Investigation and Comparison Effect of Glomus Intraradices and Glomus Mosseae to Control of Meloidogyne Javanica in Cucumis Sativus
Niloufar Mahmoudi*, Yousef Naserzadeh and Albert Engeribo
Department of Agro Biotechnology, RUDN University, Russia
Submission: September 03, 2021; Published: September 07, 2021
*Corresponding author: Niloufar Mahmoudi, Department of Agro Biotechnology, Institute of Agriculture, RUDN University, Moscow, 117198 Moscow, Russia
How to cite this article: Niloufar M, Yousef N, Albert E. Investigation and Comparison Effect of Glomus Intraradices and Glomus Mosseae to Control of Meloidogyne Javanica in Cucumis Sativus. Agri Res & Tech: Open Access J. 2021; 26 (2): 556331. DOI: 10.19080/ARTOAJ.2021.26.556331
Introduction
Cucumber (Cucumis sativus L.), from the squash family, which includes 90 genera and 750 species. Various factors reduce the yield of cucumber in different areas under the cultivation of this crop. Nematodes are important pathogens that cause great damage to agricultural products [1]. Plant root-knot nematodes are among the most destructive parasitic nematodes of various plants, especially cucumber. Different methods are used to control this nematode, but in most cases, their control is difficult due to the wide host range, short life cycle, and high rate of reproduction. Diverse organisms affect a variety of plant root-knot nematodes and reduce their population [2].
Mycorrhiza fungi are among the factors that reduce several plant diseases and can, directly and indirectly, affect the population of plant nematodes. Mycorrhiza fungi in nature in meeting the water and nutritional plant needs have an effective role and increase phosphorus, water, and minerals to increase plant growth and health. Much research has been done on the effect of mycorrhiza fungi on plant pathogens.
G. intraradices and G. mosseae are important in the interaction of pathogens with the host plant. They are released into the environment roots, soil and aerial parts of the plant in interaction with various other microorganisms survive. Species of this fungus, especially G. intraradices and G. mosseae to high reproductive rate, high ability in the use of different food sources, high power of aggression against pathogens, utilization of antagonistic mechanisms such as competition, parasitism, and antibiosis, ability to changes in the rhizosphere, production of extracellular enzymes such as amylolytic, pectolytic, proteolytic, lipolytic, Ketinolytic and cellulite as well as efficacy in stimulation growth and induction of resistance in plants are very important factors. More studies have shown that these fungi cause decrease diseases and symptoms are caused by soil pathogens [3].
Materials and Methods
Preparing M. javanica nematode population
After providing a plant sample infected with nematode, using single egg mass method, and reproducing it consecutively on the roots of cucumber, its purification and reproduction were performed. In most of the nematology investigation, the egg and larvae of nematode are used in order to create an infection. However, generally, because some eggs are not alive, using the second larvae of nematode is more common. Extracting egg and providing second larvae was done using [4] method.
Mycorrhiza fungi species preparation
Mycorrhiza fungi species G. mosseae, G. Intraradaices were taken from agricultural biotechnology laboratory and to investigate the effect of them in control of M. javanica in cucumber. Ingredients include vesicles, filaments, arbuscles, and fungal spores were mixed with sand. To mass propagate this fungus and obtaining the desired population from pure spores and disease-free, based on the advice of the method potting, was used in the greenhouse [5,6]. For this purpose, the fungi inside 5kg pots with a mixture of sand and clay in a ratio of five to one near the root of the cultivar corn single grass 704 stored and propagated for four months. The greenhouse conditions were controlled by day temperature and twenty-seven and nineteen degrees Celsius, respectively the relative humidity was sixty percent. After reproduction, for Ensuring the desired species and species, identification relying on the color scheme of the international collection of mycorrhiza fungi vesicles based at the University of West Virginia [7-10] finally, specifications species with descriptions of arbuscular mycorrhiza fungi compared and identified [11,12].
Investigation of the interaction of Glomus spp. on M. javanica nematode in greenhouse conditions
A total of six treatment combinations in the form of a complete random design in four replications in plastic pots one kilogram and one control were examined. Treatments include
a. Cucumbers inoculated with G. mosseae
b. Cucumbers inoculated with G. Intraradaices
c. Cucumbers inoculated with nematodes M. javanica
d. Cucumbers inoculated with G. mosseae and nematodes M. javanica
e. Cucumbers inoculated with G. Intraradaices and nematode M. javanica
f. Cucumbers inoculated with G. mosseae, G. Intraradaices and nematode M. javanica
g. A control.
Greenhouse environmental conditions during the maintenance period pots with a temperature range of 26 ± 2°C, sixty percent relative humidity, sixteen hours’ exposure period Irrigation was forty-eight hours a day and round. Eight weeks after inoculation of nematodes into the plants, the treatments were evaluated and indicators such as the number of nodes at the root, the number of egg and number of second instar larvae in the soil as well as reproduction index were calculated. Vegetative characteristics of the plant include root length, fresh and dry weight aerial parts, and roots were also evaluated.
Statistical analysis of data
Data analysis using statistical software, SPSS 20 analysis of variance of data and comparison of means with test Multiple LSDs performed at one and five percent levels.
Results and Discussion
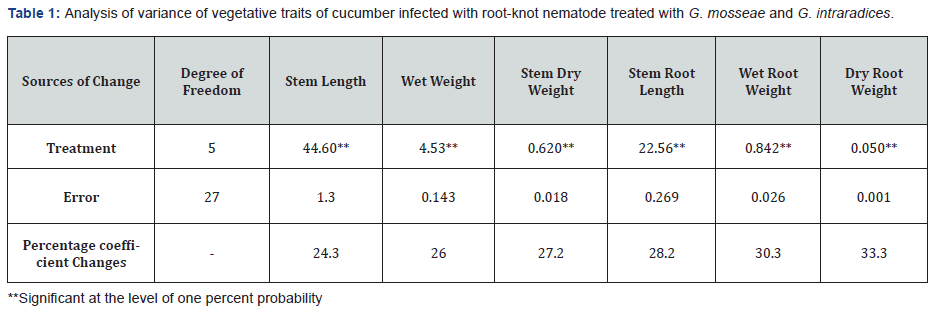
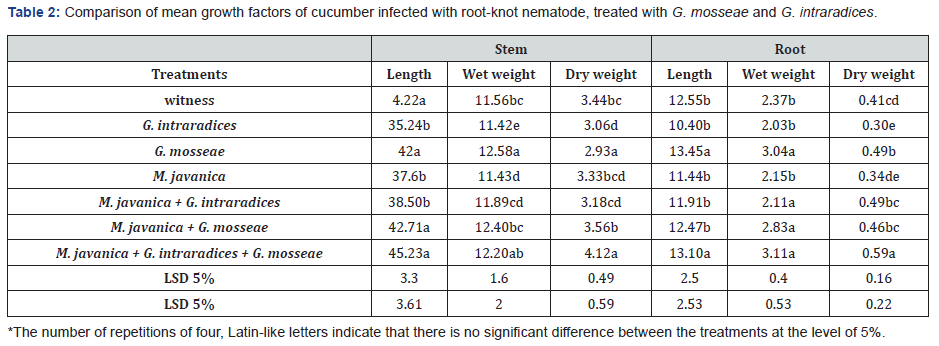
Results of analysis of variance of the stem and vegetative traits roots related to infected cucumbers root-knot nematode M. javanica with fungi G. Intraradaices and G. mosseae were treated, shows that the effect of inoculation of treatments for all traits the study is significant at the level of one percent (Table 1) As in the table of comparison of mean traits vegetative (Table 2) is observed, the most effective G. mosseae + G. Intraradaices + M. javanica treatments and in G. mosseae + M. javanica is observed. The plant has also been compared to the control. according to the results comparison of mean growth characteristics of cucumber, fungal inoculated treatments, G. mosseae even ratio untreated control treatment with nematode with higher growth were due to the improvement of plant nutritional conditions and increased absorption of minerals, especially phosphorus with, G. mosseae increasing plant growth conditions and plant tolerance to nematodes [11,13,14].
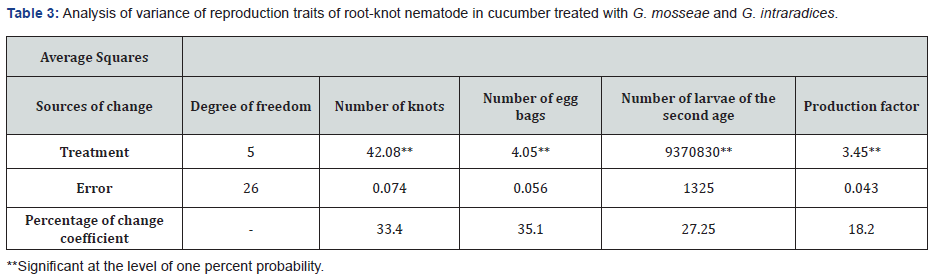
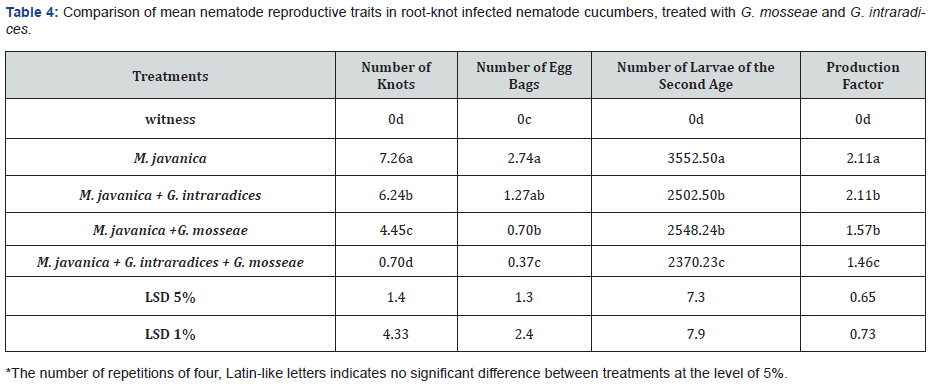
Reproductive traits of nematodes result of analysis of variance of reproductive traits of root-knot nematode M. javanica (Table 3) shows that between treatments in terms of four important traits of nematode reproduction, namely number of nodes per root, number of eggs per root, number second instar larvae in soil and nematode reproduction factor, there is a statistically significant difference at the level of one percent. With pay attention to the comparison table of the mean of the related traits reproduction of nematodes (Table 4) lowest productions of galls and Egg, as well as second instar larvae and reproductive factor respectively in G. Intraradaices and G. mosseae + M. javanica and G.mosseae + M. javanica with M. javanica treatment, have a significant difference in the level they were one percent.
Conclusion
Examination of the results of the fungal effect test of G. intraradices and G. mosseae on M. javanica showed that both fungi have a significant effect on reproductive characteristics M. javanica. Due to the interaction of fungi and nematode with the plant, the amount of knot formation on the root and the number of eggs decreases. Therefore, the fungus may infect the plant defense system stimulates. Mycorrhiza fungi with the colonization of the rhizosphere environment prevent the spread of nematodes internal tissues take root. In the present study, among the two fungi tested, G. mosseae had a greater inhibitory effect. In roots colonized by fungi, G. mosseae enzyme chitinase increases. It causes the chitin to break down the nematode egg wall and eventually destroy it becomes an egg.
Also, the increase of arginine in the plant, from prevents nematode reproduction, and on the other hand with increasing the amount of phenol in the root reduces nematode proliferation and egg bag production and number of eggs. According to the report Smith this fungus has a lot of competitive power and has the power of colonization is high on the root system Smith 1988. Therefore, it can be justified that G. mosseae with the same competitive system has been able to significant growth and development of nematodes in the roots and consequently, in inoculated treatments with G. mosseae nematode reproductive factor significantly has decreased.
References
- Mirehki K, Abdollahi M, Talaie F (2013) Effect of Trichoderma virens and Glomus mosseae in control of Meloidogyne javanica in tomato. Biolog Con Pes Pla Dise 2(1): 9-16.
- Sharon E, Chet I, Viterbo A, Bar Eyal M, Nagan H, et al. (2007) Parasitism of Trichoderma on Meloidogyne javanica and role of the gelatinous matrix. Eur J Plant Pathol 118(3): 247-258.
- Dehne HW, Backhaus GF, Baltruschat H (1987) Inoculation of plants with VA mycorrhizal fungi at inorganic carrier materials. In Mycorrhizae in the next decade, practical applications and research priorities. Proceedings of the 7th North American conference on mycorrhiza. Institute of Food an Agricultural Sciences 272-273.
- Niloufar M (2019) Effect of beta-aminobutyric acid on activity of peroxidase and polyphenol oxidase enzymes in cucumber infected with nematode Meloidogyne javanica. RUDN J Agro Anim Indus 14(1).
- Yousef N, Niloufar M, Elena P (2019) Antipathogenic effects of emulsion and nanoemulsion of cinnamon essential oil against Rhizopus rot and grey mold on strawberry fruits. Food raw mater 7(1): 210-216.
- Naserzadeh Y, Kartoolinejad D, Mahmoudi N, Zargar M, Pakina E, et al. (2018) Nine strains of Pseudomonas fluorescens and P. putida: Effects on growth indices, seed and yield production of Carthamus tinctorius L. Res Crop 19(4): 622-632.
- Cofré N, Becerra AG, Marro N, Domínguez L, Urcelay C (2020) Soybean growth and foliar phosphorus concentration mediated by arbuscular mycorrhizal fungi from soils under different no-till cropping systems. Rhizosphere 16: 100254.
- Mahmoudi N, Pridannikov M, Zargar M, Naserzadeh Y, Limantceva L, et al. (2020) Molecular diagnostics of Ditylenchus destructor based on the ITSrDNA from Iran and Russia Federation. Res Crop 21(1): 151-155.
- Błaszkowski J, Tadych M (1997) Glomus multiforum and G. verruculosum, two new species from Poland. Mycologia 89(5): 804-811.
- Naserzadeh Y, Bondarenko GN, Kolesnikova EV, Pakina EN (2021) Phylogenetic analysis and designing new primers for molecular identification of Drosophila suzukii. RUDN J Agro Anim Indus 16(2): 137-145.
- Smith SE, Gianinazzi Pearson V (1988) Physiological interactions between symbionts in vesicular-arbuscular mycorrhizal plants. Annu Rev Plant Physiol Plant Mol Biol 39(1): 221-244.
- Lawaju BR, Groover W, Kelton J, Conner K, Sikora E, et al. (2021) First report of Meloidogyne incognita infecting Cannabis sativa in Alabama. J Nematol 53: e2021-2052.
- Chiamolera FM, Martins ABG, Soares PLM, Cunha Chiamolera TPLD (2018) Reaction of potential guava rootstocks to Meloidogyne enterolobii. Revista Ceres 65(3): 291-295.
- Bellé C, Dos Santos PS, Kaspary TE (2021) First report of rice root-knot nematode, Meloidogyne graminicola, infecting Juncus microcephalus in Brazil. J Nematol 53: e2021-2031.






























