Pyroligneous Acid Produced by Rice Husk Using the Charcoal Processing System with Internal Combustion Furnace
Wei-Puo Kuo1*, Yutaka Kitamura2, Yoshiyuki Hara3, Ching-Chen Hsieh4, Yi-Hong Lin1 and Chen-Pin Chen5
1Department of Mechanical Engineering, National Pingtung University of Science and Technology, Taiwan
2Faculty of Life and Environmental Sciences, University of Tsukuba, Japan
3Hokkaido Central Agricultural Experiment Station, Japan
4Biomechatronics Engineering, National Pingtung University of Science and Technology, Taiwan
5QI-MEI Cooperative of friendly farming manager, Taiwan
Submission: December 03, 2019 Published: December 11, 2019
*Corresponding author: Wei-Puo Kuo, Department of Mechanical Engineering, National Pingtung University of Science and Technology, 1, Shuefu Road, Neipu, Pingtung 91201, Taiwan
How to cite this article: Wei-Puo Kuo, Yutaka Kitamura, Yoshiyuki Hara, Ching-Chen Hsieh, Yi-Hong Lin, Chen-Pin Chen. Pyroligneous Acid Produced by Rice Husk Using the Charcoal Processing System with Internal Combustion Furnace.Agri Res& Tech: Open Access J. 2019; 22(4): 556238. DOI: 10.19080/ARTOAJ.2019.22.556238
Abstract
This paper reports the first use of a charcoal processing system with an internal combustion furnace (CPSICF) to produce pyroligneous acid from rice husk, called rice husk vinegar. The gate width in CPSICF influenced air intake, which directly affected the temperature of the furnace that played a key role during pyrolysis. The process characteristics of rice husk vinegar collected from (A) the internal combustion furnace cover and (B) chimney are discussed separately. The total rice husk vinegar weight was 13.13-14.08 kg with cooling water but only 7.23 kg without cooling water. The pH value and acidity ranges were 3.1-3.6 and 5.9%-8.3%, respectively. The tar content was 1.3-3.9 but was 6.1 in the vinegar obtained from the chimney without cooling water. The suspended solid content was 2.1-8.5 mg/L but was 20.6 in the vinegar obtained from the chimney without cooling water. These results are similar to those for other charcoal processing systems.
Keywords:Pyroligneous acid; Rice husk; Rice husk vinegar; Charcoal processing system with internal combustion furnace; pH; Acidity; Tar content; Suspended solid
Introduction
In Taiwan, an island state in East Asia, the 2018 rice yield was 1.56 million tons. This produced 312,000 tons of rice husk, which accounts for approximately 20% of the weight of rice [1]. Rice husk can be classified as complex lignocellulosic material [2]. The husk is a hard-protective layer of the rice grain and can also be used as building material, fertilizer, insulation, or fuel [3]. Regarding its chemical composition, raw rice husk contains both organic (74%) and inorganic constituents (26%) [4]. Pyrolysis is a thermochemical technology for converting biomass into energy and chemical products [5]. Pyrolysis is the thermochemical decomposition of biomass materials at high temperatures and pressures but in an inert or oxygen-free environment [6]. Pyrolysis is divided into three modes: fast, intermediate, and slow. Pyrolysis modes and conditions can influence the relative proportions of the three products (gas, liquid, and solid) obtained [7]. This paper discusses the pyrolysis liquid called pyroligneous acid, which is a crude, condensed, highly oxygenated organic liquid byproduct of pyrolysis [8]. Pyroligneous acid, also called wood vinegar or moussaka, is an aqueous liquid produced from the slow pyrolysis of lignocellulose waste and biomass as a byproduct of charcoal production [9]. Wood vinegar is a complex mixture of polar and nonpolar chemicals with various molecular weights and compositions. The main organic component of wood vinegar is acetic acid [10]. Wood vinegar is widely applied in agriculture and affects plant growth and seed germination. Therefore, wood vinegar is used as a fertilizer and growth-promoting agent in some Asian countries [11]. Many studies have demonstrated similar effects of wood vinegar on regulating plant growth in sustainable agriculture [12], which involves “meeting society’s food and textile present needs without compromising the ability of future generations to meet their needs” [13]. Furthermore, environmentally friendly agricultural practices have drawn global attention as a promising alternative for sustainable food production [14]. In organic agriculture, numerous toxic chemicals are replaced by wood vinegar, a natural product, to combat disease and pests, stimulate plant growth, improve fruit quality, accelerate plant seed germination, and serve as an herbicide [10]. In this experiment, a Charcoal Processing System with an Internal Combustion Furnace (CPSICF) was first used to produce pyroligneous acid. The raw material was rice husk, and the final product was called rice husk vinegar. The quality of rice vinegar was examined to understand the efficiency of the CPSICF.
Materials and Methods
Materials
Rice husk was acquired from the Kaohsiung Cisman Farmer’s Association, and care was taken to avoid significant differences in experimental results due to differences in rice husk quality.
Experimental device
Figure 1 displays a schematic of the CPSICF. The main structure includes a cooling water and exhaust purification tank (1), which was used to store cooling water and purify the exhaust. The chimney (2) collected the exhaust. Rice husk was burned in the Internal Combustion Furnace (ICF, 4), and the exhaust was collected in the ICF cover (3) (Figure 1).
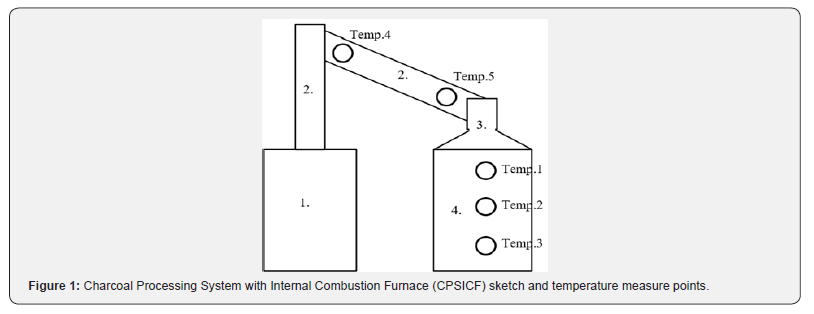
Legends:
1. Cooling water and exhaust purification tank, 2. Chimney, 3. ICF cover, and 4. ICF
Figure 2 is a detailed illustration of the CPSICF cooling system. The difference between chimneys 1 and 2 was the cooling water layer. Cooling water was used to reduce the temperature of the chimney, and the rice husk vinegar condensed in the chimney was collected (8, B). Cooling water was pumped from the tank into the cooling water input port (3). Cooling water filled the chimney (2) and exited through the above cooling water output port (4) to flow back into the tank. Water also exited through a lower cooling water output port (5) and spread on the ICF cover. The cooling water on the ICF cover exited through a cooling water recycle port (6) and back into the tank. The rice husk vinegar condensed on the ICF cover was collected (7, A).
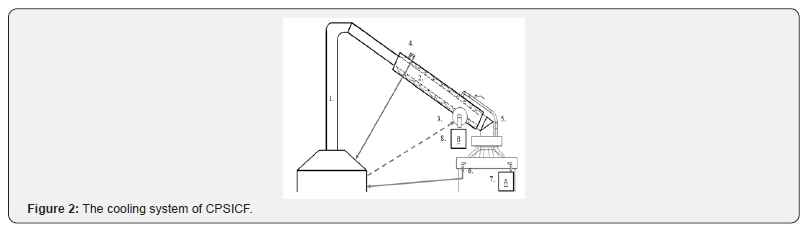
1. Chimney, 2. Chimney with cooling water layer, 3. Cooling water input port, 4. Above cooling water output port, 5. Below cooling water output port, 6. Cooling water recycle port, 7. Rice husk vinegar condensed from ICF cover (A), and 8. Rice husk vinegar condensed in chimney (B)
Experimental procedure
The CPSICF experiment was performed as follows: Approximately 40kg of rice husk was placed in the ICF and ignited with wood charcoal placed in the upper section. After the rice husk ignited, the ICF cover was covered, and the exhaust was collected. The gate width was then adjusted to the appropriate size. Using the CPSICF, rice husk vinegar could be effectively produced, and uncollected smoke could be recycled to prevent air pollution.
The experiment was conducted between April and June 2019 in Cisman District, Kaohsiung City, Taiwan. To prevent the influence of humidity changes, experiments were conducted on days with fair weather and a rainfall probability of less than 10%. The recorded temperature was approximately 23°C-33°C. To prevent external airflow from influencing the intake air volume of the CPSICF opening, the CPSICF was placed in a tin shelter. The key factor in this experiment was the air flow gate width, which affected air intake and ICF temperature. Temperature, a key factor of pyrolysis, can affect the yield and quality of rice husk vinegar [15]. Gate widths of 5, 7.5, and 10 mm were studied three times. To understand the efficiency of the cooling system, a control experiment was performed with a 5-mm gate width without cooling water. The experiment time was controlled at 21 h. The yield and quality of rice husk vinegar are discussed separately because of different collecting methods.
Temperature
Temperature measurement points, presented in Figure 1, were set in the furnace (Temp. 1-3) and recorded using a Portable Data Station (Yokogawa Electric Co., Ltd, Japan; Yokogawa XL122-D) during the experiment. In addition, the temperature of the chimney was measured at the points Temp. 4 and Temp. 5 to ensure the cooling water function of the chimney.
pH and Total Acid Content (Acidity)
The pH value was measured using a pH meter (Thermo Fisher Scientific Inc., USA; model Orion Star A329) at a liquid temperature of 20°C. The total acid content (acidity) was evaluated through titration with 0.1 N sodium hydroxide (NaOH). The content of acetic acid (CH3COOH), a major organic acid in wood vinegar, was calculated using the titrated sodium hydroxide volume [16].
Tar content
The tar content was measured by boiling 100mL of the product in a glass beaker; the residue was tar. The amount of total soluble tar indicated the presence of phenolic compounds, which may have antifungal activity and applications as wood preservatives [17].
Suspended solids
The sample (20g) was filtered through a reweighed standard glass fiber filter. The filter was then dried, and the residue on the filter was retained in a 103°C-105°C oven (CHANNEL BUSINESS CO., Ltd Taiwan; model DV-902) for 1 h. The increase in filter weight indicated suspended solids [18].
Results and Discussion
Temperature Change in ICF (Figure 3)
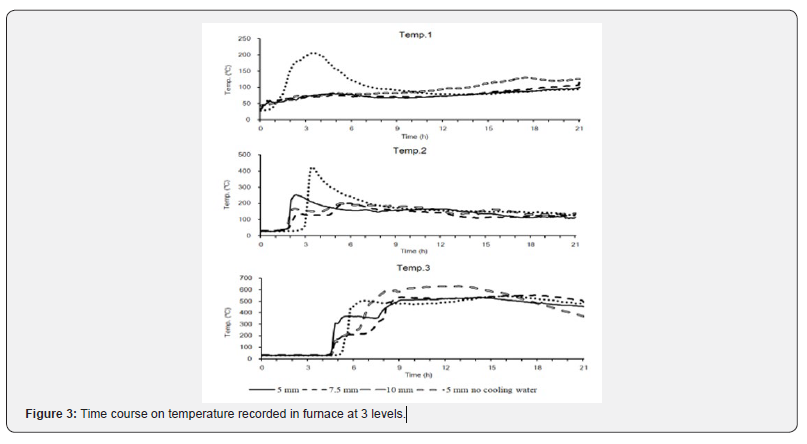
Temperature was measured at the measurement point Temp. 1 in the top layer of the furnace. Temperature increased in the beginning. With cooling water, temperature gradually increased from room temperature to 94.5°C. The differences among gate widths were nonsignificant. Without cooling water, temperature increased severely from 29.0°C to 206.0°C over 3h 30min, decreased to <95°C after 8h, and remained in the range of 78°C-95°C. Temperatures under both conditions demonstrated a similar trend after 12h. Temperature was measured at the measurement point Temp. 2 in the central layer of the furnace. With a 5-mm gate width, temperature increased from 32.0°C to 207.7°C from 3h to 12h 25min and then decreased slowly. With a 7.5mm gate width, temperature increased from 31.5°C to 328.2°C from 2h 20min to 5h 55min and then decreased slowly. With a 10-mm gate width, temperature increased from 33.8°C to 293.0°C from 1h 40min to 5h 30min, decreased to approximately 200°C at 7 h 50 min, and remained between 180°C between 200°C until 10h 20min, after which temperature decreased rapidly. Without cooling water, temperature increased from 30.6°C to 424.0°C from 2h 20min to 3h 25min and then decreased rapidly. After 7h, temperature gradually decreased until the end of the experiment. Temperature was measured at the measurement point Temp. 3 in the bottom layer of the furnace. Temperature started increasing rapidly at 6h 50min, 7h 45min, 6h 5min, and 5h 15min with gate widths of 5, 7.5, 10, and 5mm without cooling water, respectively. The highest temperatures under each condition were 579.8°C, 570.5°C, 583.4°C, and 543.6°C. With a 5-mm gate width, temperatures were maintained at more than 500°C from 9h 5min to 17h 45min and then decreased slowly. With a 7.5-mm gate width, temperatures more than 500°C were maintained from 14h 35min until the end. At a gate width of 10 mm, temperatures were maintained at more than 500°C from 7h 45min to 14h 55min and then decreased rapidly. At a gate width of 5mm without cooling water, temperatures were maintained at more than 500°C from 12h 40min to 19h 20min and then decreased slowly. Rice husk was burned from the top to the bottom of the furnace. In the top and central layers, cooling water exhibited an extreme effect on temperature, which was highest with a 5-mm gate width without cooling water. In the bottom layer, the gate width affected heating rates; with gate widths of 10 and 5mm without cooling water, peak temperatures were reached earlier than with gate widths of 5 and 7.5mm. Furthermore, at a gate width of 10mm, the temperature in the bottom layer decreased rapidly at 14h 55min, which indicated that the burning condition was decreasing. The decrease in sustained burning time might affect pyrolysis results.
Temperature change on the chimney (Figure 4)
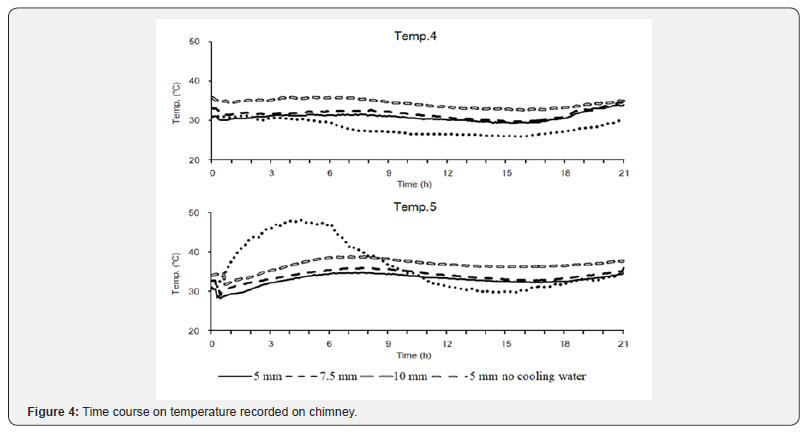
Temperature was measured at the point Temp. 4 above the chimney. The temperature change was similar to room temperature, which ranged from 23°C to 33°C. Temperature measured at Temp. 4 was not significantly different among different conditions. Temperature was measured at Temp. 5 at the bottom of the chimney. With cooling water, the temperature change among different gate widths was not significantly different. However, without cooling water, temperature increased at the beginning, reached a peak of 48.2°C after 4h 35min, and then decreased gradually. Compared with that measured at Temp. 1 in (Figure 3), without cooling water, temperature at the top of the furnace affected that at the bottom of the chimney. The cooling system could ease the burning condition and reduce temperature at the bottom of the chimney.
Weight of rice husk vinegar
Table 1 presents the relationship between gate width and physical-chemical characteristics of the rice husk vinegar. In the experiments with cooling water, the weight of A, rice husk vinegar condensed on the ICF cover, was three times more than that of B, rice husk vinegar condensed in the chimney (Figure 4). The weight change of B was nonsignificant also the chimney temperature in Figure 4. The largest total amount was collected when the gate width was 7.5 mm (14.08kg), and that collected at a gate width of 5 mm (13.92kg) was slightly less. However, the yield with a 10-mm gate width (13.12kg) was 7% less than that with a 7.5-mm gate width. Furthermore, at a 5-mm gate width without cooling water, the total yield (7.23kg) was nearly half the amount in other conditions. The weight of A (2.09kg) was less than half of that of B (5.14kg), which was different from the condition with cooling water. Comparing temperature changes in the furnace and chimney with the rice husk vinegar weight, the results revealed that cooling water that affected the temperature of the furnace and chimney also affected the weight of the rice husk vinegar (Table 1). Without cooling water, temperature in the top layer was higher than that in other conditions; this affected the amount of rice husk vinegar condensed on the ICF cover. The rest of the rice husk vinegar condensed on the chimney. Higher heating rates tend to favor higher wood vinegar yields [19]. Than & Suluksna [20] reported that increasing primary airflow initially increased each of the process rates but caused a decrease in the rates beyond a certain point. From the experimental results and studies, a gate width of 10 mm is confirmed to cause overheating.
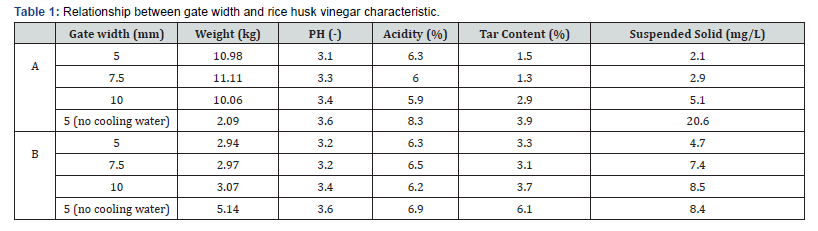
pH Value and acidity of rice husk vinegar
The pH value was not significantly different between A (3.1- 3.6) and B (3.2-3.6). The pH value increased as the gate width became larger. Moreover, the highest pH was observed in the 5-mm gate and no cooling water condition. Wood vinegar pH increased as temperature increased [21]. Vinegars with lower pH values are preferred because of their higher antibacterial abilities [22]. The acidity of the vinegar obtained from each gate width test is listed in Table 1. Comparing with Figure 3, the acidity of A without cooling water decreased as the gate width and temperature in the ICF increased. This result is consistent with the research result of Budaraga et al. [22] that total acid value decreased as pyrolysis temperatures increased. There was no interaction observed between the acidity of B and gate width. Moreover, the acidity of vinegar produced with a 5-mm gate width without cooling water was more obvious than that in the condition with cooling water. The pH value and acid content were weakly correlated because of a complex effect among the various pyrolyzate components [16].
Tar content of rice husk vinegar
Under all conditions, the tar content of B (3.1 to 3.7 and 6.1) was almost twice that of A (1.3 to 2.9 and 3.9). The least tar content was observed with a gate width of 7.5mm. Japan Pyroligneous Liquor Association, an industrial body for pyroligneous liquor traders, established that the tar content of good-quality wood vinegar should not exceed 3% [10]. The tar content of the vinegar obtained with a gate width of 5 mm without cooling water and the tar content of B did not meet this specification.
Suspended solids
Suspended solids increased as the gate width became larger. For the 5-mm gate width with and without cooling water, the suspended solids increased 10 times in A but only twice in B. Compared with (Figure 3), the more obvious the burning condition, the higher the suspended solid content would be.
Conclusion
This experiment used rice husk as raw material and a CPSICF to produce rice husk vinegar. The physical properties of rice husk vinegar obtained in this experiment were analyzed in two parts, A and B, and included weight, pH value, acidity, tar content, and suspended solids. The process characteristics are as follows:
a) The total weight of the rice husk vinegar was 13.13- 14.08kg but only 7.23kg without cooling water. The pH value,acidity, and tar content were 3.1-3.6, 5.9%-8.3%, and 1.3%- 6.1%, respectively. The suspended solid content was 2.1-8.5 mg/L but reached 20.6 mg/L in A without cooling water.
b) The temperature change during pyrolysis affected the physical properties of the rice husk vinegar. The operating procedure for the CPSICF was relatively easy, and the quality could be efficiently controlled by adjusting the gate width.
c) The quality of the rice husk vinegar obtained from the CPSICF with 5- and 7.5-mm-wide gates did not differ substantially. Quality decreased when the gate width was 10 mm. Moreover, without cooling water, rice husk vinegar quantity and quality were significantly reduced. With cooling water, one must only adjust the gate width: a gate width of 5-7.5 mm provides high-quality rice husk vinegar.
d) To achieve the goal of sustainable agriculture, the CPSICF shows potential in converting biomass into valuable products. Thus, the utility of rice husk vinegar produced by the CPSICF should be discussed in additional studies.
Acknowledgment
The author is grateful to Dr. Ching-Chen Hsieh (Biomechatronics Engineering, NPUST) for supporting this study in its entirety. This manuscript was edited by Wallace Academic Editing.
Conflict of Interest
The authors declare has no conflict of interest.
References
- Pacheco-Torgal F, Lourenco PB, Labrincha J, Chindaprasirt P, Kumar S (2014) Eco-efficient masonry bricks and blocks: Design, properties and durability (Woodhead Publishing Series in Civil and Structural Engineering Book 55). (1st), Woodhead Publishing, Cambridge, MA, USA p. 202-205.
- Risfaheri R, Hoerudin H, Syakir M (2018) Utilization of rice husk for production of multifunctional liquid smoke. J Adv Agric Technol 5(3): 192-197.
- Padinjakkara A, Thankappan A, Souza FG, Thomas S (Editors.) (2018) Biopolymers and biomaterials. (1st Edition), Apple Academic Press, New York, NY, USA pp. 147-199.
- Sankar S, Sharma SK, Kaur N, Lee B, Kim DY, et al. (2016) Biogenerated silica nanoparticles synthesized from sticky, red, and brown rice husk ashes by a chemical method. Ceram Int 42(4): 4875-4885.
- Kan T, Strezov V, Evans TJ (2016) Lignocellulosic biomass pyrolysis: A review of product properties and effects of pyrolysis parameters. Renew Sustain Energy Rev 57: 1126-1140.
- Yorgun S, Yildiz D (2015) Slow pyrolysis of paulownia wood: Effects of pyrolysis parameters on product yields and bio-oil characterization. J Anal Appl Pyrolysis 114: 68-78.
- Angin D (2013) Effect of pyrolysis temperature and heating rate on biochar obtained from pyrolysis of safflower seed press cake. Bioresour Technol 128: 593-597.
- Grewal A, Lord A, Gunupuru LR (2018) Production, prospects and potential application of pyroligneous acid in agriculture. J Anal Appl Pyrolysis 135: 152-159.
- Tiilikkala K, Fagernäs L, Tiilikkala J (2010) History and Use of Wood Pyrolysis Liquids as Biocide and Plant Protection ProductHistory and Use of Wood Pyrolysis Liquids as Biocide and Plant Protection Product. Open Agric J 4(1): 111-118.
- Theapparat Y, Chandumpai A, Faroongsarng D (2018) Physicochemistry and utilization of wood vinegar from carbonization of tropical biomass waste. In: Sudarshana P, Nageswara-Rao M, Soneji JR (Editors.), Tropical forests - New edition, IntechOpen, London, UK 163-183.
- Mingfenga W, Enchen J, Leiming X, Xiwei X, Chuang Z, et al. (2013) Components characteristics of wood vinegar from rice husk continuous pyrolysis and catalytic cracking. Appl Mech Mater 291-294: 368-374.
- Lu X, Jiang J, He J, Sun K, Sun Y (2019) Pyrolysis of Cunninghamia lanceolata waste to produce wood vinegar and its effect on the seeds germination and root growth of wheat. BioResources 14(4): 8002-8017.
- (2019) What is sustainable agriculture Retrieved from. Sustainable Agriculture Research and Education Program website:
- Lee JJ, Shon TK, Furuya T, Jin ID, Chung IK, et al. (2007) Effect of different kinds of environmental friendly materials on the growth of rice. J Fac Agr Kyushu U 52(1): 39-42.
- Hanif MU, Capareda SC, Kongkasawan J, Iqbal H, Arazo RO, et al. (2016) Correction: Effects of pyrolysis temperature on product yields and energy recovery from co-feeding of cotton gin trash, cow manure, and microalgae: A simulation study. PLoS One 11(7): e0156565.
- Mun SP, Ku CS, Park SB (2007) Physicochemical characterization of pyrolyzates produced from carbonization of lignocellulosic biomass in a batch-type mechanical kiln. J Ind Eng Chem 13(1): 127-132.
- Theapparat Y, Chandumpai A, Leelasuphakul W, Laemsak N, Ponglimanont C (2014) Physicochemical characteristics of wood vinegars from carbonization of leucaena leucocephala, azadirachta indica, eucalyptus camaldulensis, hevea brasiliensis and dendrocalamus asper. Kasetsart J - Nat Sci 48: 916-928.
- (2017) American Public Health Association, American Water Works Association WEF. Total suspended solids dried at 103–105ᵒC. In: Eaton AD, Rice EW, Baird RB (Editors.), Standard methods for the examination of water and wastewater (23rd Edition, Part 2540), American Public Health Association, American Water Works Association, Water Environment Federation, Washington, DC, USA 4-6.
- Ratanapisit J, Apiraksakul S, Rerngnarong A, Chungsiriporn J, Bunyakarn C (2009) Preliminary evaluation of production and characterization of wood vinegar from rubberwood. Songklanakarin J Sci Technol 31(3): 343-349.
- Than P, Suluksna K (2019) Experiment on influent parameters on wood vinegar burning process. IOP Confe Ser Mater Sci Eng 501(1): e012066.
- Hwang GS, Lee CM, Ho CL, Wang CH, Yu HM (2017) Study of charcoal making with michelia compressa thinned wood from afforestation on the plains. Taiwan J For Sci 32(1): 43-55.
- Budaraga K, Arnim, Marlida Y, Bunin U (2016) Liquid smoke production quality from raw materials variation and different pyrolysis temperature. Int J Adv Sci Eng Inf Technol 6(3): 306-315.






























