Simultaneous Biosorption of Chromium (III) and Chromium (VI): Application of Multiple Response Optimizations
John Babu D1Sumalatha B2,Venkata Narayana A1,Venkateswarulu T C1,King Pulipati3, Ranga Rao A1*
1Department of Biotechnology, Vignan’s Foundation for Science Technology and Research, Andhra Pradesh, India.
2Department of Chemical Engineering, Vignan’s Foundation for Science Technology and Research, Andhra Pradesh, India.
3Department of Chemical Engineering, Andhra University College of Engineering, Visakhapatnam, Andhra Pradesh, India.
Submission:October 16, 2019Published: November 06, 2019
*Corresponding author:A Ranga Rao, Department of Biotechnology, Vignan’s Foundation for Science Technology and Research, Andhra Pradesh, India. Agri
How to cite this article:John Babu D, Sumalatha B, Venkata Narayana A, King Pulipati, Ranga Rao A. Simultaneous Biosorption of Chromium (III) and Chromium (VI): Application of Multiple Response Optimizations. Serrano.Agri Res& Tech: Open Access J. 2019; 22(4): 556230. DOI: 10.19300/ARTOAJ.2019.22.556230
Abstract
This work aims, optimization of simultaneous biosorption of Cr(III) and Cr(VI) onto Gelidilla acerosa, a marine macro algae using multiple response optimization combined with central composite design of design of experiments. The three important physicochemical parameters of biosorption such as initial pH, initial concentration of metal solution and biosorbent dosage were optimized. The most favorable condition for simultaneous removal of Cr(III) and Cr(VI) were: pH 2.8, initial concentration 20 mg l-1 and biosorbent dosage 0.05g with maximum overall desirability of 1.0. Surface morphological features of biosorbent were studied using FTIR. The nature of biosorption was analyzed by fitting the equilibrium data to various adsorption isotherms and found that both Langmuir and Freundlich model were best fitted with R2: 0.99 for both chromium species. The maximum biosorption capacities predicted from Langmuir model were 285.71 mg g-1 for Cr(III) and 270.27 mg g-1 for Cr(VI). The pseudo-second order kinetic model was found to be proper approach to demonstrate biosorption kinetics indicating chemisorption with film diffusion as the rate controlling mechanism. The results demonstrated that multiple response optimization combined with central composite design using desirability function was best approach for optimization of simultaneous biosorption of Cr(III) and Cr(VI) from aqueous medium.
Keywords:Biosorption; Chromium; Central composite design; Multiple response optimizations
Introduction
Water contamination with hazardous heavy metals is one of the most important issues due to its lethal effects to human beings and aquatic ecosystem. The key root of this heavy metal contamination is intensified industrial expansion as every industrial runoff carries toxic heavy metals at different concentrations. Chromium (Cr) is the one of the most profuse metals in the earth’s crust and has gained ample attention due its potential toxic nature to human health and envir [1,2]. Though chromium exists in different oxidation states ranging from 0 to +6, hexavalent (Cr(VI)) and trivalent(Cr(III)) oxidation states have significant importance as these two states are most stable in the natural environment [3]. At lower concentrations, Cr(III) is an essential element in the body metabolism as it plays a significant role in the insulin regulation. Little dose of Cr (III) is used in supplements to treat diabetes which is known to enhances the action of insulin [4,5]. Prolonged exposure to higher concentrations of Cr(III) becomes toxic and leads to skin allergic reactions. Moreover, the microorganisms of aquatic environment may oxidize Cr(III) into Cr(VI) which further enhances the toxicity (Sari et al. 2001). Contrary to Cr(III), Cr (VI) species have high solubility and mobility in water, has no biological activity and penetrates into the human body 10,000 times quicker than Cr (III). Moreover Cr (VI) is mutagenic, carcinogenic and teratogenic to humans, animals and alters plant morphology [6-8].
Chromium is widely used in various industrial and household applications. Across the globe 80% of chromium is used in metallurgical applications [1]. In view of hazards nature of chromium species to human health and economic importance of recovering chromium from industrial wastewater, several treatment processAbstract es such as precipitation by alkaline treatment, electrochemical precipitation, ion exchange and adsorption onto nanofibers and activated carbon have been used to remove chromium from contaminated wastewater. However, all these treatment methods are either inefficient or expensive. In addition, these techniques may also generate toxic secondary sludge which needs further expensive sludge disposal strategy [9-11]. On the other hand, adsorption was emerged as a promising technology to treat large quantities of dilute effluents due to its viability, simplicity, inexpensive and eco-friendly nature [12,13]. Biosorption is adsorption of pollutants using naturally available biomasses such as agro waste, algae, microbial consortium and digested sludge. In recent decades biosorption emerged as an effective technique for removal of pollutants from dilute effluents [14,15]. Algae as a renewable, naturally abundantly available biomass, it is well reported by researchers as biosorbent for wastewater treatment. Some marine and freshwater algae (biosorption of Au, Ag, and Co) showed higher biosorption potential than synthetic adsorbents like activated carbon, nanofibers and ion-exchange membranes [16]. It was also reported that in case of highly toxic metals the uptake capacities of brown algae are higher than other algae (green and red algae) [17]. Hence marine brown algae Gelidilla acerosa(G.acerosa) was selected as biosorbent for this study.
In statistical optimization using response surface methodology (RSM), experimental matrix is generated from design of experiments (DOE), experimental results are fitted to various mathematical models and statistical analysis tools are used to verify the fitness of model and interactions among various variables and responses. Optimization of single response is comparatively simple and requires a smaller number of experiments [18]. However, in many times, more than one response must be optimized simultaneously. In multiple responses optimization, RSM is merged with Derringer’s desirability function. The desirability function transforms multiple responses into a single response case (desirability value), which can be optimized using univariate techniques [19].
Thus, this work aims to simultaneous biosorption of Cr(III) and Cr(VI) using Gelidilla acerosa (G.acerosa) as biosorbent. Independent process variables (pH, initial concentrations of chromium species and biosorbent dosage) were optimized by Central Composite Design (CCD) and multiple response optimizations. Biosorption kinetics and isotherms were also studied.
Materials and Methods
Chemicals and biosorbent
Stock solutions of Cr(III) and Cr(VI) (1000ppm) were prepared by dissolving 11.690 & 3.373 g of Cr2(SO4)3.12H2O & K2CrO4 in 1000ml of Double distilled water. Working samples of both chromium solutions were prepared from stock solutions by making accurate dilutions for each batch experiment. Brown marine macro algae (G.acerosa) was collected from Gulf of manar, Tamil Nadu, India. Collected algal plants were washed several times with normal and distilled water. Sundried algae were grounded and sieved using standard mesh. Raw biomass without any further treatment was used as biosorbent.
Experiments
The biosorption experiments were conducted by taking 100ml of working samples of different concentrations (20 to 100ppm) in an Erlenmeyer flask, pH (1 to 5) was adjusted with 0.1 normal HCl and NaOH and then pre-determined quantity of alga biomass (0.01 to 0.05g) was added. Solutions were shaken in orbital shaker at 120rpm speed. After filtration, the filtrate was analyzed for remaining chromium using Atomic adsorption spectroscopy. The chromium uptake was calculated as follows:
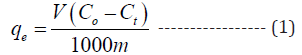
where C0 and Ct (mg l-1) are the initial and equilibrium chromium concentrations, qe is chromium uptake (mg g−1), V is working sample volume (ml) and m is weight of biosorbent (g). Triplicate experiments were performed, and average values were reported

RSM is a multi-variable optimization tool used to find the optimal response of a process which is a function of several independent variables through fitting the experimental results to polynomial equations. The three major steps of RSM include design of experimental matrix, development of mathematical model and optimization of response of interest [20,21]. In this study, pH (X1), initial chromium concentration (X2) and dosage of biosorbent (X3) are independent variables and uptakes of Cr(III) Y1 and Cr(VI) Y2 are response variables. Experiments were designed using face centered central composite design (FCCCD) of RSM. The ranges and levels (-1, 0, +1) of independent variables are presented in Table 1. The regression equation relating the independent variables and the response of interest i.e., chromium uptake is presented in the form of following equation (2).
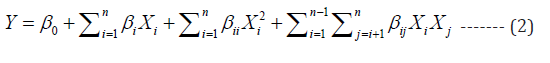
Where Y is response of system, β0 is constant, βi is linear constant, βii is quadratic constant and is independent variable. MINITAB16 was used for experiments design, response fitness analysis and simultaneous response optimization. In multiple responses optimization, the responses were converted into equivalent desirability value (di) which varies from 0 (lowest) to 1 (highest desirability). In this study, maximum metal uptake is the desirability. The individual desirability function (di(Yi) ) is shown below
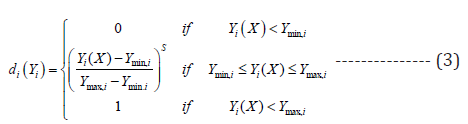
where Ymin,i and Ymax,i are minimum and maximum acceptable value of i Y , respectively. The exponent s is scale of desirability. The desirability function is linear, when s=1. The individual desirability functions are then combined as the geometric mean for calculating overall desirability function (D):
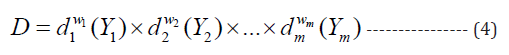
where di (Yi ) and are the desirability of response and weight of various responses, respectively (Liu and Tang 2010). In present study, Cr(III) and Cr(VI) biosorption efficiencies had equal weight ( w1= w2 = 1/2).
Kinetics of biosorption
Kinetic modeling of biosorption is very much essential to scale-up the process for industrial-scale operations. Moreover, kinetics of a reaction describes nature of the process, reaction pathways and exact interface resistance for mass transfer. Hence, fresh experiments were conducted for kinetics analysis by taking 100ml solutions (20 mg l-1) chromium species with biosorbent dosage of 0.01g at corresponding optimum pH and the data was fitted to pseudo-first-order, pseudo-second order, and intra-particle diffusion models.
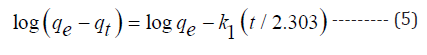
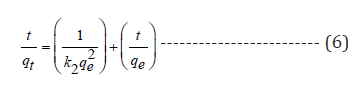

Where k1 and k2 are the first and second order rate constants, qe (mg g-1) and (mg g-1) are chromium uptake at equilibrium and time t(min) respectively, kid is diffusion constant, C is the constant related to film thickness.
Biosorption isotherms
Experimental equilibrium data was generated by conducting a set of experiments at room temperature, optimum pH and biosorbent dosage with variable initial metal ions concentrations and fitted to the Langmuir, Freundlich and Temkin isotherms and details of each isotherm are given below:
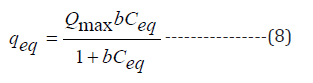

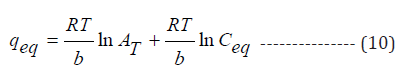
Temkin constant (l mg-1), b(J mol-1) is heat of sorption, R is universal gas constant (8.314 J mol-1 K-1) and T is temperature (K).
Results and Discussion
Central Composite Design (CCD)
Effects of the three individual variables pH, initial chromium ions concentration and biosorbent dosage on biosorption efficiency (%) were examined using CCD. Complete design matrix generated using FCCCD in terms of coded, actual values of variables and responses reported from experiments were shown in Table 2. It shows that, bisorption of Cr (III) varied from 31.54% to 93.20% and 20.63% to 81.81% for Cr (VI). Significances of model and process variables were determined by ANOVA and found that all individual variables had significant effect, whereas quadratic and interaction terms had no effect on biosorption. Low value of lack of fit and high values of R2 and R2 adj demonstrates the consistency of models. The regression models equations were:
Y1 = 12.70 + 57.781 X1 - 0.2271 X2+ 166.7 X3 -10.2780 X1*X1 + 0.000868 X2*X2 - 1017 X3*X3 - 0.00061 X1*X2 + 1.97 X1*X3 - 0.014 X2*X3 -------------------------- (11)
Y2 = 1.23 + 57.657 X1 - 0.2074 X2 + 173.0 X3 - 10.269 X1*X1 + 0.000722 X2*X2 - 1039 X3*X3 + 0.00005 X1*X2 + 1.03 X1*X3 + 0.020 X2*X3 ------------------------- (12)
where Y1 and Y2 are biosorption efficiencies of Cr(III) and Cr(VI) respectively. X1, X2 and X3 are pH, initial concentration of chromium (mg l-1) and biosorbent dosage (g) respectively. The optimum conditions for the maximum efficiency were pH: 3; biosorbent dosage: 0.03g; initial concentration: 20mg l-1 for both Cr(III) and Cr(VI), can result in maximum biosorption efficiency 93.20% (185.12 mg g-1) and 81.81% (163.63mg g-1) for Cr(III) and Cr(VI), respectively.
Effects of process variables on biosorption efficiency
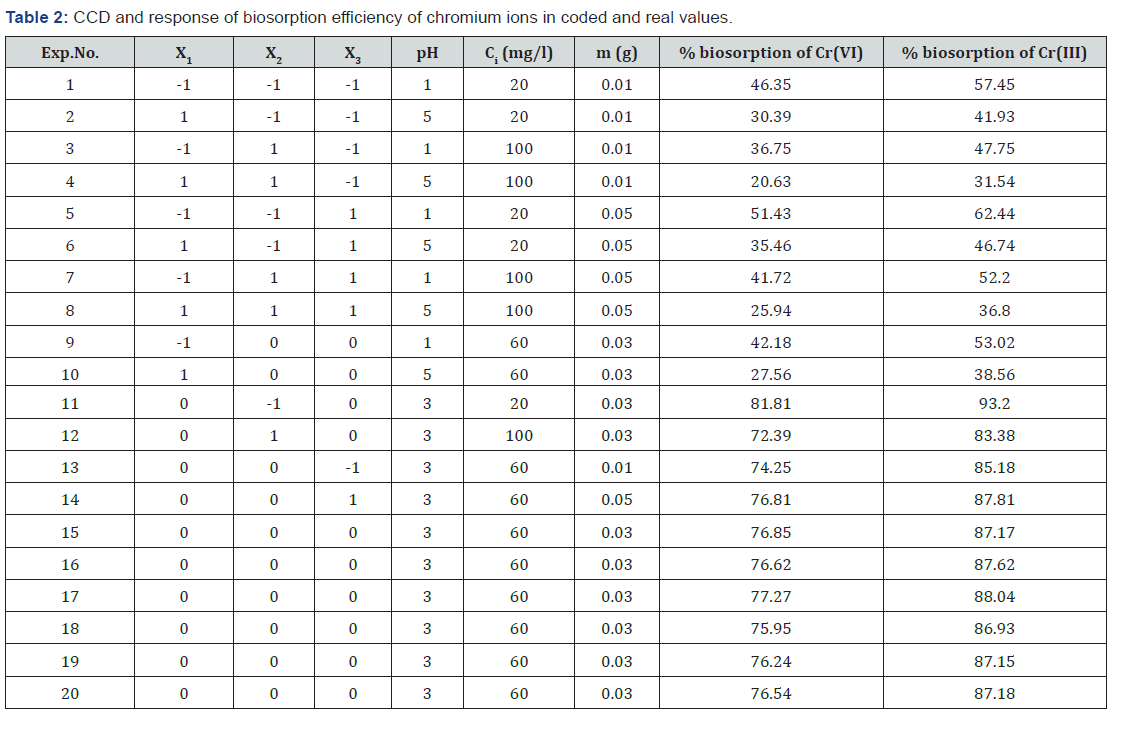
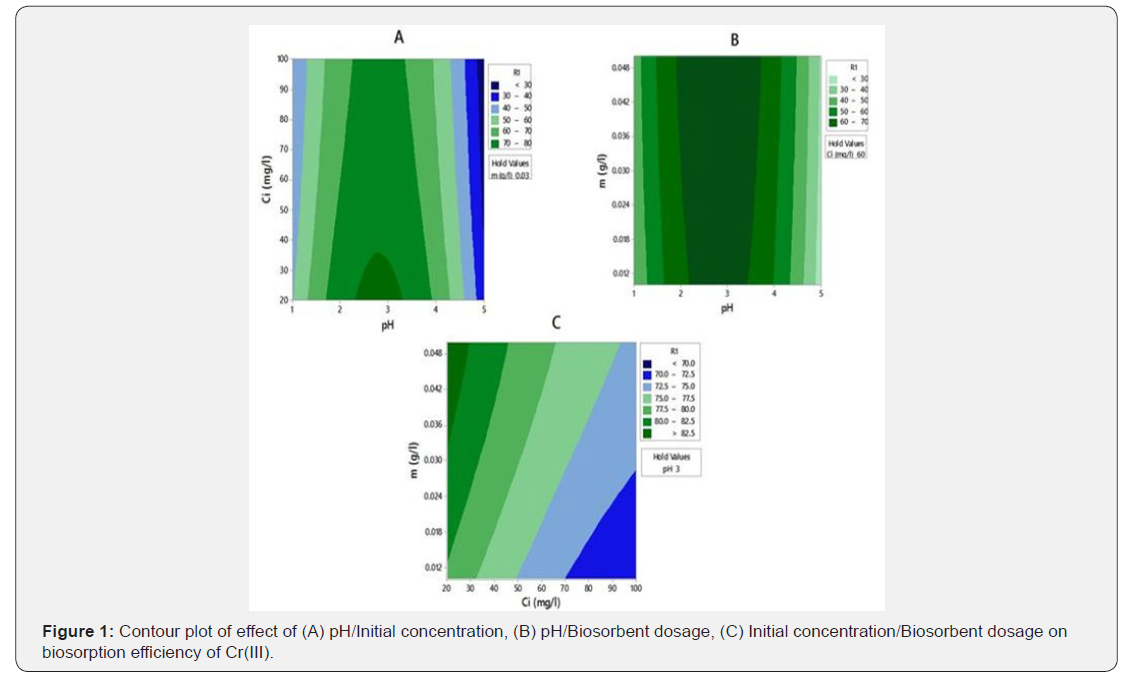
Figures 1 & 2 A & B show the influence of initial pH of chromium solution on biosorption efficiency at constant chromium concentration and biosorbent dosage. The biosorption efficiencies of Cr(III) and Cr(VI) were increased with pH(1-3) and reached maximum at pH 3. At lower pH, in the acidic medium the biomass surface will be protonated; hence the positive surface of biomass effectively holds the chromium ions. Moreover, at lower pH large number of H+ ions present in the solution will be neutralized by the OH- ions present on the surface of biomass which promotes the migration of negatively charged chromium ions towards binding sites on biomass surface. Hence these two factors promote maximum biosorption at lower pHs [22]. With increase in pH(3- 5), the surface of biosorbent becomes negatively charged which hinders biosorption through electro static repulsion, as a result biosorption efficiency decreases [23]. Effect of concentrations of chromium species on biosorption efficiency were shown in Figures 1 & 2 A & C. Biosorption efficiency was gradually decreased with the increase of concentration. At lower concentrations because of presence of good number of binding sites and less number of chromium ions biosorption efficiency is high, whereas at higher concentrations due to lack of sufficient binding sites and repulsion between biosorbed molecules and bulk phase the biosorption efficiency decreases [24]. Effects of G.acerosa dosage on chromium removal efficiency were presented in Figures 1 & 2 B & C. It was noticed that with increase of biosorbent, biosorption efficiency was increased. With the increase of biosorbent dosage number of available metal binding sites and therefore metal removal efficiency increases [25].
Simultaneous responses optimization
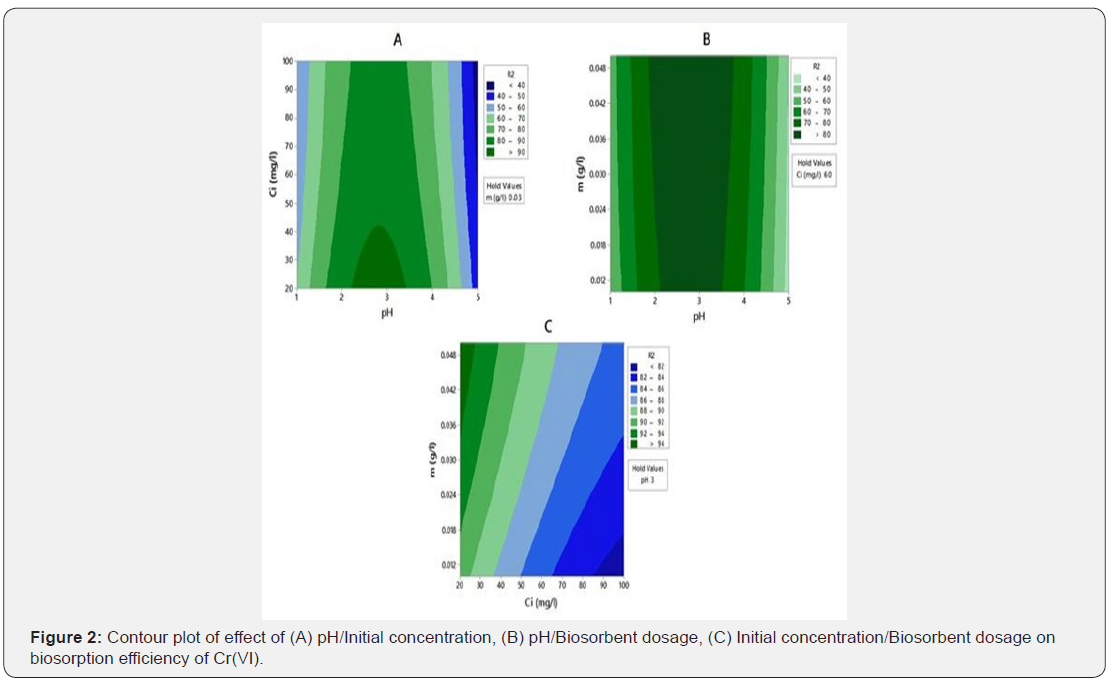
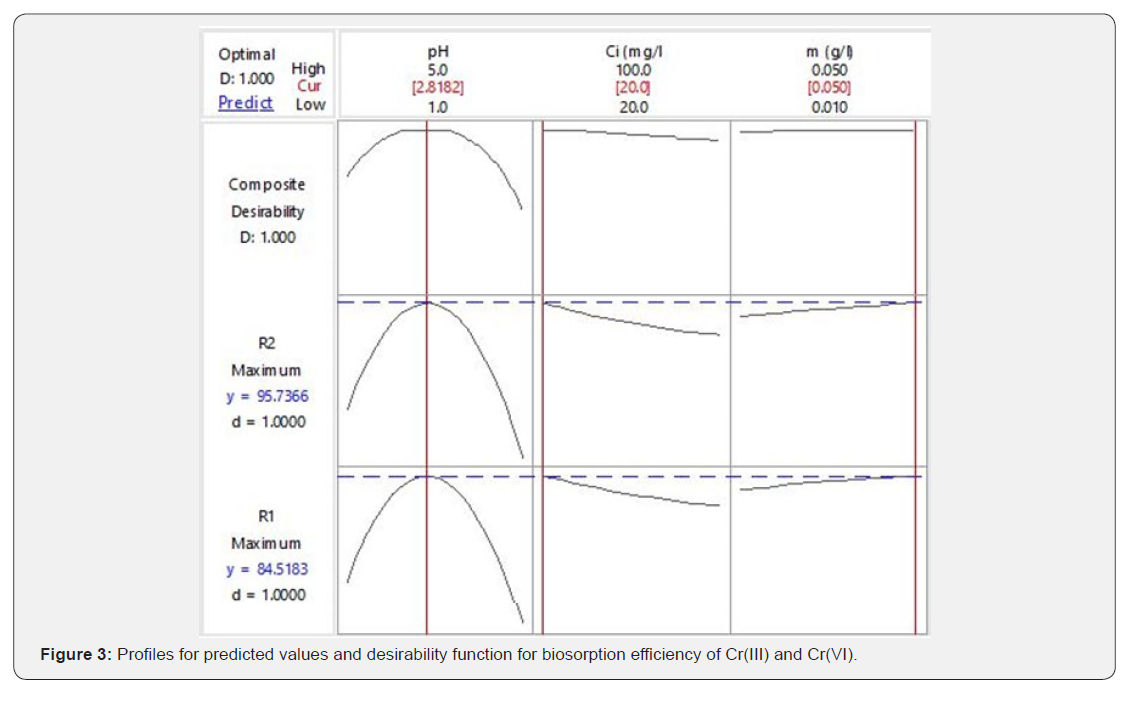
Optimization of single response using statistical optimization techniques is simple and well reported in literature, but in effluent treatment optimization of multiple responses is much needed. As shown in Table 2, maximum biosorption efficiency of Cr(III) and Cr(VI) was not under similar conditions. Therefore, multiple response optimization with desirability function was used to predict the common optimum point for simultaneous biosorption of Cr(III) and Cr(VI). The desirability values for maximum biosorption of Cr(III), Cr(VI) and overall desirability value for simultaneous maximum biosorption were calculated. Optimization of simultaneous biosorption of chromium ions with desirability function was depicted in Figure 3. As shown in the figure individual and overall desirability values of 1.0 indicate that optimization of individual responses and simultaneous biosorption of Cr(III) and Cr(VI) are effective. Hence, pH 2.8, initial chromium species concentrations 20 mg l-1 and G.acerosa dosage 0.05g were chosen as optimum conditions for simultaneous biosoprtion. At this optimum process conditions, maximal predicted responses for simultaneous biosorption were Y1 =95.73% and Y2 =84.51%. The validity of optimization process was examined by conducting a set fresh experiments (triplicate) at the above predicted values and reported 92.25% and 82.47% of Cr(III) and Cr(VI) removal respectively.
The experimental results are very closer to the predicted values. Hence, the predicted values of independent variables at maximum chromium removal were considered as optimum process conditions for biosorption of chromium onto G.acerosa.
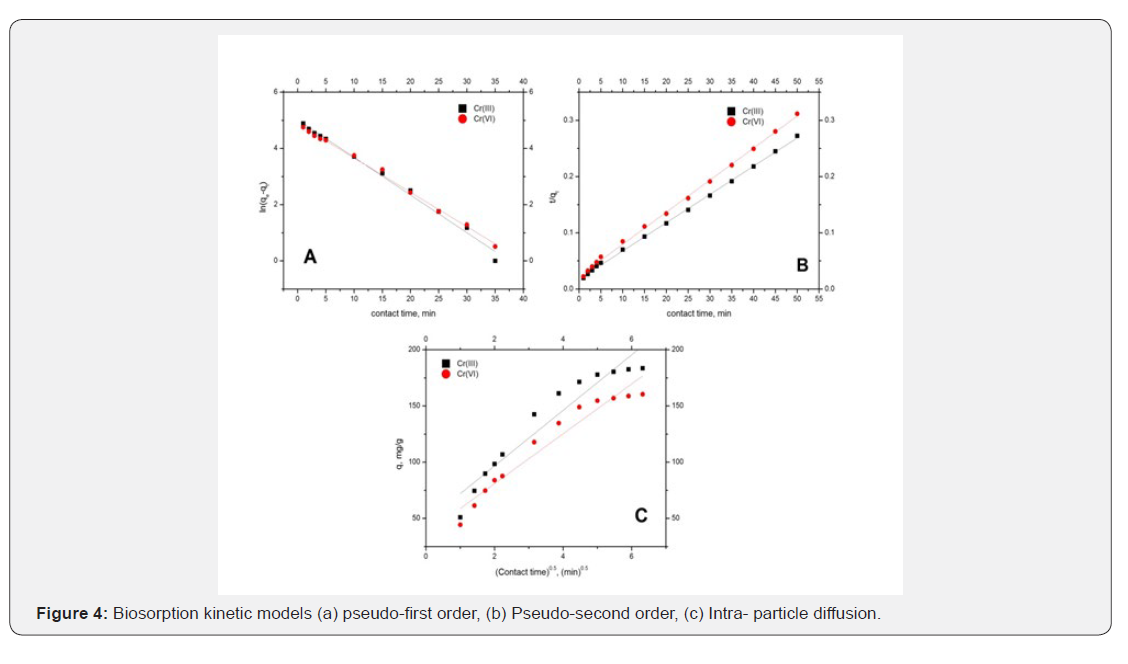
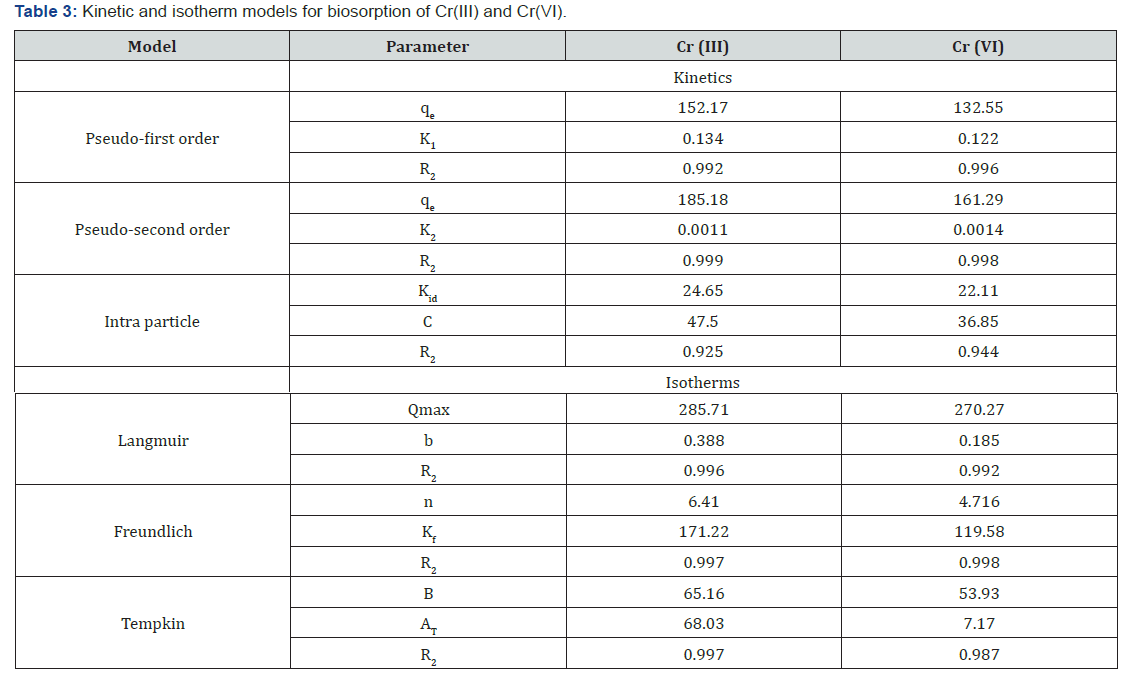
Kinetic modeling is fundamental in order to determine the affinity or capacity of the sorbent which in turn governs the residence time in the design of biosorption process. The fitness of kinetic data to pseudo first, second order rate equations and intra particle model was described in Figure 4 and corresponding rate constants, predicted chromium uptakes and R2 values were reported in Table 3. Though the values of first order model are in the range of 0.99 to 0.98 but the predicted metal uptakes are far away from the experimental values. Hence, pseudo first order rate equation is not appropriate for determining kinetics. Whereas for pseudo second order, high correlation coefficients and very close predicted and experimental chromium uptake values shows the significance of the model for representing kinetics of biosorption of chromium. Therefore biosorption of chromium ions onto G.acerosa follows pseudo second order model. The intra-pore diffusion of chromium ions was investigated by fitting the kinetic data to intra particle diffusion model Figure 5(a) shows that the kinetic data is not well correlated to the intra particle diffusion model and also plots are not passing through origin, indicating the biosorption of chromium is controlled by film diffusion.
Biosorption isotherms
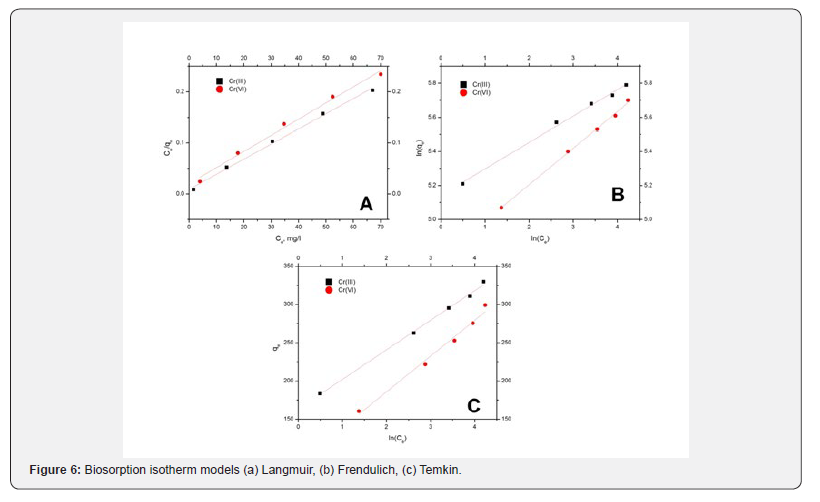
Langmuir, Freundlich and Temkin isotherms were fitted to experimental equilibrium data to illustrate the biosorption nature of chromium onto G.acerosa. The plots pertain to Langmuir and Freundlich and Temkin models were shown in Figure 6 and the equilibrium constants, regression coefficients and were shown in Table 3. The Freundlich model was better approximation to describe the biosorption isotherms than the other models.
Characterization of G.acerosa using FT-IR
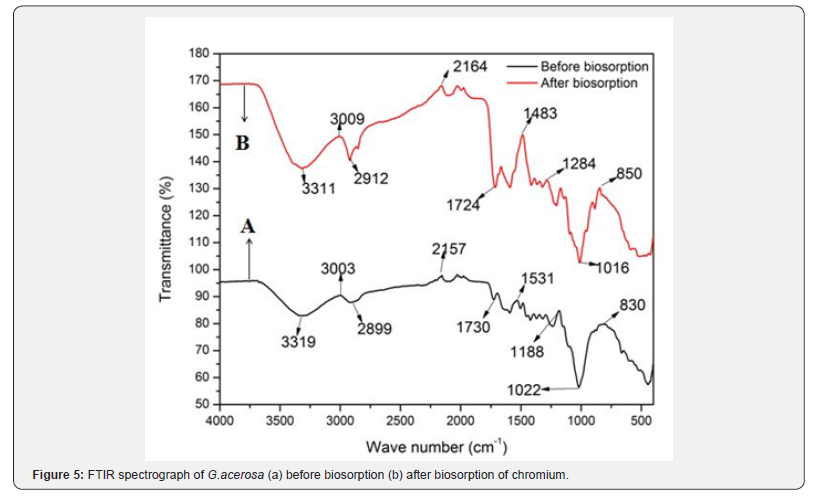
FT-IR spectra of G.acerosa before and after biosorption of chromium were recorded at frequencies from 4000 to 400 cm-1 and presented in Figure 5. The spectra disclosed presence of various functional groups like carboxyl, hydroxyl, amino and carbonyl on the biosorbent surface. Algal cell wall is composed of many functional groups like hydroxyl, carboxyl, amine, sulphate and phosphate that play major role in metal binding [26]. Several peaks observed in Figure 5(a) indicating the prevalence of various functional groups which are responsible for binding of chromium. The medium peak at 3319 cm-1 represents N-H stretching vibration that showed the presence of amine group on the surface of biosorbent. The peaks at 3003 and 2899 cm-1 represent C-H bonds that indicate the presence of carboxylic groups [26,27]. In most of the brown algae, 70% of cell wall functional groups are carboxylic and amino groups, these groups play vital role in metal binding [28]. The peaks at wave numbers of 1730, 1531 cm-1 are indicative of the presence of S, C=O and NO2 groups [29]. Strong stretching vibrations in the range of 1250-970 cm-1 are indication of the presence of alcohols and phenols. The peak at 830 cm-1 is associated with absorbance of O-H group indicating the presence of carboxylic groups. Therefore it is inferred that the sur¬face of G.acerosa comprises of functional groups like carboxylic acids, amino, alkenes and alkyl groups. Comparison of FTIR spectra of G.acerosa loaded with chromium (Figure 5(b)) with that of unloaded revealed significant changes. Formation of new peaks at 1438 and 1248 cm-1 and disappearance of peak at 1724 cm-1 are the clear indications of possible chemical reaction between chromium ions and surface functional groups. Further shifting of peaks in the range of 3500 to 2800 cm-1 pertaining to carboxylate group reveals that involvement of C-H group in adsorption of chromium ions onto the biosorbent. Moreover, shifting of peak from 830 cm-1 to 850 cm-1 also discloses the binding of chromium ions to amine group present on the surface of spent G.acerosa [31].
Conclusion
This work demonstrated the simultaneous optimization of biosorption efficiency of Cr(III) and Cr(VI) using G.acerosa by applying CCD and multiple response optimizations. The optimum process conditions for simultaneous biosorption of Cr(III) and Cr(VI) were pH 2.81, initial chromium ions concentration 20 mg l-1 and G.acerosa dosage 0.05g. The maximum biosorption efficiencies of Cr(III) and Cr(VI) reported under this optimum process conditions were 95.73% (183.83 mg g-1) and 84.51% (160.47 mg g-1) respectively. Kinetic study showed that pseudo second order model provided better fit and fitness to the intra particle diffusion model reveled that film diffusion is the biosorption rate controlling step. Isotherm study showed that Freundlich isotherm model better explains biosorption equilibrium. The combination of RSM and multiple response optimization in present work led to finding optimized conditions for the simultaneous biosorption of Cr(III) and Cr(VI). These results indicate that marine algae G.acerosa without any pretreatment is a favorable sorbent material for biosorption of Cr(III) and Cr(VI).
References
- Miretzky P, Cirelli AF (2010) Cr (VI) and Cr (III) removal from aqueous solution by raw and modified lignocellulosic materials: a review. J Hazard Mater 180(1-3): 1-19.
- Avudainayagam S, Megharaj M, Owens G, Kookana RS, Chittleborough D, et al. (2003) Chemistry of chromium in soils with emphasis on tannery waste sites. Rev Environ Contam. Toxicol 178: 53-91.
- Chen S, Yue Q, Gao B, Li Q, Xu X, et al. (2012) Adsorption of hexavalent chromium from aqueous solution by modified corn stalk: a fixed-bed column study. Bioresour Technol 113: 114-120.
- Cohen MD, Kargacin B, Klein CB, Costa M (1993) Mechanisms of chromium carcinogenicity and toxicity. Crit Rev Toxicol 23(3): 255-281.
- Losi ME, Amrhein C, Frankenberger WT (1994) Environmental biochemistry of chromium. Rev Environ Cont Toxicol 136: 91-121.
- Bennett RM, Cordero PRF, Bautista GS, Dedeles GR (2013) Reduction of hexavalent chromium using fungi and bacteria isolated from contaminated soil and water samples. Chem Ecol 29: 320-328.
- Thatoi H, Das S, Mishra J, Rath BP, Das N (2014) Bacterial chromate reductase, a potential enzyme for bioremediation of hexavalent chromium: a review. J Environ Manag 146: 383-399.
- Abyaneh AS, Fazaelipoor MH (2016) Evaluation of rhamnolipid (RL) as a biosurfactant for the removal of chromium from aqueous solutions by precipitate flotation. J Environ Manag 165: 184-187.
- Cardoso VDA, De Souza AG, Sartoratto PP, Nunes LM (2004) The ionic exchange process of cobalt, nickel and copper (II) in alkaline and acid-layered titanates. Colloids. Surf A Physicochem Eng Asp 248(1-3): 145-149.
- Kandah MI (2004) Zinc and cadmium adsorption on low-grade phosphate. Sep Purif Technol 35(1): 61-70.
- Gupta VK, Ali I (2008) Removal of endosulfan and methoxychlor from water on carbon slurry. Environ Sci Technol 42(3): 766-770.
- Ferraz AI, Amorim C, Tavares T, Teixeira JA (2015) Chromium (III) biosorption onto spent grains residual from brewing industry: equilibrium, kinetics and column studies. Int J Environ Sci Technol 12: 1591-1602.
- Gupta VK, Pathania D, Agarwal S, Sharma S (2013) Removal of Cr (VI) onto Ficus carica biosorbent from water. Environ Sci Pollut Res 20(4): 2632-2644.
- Kuppusamy S, Thavamani P, Megharaj M, Venkateswarlu K, Lee YB, et al. (2016) Potential of Melaleuca diosmifolia leaf as a lowcost adsorbent for hexavalent chromium removal from contaminated water bodies. Process Saf Environ Prot 100: 173-182.
- Spasojevic PM, Panic VV, Jovic MD, Markovic J, Van Roost C, et al. (2016) Biomimic hybrid polymer networks based on casein and poly (methacrylic acid). Case study: Ni2+ removal. J Mater Chem A 4: 1680-1693.
- Kumar KS, Dahms HU, Won EJ, Lee JS, Shin KH (2015) Microalgae-A promising tool for heavy metal remediation. Ecotoxicol Environ Saf 113: 329-352.
- Mata YN, Blazquez ML, Ballester A, Gonzalez F, Munoz JA (2008) Characterization of the biosorption of cadmium, lead and copper with the brown alga Fucus vesiculosus. J Hazard Mater 158(2-3): 316-323.
- Witek-Krowiak A, Chojnacka K, Podstawczyk D, Dawiec A, Pokomeda K (2014) Application of response surface methodology and artificial neural network methods in modelling and optimization of biosorption process. Bioresour Technol 160: 150-160.
- Grosso C, Ferreres F, Gil-Izquierdo A, Valentão P, Sampaio M, et al. (2014) Box-Behnken factorial design to obtain a phenolic-rich extract from the aerial parts of Chelidonium majus L. Talanta 130: 128-136.
- Chatterjee S, Kumar A, Basu S, Dutta S (2012) Application of Response Surface Methodology for Methylene Blue dye removal from aqueous solution using low cost adsorbent. Chem Eng J 181-182: 289-299.
- Sadhu K, Mukherjee A, Shukla SK, Adhikari K, Dutta S (2014) Adsorptive removal of phenol from coke-oven wastewater using Gondwana shale, India: experiment, modeling and optimization. Desalin Water Treat 52(34-36): 6492-6504.
- Gorzin F, Bahri RAMM (2018) Adsorption of Cr (VI) from aqueous solution by adsorbent prepared from paper mill sludge: Kinetics and thermodynamics studies. Adsorpt Sci Technol 36(1-2): 149-169.
- Hiremath PG, Theodore T (2016) Zirconium-Doped Fungal Sorbents: Preparation, Characterization, Adsorption Isotherm, and Kinetic and Mathematical Modelling Study for Removal of Fluoride. Advances in Chemistry.
- Fathi MR, Asfaram A, Farhangi A (2015) Removal of Direct Red 23 from aqueous solution using corn stalks: isotherms, kinetics and thermodynamic studies. Spectrochim Acta Part A 135: 364-372.
- Chowdhury MRI, Mulligan CN (2011) Biosorption of arsenic from contaminated water by anaerobic biomass. J Hazard Mater 190(1-3): 486-492.
- Jerold M, Sivasubramanian V (2016) Biosorption of malachite green from aqueous solution using brown marine macro algae Sargassum swartzii, Desal. Water Treat 57: 25288-25300.
- Dulla JB, Sumalatha B, King P, Yekula PK (2018) Investigation on biosorption of Cd (II) onto Gelidiella acerosa (brown algae): Optimization (using RSM & ANN) and mechanistic studies. Desalin Water Treat 107: 195-206.
- Meseguer VF, Ortuño JF, Aguilar MI, Pinzón-Bedoya ML, Lloréns M, Sáez J, et al. (2016) Biosorption of cadmium (II) from aqueous solutions by natural and modified non-living leaves of Posidonia oceanic. Environ Sci Pollut Res 23(23): 24032-24046.
- Khan TA, Mukhlif AA, Khan AA, Sharma DK (2006) Isotherm and kinetics modeling of Pb (II) and Cd (II) adsorp¬tive uptake from aqueous solution by chemically modified green algal biomass. Model Earth Syst Environ 2: 1-13.
- Liu RS, Tang YJ (2010) Tuber melanosporum fermentation medium optimization by Plackett-Burman design coupled with Draper-Lin small composite design and desirability function. Bioresour Technol 101(9): 3139-3146.
- Sari A, Mendil D, Tuzen M, Soylak M (2008) Biosorption of Cd (II) and Cr (III) from aqueous solution by moss (Hylocomium splendens) biomass: equilibrium, kinetic and thermodynamic studies. Chem Eng J 144(1): 1-9.






























