Physiological traits involved in grazing tolerance of alfalfa genotypes
Mariana Rockenbach de Ávila¹*, Raquel Esteban², Miguel Dall’Agnol³ and José F Morán4
1Embrapa Clima Temperado, Pelotas, RS, FAPEG, Brazil
²University of the Basque Country (UPV/ EHU), Spain
³Department of Forage Plants and Agrometeorology, Federal University of Rio Grande do Sul, Brazil.
4Department of Sciences, Institute for Multidisciplinary Research in Applied Biology (IMAB), Public University of Navarre (UPNA), Spain
Submission: November 24, 2020; Published: December 08, 2020
*Corresponding author: Mariana Rockenbach de Ávila, Embrapa Clima Temperado, Pelotas, RS, FAPEG, Brazil
How to cite this article: Mariana R d Á, Raquel E, Miguel D A, José F M. Physiological traits involved in grazing tolerance of alfalfa genotypes. Agri Res & Tech: Open Access J. 2020; 25 (2): 556303. DOI: 10.19080/ARTOAJ.2020.25.556303
Abstract
Alfalfa [1] is one of the most largely distributed forage legume species in the world and it is used on more hectares than any other forage legume. Even so, alfalfa production in Brazil is still limited by the low persistence of this species, especially when used for grazing. Therefore, the objective of this study was to evaluate and select grazing-tolerant germplasm by evaluating genotypes based on their physiological traits. Eight alfalfa genotypes were grown in two different and simultaneous experiments. The experiment ‘A’ evaluated physiological traits:
a) chlorophyll content
b) sum of lateral roots and shoot and
c) length of the first internode
The experiment ‘B’ consisted of a jasmonate treatment, which induced remarkable effects on alfalfa roots performance. The CRAltR genotype showed the most promising results for length of the first internode 0.429 cm at the last day of evaluation (day 15). Furthermore, this genotype obtained higher rates in the evaluation of chlorophyll. E1C4AltR in addition to being selected in previous studies for grazing tolerance showed variability for decumbent habit and intermediate values for chlorophyll. The genotypes E1C4 AltR and CRAltR presented more promising results and they will be used for further field evaluations in Brazil.
Keywords:Medicago sativa L, Forage legumes, Low persistence, Physiological traits
Introduction
Livestock for beef and dairy in the southern of Brazil are commonly produced on grazing rangelands [2] or on cultivated grasslands [3]. In the context of grassland intensification, Medicago sativa L. (alfalfa) is one of the most largely distributed forage legume species in the world and it is used on more hectares than any other forage legume [4]. Alfalfa is adapted to grow in dry, cool, or warm climates, being tolerant to extreme temperatures [5]. The quality of its herbage has a marked influence on the performance of the grazing livestock. The effects of plant maturity and nutritive value have been evaluated by numerous researchers [6]. Forage legumes boost the quality of forage intake for grazing livestock conferring multifunctional benefits to animal production [7]. In this context, alfalfa is a perennial legume of high interest, as it is rich in protein, calcium, phosphorus, and vitamins [8]. This species is also widely used as hay, though another way of using this legume is by incorporating it to grass pasture seeds. This mixture presents benefits by means of its high yields and its ability to fix nitrogen [9]. Mixtures of alfalfa and grasses reduce the risk of bloat for grazing ruminants [4] and promote a high-quality diet [10]. In southern Brazil, alfalfa grazing is increasing – even if slowly – due to farmers’ investments in soil fertility and the protein efficiency valorization offered by this legume. Even so, alfalfa production in this region is still limited by the low persistence of alfalfa, especially when used for grazing [11]. Alfalfa area could expand in this region if grazing-tolerant cultivars could be developed. Several alfalfa cultivars cannot withstand long-term continuous defoliation and must be rotationally grazed to maintain stands [12].
There is evidence of specific adaptation of decumbent alfalfa to severe continuous grazing [13]. Herbivory or removal of plant tissue can be simulated by treatment with jasmonic acid, which is a natural elicitor of defenses against herbivores [14,15]. It is well already known that plant responses to biotic and abiotic stress stimuli are controlled by defense related hormones [16]; among them, jasmonic acid (JA) is responsible for the elicitation of defenses against attack from herbivores [17]. Furthermore, ecophysiological variables have a great influence not only on the vegetative behavior of cultivated plants but also on their performance regarding productive characteristics [18], being an efficient tool for studying the behavior of plants in response to stress [19]. Assessment of leaf physiological characteristics (chlorophyll content) complement with other analysis [20] was shown to be relevant for the identification of genotypes with differential tolerances to short-term stress [21]. The adaptations to stress can be assessed by instantaneous measurements [22] that provide information about chlorophyll accumulation [23], or desirable agronomic and morphological traits [24], and are usually being applied to assess alfalfa. Besides, the gains of alfalfa yield appear to be primarily due to improved disease resistance [25]. Therefore, some of the genotypes used in this study are the results of a selection of two types of leaf diseases, which were performed by Ávila et al. [26] in field and greenhouse. The objective of this paper was to evaluate and select grazing-tolerant germplasm by evaluating genotypes (preselected in previous studies) by physiological measurement and methyl jasmonate application. This selection was made based on resistance and susceptibility towards two diseases in foliar and forage production.
Materials and Methods
Genotypes
These were the genotypes evaluated: 1= CRAltS; 2= ECAltRC; 3= CRCurSC; 4= ECCurRC; 5= CRCurRC; 6= CRAltRJ; 7= ECAltSC; 8= ECCurSC. The genotypes with the abbreviation AltR mean that they were previously selected for resistance to the leaf pathogen Alternaria alternata and AltS are susceptible (S=Susceptible) to this disease [27]. The same description and abbreviations occur for the genotypes selected for the pathogen Curvularia geniculata (CurR and CurS) [28]. The abbreviation ‘CR’ stands the genotype Crioula, which is the most widely used commercial genotype of alfalfa throughout Brazil [29]. The E1C4 genotype (abbreviation ‘EC’) was selected for the length of first short internode (E1) and selected during 4 cycles (C4). This second genotype (EC) is based on selections previously made for grazing tolerance [30].
Experimental design
Eight alfalfa genotypes were grown in two different and simultaneous experiments from January to July 2016 at the Institute for Research in Multidisciplinary Applied Biology (IMAB) (IdAB) and Universidad Pública de Navarra (UPNA), Spain. The alfalfa seeds were sterilized, mechanically scarified with sandpaper to break dormancy and then placed on water-agar plates in a growth chamber at 25°C until the radicles of the seedlings were about 10 mm long. Subsequently, seedlings were transferred to water-agar plates for seven days and lastly, transferred to their respective experiments (plates or pots) with a nutritive solution. Two experiments were performed. The Experiment ‘A’ was developed in a growth chamber and seedlings were evaluated on plates with a nutrient solution. They were subcultured in Murashige– Skoog (MS) liquid medium [31] enriched with calcium nitrate (Ca (NO3)2) and with pH adjusted to 6.5. The growing conditions were the same as for Esteban et al. [42]. The variables evaluated were:
i. Chlorophyll content
ii. Sum of lateral roots and shoot (Control and Jasmonate)
iii. Length of the first (1st) internode at the vegetative stage.
The plants and treatments from experiment ‘A’ were arranged at random (n= 20), with five plates and four plants on each plate, and every seedling was considered as one replicate of each genotype. To investigate the grazing tolerance, seedlings alfalfa were grown and evaluated in the greenhouse (Experiment ‘B’), and the variables measured were: 1) Growth habit and variables were measured: 1) Growth habit and 2) Fungal foliar diseases (Curvularia geniculata and Alternaria alternata), in order to monitor the pre-selected susceptibility or resistance [26]. Three repetitions for each genotype (total 8) were evaluated (n = 24) using the same type of substrate (organic waste, vermiculite, NPK and Limestone in the composition), temperature (maximum temperature 25 degrees) and irrigation.
Methyl Jasmonate (MeJa) Treatment
After conducting pilot tests according to Bruinsma et al. [40] and Mantyla et al. [44], finally was obtained a final solution of MeJa containing purity >95%.
Experiment ‘A’: growth chamber
The evaluated characteristics in the experiment ‘B’ were: 1) Chlorophyll content; 2) Sum of lateral roots and shoot (Control and Jasmonate) and 3) Length of the 1st internode. The radicles alfalfa was transferred into a plate containing nutritive solution with nitrate as nitrogen source.
To assess the length of the internode and Sum of Lateral roots pictures were taken after 7 days after transfer to nutritive solution plates to measure the size of the first (1st) internode and cumulative sum of Lateral roots and shoot (length in millimetres) from all alfalfa genotypes. Resources of ImageJ software was used to measure both variables. The software’s image analysis techniques allow to accurately measure all parts of the plant [31]. Shorter internode lengths were considered positive because they are indicative of adaptation to grazing. with this character considered as a complementary morphological marker to select alfalfa populations more adapted to grazing [32].
On the 7th day after transfer to nutritive solution plates, chlorophyll content measurements were measured for the alfalfa seedlings. The chlorophyll content was measured at the end of the study because the seedlings were in petri dishes and we needed to manipulate them. A chlorophyll meter (CCM 200, ADC, UK) was used and three readings from each seedling (three leaflets).
Experiment ‘B’: greenhouse
Seed of all genotypes (n=24) were germinated in petri dishes and later transplanted for pots grown with commercial substrate and kept in greenhouse with controlled temperature and humidity. All received the same substrate and irrigation management.
Growth habit
Two measurements of growth habit were realized in all plant at different weeks and stages of plant development (both seedlings with vegetative stage), after 30 and 45 days of cultivation (two measurements) in a controlled environment. Visual estimation was used to assess for erect or decumbent phenotypes.
Leaf lesions
It was established the visual scale methodology for the foliar disease. The percentage of leaf area damaged was visually estimated for all leaflets on each seedling, being eight genotypes and 3 repetitions (n=24). For each seedling, the average of all leaflets was used. This monitoring was carried out to ensure the resistance or susceptibility existing in the genotypes used for use in this study.
Statistical analysis
Statistical analysis was performed by analysis of variance (ANOVA) using the statistical package SAS software V. 9.1. When statistical differences were detected in the analysis of variance at P<0.05, means were compared by the Tukey significant difference (HSD) test at 5% of significant level. The variables were tested for a normal distribution using the Shapiro-Wilk test at P > 0.05.
Results and Discussion
The results demonstrated a difference between genotypes (P<.001) in both experiments ‘A’ and ‘B’. That is, the results related to the growing habit of the alfalfa seedlings evidenced that the genotypes Crioula AltR and Crioula AltS had genetic variability for decumbent habit, ideal when selecting for grazing tolerance. Furthermore, this genotype obtained higher rates in the evaluation of chlorophyll, even with the inclusion of treatment with jasmonate (Figure 1C). In relation to experiment ‘A’, there was no presence of leaf lesions in any of the seedlings (zero), therefore, the data are not presented. It is noteworthy, however, that inside the greenhouse, the temperature and water factors were controlled, which allowed excellent sanity in the plants (100%).
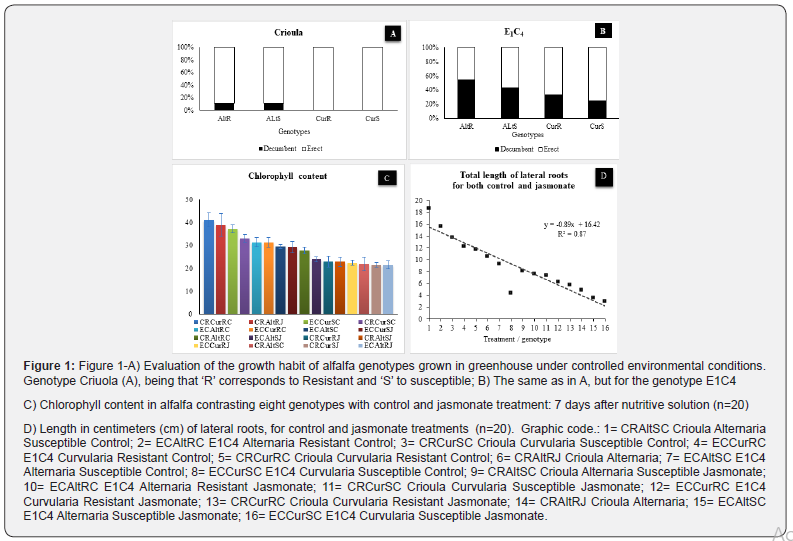
Another promising genotype, for both variables, is the E1C4 AltR, which in addition to being selected in previous studies for grazing tolerance, also showed variability for decumbent habit and intermediate values for chlorophyll. Improved grazing tolerance is often an important goal in forage breeding programs [32]. It is also important to note that this genotype also has the characteristic of resistance to foliar disease (AltR), adding favourable characteristics. Alfalfa has some problems for achieving high production and persistence, some of them related to damage caused by foliar diseases [33,34]. Therefore, it is important to select a genotype that has this resistance characteristic already fixed. Without jasmonate treatment, all genotypes developed the better behaviour in terms of the maximum production roots, except the susceptible genotype with code 8 (Figure 1D). The genotype E1C4, is the result of a selection using the length of the 1st t seedling internode as a morphological marker. With four selection cycles for the shortest trait of the 1st internode, it was possible to identify progress and genetic gain in the selection of this alfalfa germplasm [28].
One of the factors that contributed to the low persistence of forage legumes is the little attention of breeding programs regarding their use for grazing [35], that is why the importance of this work. The Crioula cultivar is the most used in the country for decades, however, it is s considered hay type due to its habit of erect growth and limited tolerance to severe defoliation [36], needing to evolve at that point to expand its use (grazing). According to same author, the length of the seedling internode can be used complementarily to select alfalfa populations with greater grazing aptitude, which would make it possible to obtain populations with higher forage production, drought tolerance and persistencel [37-39]. The CRAltR genotype showed most promising results (P<0.005) for both length of the 1st internode (0.429 cm) at the last day of evaluation. This genotype will be used for subsequent research and selection cycles in the Universidade Federal do Rio Grande do Sul (UFRGS), Experimental Station in Brazil. Taken together, our results showed that there is a high variability in Crioula lfalfa genotypes, which has been proven in studies from different countries [40-45].
References
- Ahmed LQ, Durand JL,EscobarGutiérrez, Abraham J (2019) Genetic diversity of alfalfa (Medicago sativa) in response to temperature during germination. Seed Science and Technology 47(3): 351-356.
- Nabinger C, Ferreira ET, Freitas AK, Carvalho PDF, SantAnna, et al., (2009). Produção animal com base no campo nativo: aplicações de resultados de pesquisa. Campos Sulinos, conservação e uso sustentável da biodiversidade’. Eds VDP Pillar, SC Müller pp. 175-198.
- Fontaneli RS, Meinerz GR, dos SANTOS HP, Biazus V, FAVERO D, Rebechi IDA (2016). A contribuição das forrageiras de inverno para a pecuária de leite. Embrapa Trigo-Capítulo em livro científico (ALICE).
- Smith Jr SR, Bouton JH, Singh A, McCaugheyWP (2000) Development and evaluation of grazing-tolerant alfalfa cultivars: A review. Canadian Journal of Plant Science 80(3): 503-512.
- Barnes DK, CC Sheaffer (1995) AlfalfaIn: RF Barnes, DA Miller, CJ Nelson (Eds.) Forages. Vol.1: An introduction to Grassland Agriculture. Iowa State Univ. Press, Ames lA.
- Beauchemin KA, Iwaasa AD (1993) Eating and ruminating activities of cattle fed alfalfa or orchard grass harvested at two stages of maturity. Canadian Journal of Animal Science 73: 79-88.
- Mallarino AP, WFWedin, CH Perdomo, RS Goyenola, CP West (1990) Nitrogen transfer from white clover, red clover, and birdsfoot trefoil to associated grass. Agronomy Journal 82(4):790-795.
- Pompeu RCFF, Uchoa FC, Neiva JNM, Oliveira Filho GD, Paula Neto FD, et al., (2003) Produção de matéria seca e qualidade de quatorze cultivares de alfafa (Medicago sativa L.) sob irrigação no Estado do Ceará. Embrapa Caprinos e Ovinos-Artigo em periódico indexado (ALICE).
- Van Keuren RW, GC Marten (1972) Pasture production and utilization. In: Alfalfa Science and Technology. Agronomy Monograph 15. pp 641-658. American Society of Agronomy, Madison, WI.
- Tufarelli V, Ragni M, Laudadio V (2018) Feeding forage in poultry: a promising alternative for the future of production systems. Agriculture 8(6): 81.
- Bliss RM (2003) Flowering alfalfa breaks barriers. In: Sowers, Robert (Ed.) Agricultural research. Agricultural Research Service vol. 51, no. 10. US Department of Agriculture, Washington, DC, USA, pp. 8-10.
- Brummer EC, Bouton JH (1991) Plant traits associated with grazing‐tolerant alfalfa. Agronomy Journal83(6): 996-1000.
- Annicchiarico P, Scotti C, Carelli M, Pecetti L (2010). Questions and avenues for lucerne improvement. Czech Journal of Genetics and Plant Breeding 46(1): 1-13.
- Van Kleunen M, Ramponi G, Schmid B(2004) Effects of herbivory simulated by clipping and jasmonic acid on Solidago canadensis. Basic and Applied Ecology 5(2): 173-181.
- Henkes GJ, Thorpe MR, Minchin PE, Schurr U, Roese US (2008) Jasmonic acid treatment to part of the root system is consistent with simulated leaf herbivory, diverting recently assimilated carbon towards untreated roots within an hour. Plant, Cell & Environment 31(9): 1229-1236.
- Pieterse CMJ, LeonReyes A, Van der Ent S, Van Wees SCM (2009) Networking by small-molecule hormones in plant immunity. Nat Chem Biol 5: 308-316.
- Wasternack C (2007) Jasmonates: an update on biosynthesis, signal transduction and action in plant stress response, growth and development. Annu Bot 100(4): 681–697.
- Nogueira RJ MC, Silva Jr JFD (2001) Resistência estomática, tensão de água no xilema e teor de clorofila em genótipos de gravioleira. Scientia Agricola 58(3): 491-495.
- Mills D, Benzioni A (1992) Effect of NaCl salinity on growth and development of jojoba clones: II. Nodal segments grown in vitro. Journal of Plant Physiology 139(6): 737-741.
- Pierre Marraccini, Felipe Vinecky, Gabriel SC Alves, Humberto JO Ramos, Sonia Elbelt, et al., (2012) Differentially expressed genes and proteins upon drought acclimation in tolerant and sensitive genotypes of Coffea canephora. Journal of Experimental Botany 63(11): 4191-4212.
- Paulo EMSilva, Paulo CCavatte, Leandro EMorais, Eduardo FMedina, Fábio MDaMatta (2013) The functional divergence of biomass partitioning, carbon gain and water use in Coffea canephora in response to the water supply: implications for breeding aimed at improving drought tolerance. Environmental and Experimental Botany 87: 49-57.
- Alvarez S, Rodríguez P, Broetto F, SánchezBlanco MJ (2018) Long term responses and adaptive strategies of Pistacia lentiscus under moderate and severe deficit irrigation and salinity: osmotic and elastic adjustment, growth, ion uptake and photosynthetic activity. Agricultural Water Management 202:253-262.
- Badran AE, ElSherebeny EAM, Salama YA (2015) Performance of some alfalfa cultivars under salinity stress conditions. Journal of Agricultural Science 7(10):281-290.
- Acharya JP, Lopez Y, Gouveia BT, de Bem Oliveira I, Resende MF, et al., (2020) Breeding Alfalfa (Medicago sativa L.) Adapted to Subtropical Agroecosystems. Agronomy 10(5):742.
- Lamb JFS, CC Sheaffer, LH Rhodes, RM Sulc, DJ Undersander, etal., (2006) Five decades of alfalfa cultivar improvement: Impact on forage yield, persistence, and nutritive value. Crop Science 46(2):902-909.
- Avila MR, DallAgnol M, Martinelli JA, Silva GBD, Bremm C, et al., (2017) Selection of alfalfa genotypes for resistance to the foliar pathogen Curvularia geniculata. Anais da Academia Brasileira de Ciências, 89(3): 1801-1813.
- Ávila MR, Dallagnol M, Martinelli JA, da Silva GBP, Koshikumo ESM, et al., (2015) First report in Southern Brazil of Alternaria alternata causing Alternaria leaf spot in alfalfa (Medicago sativa). African Journal of Agricultural Research 10(6): 491-493.
- Kopp MM, Pereira AV, Ferreira R de P (2011) Cultivares de alfafa no Brasil. Embrapa Pecuária Sul-Capítulo em livro científico (ALICE).
- SaraivaÁvila KM, de Ávila MR, Dall'Agnol M, SchneiderCanny R,Pereira E (2019) Seleção de alfafa para aptidão ao pastejo. Revista Científica Agropampa 1(1): 93-102.
- Toshio Murashige, Folke Skoog (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Plant Physiol 15(3): 473-479.
- Jinhua L, Zhenqing L, Jizhou R (2005) Effect of grazing intensity on clonal morphological plasticity and biomass allocation patterns of Artemisia frigida and Potentilla acaulis in the Inner Mongolia steppe. New Zealand Journal of Agricultural Research 48(1): 57-61.
- Hopkins AA (2005)Grazing tolerance of cool-season grasses planted as seeded sward plots and spaced plants. Crop Science 45(4): 1559-1564.
- Shangli Shi, Lili Nan, Kevin Smith (2017) The current status, problems, and prospects of alfalfa (Medicago sativa L.) breeding in China. Agronomy 7(1): 1.
- Qin F, Liu D, Sun B, Ruan L, Ma Z, Wang H(2016)Identification of alfalfa leaf diseases using image recognition technology. PLoS One 11(12): e0168274.
- Perez NB, DallAgnol M (2009) Características morfológicas de plantas de alfafa relacionadas à aptidão ao pastejo. Revista Brasileira de Zootecnia, Viçosa 38(3).
- Favero D, SchefferBasso SM, Carneiro CM (2009) Morphological development of Alfagraze cultivar and Crioula alfalfa populations selected at the seedling stage. Revista Brasileira de Zootecnia 38(4): 605-610.
- Strbanovic R, Stanisavljevic R, Dukanovic L, Postic D, Markovic J, et al., (2017) Variability and correlation of yield and forage quality in alfalfa varieties of different origin. Journal of Agricultural Sciences 23(1): 128-137.
- Otero A, Castro M(2019) Variability of Alfalfa (Medicago sativa L.) Seasonal Forage Production in the Southwest of Uruguay. Agrociencia Uruguay 23(1): 1-11.
- Stawarczyk M, Stawarczyk K (2015) Use of the ImageJ program to assess the damage of plants by snails. Chemistry-Didactics-Ecology-Metrology 20(1-2): 67-73.
- Bruinsma M, Posthumus MA, Mumm R, Mueller MJ, van Loon JJ, et al., (2009) Jasmonic acid-induced volatiles of Brassica oleracea attract parasitoids: effects of time and dose, and comparison with induction by herbivores. Journal of Experimental Botany 60(9): 2575-2587.
- Paulo C de F Carvalho, Vivian Fisher, Davi T dos Santos, Andréa ML Ribeiro, Fernando LF de Quadros, et al.,(2006)Produção Animal No Bioma Campos Sulinos. Brazilian Journal of Animal Science 35: 156-202.
- Esteban R, Royo B, Urarte E, Zamarreno AM, GarciaMina JM, et al.,(2016) Both free indole-3-acetic acid and photosynthetic performance are important players in the response of Medicago truncatula to urea and ammonium nutrition under axenic conditions. Frontiers in Plant Science 7:140.
- Daniela Favero, Simone Meredith SchefferBasso, Miguel Dall Agnol,Daniquelen Seco (2008) Desempenho de populações de alfafa sob desfolhação. Revista Brasileira de Zootecnia, Viçosa37(4): 589-595.
- Mäntylä E, Blande JD, Klemola T (2014) Does application of methyl jasmonate to birch mimic herbivory and attract insectivorous birds in nature? Arthropod-Plant Interactions 8(2): 143-153.
- Mortenson MC, GE Schuman, LJ Ingram V Nayigihugu, BW Hess (2005) Forage production and quality of a mixedgrass rangeland interseeded with Medicago sativa ssp. falcata. Rangeland Ecology & Management 58(5):505-513.
- Vough LR, AM Decker, TH Tailor (1995) Forage establishment and renovation. In: R.F Barnes, D.A. Miller, and C J. Nelson (Eds.) Forages. Vol 2: The science of grassland agriculture. Iowa State Univ. Press, Ames, lA.






























