Safety Limits for Using Ostrich Olein in Frying Process
Amany M Basuny1*,Amal R Tageldeen2,Marwa A El- Ghonamy2
1Department of Biochemistry, Beni-Suef University, Egypt
2Department of Oils & Fats, Food Technology Research Institute, Agricultural Research Center, Egypt
Submission:September 09, 2019Published: October 21, 2019
*Corresponding author:Amany M Basuny, Department of Biochemistry, Beni-Suef University, Egypt Agri Res
How to cite this article:Amany M Basuny, Amal R Tageldeen, Marwa A El- Ghonamy. Safety Limits for Using Ostrich Olein in Frying Process.Agri Res& Tech: Open Access J. 2019; 22(4): 556223. DOI: 10.19230/ARTOAJ.2019.22.556223
Abstract
Ostrich (Struthio camelus) oil is used as a new source of animal oil. Physical and chemical characteristics for the ostrich oil, profile of fatty acid (%) and unsaponifiable matter (%) were analysis. Ostrich oil was fractionated to two phases (liquid and solid fractions) after fractionated some characteristics of two fractions was measured. Blending process of sunflower oil with liquid fraction (named ostrich olein) showed that oxidative stability, which was evaluated by the Rancimat method at 100°C, was obtained from ostrich oil by dry fractionation. The results indicated that blending process of sunflower oil with liquid fraction (ostrich olein) on effective method to prepare more stable vegetable oils. Ostrich olein was mixed separately with sunflower oil at (75:25, 50:50 and 25:75, v/v). The frying process was conducted at 180°C ± 5°Cfor 16hr/ 4hr per day. Some physical and chemical characteristics of non-fried and fried oil mixture were measured at various heating periods. The results demonstrate that mixing ostrich olein with sunflower oil increased the stability and hence improved the quality of sunflower oil during frying process.
Keywords:Ratite family; Ostrich oil; Fractionation; Blending process, Frying technique
Introduction
Deep-fat frying is one of the best cooking processes to make palatable foods with golden color, exquisite flavor, and attractive surface through a total submersion of nourishment materials in frying oil. During frying processes, heat is moved from oil to sustenance food materials, and water in singed items dissipates at the same time with the items the oil [1]. During commercial frying technique, frying oils are typically more than once utilized, thus changing quality of fried foods with development of non-volatiles and volatiles components, some of which are potentially harmful to human health. Thusly, it is critical to screen changes in quality of frying oils [2]. Physical properties such as color index and viscosity, and chemical characteristics such as acid value, total polar compound, and p-anisidine value have been generally considered to assess the quality of frying oils [3]. As total polar compound has been a reasonable factor to evaluate quality of frying oils, acceptable limits of total polar compound have been suggested to be 24-27% (w/w). In Korea, acid value has been used to monitor quality of oils with legal rejection limit of frying oil being 3.0 acid values. Volatile compounds, particularly aldehydes, shaped formed during frying technique of oils are a conclusive factor for flavor of fried products even at low concentrations. Along these lines, measurements of volatile compounds in frying oils would be significant to screen their quality [4].
During frying technique, hydrolysis, oxidation, and polymerization are formed of the oils. These chemical reactions of frying oils mostly depend on presence of fatty acid compositions and antioxidant contents. Its realized that the higher unsaturated fatty acids exist in the oils, the faster thermal oxidation occurs during frying. It has been recommended that fats and oils with high amounts of saturated fatty acids, example coconut oil and palm oil, be utilized in frying in substitution for traditionally frying oils with high amounts of unsaturated fatty acids [5]. Ostrich bird (Struthio camelus) is an enormous flightless flying creature local to African Grassland and Desert. Wild ostrich has been recorded in the substance of Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES) since 1973. Since it gives numerous things of extraordinary incentive to man, as a wellspring of ostrich meat, leather, feather, egg and fat, in excess of 50 nations have been advancing the ostrich ventures enthusiastically on the worldwide scale [6]. Extracted oil from ostrich was used by ancient cultures like a cosmetic as well as in the remedy of burns and lesions. Cleopatra is thought to have used ostrich oil as part of her beauty regiment for maintaining her beauty and attractiveness in the desert climate. Most of the ostrich oil benefits are mainly caused by the ω-fatty acids contained in the oil [7,8]. Ostrich oil comes from the fat of the ostrich. This biggest flightless bird in the world, the Ostrich, has been a valuable source of food and other purposes for thousands of years for many native tribes [9]. For centuries ostrich oil has been used by Egyptian, Roman and African cultures for the relief of dry skin, burns, lesions, eczema, sunburn and even dry hair [10]. Ostrich oil It’s a great source of Omega 3, 6 and 9 which contain vital fatty acids for the growth of new skin cells and is naturally rich in vitamins well known for skin rejuvenating. Due to the way that tamed ostriches are being fed with an all- natural diet without any animal by-products, the natural oil is fantastic for your skin. It infiltrates into the skin and provides a moisture layer without clogging skin pores and leaves your skin without feeling greasy. It can be combined with a variety of essential oils to sooth aches and pains. At our Safari Ostrich Leather Shop, we stock a brand-new range of modern cosmetics under the name of Struis Lux. These exclusive products are all based on ostrich oils. The Ostrich oil items extend from Hand creams, body creams, day and night lotions and ostrich balsam [10-13]. The extracted oil from ostrich contains 28% saturated fatty acids, mostly as palmitic acid (20%) and stearic acid (8%). The polyunsaturated fatty acid is about 20% linoleic acid and 2% linolenic acid and the oil is high in oleic acid [11,12]. Ostrich oil was used as a new source of dietary fat [14] and was also introduced in the production of healthy biscuits [14]. However, there is less study data elucidating its fatty acid composition and physicochemical properties being reported, which limits the utilization of this natural product. Such characterization is indispensable for the in‐depth pharmacological research and quality control of ostrich fat. Therefore, the present study was focused on collecting the basic information of ostrich oil in terms of fatty acid composition and physicochemical properties. Separate into two distinct phases, a liquid phase and a solid phase and study characteristics of two phases. Also, assess the ostrich olein mixed with sunflower oil for deep-fat frying and to extent the shelf- life of oils.
Materials and Methods
Materials
Source of fats:
Ostrich fat (Sturuthio canelus) was obtained from Faculty of Agriculture Al-Azhar University, Cairo, Egypt, and sunflower oil was obtained from Savola Sim Misr 10th Ramadan, Egypt.
Solvents:
All solvents used throughout the whole work were analytical grade and distilled before use.
Methods
Rendering of fats:
Ostrich was extracted from fat tissues by dry rendering method reported by [15]. The rendering process was conducted at 90oC for 3 hrs. After cooling at 50oC, the fats were filtered through a Whitman No.1 filter paper and kept in brown bottles at 5oC until analysis.
Fractionation of ostrich oils:
OFat was fractionally for obtaining two fractions of the fat [15]. The liquid fraction (called olein) and solid fraction (called stearin).
Blending of olein fraction and sunflower oil:
The oil blending was prepared as follows:
a) 100 % ostrich.
b) 100 % sunflower oil.
c) 100 % liquid fraction.
d) 75 % liquid fraction + 25 % sunflower oil.
e) 50 % liquid fraction + 50 % sunflower oil.
f) 25 % liquid fraction + 75 % sunflower oil.
Physico- chemical properties of fats and blends:
Refractive index, melting point, smoke point, color, acid value, peroxide value, iodine value, polar content, insoluble polymer content, oxidized fatty acids, saponification number and unsaponifiable matter content were determined according to [16]. Fatty acid composition of the samples was determined by analyzing. The fatty acid methyl esters by gas liquid chromatography according to [17], unsaponifiable matter composition of the samples was determined by analyzed using gas liquid chromatography according to [18] and oxidative stability by Rancimat method at 100oC ± 2oC were determined according to the method of [19]. Frying process: Ostrich olein, sunflower oil and their blends were sued for frying potato chips as follows: A known amount (ca. 2kg) of each system was placed separately in a stainless-steel pan fryer (25cm diameter X 20cm height). The oils and their mixtures were separately heated at 180°C ± 5°C, then a lot of potato chips (2mm thickness) previously soaked in sodium chloride solution (10%, w/v) were fried. After frying of potato chips and at end of each day, sample oils were withdrawn and stored in brown bottles at 20°C until analysis.
Statistical analysis:
A one-way ANOVA followed by Duncan’s multiple range test (DMRT) were performed using SPSS 11.00 (SPSS Inc., Chicago, IL, USA) to analyze and compare the data. Results were presented as mean ± SD and P- values ≤ 0.05 were regarded as statistical significance.
Results and Discussion
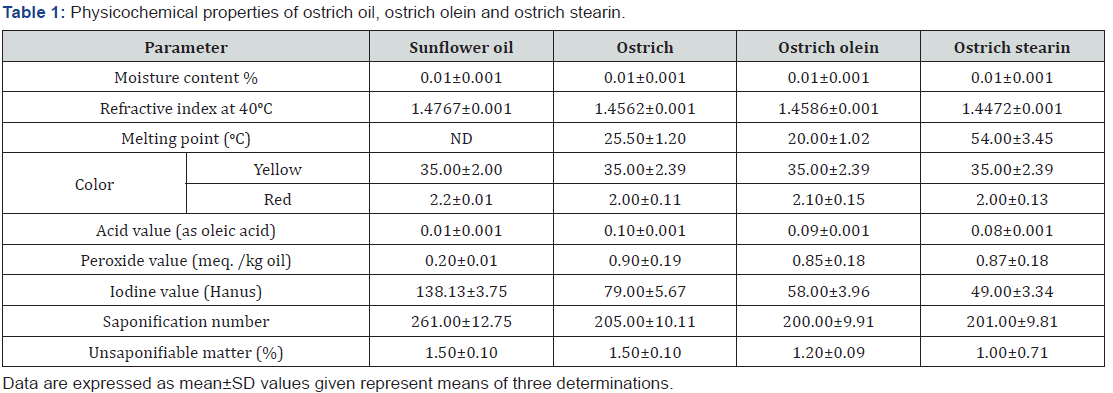
Physicochemical properties of ostrich oil, ostrich olein and ostrich stearin
The tabulated data in Table 1 showed that the moisture and volatile matter contents% and some physical and chemical properties of ostrich oil and its factions. From results in Table 1 the moisture and volatile matter content (%) within the ostrich oil and 2 fractions (ostrich olein and stearin) was 0.01 %. Whereas, the refractive index values at 40oC of ostrich oil and 2 fractions (ostrich olein and stearin) were 1.4562±0.001, 1.4586±0.001 and 1.4472±0.001, respectively. Data in Table 1 showed that the colour index of ostrich oil and 2 fractions (ostrich olein and stearin) were underneath the limitation that rumored by the [20], that mentioned that the red were 00.00, 2.20 and 2.50 at yellow 35 for edible oils. Freezing point of ostrich oil was less than those different fats. This information is comparable and therefore the proven fact that the ostrich oil is semi-solid at room temperature. The results indicated that the definite quantity (as nothing oleic acid) of ostrich oil was lower (0.10 %) than 2 fractions (0.27 and 0.30); respectively. The peroxide value of ostrich oil was 0.90 compared with 2 fractions that were, 1.70 and 1.85 meq. /kg sample, respectively. The iodine value of ostrich oil (79.00 gI2) is more than that of 2 fractions (53.00, and 56.00), respectively. Saponification number and unsaponifiable matter of ostrich oil (205.00 and 1.5%; respectively) were more those of 2 fractions shown in Table 1. These results are agreement thereupon rumored by [18].
Fatty acid composition of ostrich oil

Fatty acid composition of ostrich oil, ostrich olein and ostrich stearin were known by gas liquid chromatography and the obtained results are tabulated in Table 2. It may be noticed that monounsaturated fatty acid is found to be the dominant unsaturated fatty acid in ostrich oil and ostrich olein, which portrayed concerning (46.75±3.17%). Palmitic acid was found additionally to be the dominant saturated fatty acid in ostrich oil and ostrich stearin (28.50±1.24%). The results agree thereupon reported by [21,22].
Oxidative stability of ostrich oil, olein fraction, sunflower oil and their blends
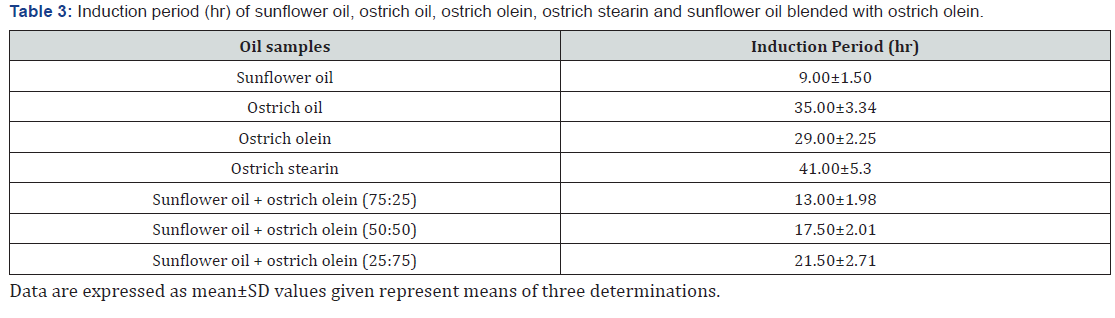
The changes in oxidative stability (Rancimat methodology at 100oC ± 2oC hrs) of ostrich oil, sunflower-seed oil (SO), stearin fraction (OF) and blends (SO/OF) are conferred in Table 3. Ostrich oil and stearin fraction were the foremost stable samples having an induction period was 35.00±3.34 and 41.00±5.3hrs, whereas sunflower-seed oil was the smallest amount stable sample with induction period 9.00±1.50hrs. Results in Table 3 shows that the oxidative stability of therefore supported induction amount was considerably enlarged by mixing with ostrich oil and its derivatives. The rise in oxidative stability was proportional to the rise of proportion within the blends. This study showed that combining therefore with completely different proportions of obtained from ostrich oil by dry fraction crystallization provides a good, simple, safe and accessible methodology to arrange additional stable edible oils are often employed in manufacturing French cooked potatoes.
Quality assurance for fried ostrich olein, sunflower oil and their blends during frying

Refractive index: Figure 1 show that the refractive index values for the fresh ostrich olein, sunflower oil and their mixtures (75:25, 50:50 and 25:75 v/v). The values demonstrate that ostrich olein refractive index value was higher than that of the refractive index of sunflower oil and blended samples. There is a strong relationship between the refractive index and iodine value, with higher iodine values would have higher refractive index and this fact is in line with data of the present work. The value of refractive index of fried ostrich olein, sunflower oil and blended of them indicate a linear relationship between their refractive indices and frying time. The increase of the refractive index values over frying time for the oil systems was in the order: Ostrich olein > ostrich olein + sunflower oil blends (50:50 v/v) > sunflower oil + ostrich olein (75:25 v/v) > sunflower oil + ostrich olein (75:25 v/v) > sunflower oil. This sequence is in line index values for the conjugated compounds are higher than that of their non-conjugated isomers. Its established that during oil frying some of the non-conjugated double bonds are converted to conjugated ones and this process cause an increase in the refractive index value [15].
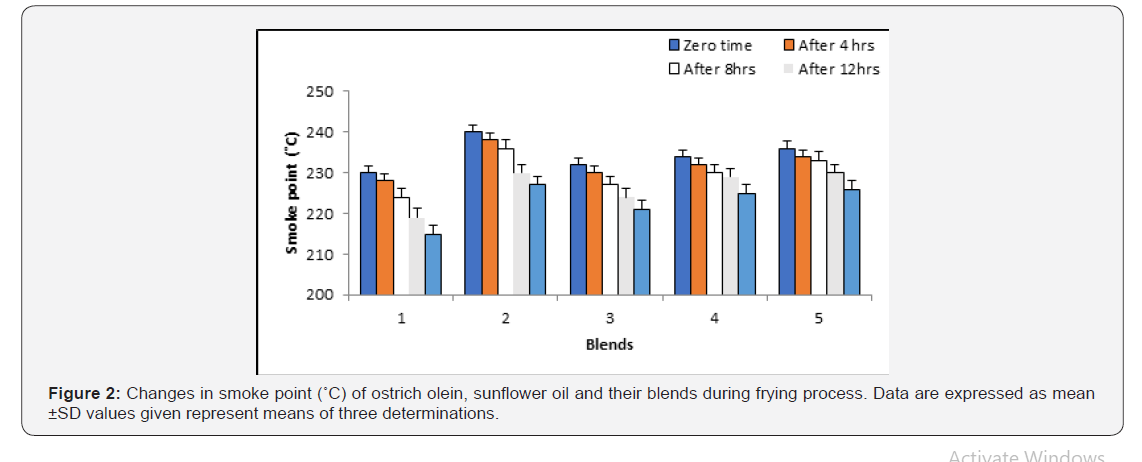
Smoke point (oC): Figure 2 shows the changes in smoke point of fresh, fried oils and their blends at various periods compared with those at zero time. The values of smoke point of fried ostrich olein were gradually decrease compared with sunflower oil. It is worth noting that the smoke points of fried ostrich oil mixed with sunflower oil at various levels (75:25, 7:3 and 50:50, v/v) were generally lower than ostrich olein alone.
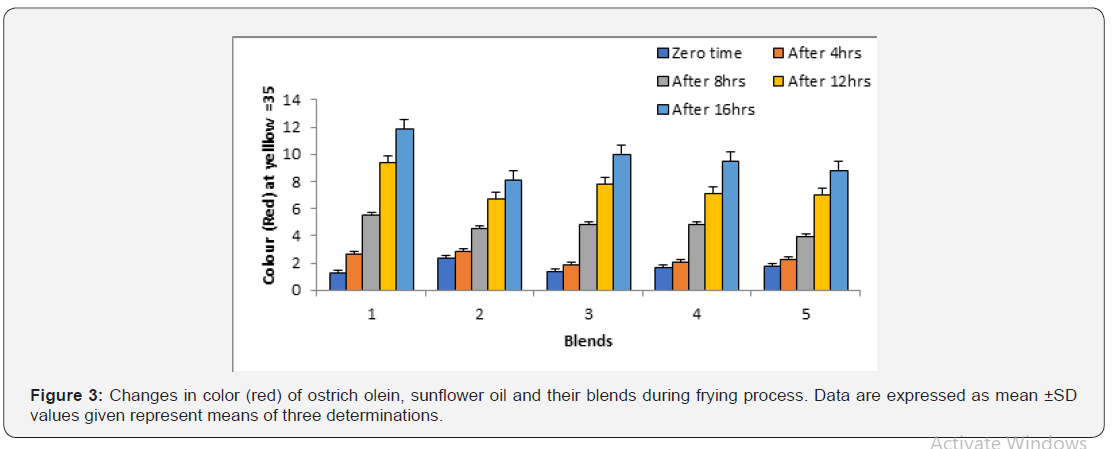
Color index:
In most cases two types of colored glasses of Lovibond tintometer, i.e., yellow and red, were used to measure the color of the oils. The yellow glasses were fixed at a value of 35 and the variation in oil color was matched with red glasses. Figure 3 illustrated that the initial red colors for ostrich olein, sunflower oil were 1.70 and 2.10 respectively. As a general trend, the intensity of the red color in all oil systems was increased as the frying time increased. The darkness of oil color due to frying at 180°C ± 5°C was arranged according to the sunflower > sunflower oil + ostrich olein blended (75:25, v/v) > sunflower oil + ostrich olein (75:25, v/v) > sunflower oil + ostrich olein (50:50, v/v) > ostrich olein. Accordingly blending ostrich olein with sunflower oil produced lighter frying media.
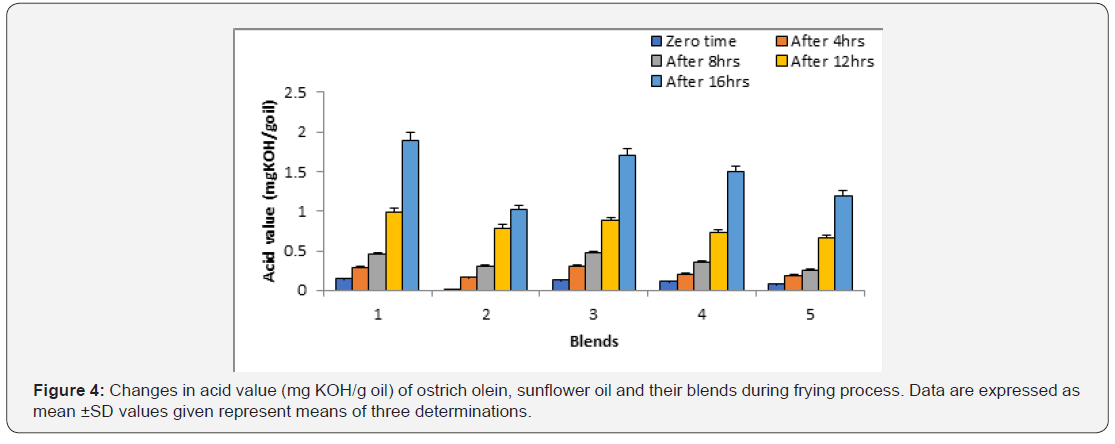
Acid value:
Acid value is one of the indicators used to assess oil quality. Figure 4 shows the changes of acid values for of fried under study. The acid value of fried oils showed gradually increase with frying time. Hence, the increase of the acid value was in the order: sunflower oil > sunflower oil + ostrich olein mixture (75:25, v/v) > sunflower oil + ostrich olein (75:25, v/v) > sunflower oil + ostrich olein (50:50, v/v) > ostrich olein. These findings demonstrate the improvement in sunflower oil quality during frying at 180°C ± 5°C when mixed with ostrich olein.
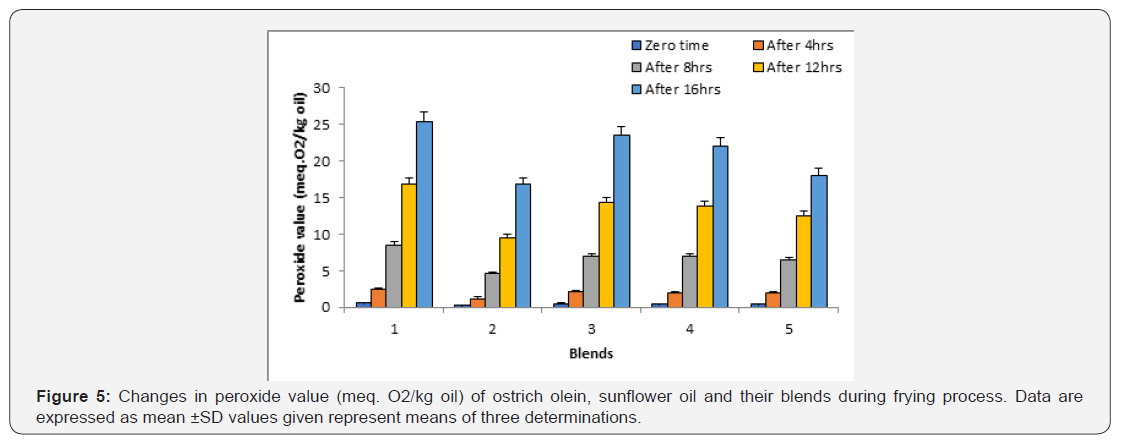
Peroxide value:
This fat constant indicator the primary oxidation products of the oils (hydroperoxides). Table 1 show the peroxide value of ostrich olein and sunflower oil at the beginning of the experiment and were 0.20 and 0.33 meq. O2 / kg oil, respectively. The peroxide value of these oils was within the recommended values for human consumption. The changes in the peroxide values of fried ostrich olein, sunflower oil and their blends are shown in Figure 5. The changes in peroxide value of the fried oils were progressively and significantly increased during frying process. The values of peroxide values for the oils at the end of frying indicate that the increase of peroxide value was in the in the order: sunflower oil > sunflower oil + ostrich olein mixture (75:25, v/v) > sunflower oil + ostrich olein (75:25, v/v) > sunflower oil + ostrich olein (50:50, v/v) > ostrich olein. It’s well known that the degree of oil oxidation is obviously dependent upon oil unsaturation and this order is in line with this fact. In other words, mixing ostrich olein with sunflower oil lowered the peroxide value of sunflower oil during frying process and hence increases the stability of sunflower oil during frying [23].
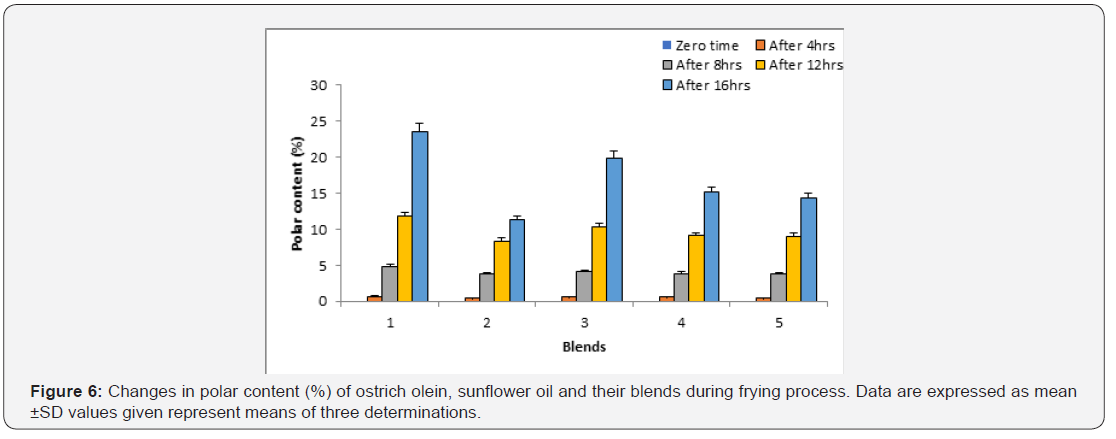
Polar content:
Changes in polar content of ostrich olein, sunflower oil and blends of them (7:25, 75:5 and 50:50 v/v) are show in Figure 6. At zero time, no detectable polar compounds were found. Frying of ostrich olein, sunflower oil and their blends at 180°C ± 5°C for 4hr/5 day caused increase in the polar compounds content of all oil systems. The increases of polar compounds contents of the oil systems were in the order: sunflower oil > sunflower oil + ostrich olein mixture (75:25, v/v) > sunflower oil + ostrich olein (75:25, v/v) > sunflower oil + ostrich olein (50:50, v/v) > ostrich olein. In addition, blending ostrich olein with sunflower oil induced lowering effect on the formation of polar content [24].
Polymer content:
At zero time, no detectable polymer compounds were found for ostrich olein, sunflower oil and its admixtures were nil Figure 7. The changes in polymer contents of the fried oil systems showed increases with time. The increases of polymer contents of the oil systems were in the order: sunflower oil > sunflower oil + ostrich olein mixture (75:25, v/v) > sunflower oil + ostrich olein (75:25, v/v) > sunflower oil + ostrich olein (50:50, v/v) > ostrich olein. These results indicate that blending ostrich olein with sunflower oil at different ratios possessed lower effect on the formation of polymers during frying process. It is well established that the degree of polymer formation is largely depends on the oil unsaturation [25].
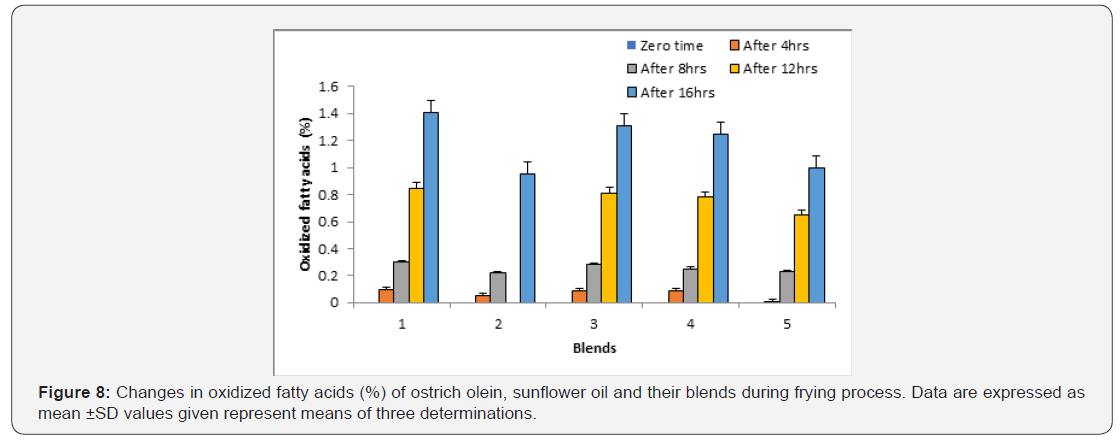
Oxidized fatty acids:
The results in Figure 8 showed the formation of oxidized fatty acids in all oil systems. The changes in oxidized fatty acids of the fried oil systems showed increases with time. Accordingly, the increase of the oxidized fatty acids contents were in the order: sunflower oil > sunflower oil + ostrich olein mixture (75:25, v/v) > sunflower oil + ostrich olein (75:25, v/v) > sunflower oil + ostrich olein (50:50, v/v) > ostrich olein. These results showed that blending ostrich olein with sunflower oil at different ratios led to the decrease of oxidized fatty acids contents during frying at 180°C ± 5°C.

References
- Mba OI, Dumont MJ, Ngadi M (2016) Deterioration kinetics of crude palm oil, canola oil and blend during repeated deep-fat frying. J Am Oil Chem Soc 93: 1243-1253.
- Warner K (2001) Chemical and physical reactions in oil during frying 1: Frying Technology and Practices. Eds. MK Gupta K, Warner PJ. White, AOCS press. Champaign (USA) pp. 16-28.
- Achir NK, Chipeaux WC, Trezzani I (2006) Effect of energy transfer conditions on the chemical degradation of frying oil. Eur J Lipid Sci Technol 108: 999-1006.
- Tyagi VK, Vasihtha AK (1996) Changes in the characteristics and composition of oils during deep-fat frying. J Am Oil Chem Soc 73: 799-706.
- Yu K, Cho H, Hwang H (2018) Physicochemical properties and oxidative stability of frying oils during repeated frying of potato chips 27(3): 651-659.
- Gegner L (2001) Ratite producton: Ostrich, emu and rhea, Livestock production Guide. ATTRA-National Sustainable Agriculture Information Services.
- International Ostrich Oil Standards (1998): American Emu Association oil standards. AEA News 5(7).
- Grompone AM, Irigaray B, Gil M (2005) Uruguayan nandu (Rhea Americana oil: A comparison with emu and ostrich oils. J Am Oil Chem Soc 82(9): 687-689.
- Chris P, Slates C (1999) Uses and benefits of emu oil. Nutrition & Food Science pp. 38-42.
- Sales J, Franken L (1996) Ostrich fat. Australian Ostrich Association Journal 37: 39-45.
- Brown MA, Craig-Schmidt MC, Smith PC (1995) Fatty acid composition of emu (Dromaius navaehollandiae) oil. INFORM 6: 470.
- Krawezk T (1997) Emu: premier oil bird. INFORM 8: 246-252.
- Craig-Schmidt MC (1999) Ratite oils: composition and claimed beneficial effects. Lipid Technol Newsletter 8: 80-83.
- Basuny AM (2007) Ostrich oil: A new source of animal fats. Journal of Biological Chemistry & Environmental Science 2: 217-235.
- Bailey (2005) Industrial oil and products. In: Hui YU, (5th), Wiley Internet. Science Publication John Wiley and Sons Inc pp. 499.
- AOAC (2016) Official Methods of Analysis Association of official Analytical Chemists, 20th Ed Association International, Arlington, Virginia, USA.
- Farag RS, Abdel-Rahim EA, Elsharabasy AMm Hewedy FM, Ragab AA (1984) Biochemical studies on lipids of hen’s egg during incubation. Seifen de-fette Wachse 100: 63.
- Mordert F (1968) Comparison and study of methods of obtaining Unsaponifiable matter. J Crops Grasses 6: 389-397.
- Evangelisti F, Zunin P, Tisconia E, Petacchi R, Drava G, Lanteri S (1997) Stability to oxidation of virgin olive oils as related to olive condition: study of polar compounds by chemometric methods. J Am Oil Chem Soc 74 (8): 117-121.
- Egyptian Standard Specification (1993): Egyptian standard (49 /1993) for Oils Egyptian Organization for Standardization and quality Control. Ministry of Industry Cairo Egypt.
- Bailey (1996) Industrial oil and products. In: Hui YU, (5th), Wiley Internet. Science Publication John Wiley and Sons Inc p. 20.
- Mandal AB, Wadhwa M, Bakshi MPS (2016) Animal Feeding - Concepts and Practices. Published by Satish Serial Publishing House Azadpur Delhi-110033 India.
- Nayak PK, Dash U, Rayaguru K, Krishnan KR (2016) Physio-chemical changes during repeated frying of cooked oil: a review. J Food Biochem 40: 371-390.
- Basuny AM, Arafat SM, Soliman HM (2017) Biological evaluation of ostrich oil and using it for production of healthy biscuit. Egyptian Journal Chemistry 60(6): 1091-1099.
- Kochar SP, Gertz C (2004) New theoretical and practical aspects of the frying process. Eur J Lipid Sci Technol pp. 722-727.






























