Assessment of the Physicochemical and Microbiological Quality of Sachet Water Sold in Kumbotso LGA, Kano State, Nigeria
Mohammed M1*, Mamudu H Badau2, Amin O Igwegbe2, Florence J Maina3 and Fatima <
1Department of Food Science and Technology, Faculty of Agriculture, Bayero University Kano, Kano State, Nigeria
2 Departent of Food Science and Technology, Faculty of Engineering, University of Maiduguri, Borno State, Nigeria
3Department of Food Science and Technology, Federal Polytechnic, Mubi, Adamawa State, Nigeria
Submission: April 01, 2020; Published: April 08, 2020
*Corresponding author: Mohammed M, Department of Food Science and Technology, Faculty of Agriculture, Bayero University, Nigeria
How to cite this article: Mohammed M, Mamudu H B, Amin O I, Florence J M, Fatima A. Assessment of the Physicochemical and Microbiological Quality of Sachet Water Sold in Kumbotso LGA, Kano State, Nigeria. 2020; 24(2): 556256. DOI:10.19080/ARTOAJ.2020.24.556256
Abstract
The rise in number of sachet water producers in Kano state calls for continuous and vigorous surveillance of the quality of water consumed by the populace to ensure compliance with the WHO and NAFDAC standards for such products. This study assessed the quality of sixteen brands of sachet water sold in Kumbotso LGA of Kano State, Nigeria. Physical assay, physico-chemical, heavy metal contents and microbiological quality assessments of the water samples were carried out. Not all the water samples examined contained any information on their manufacturing and/or expiry dates. The pH, temperature, conductivity and total dissolved solids (TDS) of all the sachet water brands conformed to the WHO standards. All the five heavy metals (arsenic, lead, iron, copper and zinc) analyzed are observed to be within the safety limits recommended by WHO, however Coliforms and some other pathogenic bacteria were detected in 31% of the sachet water brands; leading to the conclusion that 69% of the sachet water sold in Kumbotso LGA of Kano State is safe for human consumption.
Keywords: Kumbotso; Quality; Sachet water; Arsenic; Lead
Introduction
Water is a liquid that is essential to life and makes up a large part of the body. Bateman and Sargeant, (2006) and [1] reported that the human body is made up of more than 65% of water. Man’s continued existence and human physiology depends on the availability of good quality drinking water [2,3]. A large number of the world’s population lacks access to safe drinking water. Of the more than six billion people on earth, more than one billion (one in six) lack access to safe drinking water [1], especially in developing countries such as Nigeria. The adequate supply of clean, fresh and safe drinking water is of great importance to all human beings. Water consumers are frequently unaware of the potential health risks associated with exposure to water borne contaminants, which have often led to diseases like diarrhoea, cholera, dysentery, typhoid fever, Legionnaire’s disease and parasitic diseases [4]. Water quality can be determined using a variety of physico-chemical and microbiological processes. Good quality water should be odourless, colourless or transparent, tasteless, and free from faecal pollution [5]. It appears colorless in small quantities although in larger bodies there is an inherent blue hue [6].
Sachet water, popularly known as ‘pure water’ in Nigeria, is any commercially treated water, manufactured, packaged and distributed for sale in sealed food grade containers and is intended for human consumption [7]. Sachet water was introduced into the Nigerian markets as a less expensive means of accessing good quality drinking water than bottled water [3,8]. However, several studies have shown that packaged water can be contaminated with bacteria at various stages of production [9-11] and during handling. It can also be contaminated with toxic substances depending on the original source of the water. The use of chemical disinfectants in water treatment or construction materials used in water supply system usually results in the formation of the chemical by-products, some of which are potentially hazardous [12,13]. Drinking water can serve as a vehicle for disease transmission, it has indeed, been implicated in many foodborne illnesses [14-16]. [1], reported pH values below the WHO maximum permissible limits of 6.5 in some sachet water samples, this affects disinfection efficiency and may have an indirect effect on human health. Due to the perceived monetary benefits or gains in the production of sachet water, a large number of people have engaged in the business of its production, this can lead to introduction of substandard and fake or misbranded products into the market [11]. Poor manufacturing and handling practices such as production of the water under unhygienic environment, sharp practices, poor hygiene of factory employees and / or vendors and non-adherence to WHO or NAFDAC regulations [4], can lead to production of unsafe sachet water. These concerns led to the need for continuous surveillance of the quality of water consumed in Kano State. Thus, the intent of this study is to assess the physico-chemical and microbiological quality of selected brands of sachet water sold for human consumption in Kumbotso Local Government Area of Kano State, Nigeria.
Materials and Methods
The Sample Collection
Sixteen (16) different brands of sachet water samples were randomly purchased directly from both street hawkers and shops in Kumbotso LGA of Kano State, Nigeria. Samples of similar brands were pooled and analyzed, and in triplicates from different street hawkers and shops in the study area, forty-eight (48) samples were used for this study, they were labeled appropriately. To avoid contamination, the edges of the sachets’ packets were cut using a sterile scissors and emptied into a previously cleaned and sterilized beakers as described by Airaodion et al. [17] and kept at room temperature; samples were analyzed 2 to 4 hours after collection, and the analyses included physical, chemical (assessment for trace and heavy metals) and microbiological quality, analyses were carried out in triplicate.
Water Analysis
All the glassware used in this experiment were washed with distilled water and sterilized in an autoclave at 121oC for 15 minutes. The sachet water packets were physically examined information such as product name, manufacturers address, batch number, NAFDAC registration number, manufacturing and expiry dates were recorded for each sample as described by Oyekuet al. [18] and Airaodion et al. [17].
Temperature and pH Determination
The temperature of each brand of the water samples was measured using a mercury bulb thermometer in oC, whereas the pH of the samples was determined using a digital pH meter (Clida Instruments PHS-25 Precision pH/mV meter). 100ml of each brand of the water samples was poured into a 150ml beaker for the determination. The pH meter was standardized with buffer solutions of pH values between 4 and 9 as described by Igwegbe et al. [19]. Moreover, the pH of the samples was determined by placing the electrode of the pH meter into the samples and recording the readings. The electrode was rinsed several times after each determination with distilled water. The determinations were carried out in triplicates.
Turbidity Test
Turbidity of the water samples was determined using a turbidometer (HANA Instrument HI93703) the values obtained were expressed as Nephelometric Turbidity Unit (NTU) [3,20-22].
Conductivity and Total Dissolved Solids (TDS)
Determination of conductivity was carried out using a digital conductivity meter Model 4520 JENWAY and, the total dissolved solids for each water sample was determined mathematically as a product of conductivity multiplied by a constant value of 0.6 [3,23], that is: TDS = conductivity × 0.6.
Determination of Heavy Metals
The heavy metals analyzed were arsenic (As), iron (Fe), copper (Cu), zinc (Zn) and lead (Pb). Sample collection and handling was as described by Igwegbe et al. [24]; and the metals were quantified using a Microwave Plasma Atomic Emission Spectroscopy (Agilent Technologies 4210 MP-AES, FDGS Innovative Gas Company). This instrument uses a microwave excitation assembly to create a concentrated axial magnetic field around a conventional torch, which focuses the microwave energy where it is needed to produce a toroidal plasma with a cooler central channel that is suitable for stable introduction of liquid samples using a conventional sample introduction system. The intensity of each emitted line is directly proportional to the concentration of a particular metal element [25,26]. An Agilent 4210 MP-AES has a nitrogen plasma gas supplied via an Agilent 4107 Nitrogen Generator. Procedural blanks, calibration solutions, sample solutions, and reference sample solutions were analyzed in that order as described by Vudagandla et al. [26].
Microbial Analysis of the Water Samples
For the bacteriological analysis, Nutrient Agar was used to determine the total bacteria present in the water samples, MacConkey Agar was used for the determination of Coliforms, while Salmonella-Shigella Agar was used to enumerate Salmonella and Shigella [27]. All glassware - including Petri-dishes, test tubes, pipettes, flasks and bottles used in the microbial analysis were sterilized in a hot oven at 170°C for several hours, while the media and distilled water were sterilized by autoclaving at 121°C for 15 min and at 15 psi [24]. Each medium was prepared according to the manufacturer’s instruction. Plating was carried out in triplicate and pour plate method was used to make the viable counts. The samples were incubated at 37°C for 24-48 hours at the end of which the viable colonies in the three plates were counted and the mean was calculated and expressed as colony forming units (cfu/ ml).
Results and Discussion
Results obtained from the physical assessment of the sachet water samples are presented in Table 1. The National Agency for Food, Drug Administration and Control (NAFDAC) requires that sachet water packets must contain information such as product name, manufacturers name and address, NAFDAC registration number, batch number, manufacturing and expiration dates [17,28,29]. All of the sixteen samples analyzed had the product’s name and address of the manufacturer, and NAFDAC registration number boldly written on the packets, while only two samples had the batch number indicated on the packet. 100% of the samples did not contain any information on their manufacturing and expiration date. Lack of information on the manufacturing and expiration dates makes it impossible to know the shelf life of such products, thus posing a threat to health of the consumers when they consume a product that may have passed its best before date. In addition, without any information on the batch number, tracking the source and realling of defective product will be impossible.
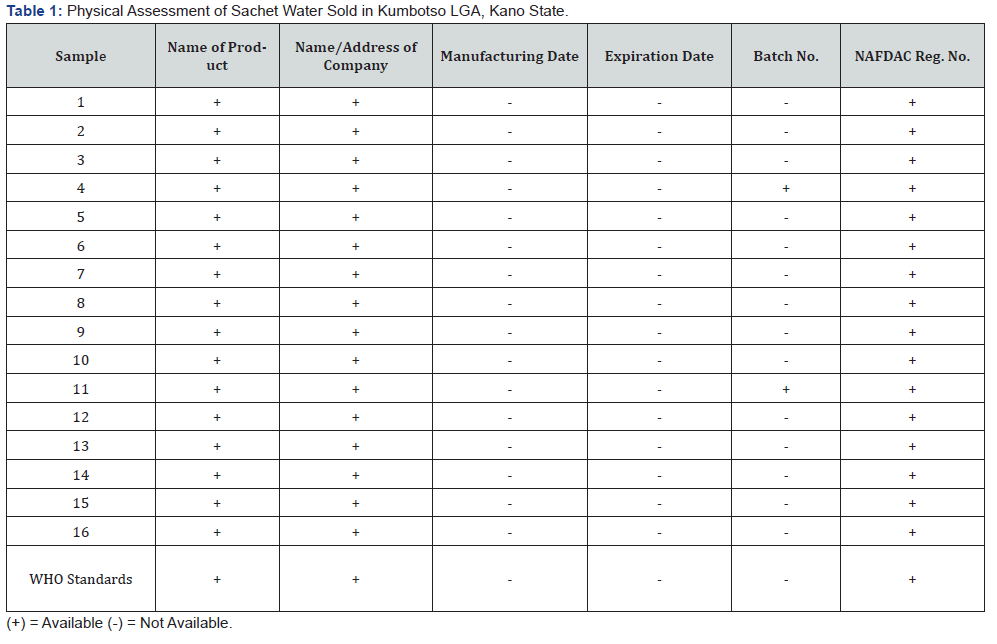
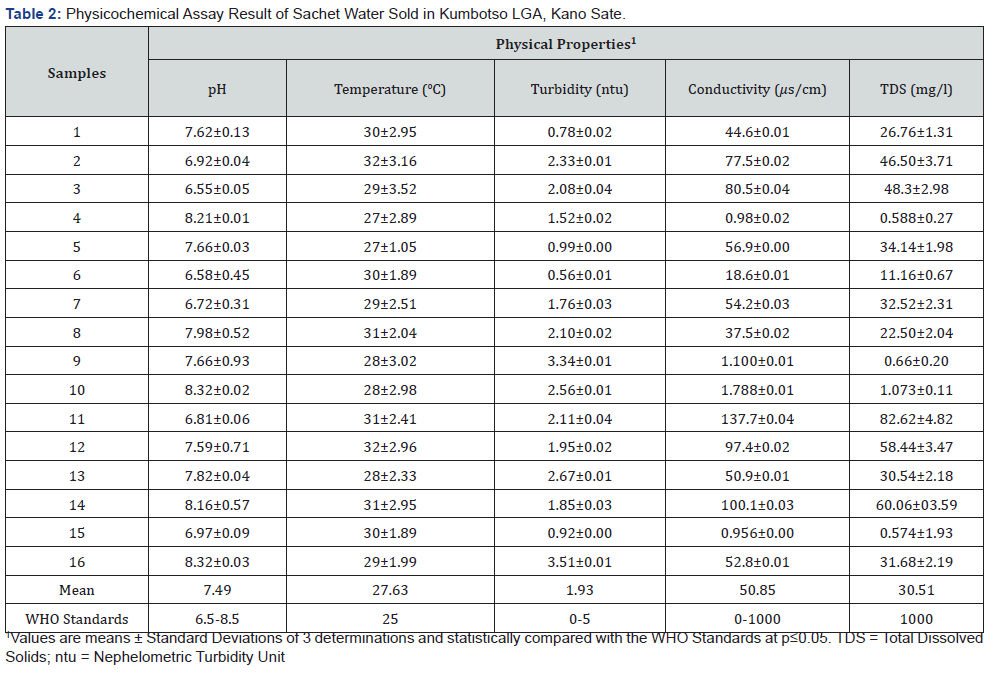
Also, noncompliance to NAFDAC guidelines and sharp practices by some sachet water manufacturers should be a source of great concern to all stakeholders in the food industry because it can lead to easy spread of epidemics such as cholera and typhoid fever. Similarly, the results of the physicochemical assessment of the sachet water samples are shown in Table 2. The pH, turbidity, total dissolved solids and conductivity of the water samples were observed to within the ranges recommended by WHO. The highest and lowest pH values recorded were 8.32 and 6.55, respectively; while the highest value for turbidity was 3.51ntu. On the other hand, the highest and lowest conductivity values recorded were 137.7 and 0.98 μs/cm, respectively; whereas the highest recorded value of the total dissolved solids (TDS) was 82.62mg/l. All the sixteen brands of the sachet water samples had temperatures higher than the WHO standard, this could be due to the fact that most of the brands of the sachet water sold in the study area are transported and stored using open vehicles and under the sun and/or shades, respectively.
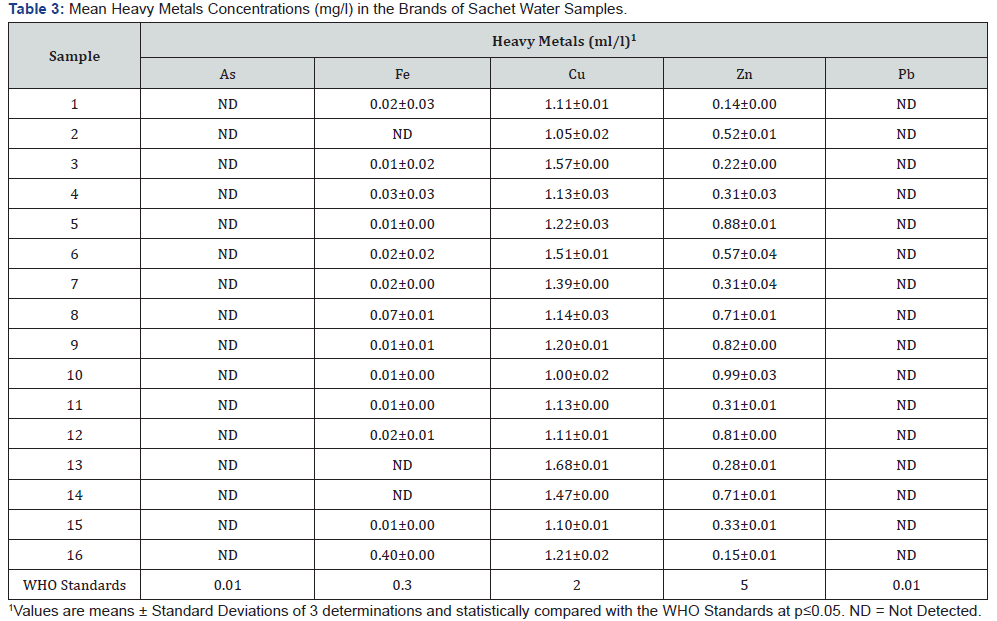
The sources of heavy metals that could contaminate drinking water supplies include industrial and domestic wastes as well as the release of heavy metals into streams, lakes, rivers, and groundwater because of acid rain [30]. In general, a strong relationship between contaminated drinking water with heavy metals and the incidence of chronic diseases such as heart diseases, stroke, cancer, renal failure, liver cirrhosis, hair loss, and chronic anemia has been documented [31]. Arsenic and lead were not detected in any of the brands of sachet water samples as shown on Table 3, while copper and zinc were detected, though below the maximum concentration limits permissible by WHO. However, high level of iron (Fe) was recorded in only one sampled brand. It should be noted that continuous consumption of the water with high level of iron may lead to detrimental effects in humans.
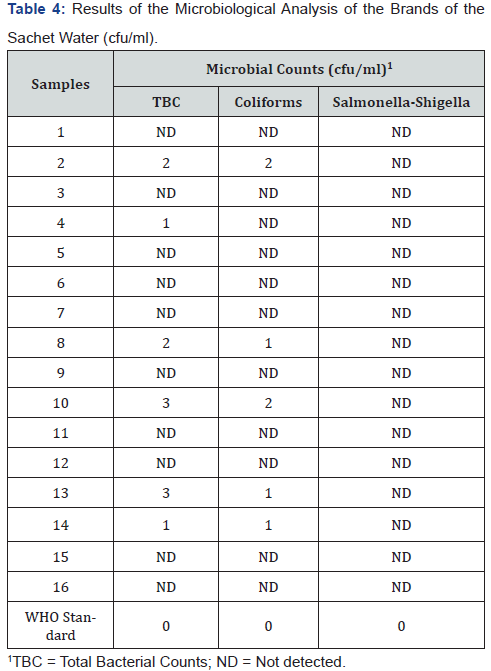
The presence of indicator organisms such as Coliforms in treated drinking water is a measure of its sanitary quality [17,32]. The results of microbiological analysis carried out on the sixteen brands of the sachet water samples presented in Table 4. From the Table, it can be observed that Salmonella and Shigella were not detected in any of the brands investigated however, 31% (five out of the sixteen brands) contained varying numbers of total plate counts including Coliforms. The implication of this result is that the five samples found to contain total bacteria count are the same ones that contained Coliforms, this is an indication that the affected brands of the sachet water were not suitable for human consumption. The WHO standards for these microorganisms were set at 0.00cfu/100ml [17,33]
Conclusion
The physical assay carried out in this study informed us that none of the manufacturers of the sixteen sachet water brands provides all the required information recommended by WHO and NAFDAC for sachet water. Physicochemical analysis such as pH, conductivity, turbidity, total dissolved solids all conform to the WHO standards, whereas the temperatures recorded were all higher than that recommended by the WHO. Four of the five heavy metals (arsenic, lead, copper and zinc) analyzed where within the WHO permissible limits, whereas only one brand of the sachet contained a high-level ofiron (Fe); while five brands varied significantly from the WHO standards. It was concluded that those brands that varied from the WHO Standard were not fit for human consumption because of their contents of not only high total plate counts but also their content of Coliforms. Although, 69% of sachet water brand sold Kumbotso LGA of State of Kano State were found to be safe, there is need for continued surveillance and enforcement of safe drinking water standards to protect the health of the consumers [34,35].
Acknowledgement
We extend our special thanks and gratitude to the Managements of Department of Food Science and Technology, Faculty of Agriculture, Bayero University of Kano, Kano State, and the Department of Food Science and Technology, Faculty of Engineering, University of Maiduguri, Borno State, Nigeria, for providing the resources and enabling environments for the conduct of this study
References
- Uduma AU, Uduma B (2014) Physicochemical Analysis of the Quality of Sachet Water Consumed in Kano Metropolis. American Journal of Environment, Energy and Power Research 2(1): 1-10.
- Lamikanra A (1999) Essential Microbiology for Students and Practitioners of Pharmacy. Medicine and Microbiology, 2nd Amkra books, Lagos pp. 406-410.
- Sheshe MU, Magashi AM (2014) Assessment of Physicochemical Quality of Sachet Water Produced in Selected Local Government Areas of Kano Metropolis. Kano State - Nigeria 7(2): 31-35.
- Omalu ICJ, Eze GC, Olayemi IK, Gbesi S, Adeniran LA, et al. (2010) Contamination of Sachet Water in Nigeria : Assessment and Health Impact. 9(4): 9-11.
- Ezeugwunne IP, Agbakoba NR, Nnamah NK, Anhalu IC (2009) The Prevalence Bacteria in Packaged Sachet Water Sold in Nnewi, South East, Nigeria. World Journal of Dairy and Food Science 4(1): 19-21.
- Roos YH, Finley JW, De Man JM (2018) Water. In: De Man JM, Hurt WJ, Finley JW, Lee CY (Eds.). Principles of Food Chemistry. 4th Springer International Publishing AG. Cham Switzerland pp. 1-38.
- Denloye SA (2004) Quality Parameters for Packaged Water, NAFDAC Laboratory Experience. IPAN News.
- Ogundipe S (2008) Safe Water: So near, yet so far. Vanguard Newspapers (Home Ed.) Section C: 15 (Col. 9 and 10).
- Gangil R, Tripathi R, Patyal A, Dutta P, Mathur KN (2013) Bacteriological Evaluation of Packaged Bottled Water sold at Jaipur City and its Public Health Significance. Veterinary World 6(1): 27-30.
- Dodoo DK, Quagraine EK, Okai Sam F, Kambo DJ, Headley JV (2006) Quality of ‘Sachet’ Waters in the Cape Coast Municipality of Ghana. Journal of Environmental Science and Health-Part A: Toxic Hazardous Substances and Environmen- tal Engineering 41(3): 329-342.
- Halage AA, Ssemugabo C, Ssemwanga DK, Musoke D, Mugambe RK, et al. (2015) Bacteriological and Physical Quality of Locally Packaged Drinking Water in Kampala , Uganda.
- International Agency for Research on Cancer (IARC) (1987) Overall Evaluations of Carcinogenicity. An updating of IARC Monographs volumes 1-42. IARC Evaluation of Carcinogenic Risk to Humans. IARC, lyon. pp. 230-232.
- Amercan Waterworks Association (AWA) (1991) Health Effect of Disinfectants and Disinfection by Products. Denver Co. New York pp. 86-98.
- Degremont J (1991) Water Hreatment handbook. (6th ed). Lavoisier, Paris p. 10-15.
- Jay JM, Loessner MJ, Golden DA (2005) Modern Food Microbiology. 7th Edition, Springer Science + Business Media Inc, New York, USA.
- Reiman HP, Cliver DO (2006) Foodborne infections and Intoxications. 3rd Elsevier Inc, London.
- Airaodion AI, Ewa O, Awosanya OO, Ogbuagu EO, Ogbuagu U, et al. (2019) Assessment of Sachet and Bottled Water Quality in Ibadan. Nigeria.
- Oyeku OM, Omowunmi OJ, Kupoluyi OJ, Kupoluyi CF, Toye OE (2001) Wholesomeness Studies of Water Produced and Sold in Plastic Sachets (Pure Water) in Lagos Metropolis. Nigerian Food Journal (19): 63- 69.
- Igwegbe AO, Kassum AL, Maina FJ, Bristone C, Abubakar F, et al. (2019) Effects of sodium citrate and garlic on pH and microbial stability of smoke-dried meat stored at ambient temperatures. In: Health, Environment and Sustaniable Development: A Book of Readings in Honour of Professor Ibrahim Njodi, Chapter 23. Opara JA (ed.). University of Maiduguri Press, pp. 226-240.
- Essien EB, Olisah AC (2010) Physicochemical and microbiological quality of water samples in three Niger Delta States, Nigeria. Journal of Pharmacy Research 8(3): 1844-1847.
- Dinrifo RR, Babalunde SO, Bankole YO, Demu QA (2010) Physicochemical Properties of Rain Water collected from some Industrial Areas of Lagos State, Nigeria. European Journal of Scientific Research 41(3): 383-390.
- Olaoluwa OJ, Olubukola OA, Deborah DO, Oluwanike O, Oluwaloyin I et al. (2010) Incidence of Drug Resistant Bacteria and Physicochemical Properties of Ero Dam, Nigeria. Report and Opinion 2(12): 78.
- APHA (1985) Standard Methods for the Examination of Water and Wastewater. 19th
- Igwegbe AO, Negbenebor CA, Chibuzo EC, Badau MH (2014) Effects of season and location on heavy metal contents of fish species and corresponding water samples from Borno State of Nigeria. Global Advanced Research Journal of Medicine and Medical Sciences 3(3): 64-74.
- Kamala CT, Balaram V, Dharmendra V, Satyanarayanan M, Subramanyam KSV (2014) Application of Microwave Plasma Atomic Emission Spectrometry (MP-AES) for Environmental Monitoring of Industrially Contaminated Sites in Hyderabad City. Environmental Monitoring and Assessment 186(11): 7097-7113.
- Vudagandla S, Nadavala SK, Vummiti D, Mohammad A, Vysetti, B, et al. (2017) Determination of Boron, Phosphorus, and Molybdenum Content in Biosludge Samples by Microwave Plasma Atomic Emission Spectrometry (MP-AES). Applied Sciences 7(3): 264.
- Kigigha LT, Samson A, Sylvester CI, Ebinyo RA (2018) Microbial Assessment of Zobo Drink Sold in Some Locations in Yenagoa Metropolis. Nigeria 7: 470-476.
- Dada AC (2009) Sachet Water Phenomenon in Nigeria: Assessment of the Potential Health Impacts. African Journal of Microbiology Research. 3(1): 15-21.
- Musa U, Aliyu MA, Sadiq MM, Mohammed IA, Manase A (2014) Quality Assessment of Sachet Water in Minna Metropolis of Niger State, Nigeria. Proceedings of 44th Annual Conference, Exhibition of Nigerian Society of Chemical Engineers (NSChE), Owerri, Imo State, Nigeria.
- Mellor A (2001) Lead and Zinc in the Wallsend Burn, An Urban Catchment in Tyneside, UK. Science of the Total Environment 269(1-3): 49-63.
- Salem HM, Eweida AE, Faraq A (2000) Proceedings of the International Conference for Environmental Hazard MiTigaDamtion. Cairo, Egypt.
- Ashbolt NJ, Grabow WK, Snossi M (2001) Indicators of Microbial Water Quality. In: Fewtrell L, Bartram J (Edts.), Water Quality Guidelines: Guidelines, Standards and Health, World Health Organisation Water Series, IWA Publishing, London, pp. 289-316.
- WHO (2001) Guidelines for Drinking Water Quality: Microbial Methods. (2nd), World Health Organization, Geneva, Switzerland, Europe.
- AOAC (2000) Official Methods of Analysis. 20th AOAC International, Gaithersburg, MD.
- Heather B, Howard SKM (2006) Dictionary of Food Science and Nutrition.






























