Effect of 2,4-dichlorophenoxyacetic on in Vitro Callus Induction Using Leaf of Rocoto (Capsicum Pubescens Ruiz & Pav.) Cv. Serrano
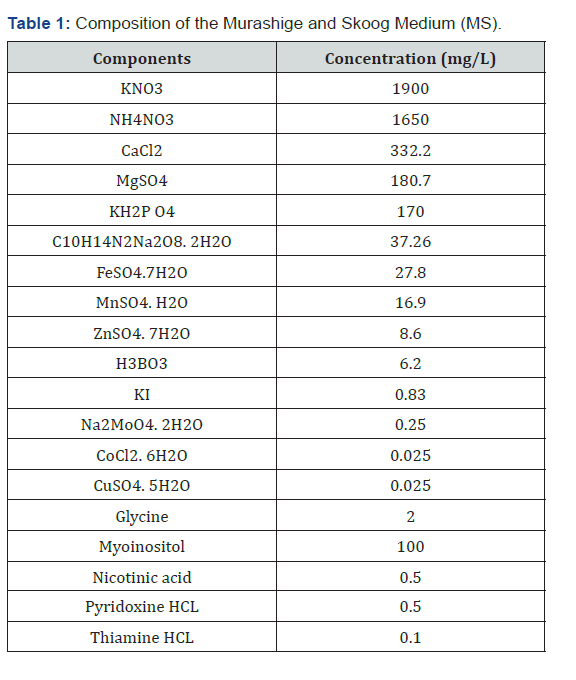
Hernández Amasifuen Angel David*Argüelles Curaca Alexis, Cortez Lázaro Anthony Apolinario, Barreto Romero Andrea Alhely, Díaz Pillasca Hermila Belba,Silva Vergara Mishell Zaori
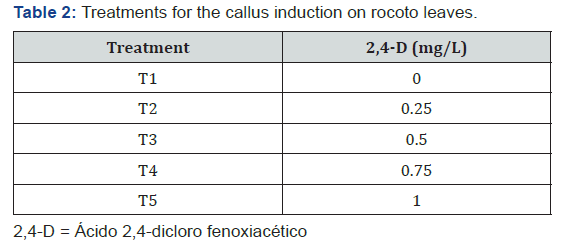
Department of Plant Biotechnology Laboratory of the Professional School of Biology with a mention in Biotechnology, José Faustino Sánchez Carrión National University, Peru
Submission:October 16, 2019Published: October 29, 2019
*Corresponding author:Hernández Amasifuen Angel David, Department of Plant Biotechnology Laboratory of the Professional School of Biology with a mention in Biotechnology, José Faustino Sánchez Carrión National University, Peru
How to cite this article:Hernández Amasifuen Angel David, Argüelles Curaca Alexis, Cortez Lázaro Anthony Apolinario, Barreto Romero Andrea Alhely,Díaz Pillasca Hermila Belba, Silva Vergara Mishell Zaori. Effect of 2,4-dichlorophenoxyacetic on in Vitro Callus Induction Using Leaf of Rocoto (Capsicum Pubescens Ruiz & Pav.) Cv. Serrano.Agri Res& Tech: Open Access J. 2019; 22(4): 556229. DOI: 10.19290/ARTOAJ.2019.22.556229
Abstract
The rocoto (Capsicum pubescen Ruiz & Pav.) is a plant of the genus Capsicum with center of origen in Perú, Used in national cuasina and of great valué for its pharmaceutical and medicinal propretores, relatad to capsaicin. In the present work a methodology was developed to determine the effect of 2,4-dichlorophenoxyacetic on in vitro callus induction from leaves in rocoto cv. Serrano. Were used seedlings rocoto cv. Serrano germínate in vitro, and subsequently the youngest leaves were selected, which were sectioned and placed in the different culture media. Five treatments with different concentrations of 2,4-D were evaluated. The best results were obtained with treatments T4 and T5 for the callus induction in 21 days allowed 72% callus proliferation. This study, a pioneer for the species, opens expectations for the potential use of callus of rocotos in other applications such as the production of protoplasts, somatic hybrids, somatic embryos and their use in polyploidization.
Introduction
The rocoto (Capsicum pubescens Ruiz & Pav.) is a herbaceous plant that belongs to the Solanaceae family, whose center of origin is Peru, when found wild in the high jungle and mountains. This species differs from other Capsicum by presenting purple or purple flowers and also by its black seeds. The fruit can vary in shape, size and color, but it is characterized by its itching, so it is widely used in Peruvian cuisine. In addition, rocoto has pharmaceutical and medicinal properties, related to capsaicin [1].
National rocoto production is increasing every year mainly due to the gastronomic boom that has been taking place and in which rocoto has become a flagship product. But it should also be considered that this crop is susceptible to diseases such as risotonia solanacearum, anthracnose, wilting and root rot, among others. It is also susceptible to viruses that can cause yellowing of the ribs, deformations in leaves and fruits, dwarfism, lack of vigor and fall of the leaves. The causes of these symptoms are usually related to Tomato Black Plague Virus (TSWV), Alfalfa Mosaic Virus (AMV), Mottled Pepper Virus (PMMoV), Peruvian Tomato Virus (MTV) and Tomato Mosaic Virus (ToMV), the latter by dissemination on contact between plants, also by seeds from infected cultivars or seedlings [2, 3]. Being important to strengthen the rocoto value chain is to obtain seeds or seedlings free of pathogens, and there fore have great production. Within the alternatives of aseptic plant material, biotechnological tools are present, with tissue culture techniques, being the technique of callogenesis or embryogenesis often inducing somaclonal variation [4]. In this way, genotypes of good quality can be multiplied, as has been developed in the tissue culture and regeneration of in vitro seedlings of different species of the Solanaceae family [5], but no tissue culture work has been reported. in C. pubescens. The present investigation was carried out with the objective of developing a methodology to determine the effect of 2,4-dichlorophenoxyacetic on in vitro callus induction from leaf segments in rocoto cv. Serrano.
Materials and Methods
This research was carried out in the facilities of the plant biotechnology laboratory of the professional school of Biology with a mention in Biotechnology, located at the José Faustino Sánchez Carrión National University, Huacho, Lima, Peru.
Disinfection of plant material
Rocoto seeds were used, which are kept in the greenhouse of the Plant Biotechnology laboratory of the School of Biology. These seeds were first washed with water plus detergent for 5 minutes, and then began the disinfection process, in which used the protoAbstract col described by Argüelles et al [6], which consists of first immersing the seeds in 70% ethanol for one minute, then all the material was passed to the laminar flow chamber where the disinfection was continued, then submerged in a hypochlorite solution of 2% sodium for 15 minutes under constant stirring. After the established time, three rinses were made with sterile distilled water, then they were cast on filter paper and 3 seeds were placed pertest tube with MS culture medium. They were kept in total darkness at a constant temperature of 27°C, with relative humidity of 75 ± 2%. The MS culture medium consisted of salts and vitamins described by Murashige and Skoog [7] (Table 1), but was used at half of its concentration; 7g/L of agarose, 30g/L of sucrose were added and the pH was adjusted to 5.7. Subsequently, it was sterilized in an-autoclave at 1.2 Bar pressure and a temperature of 121°C for 20 minutes.
Callus induction
From the germinated seedlings in vitro they selected the youngest leaves to be used in the experimentation, which were segmented and five segments per plate were introduced with MS culture medium (Table 1), which was used at half of its concentration and It was supplemented with five 2,4-D treatments (Table 2), in addition all treatments consisted of 30g/L sucrose and 7g/L agarose. All plates were incubated at 25°C, in total darkness and with relative humidity of 75 ± 2% for 21 days.
Experimental design and data análisis
Statistical analyzes were performed using the SPSS software (version 21.0 for Windows). Each treatment consisted of 10 plates with five explants each, using each explant as an experimental unit. To observe the significant differences in the induction percentages, an Analysis of Variance (ANVA) was used and the comparison between the means was made according to the Tukey test (p≤0.05).
Results

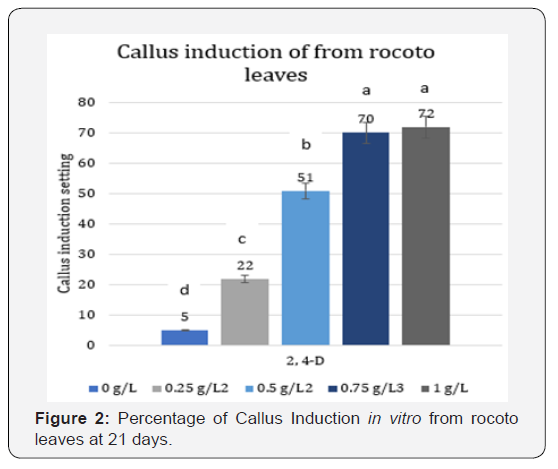
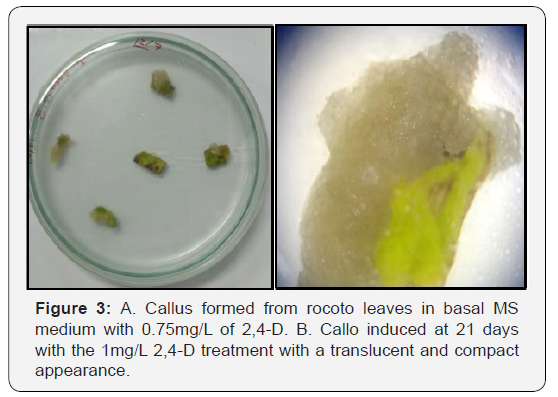
With respect to the disinfection of rocoto seeds, the methodology used allowed to obtain 100% free of contamination. In callus induction from the rocoto leaf segments, callus formation was observed from day 11 on the induction medium T4 and T5 (Figure 1), From day 15 on induction media, it was observed that the first 4 treatments had callus formation, but treatments T4 and T5 had a percentage of callus induction greater than 50%, while treatment T1 did not present callus induction. At day 21, the treatment with greater callus induction was obtained and in less time the treatments with 0.75 and 1mg/L of 2,4-D with 72% (Figure 2) with a translucent and compact appearance (Figure 3).
Discussion
The method of disinfection of rocoto seeds was effective when immersed in 70% ethanol and then with sodium hypochlorite, which is a substance commonly used in the superficial disinfection of plant material in the process of introduction in the in vitro culture for presenting A positive effect in the elimination of resistant microorganisms, due to the effects of chlorination [8], is effective in most pathogenic bacteria, but with an unpredictable action against fungi and viruses. The death of microorganisms is due to the direct combination of chlorine with the proteins of cell membranes and enzymes, this because chlorine destroys organisms by being inactivated by oxidation of cellular material. Likewise, in the presence of water gives off nascent oxygen (O2) that oxidizes organic matter [9]. In addition, stirring the explants together with the disinfectant helps to reduce the surface tension of the water, thus allowing greater contact of the explant with the disinfectant solution [10,11]. On the other hand, ethanol acts by denaturing proteins, dissolving lipid layers and as a dehydrating agent, it is lethal to bacteria, but irregular to fungi and viruses, it does not act on spores; when combined with antiseptics of another group has a greater germicidal action [9]. It was possible to induce callus from the rocoto leaf segments in all treatments, with a translucent and compact appearance. Callus induction is highly dependent on the type of explant and hormone used, in the case of the leaves, unlike other explants, a clear increase in callus is seen when the concentration of 2,4-D is increased, in addition to This type of hormone influences the appearance of calluses [12]. The culture medium with different concentrations of growth regulators allows to have different responses in the explants, in this case the addition of auxins, which has the compounds that have the greatest influence on cell development and differentiation. This is due to the fact that in vitro culture these endogenous compounds present drastic changes in the cellular environment, which generates a stress effect that can cause a cellular reorganization and form a mass of undifferentiated cells [13]. The greatest number of calluses formed were obtained from the treatment added with 0.75 and 1mg/L of 2,4-D, in comparison to other authors that recommended the addition of a high combination of cytokinins and auxins, or only the addition of auxins can favor the callus induction, in addition to the appearance of callus is related to the type of hormone used during induction. In the case of the growth regulator that has more reports of callus induction is auxin 2,4-D [14-18]. These results represent an important advance in the use of biotechnological techniques for the genetic improvement of rocoto, which opens expectations for the potential use of callus of this species for production of production of protoplasts, somatic hybrids, somatic embryos and their use in polyploidization
Conclusion
A methodology was developed for the callus induction in vitro from leaves in rocoto cv, Serrano, using basal MS medium supplemented with 0.75 and 1mg/L of 2,4-D. for the highest percentage of callus formation from the second week.
Acknowledgement
The authors thank Ing. Angel Abel Hernández Cotrina and Lic. Lastenia Amasifuen Ochavano for providing access to the cultivation areas and supporting the collection of samples that were donated to the Plant Biotechnology laboratory.
References
- Caballero B, Márquez C, Alberto B (2017) Effect Of The Freeze - Drying On The Functional Properties Rocoto Chili Pepper (Capsicum Pubescens) Revista U.D.C.A Actualidad & Divulgación Científica 20(1): 111-119.
- Lizarraga L, Aragón L (2012) Respuesta de 5 especies de Capsicum spp. A Phytophthora capsici Leon, bajo condiciones de invernadero, en los laboratorios de fitopatología de la UNALM-Lima. Tesis de Ingeniera. Universidad Nacional de San Antonio Abad del Cusco. Cusco. Perú.
- Hernández A, Pineda A, Noriega H (2019) Aislamiento e identificación de Fusarium oxysporum obtenidos de zonas productoras de “ají paprika” Capsicum annumm (Solanaceae) en el distrito de Barranca, Perú. Arnaldoa 26 (2): 689-698.
- Orlinska M, Nowaczyk P (2015) In vitro plant regeneration of 4 Capsicum spp. genotypes using different explant types. Turkish Journal of Biology 60-68.
- Sanatombi K, Sharma GJ (2007) Micropropagation of Capsicum annuum Notulae Botanicae Horti Agrobotanici Cluj-Napoca 35: 57-64.
- Argüelles A, Domínguez G, Gózalo A, Díaz H (2019) Inducción de germinación in vitro de semillas de rocoto (Capsicum pubescens). IV Congreso Peruano de Biotecnología y Bioingenierí Libro de Resúmenes 8-11.
- Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiology Plant 15(3): 473-497.
- Ramírez M, Urdaneta A, León S (2002) Establecimiento in vitro de explantes adultos del guanabo (Annona muricata L.) tratados con hipoclorito de sodio. Rev Fac Agron 19(1): 1-8.
- Folgueras M, Herrera L, Carrazana D (2001) La contaminación microbiana en la micropropagación in vitro de las raíces y tuberculos troplicales. Taller Internacional 183-185.
- Abdelnour A, Escalante V (1994) Conceptos básicos del cultivo de tejidos vegetales. Centro Agronómico de Investigaciones y Enseñanza Turrialba Costa Rica 38.
- Hernández A, Pineda A, Díaz H (2019) Efecto de la luz y del ácido giberélico en la germinación in vitro de Capsicum annuum cv. Papri King. Biotecnología Vegetal 19(2): 43-51.
- Rodríguez M, Latsague M, Chacón M, Astorga P (2014) Inducción in vitro de callogénesis y organogénesis indirecta a partir de explantes de cotiledón, hipocótilo y hoja en Ugni molinae. Bosque 35(1): 111-118.
- Fehér A, Pasternak T, Dudits D (2003) Transition of somatic plant cells to an embryogenic state. Plant Cell Tissue Organ Cult. 74(3): 201-228.
- Shiram V, Kumar V, Shitole M (2008) Indirect organogénesis and plant regeneration in Helicteres isora L., an impor tant medicinal plant. In vitro Cellular and Developmental Biology-Plant 44(3): 186-193.
- Larson C, Gómez C, Sánchez M, Ríos D (2006) Inducción de caulogénesis indirecta en Eucalyptus globulus. Bosque 27(3): 250-257.
- Meiners J, Schwab M, Szankowski I (2007) Efficient in vitro regeneration systems for Vaccinium species. Plant Cell, Tissue and Organ Culture 89(2-3): 169-176.
- Hernández A, Díaz H (2019) Inducción in vitro de callo embriogénico a partir del cultivo de anteras en “papa amarilla” Solanum goniocalyx Juz. & Bukasov (Solanaceae). Arnaldoa 26(1): 277-286.
- Feeney M, Bhagwat B, Mitchell J, Lane W (2007) Shoot regeneration from organogenic callus of sweet cherry (Prunus avium L.). Plant Cell, Tissue and Organ Culture 90: 201-214.






























