Morphological Characteristics of Reproductive System of the Codling Moth Cydia Pomonella
Suqin Shang1*, Ning Liu1, Weizhen Li1, Jing Jiang Zhou1,2*
1ollege of Plant Protection, Gansu Agricultural University/Biocontrol Engineering, China
2Laboratory of Crop Diseases and Pests of Gansu Province, China
2State Key Laboratory of Green Pesticide and Agricultural Bioengineering, Ministry of Education, Guizhou University, Huaxi District, Guiyang, China
Submission: April 29, 2021; Published: May 24, 2021
*Corresponding author: Suqin Shang, College of Plant Protection, Gansu Agricultural University/Biocontrol Engineering, China Jing-Jiang Zhou, State Key Laboratory of Green Pesticide and Agricultural Bioengineering, Ministry of Education, Guizhou University, Huaxi District, Guiyang, China
How to cite this article: Suqin S, Ning L, Weizhen L, Jing Jiang Z. Morphological Characteristics of Reproductive System of the Codling Moth Cydia Pomonella. Agri Res & Tech: Open Access J. 2021; 25 (5): 556320. DOI: 10.19080/ARTOAJ.2021.25.556320
Abstract
Forest management system as a component of ungulate pest management. Wild ungulates are an important site factor in forests and landscapes. Impacts of ungulates on forest vegetation and the effect of hunting systems are often investigated and discussed. In contrast, the impact of silvicultural systems on forest damage by ungulates is much less conscious. Therefore, in a long-term research program in Central Europe since the 1990ies two questions were investigated additionally, (i) in which way can forest management systems and landscape structures influence the interactions between ungulates and forest vegetation, and (ii) how can silvicultural practices modify the susceptibility of forests to damage by ungulates. The results showed a strong impact of these factors on the emergence of game damage to forests. Factors that are often underestimated in practice. Recommendations for a coordinated management of different land users to avoid ungulate damage to forest vegetation and to preserve biodiversity were made.
Keywords:Forestry; Silviculture; Ungulates; Game damage; Wildlife ecology; Integrated management
Introduction
The codling moth Cydia pomonella L. (Lepidoptera, Tortricidae) was originally found in the southeast regions of Europe and then spread to the apple tree Malus pumila Miller around the world. In China, it was first discovered in Korla city, Xinjiang province in 1953 [1,2]. It shows a remarkable ability to adapt to a variety of climatic and trophic conditions [3], even those in North America, South American, and South Africa. The codling moth is one of the main quarantine insect pests of fruit trees in the world (Jin 1997) and a key pest of the plant family Rosaceae. It causes a great damage to pome fruits (apple, pear and quince); [4] and occasionally apricots [2,5]. It has been also found in walnut orchards of the tree family Juglandaceae. Recently, it was reported to harm members of plant family Magnoliaceae [6].
The strong reproductive ability is one of the main reasons for their ability to cause serious damages [2]. Under laboratory conditions, each female adult can lay 12 to 345 eggs [7-10]. The eggs were always laid in fruits, as well as on leaves and twigs of trees, rarely on petioles or stems because of their oviposition behaviours [5]. Therefore, the egg mortality is low (4.7-6.9%) in integrated control orchards due to unviability to nature enemies and other animals [5], causing serious fruit dropping and rotting by the larvae feeding on fruits [11], (Du et al. 2012). The morphological characteristics of reproductive systems are of significance to predict the emergence period and population trend, and to determine the optimum timing for pest control and to identify species [12-16], especially in the cases of Lepidoptera pests, such as Grapholita molesta [17], Palumbina spp. [12], Spodoptera exigua [12]. Besides, insect genitalia are often species-specific, and their structures are often more divergent among closely related species than other structures such as legs, antennae, and eyes [18], which make genitalia more useful in distinguishing closely related species [18].
Allman (1930) and Wiesmann (1935) [19, 20] described the gross morphology of the external and internal reproductive systems of C. pomonella females. Williams (1941) compared the structure of the codling moth spermatophore with those of other Tortricoidea species. Ferro and Akre (1975) [4] illustrated both female and male reproductive systems of the codling moth Laspeyresia pomonella [4]. Since then, mostly studies have focused on their chemical control. There are very few reports about the reproductive characters of this species. Until 2018, the process of egg maturation and classification of ovary development in the codling moth were described and reported [21]. In the present study, the C. pomonella reproductive systems from a Chinese codling moth population in Gansu Province were dissected. The morphology of each parts was described for the first time in detail. The study aims to provide morphological information for identification and support for the prevention and prediction of the codling moth.
Methods and Materials
Codling moth colony
The codling moth Cydia pomonella was collected from an abandoned apple orchard in Wuwei City, Gansu Province of China. The larvae were reared in fruits, the emerged adults were reared with 10% honey water in Laboratory under the condition of 25±1℃ temperature, 75±5% relative humidity and a photoperiod of 16h light: 8h dark.
Reproductive system dissection and observation
The 1-2 d old female and male adults were used. The inner organ was prepared by putting the abdomen in Bouin’s fluid for 30 minutes in a centrifuge tube, and then kept in Ringer’s saline for dissection. The genitalia were treated by boiling with 10%NaOH for 5-10 min to remove all soft tissue, and then kept in 75% alcohol ready for observation.
The reproductive system was observed under Zeiss SteREO Discovery V12 stereoscopic microscope. The photographs were taken with Carl Zeiss CCD AxioCam MRc 5 digital cameras. All images were processed by using Adobe Photoshop CS6.
Results and Analysis
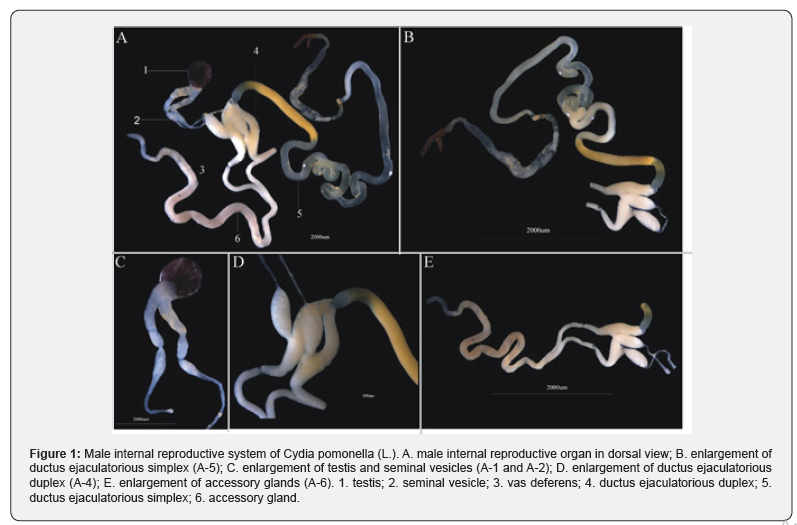
Male internal reproductive organs
The male internal reproductive organ consisted of a testis (Figure1: A-1), a pair of seminal vesicles (Figure1: A-2), a pair of vas deferens (Figure1: A-3), a ductus ejaculatorious duplex (Figure1: A-4) and a ductus ejaculatorious simplex (Figure1: A-5), a pair of accessory glands (Figure1: A-6). The testis is the main sperm productive structure and formed by two testicular follicles enveloped in the lavender adventitia. The seminal vesicles are constricted in the middle and swollen at apex. The vas deferens are the access to ductus ejaculatorious duplex for sperms. The ductus ejaculatorious simplex can be divided into four sections: the first section is transparent, thick and short; the second section is translucent, as thick as the first but longer; the third section is transparent and the longest one; the fourth section is narrowed at the base and then distorted at the connecting section with the aedeagus.

Male external reproductive organs
The male genitalia mainly consist of a pair of valvae (Figure 2A-6 and B), a pair of aedeagus (Figure 2A-3, D and E) and other accessory structures. The valvae are ear shaped. The end of the valvae (Figure 2: A-6) is round, densely covered with thin and irregular hairs and finger like ventral process (Figure 2: A-7). The uncus (Figure 2: A-5) is short. The aedeagus is cecum-like with strong sclerotization and seven cornuti dorsally. Five of the cornute are close to the apex and the other two are in the middle (Figure 2: E). The juxta (Figure 2: F) is V-shaped and membranous.

Female internal reproductive organ

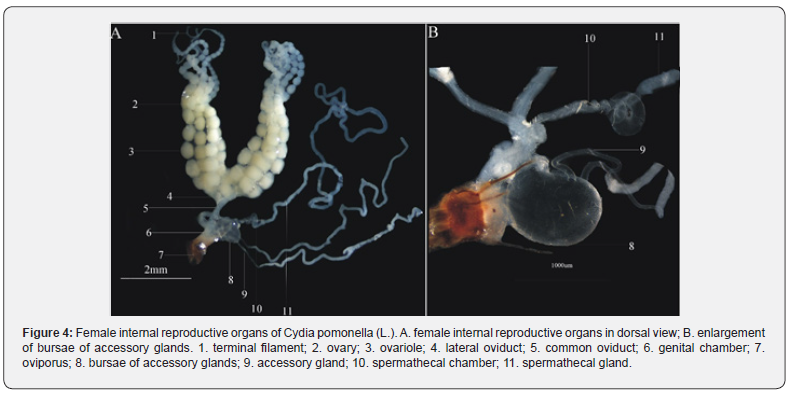
The female internal reproductive organ (Figure 3A) consists of a pair of ovaries (Figure 4: A-2), a pair of lateral oviducts (Figure 4: A-4), a common oviduct (Figure 4: A-5), a bursa of accessory glands (Figure 4: A-8), accessory glands (Figure 4: A-9), a spermathecal (Figure 4: A-10) and a spermathecal gland (Figure 4: A-11). The ovaries are symmetrical, and each ovary has four polytrophic ovarioles. The ovarioles twin together through tracheoles, merge at the base, then incorporate into lateral oviducts. The common oviduct is thin and straight initially, becomes widen and short afterward. The ovarioles (Figure 4: A-3) are divided into four parts: terminal filament (Figure 4: A-1), germarium, vitellarium and pedicel. The two accessory glands are connected with the bursae of accessory glands.
Female external reproductive organs
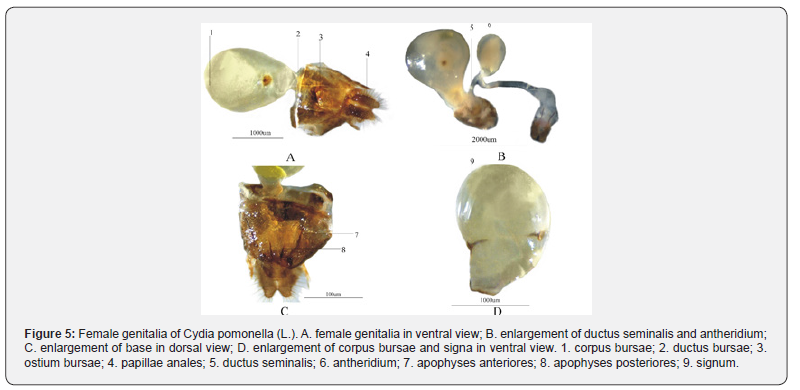
The female external reproductive organs (Figure 3B) consist of a corpus bursa (Figure 5: A-1), a ductus bursa (Figure5: A-2), papillae anales (Figure5: A-4), a ductus seminalis (Figure5: B-5), antheridium (Figure5: B-6) and accessory structure, apophyses anteriores (Figure5: C-7), apophyses posteriors (Figure5: C-8), and two horn-shaped signa (Figure5: D-9). The ductus bursae (Figure 5: A-2) is short, the ostium bursae (Figure5: A-3) is flat and broad. The length of apophyses anteriores is longer than that of apophyses posteriores. Both anus and oviporus have an opening between the papillae anales. The antheridium (Figure5: B-6) is oval and milky and located in the section of the ductus seminalis.
Disscussion
The morphological differences of insect reproductive systems mainly lie on the shape and number of ovary and testis [18, 22], the presence of antheridium, the shape of corpus bursae and signum. In our study of the codling moth, the two testicular follicles are closely connected to each other like a single organ and enveloped in the lavender adventitia, which is similar to those of most species in Lepidoptera [23-26], but inconsistent with the structure of Heliothis zea [27,28] and Papilio xuthus [29]. Moreover, the corpus bursae of the codling moth have a pair of horn-shaped signa instead of cysticerci, but the corpus bursae have cysticerci in Pyrausta nubilalis, Catopsilia Pomona and Colias fieldii [25, 26, 30]. In addition, some species such as Lipographis and Etielloides have no signa [31,32]. Wiesmann (1935) [20] illustrated the accessory gland reservoir being bifurcate, but no reservoirs were bifurcate in our study and nor in Ferro’s sample [4].
In addition, the aedeagus of the codling moth has 7 cornuti, among which five are close to the end and two are in the middle. But Ferro (1975) [4] described 6 large cornuti (spines) that radiate from the endophallus and evert from the aedeagus into the female cervix bursae during copulation. It is probably due to different geographical populations or may be because of intraspecific variations. Moreover, in the process of sexual maturation, we observed that an antheridium is formed in the section of ductus seminalis between corpus bursae and ostiumbursae. Originally, the antheridium is small and transparent, then it becomes large, milky color, and finally turn into large, yellowish color. This feature can also be found in those species in the genus Eurema but missing in other genera of subfamily Coliadinae [26]. In the present study, we have clarified the structural characteristics of both internal and external reproductive organs of the codling moths of a Chinese population. We will conduct the spermatogenesis in the near future in order to study futher the reproductive isolation mechanism and reproductive biology of the codling moth, and to provide more guidance for integrated control them [33-36].
Acknowledgements
We are grateful to our research members Caihong Zhang and Xingbo Wu for their helpful work in field and laboratory. This research was supported by the foundation of College Student's Platform for Innovation and Entrepreneurship of Gansu province (No.201610733002) and the Fund for key course construction of postgraduate of Gansu Agricultural University (No. GSAUZDKC-2016). Program of Introducing Talents to Chinese Universities (111 Program No. D20023) to JJZ.
Notes
All authors declare no conflicts of interest.
References
- Zhang X Z (1957) The new discovery of Carpocapsa pomonella L. in China. Acta Entomologica Sinaca 7: 467-472.
- Wang X T, Piao M H Advanced on quarantine pest codling moth Cydia pomonella in China. Scitific research in Agriculture and Forestry, 250–43+.
- Audemard H (1991) Population dynamics of the codling moth. In: World crop pests. Tortricid pests. Their biology, natural enemies and control. van der Geest LPS, Evenhuis HH [Eds.] Elsevier, Amsterdam: 329-338.
- Ferro D N, Aker R D (1975) Reproductive Morphology and Mechanics of Mating of the Codling Moth, Laspeyresia pomonella. Ann. Entomol. Soc. of America 68(3): 417-423.
- Wood TG (1965) Field observations on flight and oviposition of codling moth (Carpocapsa Pomonella (L)) and mortality of eggs and first-instar larvae in an integrated control orchard. New Zealand Journal of Agricultural Research 8: 1043-1059.
- Salinas Castro A, Sandi MTMP, Ramírez Reyes T, Luna Rodríguez M, Trigos Á (2014) An unusual food plant for Cydia pomonella (Linnaeus) (Lepidoptera, Tortricidae) in Mexico. Revista Brasileira de Entomologia. 2014, 58(3), 261-264.
- Isely D (1938) Codling Moth Oviposition and Temperature. Journal of Economic Entomology 356-359.
- Vickers R A (1997) Effect of Delayed mating on oviposition pattern, fecundity and fertility in codling moth, Cydia pomonella (L.) (Lepidoptera: Tortricidae). Australian Journal of Entomolgy 36: 179-182.
- Saethre MG, Hofsvang T (2002) Effect of Temperature on Oviposition Behavior, Fecundity, and Fertility in Two Northern European Populations of the Codling Moth (Lepidoptera: Tortricidae). Environmental Entomology 31(5): 804-815.
- Blomefield TL, Giliomee JH (2011) Effect of Temperature on the Oviposition, Longevity and Mating of Codling Moth, Cydia pomonella (L.) (Lepidoptera: Tortricidae). African Entomology 19: 42-60.
- Barnes MM (1991) Codling moth occurrence, host race formation and damage van der Geest LPS, Evenhuis HH (Eds.) Tortricid Pests: Their Biology, Natural Enemies and Control. Amsterdam: Elsevier: 313-
- Leather SR, Hardie J (1995) Insect Reproduction. CRC press, London 85 pp.
- Jin T, Feng SZ, Jin QA, Wen HB, Lin YY, et al., (2013) The reproductive system and fecundity of the coconut leaf beetle, Brontispa longissima. Chinese Journal of Applied Entomology 50(5): 1280-1288.
- Chen L, Cai DC, Chen Q, Tang C, Feng G, et al., (2009) The reproductive system and reproductive biology of the allitator weed flea bettle, Agasicles hygrophila (Coleoptera: Chrysomelidae). Acta Entomologica Sinica 52(11): 1255-1260.
- Li DZ, Lou YX, Chen YK, Lian GY, Liu ZG, et al., (2012) A three-dimensional, visualized, digital model of the genital system of the male oriental migratory locust: Locusta migratoria manilansis. Chinese Journal of Applied Entomology 49(1): 248-254.
- Wang XH, Xu HF, Xu YY, Liu Y, Zhou Z (2003) The structures and developmental progress of reproductive system of beet armyworm, Spodoptera exigua (Hübner), and their use in forecast. Acta Phytophylacica Sinica 30(3): 261-266.
- Zhang Z, Men L, Peng Y, Li J, Deng A, et al., (2017) Morphological differences of the reproductive system could be used to predict the optimum Grapholita molesta (Busck) control period. Scientific Reports 7: 1-14.
- Eberhard WG (2009) Evolution of genitalia: Theories, evidence, and new directions. Genetica 138: 5-18.
- Allman SL (1930) Studies of the anatomy and histology of the reproductive system of the female codling moth, Carpocapsa pomonella (Linn.). Univ. Calif. Pub. Entomol 5: 135-64.
- Wiesmann R (1935) Untersuchungen über den weiblichen Genitalapparat, das Ei und die Embryonalent wicklung der Apfelwicklers Carpocapsa (Cydia) pomonella L. Mitt. Schweiz. Entomol. Ges 16: 370-377.
- Liu N, Zhang CH, Zhang Y, Wu XB, Shang SQ (2018) Study on the process of egg maturation and classification of ovary development in codling moths (Cydia pomonella L.). Journal of Fruit Science 35(9): 1098-1104.
- Song HJ, Bucheli SR (2010) Comparison of phylogenetic signal between male genitalia and non-genital characters in insect systematics. Cladistics 26: 23-35.
- Liu KK, Xiao YH, Cheng YX, Luo LZ, Xu X (2013) The structure and morphology of the reproductive system of adult male Loxostege sticticalis L. Chinese Journal of Applied Entomology 50(6): 1692-1699.
- Cao S, Zhang YL (2012) Anatomy of the reproductive system of Pieris melete Ménétriés (Lepidoptera: Pieridae). Journal of Northwest A & F University (Nat. Sci. Ed.) 40(9): 77-82.
- Meng YF, Shang SQ, Zhang YL (2013) The morphology of the reproductive system of Colias fieldii. Chinese Journal of Applied Entomology 50(3): 813-817.
- Wang XF, Shang SQ, Zhang YL (2010) Structure of the female reproductive system of Catopsilia pomona. Chinese Bulletin of Entomology 47(6): 1170-1173.
- Callahan PS, Cascio T (1963) Histology of the Reproductive Tracts and Transmission of Sperm in the Corn Earworni, Heliothis zea. Annals of the Entomological Society of America 56: 535-556.
- Callahan PS, Chapin JB (1960) Morphology of the reproductive systems and mating in two representative members of the family Noctuidae, Pseudaletia unipuncta and Peridroma margaritosa with comparison to Heliothis zea. Annals of the Entomological Society of America 53(6): 763-782.
- Kong H L, Liang XC, Luo YZ (2006) Anatomy of the Reproductive system of Papilio xuthu Linnaeus. Journal of Yunnan Agricultural University 21(4): 459-462.
- Ye YL, Lu JR (1964) The inner reproductive organ and sperms structure of Pyrausta nubilalis Hübner. Chinese Bulletin of Entomology 4: 152-154.
- Ren YD, Li HH (2006) A taxonomic review of the genus Etielloides Shibuya (Lepidoptera: Pyralidae: Phycitinae) from China, with description of a new species. Oriental Insects 40: 135-144.
- Ferris DC (2012) A New Phycitine Species from New Mexico (Pyraloidea: Pyralidae: Phycitinae). Journal of the Lepidopterists' Society 66(2): 76-80.
- Lee GE, Li HH (2018) Ovoviviparity and morphology of the female reproductive system in Palumbina (Lepidoptera, Gelechiidae, Thiotrichinae). Journal of Asia-Pacific Entomology 21(3): 772–777.
- Wang XF (2010) The female reproductive system of subfamily Coliadinae from China (Lepidoptera: Pieridae). Master dissertation. Northwest A & F University, Shaanxi: Yangling.
- Kuang XQ, Calvin DD, Knapp MC, Poston FL (2004) Female european corn borer (Lepidoptera: Crambidae) ovarian developmental stages: their association with oviposition and use in a classification system. Ecology and Behavior. 97(3): 828-835.
- Zhu HF, Wang LY (1997) Fauna Sinica Insecta, Lepidoptera: Sphingidae. Beijing, Science Press 11: 757pp.






























