of phloem sap, the honeydew excreted from B. tabaci support the growth of sooty mold fungi (Capnodium sp.), that inhibits host photosynthesis [11] damaging the processing and depreciating the fiber due to the lack of the product standardization [12,13]. The wide distribution of the whitefly is also related to the agricultural production system, such as the succession and sowing of host crops, in addition to wild species that contribute to the continuous population growth of B. tabaci as well.
Biopesticides containing organisms entomopathogens are environmentally friendly and often decompose quickly, resulting in lower exposures and avoiding the population problems caused by synthetic chemical pesticides [14]. Bacillus thuringiensis Berliner (Bacillaces: Bacillaceae) (Bt), is an entomopathogenic bacterium, which has been widely used worldwide in spray able biopesticides formulations and in transgenic Bt-plants. The spores and the toxins crystals used in formulations provide good protection of plants crops against attack by insect pest, and control of insect species of the orders Lepidoptera, Diptera, Coleoptera [15]. Some of isolates are also active against other insect orders such as Hemiptera, Hymenoptera, Homoptera and Orthoptera [16-19].
The use of strains and derived products of Bt has been previously proposed as a feasible alternative to manage B. tabaci [8,20-23] in bioassays realized with Bt isolates and toxins that were exposed on the leaves surface and on insect bodies. However, no commercial bioinsecticide have been produced using this bacterial to control B. tabaci so far [8]. Among these reports, there are few studies that investigated endophytic Bt against sucking pests [24] and none with B. tabaci. Studies indicate the possible success of this technology demonstrating that Bt colonizes cotton and cabbage tissues and, therefore, could be available to insects that feed on them, such as Spodoptera frugiperda (JE Smith) (Lepidoptera: Noctuidae) and Plutella xylostella (L.) (Lepidoptera: Plutellidae), respectively [25]. The use this Bt technology can help control P. xylostella in cabbage plants and achieved control of Hypsipyla grandella Zeller (Lepidoptera: Pyralidae) in mahogany seedlings [26,27]. There is a close relationship between the Bt strain and the cotton variety, and that Bt can colonize plants endophytically, acting simultaneously to promote plant growth and potentially control insects [28]. The endophytic form of Bt can be an alternative that could decrease the problem of the bacteria’s sensitivity to ultraviolet (UV) light and rainwater.
For this reason, the aim of this work was to evaluate the capacity to recovery Bt from nymphs and adults of B. tabaci ‘B byotipe’ fed on cotton plants inoculated with the Btk-gfp (B. thuringiensis subsp. kurstaki: green fluorescent protein) strain, which encodes the GFP gene. In this way, to verify the ability of this sucking pests to acquire sap Bt. Other objective was to evaluate the oviposition comparing untreated plants to those treated with Bt strains.
Materials and Methods
Rearing of B. tabaci
The adult whiteflies were obtained from a stock colony maintained on cabbage plants Brassica oleracea var. acephala (ISLA Seeds, Rio Grande do Sul State, Brazil). Eight plants were kept in cages built with PVC pipes and voile type fabric in greenhouse conditions at (28±2)°C and (60±10) % humidity. The plants were irrigated and replaced as needed to maintain their nutritional quality
Acquisition and recovery of Btk-gfp by B. tabaci fed on cotton plants inoculated
To prepare the plant material, 4 pots in polyethylene (dimensions 20×32cm), containing 3 cotton plants (variety BRS 8H) at 18 days after sowing, were filled with 500g of sterile soil and BioPlant® (Minas Gerais, Brazil) substrate in 2:1 ratio. The microorganism strain, which encodes the GFP gene, Btk-gfp (B. thuringiensis subsp. kurstaki: green fluorescent protein) was grown in liquid medium supplemented with erythromycin (10μg mL-1) and incubating the culture for 48h., 200rpm at 28°C [29]. Then 5mL of the obtained bacterial suspension was inoculated into the soil close to the root of each plant. Two pots were used to inoculate the bacteria containing the fluorescent protein and the negative control using sterile water.
Five days after the inoculation, 35 non-sexed adult B. tabaci were confined to cotton plants in a cage made with voile fabric and a wire ring for 48hours to permit feeding and oviposition. The soil was covered to avoid contact with flies. After this period, the adults were collected with the aid of an insect aspirator, and the plants were transferred to (0.3×0.8×0.3m) cages also covered in voile to observe the Btk-gfp ingestion by B. tabaci nymphs (about 80 individuals collected from cotyledon leaves and the first true leaf node) that occurred 11 days after the end of the adult exposure.
The insects’ adults were superficially disinfected with 70% alcohol, 2% hypochlorite, and sterile distilled water for 10seconds in each solution and powdered in the presence of 0.9% NaCl. An aliquot of 25μL was seeded in triplicate in Petri dishes containing Embrapa selective agar medium (erythromycin at a concentration of 10 μg/mL) and incubated at 28°C for 48h at 200rpm [29]. After this period with Drigalski (1μL) cell spreader, a portion of this bacterium was collected, and its colonies were visualized in the dark with fluorescence microscopy in a Zeiss Axiophot (Jena, Germany) microscope using a 475-550nm filter, to detect the presence the GFP in vegetative cells. The same process was conducted for nymphs, but without disinfestation.
Bemisia tabaci oviposition bioassay on cotton plants inoculated with Bt
The Bt strains used in this bioassay were randomly selected with different serotypes and strains tested to promote cotton plant growth and against S. frugiperda [28], and in cabbage seedlings [26]. Ten strains were tested: S456 (B. thuringiensis subsp. entomocidous), S546 (B. thuringiensis subsp. kurstaki), S599 (B. thuringiensis subsp. kenyae), S655 (B. thuringiensis subsp. alesti), S907 (subsp. not determined), S1166 (B. thuringiensis subsp. muju), S1450 (B. thuringiensis subsp. kurstaki HD1 (Btk)), S1806 (B. thuringiensis subsp. japonensis), S1905 (B. thuringiensis subsp. kurstaki), and S2122 (B. thuringiensis subsp. kurstaki). The strain S1450 HD1 (Btk) obtained from the Collection of B. thuringiensis and Lysinibacillus sphaericus at the Pasteur Institute, France, and the others strains from the Bank of Bacteria of Invertebrates at Embrapa Genetic Resources and Biotechnology, located in Brasília,Federal District at Brazil.
For the oviposition bioassay, a new cage was assembled like the breeding cage and under the conditions described above, now containing six cabbage plants infested with B. tabaci adults. The cotton plants were placed in pots (80mL) containing a mixture of sterile soil and Bioplant® substrate in a 2:1ratio. After the 15- day period of emergence, the plants were ready for testing. The inoculum was prepared by adding a strip of filter paper containing spores (stored in the bacteria bank) to 15mL of Embrapa medium and incubating the culture for 72h., 200rpm, 28°C [29]. The growth was observed by a phase contrast optical microscope system to verify the bacterium structures in the formation of spores and crystals.
The cotton plants were inoculated with 5mL of this bacterial suspension onto the soil close to the roots, and the plants were allowed to sit for 48h before exposure to insects. The plants with bacteria were placed in rows in the cage for the bioassay so they were between the whitefly-infested plants. The system remained for a period of 12hours for oviposition, when the plants were carefully removed from the cages to count the eggs and examined microscopically with the aid of a stereoscopic magnifying glass (Leica Microsystems, Wetzlar, Germany) at x 40 magnification. The control treatment consisted of plants treated only with sterile water and plants inoculated with Embrapa medium without the bacteria. For each strain was prepared five replicates in a completely randomized design. The data were submitted to ANOVA variance test and Tukey test (p<0.05) using the Sisvar statistical program.
Results
The ability to acquire and recovery Btk-gfp by B. tabaci was tested. Bacterial growth was obtained from macerated B. tabaci adults and nymphs that had been fed on plants treated with Btkgfp (Figures 1 & 2). The bacterial growth obtained from adult and nymphs was observed with epifluorescent lighting and showed cells that express green fluorescent protein. Plant roots probably absorb Bt bacteria, making them available for food in adult hood and immature whitefly. The insects collected from control plants did not display bacterial growth.

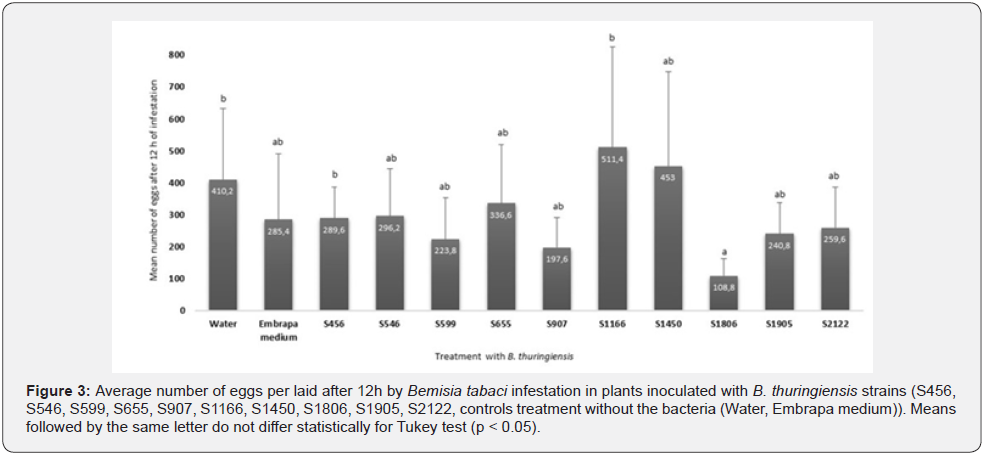
In the test with different serotypes and Bt strains the oviposition of B. tabaci on inoculated cotton plants was evaluated. After 12 hours of infestation, interesting results between treatments were observed (Figure 3). The plants with strain S1806 exhibited a lower number of B. tabaci eggs compared to the control with only water, and strains S456 and S1166 (F = 1.794; p = 0.08145). However, this strain was similar with other tested isolates and the control with the culture medium used to inoculate the roots of the plants. Plants containing inoculum with strains S1166, S1450, and S655 showed a greater egg density with 511.4; 453.0, and 336.6 eggs per plant, respectively. The least eggs were noted for S1806 with 108.8 eggs and S907 with 197.6 eggs per plant. The treatment containing strains S599, S1905, S2122, S456, S546 and the control with Embrapa medium varied between 223.8 and 296.2 eggs per plant
Discussion
Bemisia tabaci ‘B biotype’ is an important pest of cotton crops [4,5] wich has developed resistance to the chemical insecticides [8,9] affecting the agricultural economy [30]. An alternative could be the use of B. thuringiensis, but until now, there has not been commercial products containing these bacteria to B. tabaci. Bacillus thuringiensis insecticidal proteins are used as active components of biopesticides and as protectors incorporated into the plant in transgenic crops. One of the most relevant attributes of these Bt protein-based insecticidal technologies is their high specificity, which guarantees the absence of harmful effects on non-target insects, vertebrates, and the environment [31].
The use of endophytic organisms can bring benefits in biological control of plant pathogens and insect-pest [32-34]. The adoption of this technology has been investigated using Bt with relevant control results on important pest in agriculture [24-28], but no study verified this effect on B. tabaci. In this work, Btkgfp recovery and acquisition capability of B. tabaci fed in cotton plants the sap Bt was checked. The collected cells from the bodies of adults and immature stages of the whitefly produced bacterial colonies morphologically similar to Bt after maceration, plating and visualization through optical fluorescence microscopy. This practice was used with efficiently to confirmation of the colonization of Bt strains in plants and to demonstrate the toxic action of Bt against the sucking pest Aphis gossypii Glover (Hemiptera: Aphididae) [35].
Another study demonstrated the ability of Bt strains to translocate from roots to shoots in citrus seedlings, with capacity of use in control of the phloem-feeding insects Diaphorina citri Kuwayama (Hemiptera: Lividae) that feed on the plant’s shoots [24]. In summation, the inoculated Bt had been absorbed by the roots and transported to the aerial part of the plant by the xylem vessels [24,25,28], but the mechanisms of transport are not yet elucidated. This work suggests that this transport could also occur for the plant phloem, which is the preferred feeding place for B. tabaci, even though it often tastes the xylem vessels [36].
Some reports have demonstrated toxicity activity of the Bt strains or toxins to hemipterans. Nymphs of whitefly treated directly with the suspension containing the complex crystal-spore of Bt showed a 90% major mortality [8]. In that research, the strain GP139 of Bt was used to show the toxic activity against B. tabaci [37], in addition the high virulence of this Bt strain observed in bioassays performed with Myzus persicae Sulzer (Hemiptera: Aphididae) [30]. Bacillus thuringiensis isolates were selected for their high insecticidal potential against nymph’s whitefly under in vitro conditions [38]. Native isolates of Bt from soil environments caused up 50 to 70% mortality of B. tabaci nymphs [20] and mortality with superior percentage of 90% [22]. Consistent effects were found from isolates and supernatants derived products of Bt culture on adult repellence, oviposition deterrence and nymphal mortality of B. tabaci [21].
In these reports, the studies evaluated strains, toxins and derived products of Bt against B. tabaci by immersion or spraying of the bacterial suspension in surface of leaves and insect bodies [8,20,22,30,37,38]. But the action of Bt sap has not been reported to this pest.
The oviposition test comparing untreated cotton plants to those treated with serotypes and Bt strains, showed a lower eggs number to the S1806 strain. It is supposed that the colonizing capacity of Bt inside the cotton plant can interfere in the oviposition of the whitefly, which feeds on those structures, with potentially insecticide activity. Determining which mechanisms were responsible for the difference in oviposition is still premature. However, herbivores can use plant volatiles to locate their hosts [39], and cotton plants treated with growth-promoting bacteria can modify the volatile profile.
The effects of the volatiles on plants treated with the beneficial bacteria can reduce the oviposition of pest insects [40]. Females during their search for oviposition lay their eggs on healthier plants for better-quality larval development. Plants treated with Bacillus subtilis showed low populations of whitefly in greenhouse [41]. Another research group reported that the treatment of bacteria in tomato plants reduced the emergence of whitefly adults and that this suppression was due to jasmonic acid responses [42] in plantinduced systemic resistance. Many studies have reported the use of Bt as an insecticidal product or the insertion possibility of its toxins into plants to provide resistance to susceptible insects. However, little is known about how plant-colonizing bacteria act against harmful insects that feed on them. Reports discuss metabolically active form of Bt in the phylloplane [43].
The resistance induced by microorganisms to insects varies between studies, due to different hosts, different plants, insects, and microbial inoculants [44,45]. Therefore, different responses may be found for insect pests interacting with the plant treated by the bacterium. The effects of treatment with bacteria on insect pests may also vary in relation to insect eating habits, whether it is a specialist or a generalist. Beneficial bacteria that have some effect in promoting plant growth appears to negatively affect the development of insect pests [24,26,28,32]. The recovery capacity of Bt by adult and immature B. tabaci demonstrates the possibility of exploring the Bt sap as an alternative biological control technology for this insect, in an endophytic way in the plant. The result obtained opens new perspectives for studying the interaction of endophytic Bt in plants for biological control of sucking pests of great agronomic interest.
Conclusion
Bemisia tabaci ‘B biotype’ are able to acquire Bt inoculated in cotton plants by feeding on their structures. Plants treated with Bt strain S1806 had a lower number of B. tabaci eggs than plants that received only sterile water.
Acknowledgement
The authors gratefully thank the Coordination of Improvement of Higher Education Personnel (CAPES, in Portuguese) for financial support received by first author and to Dr. Francisco Aragão (Embrapa Cenargen) for your help in starting the whitefly breeding.
References
- Miranda JE (2010) Manejo integrado de pragas do algodoeiro no cerrado brasileiros. Embrapa, Circular Técnica 131 (CNPA) (infoteca-e), Embrapa Algodão, Campina Grande, Paraíba, Brazil, p 37.
- de Lima IS, Degrande PE, Miranda JE, dos Santos WJ (2013) Evaluation of the boll Weevil Anthonomus grandis Boheman (Coleoptera: Curculionidae) Suppression Program in the state of Goiás, Brazil. Neotrop Entomol 42(1): 82-88.
- Naranjo SE, Ellsworth, PC, Frisvold GB (2015) Economic value of biological control in integrated pest management of managed plant systems. Ann Rev Entomol 60: 621-645.
- Campos ZR, Boiça Júnior AL, Lourenção AL, Campos AR (2009) Parâmetros biológicos de Bemisia tabaci (Genn.) biótipo B (Hemiptera: Aleyrodidae) em genótipos de algodoeiro. Bragantia 68(4): 1003-1007.
- Farooq M, Shakeel M, Shahzad U, Khan BS, Shahid MR, et al. (2021) Comparative Demographic Traits of the Whitefly (Bemisia tabaci) B Biotype against different Host Plants. Intern J Agricul Biol 25(2): 460-468.
- Rodrigues SMM, Vivan LM (2007) A Mosca Branca (Bemisia tabaci Biótipo B) no Mato Grosso. Embrapa, Circular Técnica 111 (CNPA) (infoteca-e), Embrapa Algodão, Campina Grande, Paraíba, Brazil p 9.
- Velasco L, Ruiz L, Galipienso L, Rubio L, Janssen D (2020) A Historical Account of Viruses in Intensive Horticultural Crops in the Spanish Mediterranean Arc: New Challenges for a Sustainable Agriculture. Agronomy 10(6): 860.
- Salazar Magallon JA, Hernandez Velazquez VM, Alvear Garcia A, Arenas Sosa I, Peña Chora G (2015) Evaluation of industrial by-products for the production of Bacillus thuringiensis strain GP139 and the pathogenicity when applied to Bemisia tabaci nymphs. Bulletin of Insectology 68(1): 103-109.
- Castle SJ, Prabhaker N (2013) Monitoring changes in Bemisia tabaci (Hemiptera: Aleyrodidae) susceptibility to neonicotinoid insecticides in Arizona and California. J Econ Entomol 106(3):1404-1413.
- Navas Castillo J, Fiallo Olivé E, Sánchez Campos E (2011) Emerging virus diseases transmitted by whiteflies. Annual Rev Phytopathol 49: 219-248.
- Johnson MW, Capraio LC, Lynch JA, Tabashnik BE, Welter SC, et al. (1991) Yield response of fresh market tomatoes to greenhouse whitefly infestations in Hawaii. In Pro. Agricultural Development in the American Pacific Crop Protection Conference University of Hawaii, Honolulu, HI, USA pp 101-103.
- Toscano LC, Santos TMD, Boiça Júnior AL (2003) Preference of Bemisia tabaci biotype B oviposition in cotton cultivars. Pesq Agrop Bras 38(1): 155-160.
- Lourenção AL, Yuki VA, Alves SB (1999) Epizootics of Aschersonia cf. goldiana on Bemisia tabaci (Homoptera: Aleyrodidae) biotype B in the State of Sao Paulo, Brazil. Anais Sociedade Entomol Brasil 28(2): 343-345.
- Ganga GC, Arjyal C (2020) Field Evaluation of Native B. thuringiensis isolates Against Aphids (Aphis fabae). Tribhuvan Univ J Microb 7: 115-122.
- Xu C, Wang BC, Yu Z, Sun M (2014) Structural insights into Bacillus thuringiensis Cry, Cyt and Parasporin toxins. Toxins 6(9): 2732-2770.
- Porcar M, Grenier AM, Federici B, Rahbé Y (2009) Effects of Bacillus thuringiensis δ-endotoxins on the pea aphid (Acyrthosiphon pisum). App Environ Microb 75(14): 4897-4900.
- Palma L, Muñoz D, Berry C, Murillo J, Caballero P (2014) Bacillus thuringiensis toxins: an overview of their biocidal activity. Toxins 6(12): 3296-3325.
- Schnepf E, Crickmore N, Van Rie J, Lereclus D, Baum J, et al. (1998) Bacillus thuringiensis and pesticidal crystals proteins. Microbiol Mol Biol Rev 62(3): 775-806.
- Pardo López L, Soberón M, Bravo A (2013) Bacillus thuringiensis insecticidal three-domain Cry toxins: mode of action, insect resistance and consequences for crop protection. FEMS Microbiol Rev 37(1): 3-22.
- Torres Cabra E, Hernandez Fernandez JA (2019) Larvicidal activity of Bacillus thuringiensis Colombian native strains against Bemisia tabaci (Hemiptera: Aleyrodidae). J Plant Protect Res 59(4): 503-511.
- Sanchéz ER, Ramírez AG, Ramírez AR, Angulo MG, Suárez JMT, et al. (2019) Biological activity of Bacillus thuringiensis culture supernatant on Bemisia tabaci and its parasitoid Eretmocerus eremicus. Trop Subtrop Agroecosyst 22(1): 213-218.
- El Assal SED, Youssef NA, Ami, GA (2013) Isolation and identification of locally isolated bacterial strains effective against whitefly Bemisia tabaci. Arch Agron Soil Sci 59(6): 779-790.
- Al Shayji Y, Shaheen N (2008) Isolation of Bacillus thuringiensis strain from Kuwait's soil effective against whitefly nymphs. J Insect Sci 8(4):4.
- Dorta SD, Balbinotte J, Monnerat R, Lopes JR, da Cunha T, et al. (2020) Selection of Bacillus thuringiensis strains in citrus and their pathogenicity to Diaphorina citri (Hemiptera: Liviidae) nymphs. Insect sci 27(3): 519-530.
- Monnerat RG, Soares DM, Capdeville G, Jones G, Martins ES, et al. (2009) Translocation and insecticidal activity of Bacillus thuringiensis bacteria living inside of plants. Microb Biotech 2(4): 512-520.
- Praça LB, Gomes ACMM, Cabral G, Martins ES, Sujii ER, et al. (2012) Endophytic colonization by Brazilian strains of Bacillus thuringiensis on cabbage seedlings grown in vitro. Bt Res 3(3): 11-19.
- Castro MT, Montalvão SCL, Monnerat RG (2019) Control of mahogany shoot borer, Hypsipyla grandella (Lepidoptera: Pyralidae), with Bacillus thuringiensis in a systemic way. Nativa 7(4): 426-430.
- da Costa FSS, Praça LB, Gomes ACMM, dos Santos RC, Soares CMS, et al. (2020) Bacillus thuringiensis Effect on the Vegetative Development of Cotton Plants and the Biocontrol of Spodoptera frugiperda. Agronomy 10(12):1889.
- Monnerat RG, Batista AC, Medeiros PT, Martins ES, Melatti VM Praça, et al. (2007) Screening of Brazilian Bacillus thuringiensis isolates active against Spodoptera frugiperda, Plutella xylostella and Anticarsia gemmatalis. Biol Control 41(3): 291-295.
- Torres Quintero MC, Arenas Sosa I, Hernández Velázquez VM, Suárez Rodríguez R, Peña Chora G (2016) Characterization of Bacillus thuringiensis (Bacillaceae) strains pathogenic to Myzus persicae (Hemiptera: Aphididae). Florida Entomol 99(4):639-643.
- Jurat Fuentes JL, Crickmore N (2017) Specificity determinants for Cry insecticidal proteins: Insights from their mode of action. J Invert Pathol 142: 5-10.
- Pineda A, Zheng SJ, van Loon JJ, Dicke M (2012) Rhizobacteria modify plant-aphid interactions: a case of induced systemic susceptibility. Plant Biol 14(1): 83-92.
- Lacava PT, Azevedo JL (2013) Endophytic bacteria: a biotechnological potential in agrobiology system. In: Maheshwari D, Saraf M, Aeron A (Eds) Bacteria in agrobiology: crop productivity. Springer, Berlin, Heidelberg, Germany p 1-44.
- Li H, Soares MA, Torres MS, Bergen M, White Jr JF (2015) Endophytic bacterium, Bacillus amyloliquefaciens, enhances ornamental hosta resistance to diseases and insect pests. J Plant Interact 10(1): 224-229.
- Melatti VM, Praça LB, Martins ES, Sujii E, Berry C, Monnerat RG (2010) Selection of Bacillus thuringiensis strains toxic against cotton aphid Aphis gossypii, Glover (Hemiptera: Aphididae). BioAssay 5(2): 1-4.
- Moreno Delafuente A, Garzo E, Moreno A, Fereres A (2013) A plant virus manipulates the behavior of its whitefly vector to enhance its transmission efficiency and spread. Plos One 8(4): 1-10.
- PEÑA G, BRAVO A (2002) Discovery of a new Bacillus thuringiensis toxins. In Biotechnology of Bacillus thuringiensis and its environmental impact (Akhurst RJ, Beard CE, Hughes P, Eds). Proceedings of the 4th Pacific Rim Conference, CSRO pp 106-111.
- Gebremariam A, Chekol Y, Assefa F (2021) Isolation, characterization, and bio-insecticidal efficiency of Ethiopian isolates of Bacillus thuringiensis against Galleria mellonella L. (Lepidoptera: Pyralidae) and tomato whitefly, Bemisia tabaci (Genn.) (Hemiptera: Aleyrodidae). Egypt J Biol Pest Control 31(1): 1-12.
- Dicke M, Van Loon, JJA (2000) Multitrophic effects of herbivore-induced plant volatiles in an evolutionary context. Entomol Exp Appl 97: 237-249.
- Shavit R, Ofek Lalzar M, Burdman S, Morin S (2013) Inoculation of tomato plants with rhizobacteria enhances the performance of the phloem-feeding insect Bemisia tabaci. Front Plant Sci 4: 1-12.
- Hanafi A, Traoré M, Schnitzler, WH, Woitke M (2007) Induced resistance of tomato to whiteflies and Phytium with the PGPR Bacillus subtilis in a soilless crop grown under greenhouse conditions. Acta Hortic 474: 315-322.
- Valenzuela Soto JH, Estrada Hernandez MG, Ibarra Laclette E, DéLano Frier JP (2010) Inoculation of tomato plants (Solanum lycopersicum) with growth promoting Bacillus subtilis retards whitefly Bemisia tabaci development. Planta 231(2): 397-410.
- Bizzarri MF, Bishop AH (2007) Recovery of Bacillus thuringiensis in vegetative form from the phylloplane of clover (Trifolium hybridum) during a growing season. J Invertebr Pathol 94(1): 38-47.
- Van Oosten VR, Bodenhausen N, Reymond P, Van Pelt JA, Van Loon LC, et al. (2008) Differential effectiveness of microbially induced resistance against herbivorous insects in Arabidopsis. Mol Plant Microbe Interact 21(7): 919-930.
- Stout MJ, Thaler JS, Thomma B (2006) Plant-mediated interactions between pathogenic microorganisms and herbivorous arthropods. Annu Rev Entomol 51: 663-689.






























