Soil as a Huge Laboratory for Microorganisms
Sachidanand B1, Mitra NG1,Vinod Kumar1,Richa Roy2 and Mishra BB3*
1Department of Soil Science and Agricultural Chemistry, Jawaharlal Nehru Krishi Vishwa Vidyalaya, India
2Department of Biotechnology, TNB College, India
3Haramaya University, Ethiopia
Submission: June 24, 2019; Published: September 17, 2019
*Corresponding author: Mishra BB, Haramaya University, Ethiopia
How to cite this article: Sachidanand B, Mitra NG, Vinod Kumar, Richa Roy, Mishra BB. Soil as a Huge Laboratory for Microorganisms. Agri Res& Tech: Open Access J. 2019; 22(4): 556205. DOI: 10.19080/ARTOAJ.2019.22.556205
Abstract
Biodiversity consisting of living organisms both plants and animals, constitute an important component of soil. Soil organisms are important elements for preserved ecosystem biodiversity and services thus assess functional and structural biodiversity in arable soils is interest. One of the main threats to soil biodiversity occurred by soil environmental impacts and agricultural management. This review focuses on interactions relating how soil ecology (soil physical, chemical and biological properties) and soil management regime affect the microbial diversity in soil. We propose that the fact that in some situations the soil is the key factor determining soil microbial diversity is related to the complexity of the microbial interactions in soil, including interactions between microorganisms (MOs) and soil. A conceptual framework, based on the relative strengths of the shaping forces exerted by soil versus the ecological behavior of MOs, is proposed. Plant-bacterial interactions in the rhizosphere are the determinants of plant health and soil fertility. Symbiotic nitrogen (N2)-fixing bacteria include the cyanobacteria of the genera Rhizobium, Bradyrhizobium, Azorhizobium, Allorhizobium, Sinorhizobium and Mesorhizobium. Free-living or associative N2-fixing bacteria belonging to the species Azospirillum, Enterobacter, Klebsiella and Pseudomonas have been shown to attach to the root and efficiently colonize root surfaces. Free-living soil bacteria play a vital role in plant growth, usually referred to as plant growth promoting rhizobacteria (PGPR) are capable of promoting plant growth by colonizing in plant root zone. These are associated with the rhizosphere, which is an important soil ecological environment for plant-microbe interactions. Plant growth promotion and development can be facilitated both directly and indirectly. Indirect plant growth promotion includes the prevention of the deleterious effects of phytopathogenic organisms. This can be achieved by the production of siderophores, i.e. small metal-binding molecules. Biological control of soil-borne plant pathogens and the synthesis of antibiotics have also been reported in several bacterial species. Another mechanism by which PGPR can inhibit phytopathogens is the production of hydrogen cyanide (HCN) and /or fungal cell wall degrading enzymes, e.g., chitinase and ß-1, 3-glucanase. Direct plant growth promotion includes symbiotic and non-symbiotic PGPR which function through production of plant hormones such as auxins, cytokinins, gibberellins, ethylene and abscisic acid. PGPR also help in solubilization of mineral phosphates and other nutrients, enhance resistance to stress, stabilize soil aggregates, and improve soil structure and organic matter content. This review indicates that soil environment drivers and their interactions may cause changes in soil microbial community and activity overall abundance; however, for this addition of organic matter, loosening the soil mass, providing the optimum moisture in soil, reducing the heavy agricultural equipment, replacing chemicals with alternative source of manure, could be few steps towards ecofriendly soil with high microbial population. These results illustrate the potential for complex community changes in terrestrial ecosystems under soil management scenarios that alter numerous factors simultaneously.
Keywords:Soil MOs, Shelter, PGPR, Phytohormone, Symbiotic/Non-Symbiotic N2 Fixation, Bacteria, Fungi, Archaea, Algae, Protozoa, Nematodes, Mites, Springtails, Spiders, Insects, Earthworms, Arthrobacter, Seudoponas, Azrobacterium, Microflora, Microfauna, Photoheterotrophs, Chemoheterotrophs
Introduction
Soil is a strange full of wisdom particularly in terms of biodiversity. Soil biota consists of the MOs (bacteria, fungi, archaea and algae), soil animals (protozoa, nematodes, mites, springtails, spiders, insects and earthworms) and plants, living all or part of their lives in or on the soil or pedosphere. Millions of species of soil organisms exist but only a fraction of them have been cultured and identified. MOs (fungi, archaea, bacteria, algae and cyanobacteria)are members of the soil biota but are not members of the soil fauna. The soil fauna is the collection of all the microscopic and macroscopic animals in a given soil. The size of a soil organism can restrict its location in the soil habitat. Smaller members of the microfauna like nematodes are basically aquatic organisms that live in the thin water films or capillary pores of aggregates preying or grazing on other aquatic microfauna such as amoebas. Soil has a direct effect on the environmental conditions, habitat and nutrient sources available to the soil biota. The term pedosphere is often used interchangeably with soil and captures the concept that the soil is a habitat where the integration of spheres occurs. Thesespheres include the lithosphere, atmosphere, hydrosphere and thebiosphere. Numerous biogeochemical processes regulated by soil biota occur in the pedosphere.
The soil represents a favorable habitat for MOs and is inhabited by a wide range of MOs, including bacteria, fungi, algae, viruses and protozoa. The physical structure, aeration, water holding capacity and availability of nutrients are determined by the mineral constituents of soil, which are formed by the weathering of rock and the degradative metabolic activities of the soil MOs. Soil microbiology is the study of organisms in soil, their functions, and how they affect soil properties. It is believed that between two and four billion years ago, the first ancient bacteria and MOs came about in earth’s oceans. These bacteria could fix N2, in time multiplied and as a result released oxygen into the atmosphere.This led to more advanced MOs. Soil biology plays a vital role in determining many soil characteristics. The decomposition of organic matter by soil organisms has an immense influence on soil fertility, plant growth, soil structure, and C storage. As a relatively new science, much remains unknown about soil biology and their effects on soil ecosystems. The soil is home to a large proportion of the world’s biodiversity
Nutrients are extremely important and directly influence growth, yield and quality of crops. Soil MOs can provide nutrients to plants through the fixation of atmospheric N2 and or by enhancing nutrient mobilization/uptake through their biological activities such as mineralization, and through the production of siderophores, organic acids and phosphatases. As microbial number and activity are more intense in the rhizosphere than in soil it hardly seems conceivable that the development of rhizosphere microflora would not directly affect the development of roots. However, particularly attention has been paid to the beneficial effect exerted by rhizosphere inhabitants. Typical rhizosphere bacteria such as Arthrobaeter, Seudoponas and Azrobacterium were found long ago to be able to produce substances promoting plant growth [1]. Ectomycorrhizal fungi also provide the host plant with phytohormones and growth-regulating vitamins B [2]. Understanding soil ecology is considered to be important for the sustainability of ecosystems as well as for the restoration of disturbed habitats. Soil MOs contribute to soil quality and play an important role in soil ecosystem processes, such as nutrient cycling, organic matter decomposition and bioremediation. Both changes in soil microbial communities resulting from ecosystem management and global change can have significant impacts on ecosystem dynamics [3], and microorganisms respond sensitively to changes and environmental stress because they have intimate relations with their surroundings [4], therefore, interest in quantifying impacts on the biotic and abiotic component has increased with concern for the sustainability of agricultural ecosystem [3].
Classification of soil biota
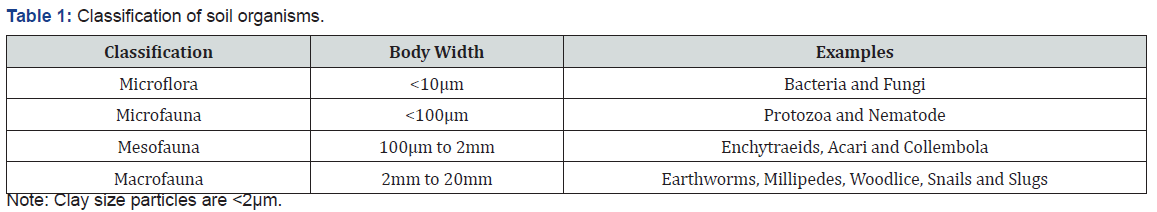
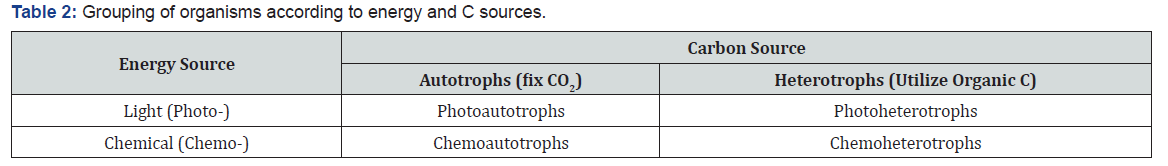
Soil organisms constitute the living organic component of soil and range in size across five orders of magnitude (viz. 10-7 to10-2m). Organisms present in soil are categorized into two main groups: Soil flora and fauna. These again divided into subgroups depending upon their size, soil macro-flora and micro-flora andsoil macro-fauna and micro-fauna. Soil organisms can be classified based on their size (Table 1). Sizes for soil organisms rangefrom 1μm to 20mm. All organisms need organic C to build their cells. Chemoheterotrophs obtain the organic C from organismswhich convert carbon dioxide (CO2) to organic compounds with either light energy (photoautotrophs) or chemical energy(chemoautotrophs). Photoheterotrophs utilize light energy and also utilize organic C. Organisms can be classified based on theenergy and C sources that they use (Table 2). Photoautotrophs conduct photosynthesis, a process in which they utilize atmospheric CO2 and water to produce organic C and oxygen in the presence of light. Photosynthesis is conducted by bacteria, algae and the higher plants, and is the foundation of food webs in diverse ecosystems. Photosynthesis is a biological process of converting CO2 from the atmosphere and is a link to terrestrial andaquatic ecosystems, which is represent by a reaction as:
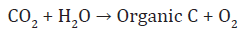
Photoheterotrophs utilize organic C compounds can alsoconduct photosynthesis. Therefore, it is possible for organisms,such as green and purple bacteria, to utilize organic and inorganic forms of C. Chemoautotrophs generate energy through theoxidation of inorganic compounds. Nitrifying bacteria can oxidize ammonium to nitrate under aerobic conditions and use the energy to convert CO2 to organic C. The reaction for these processes are:
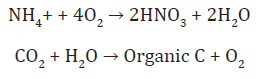
Aerobic chemoheterotrophs, most bacteria, fungi, protozoaand soil fauna, utilize organic C compounds for biomass and energy production, which is represents a reaction as:
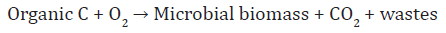
This process results in the conversion of organic compoundsinto CO2 under aerobic conditions. Oxygen is the terminal electron acceptor resulting in the formation of water. Anaerobic chemoheterotrophs, mostly heterotrophic bacteria, utilize organic C compounds for biomass and energy production but use other inorganic compounds, such as nitrate, as terminal electron acceptors. The reaction for denitrification is:
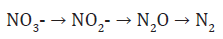
This process utilizes nitrate as a terminal electron acceptor, resulting in the formation of nitrous oxide (N2O) and dinitrogen gas (N2). As nitrous oxide is a greenhouse gas, this microbial process has a significant impact on radiative forcing of the troposphere. A diverse biological community in soils is essential to sustaining a healthy environment for plant roots. There may be over 100,000 different types of organisms living in soils. Most are providing numerous functions that assist plants, such as making nutrients more available, producing growth-stimulating chemicals and helping form soil aggregates. In a teaspoon of agricultural soils it is estimated that there are from 100million to 1billion bacteria, several yards of fungi, and several thousand protozoa. It may hold 10 to 20 bacterial-feeding nematodes and a few fungal-feeding and plant parasitic nematodes. Arthropods can number up to 100 square-1 foot, and earth worms from 5 to 30 square-1 foot.
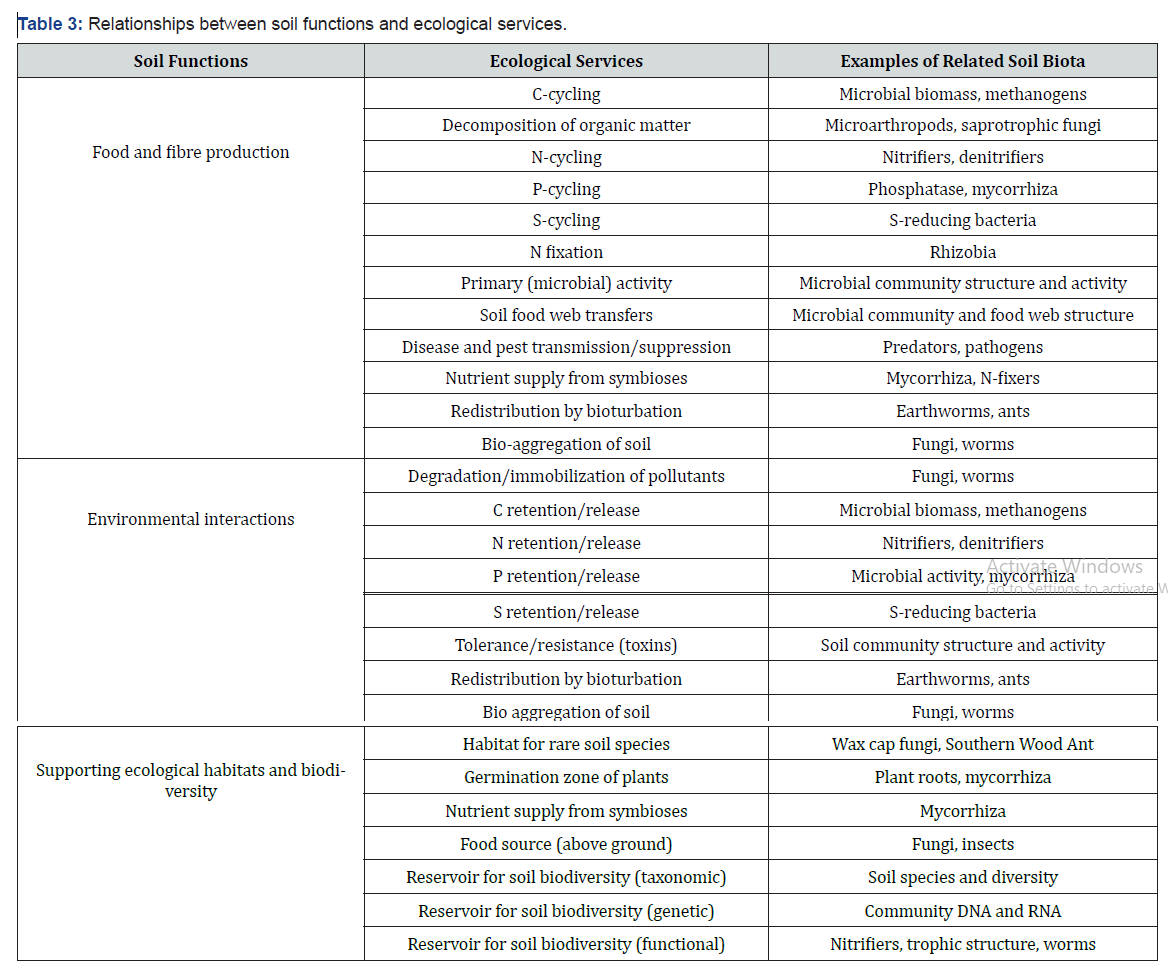
Of all the organisms in soils, only a small number of bacteria, fungi, insects and nematodes might harm plants in any given year. Diverse populations of soil organisms maintain a system of checks and balances that can keep disease organisms or parasites from becoming major plant problems. Some fungi kill nematodes and others kill insects. Still others produce antibiotics that kill bacteria. Protozoa feed on bacteria and may attack fungi. Some bacteria kill harmful insects. Many protozoa, springtails and mites feed on disease-causing fungi and bacteria. Beneficial MOs, such as fungus Trichoderma and bacteria Pseudomonas (P.) fluorescens, colonize plant roots and protect them from attack by harmful organisms. Some of these organisms, isolated from soils, are now sold commercially as biological control agents. The effects of bacteria and fungi that suppress plant disease organisms are thought to arise from competition for nutrients, production of antagonistic substances, and /or direct parasitism. In addition, a number of beneficial soil organisms induce the immune systems of plants to defend the plants. Also, roots of field crops usually have their own characteristic microbial communities with numerous interactions (Table 3).
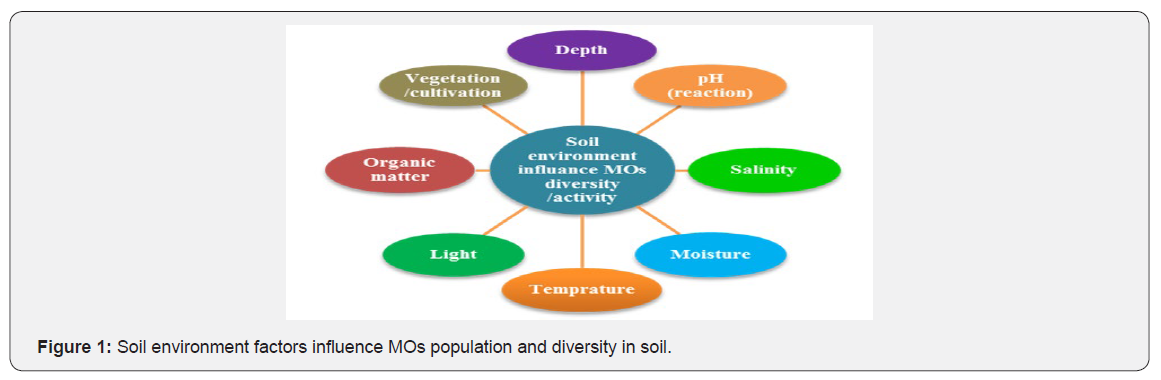
Soil organisms contribute to the maintenance of soil quality because they control many key processes. Soil MOs and their communities are continually changing and adapting to changes in their environment. A high quality soil is biologically active and contains a balanced population of MOs. In ecological theory, succession indicates the somewhat orderly and predictable species changes in space and time following the colonization of a new environment [5], and it has been one of the central ecological topics for over a century [6]. Several studies, the use of space-fortime substitution (chronosequence) succession, despite recent criticisms [7], has offered a unique opportunity to examine soil macro-community development in terms of biodiversity [8] nutrient cycling [9], natural and anthropogenic disturbances [10] and land-use changes [11]across multiple time scales. The physico-chemical and structural characteristics of soil provide many microenvironments in which complex bacterial populations can evolve [12]. As well as the physical properties of the soil, bacteria are also influenced by nutrient availability resulting from rhizodeposition and decomposition [13]. The diversity, abundance and activity of bacterial communities are therefore structured in relation to depth, since the primary source of nutrient input in grasslands is above ground. Several authors have attempted to examine the vertical distribution of bacteria in soils [14]. Bardgett et al. [15] in their study of grassland sites in Snowdonia reported that microbial biomass and activity (CO2 evolution) was highest in the top 5cm of soil and decreased down to a depth of 15 cm. There are many soil factors (Figure 1) which affect the activity of the soil MOs [16].
Soil depth
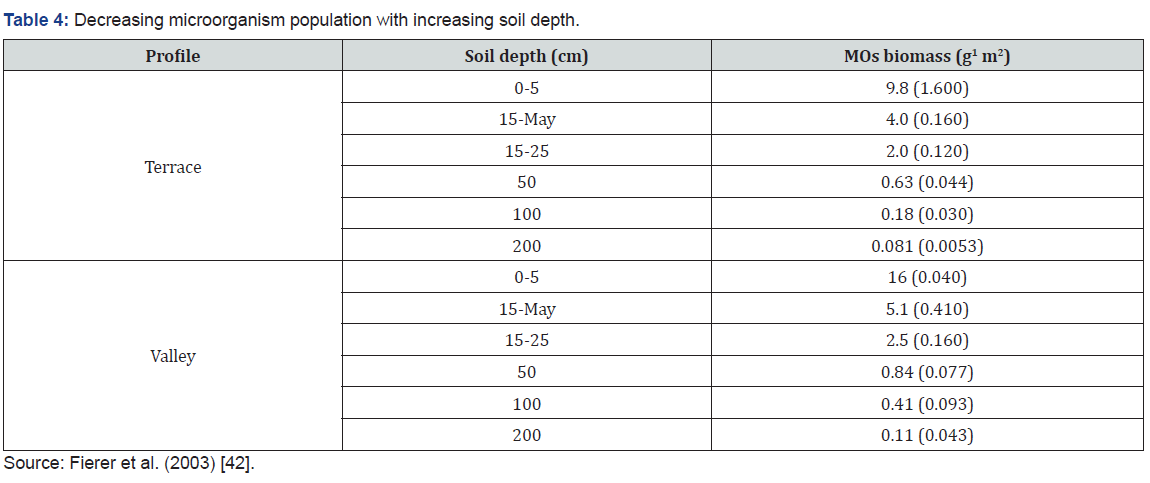
MOs are very diverse and include all the bacteria, archaea and almost all the protozoa. They also include some fungi, algae, and certain animals such as rotifers. MOs live in every part of the biosphere, including soil, hot springs, “7 miles deep” in the ocean, “40 miles high” in the atmosphere and inside rocks far inside within the Earth’s crust. Soil profiles are often many meters deep and soil varies from place to place, but with the majority of studies in soil microbiology focusing exclusively on the soil surface, we know very little about the nature of the microbial communities inhabiting the deeper soil horizons. These subsurface microbes play an important role in soil formation, ecosystem biogeochemistry, contaminant degradation, and the maintenance of ground water quality [17,18]. Fiere et al. [19] show that Gram-positive bacteria and actinomycetes tended to increase in proportional abundance with increasing soil depth, while the abundances of Gram-negative bacteria, fungi, and protozoa were highest at the soil surface and substantially lower in the subsurface. The vertical distribution of these specific microbial groups can largely be attributed to the decline in C availability with soil depth (Table 4). We expect the strong resource and environmental gradients found within the soil profile to cause microbial community composition to change with soil depth. As we examine progressively deeper layers in the soil profile, the quantity and quality of C substrates decline, while soil moistures and temperatures become less variable. Surface soils are rich in available C substrates from the input of root exudates, surface litter and root detritus. In contrast, the rates of C input to the lower horizons are generally low and the C tends to be of limited lability [20,21].
In a similar manner to temperature, organisms have a range of pH at which they are active. Within certain limits, organisms can tolerate extremes but this normally requires the cell to use energy in maintaining the correct internal cellular pH (pH 7.0). A few organisms (bacteria and archaea) can tolerate very extreme pH values such as pH 1 or pH 11 but these are extreme conditions are not found in agricultural soils. Larger soil animals may be more sensitive to pH than MOs. Earthworms being sensitive to low pH are not active in forest or peat land systems (pH<5) their function is replaced by other organisms (enchytraeids in forests) or the soil forms in a different manner (peatlands). pH directly affects the solubility of elements. At acidic pH, aluminium becomes more soluble and hence more available to the organisms with increased toxicity. Essential minerals can become unavailable at extremes of pH. For example, phosphorus (P) and manganese (Mn) become increasingly unavailable at high pH values.
Soil Salinity
Salinization consists of an accumulation of water soluble salts in the soil. These salts include the ions potassium (K+), magnesium (Mg2+), calcium (Ca2+), chloride (Cl-), sulfate (SO4 2-), carbonate (CO3 2-), bicarbonate (HCO3 -) and sodium (Na+). Sodium accumulation is also called sodification. High Na+ contents result in destruction of the soil structure which, due to a lack of oxygen, becomes incapable of assuring plant growth and animal life. The microbial communities of the soil perform a fundamental role in cycling nutrients in the volume of organic matter in the soil and in maintaining plant productivity. Thus it is important to understand the microbial response to environmental stress, such as high concentrations of heavy metals of salts, fire and the water content of the soil. Stress can be detrimental for sensitive MOs and decrease the activity of surviving cells, due to the metabolic load imposed by the need for stress tolerance mechanisms [22]. In a dry hot climate, the low humidity and soil salinity are the most stressful factors for the soil microbial flora, and frequently occur simultaneously. Saline stress can gain importance, especially in agricultural soils where the high salinity may be a result of irrigation practices and the application of chemical fertilizers. Research has been carried out on naturally saline soils, and the detrimental influence of salinity on the soil microbial communities and their activities reported in the majority of studies [23,24]. The effect is always more pronounced in the rhizosphere according to the increase in water absorption by the plants due to transpiration. The simple explanation for this is that life in high salt concentrations has a high bio-energetic taxation, since the MOs need to maintain osmotic equilibrium between the cytoplasm and the surrounding medium, excluding Na+ ions from inside the cell. As a result, energy sufficient for osmo-adaptation is required [25]. There is a significant reduction in the total fungal count in soils salinized with different concentrations of sodium chloride. Similarly, with an increase in the salinity level to above 5%, the total count of bacteria and actinobacteria was drastically reduced [26].
Soil moisture
Soil moisture affects the soil biota in two ways. Biologically water is essential for life and for enzyme activity and metabolism and, is a solvent for biological nutrients and other chemicals. Physically, soil moisture affects soil temperature (water is good conductor of heat) and soil aeration. The degree at which soil pores are filled affects the movement and predation of MOs in soil. In very dry soil, plants may not be able to extract sufficient water through the roots because of the energy it takes to remove water from the small pores. This is known as the permanent wilting point, beyond which the plant cannot recover. Conversely, under wet conditions, oxygen does not diffuse through the soil as readily so the levels available to organism may become depleted leading to anaerobic conditions. Fungi tend to be more resistant to water stress than bacteria although under extreme conditions, some bacteria can form resistant endospores which allow the organism to survive until more suitable conditions arise. Chemicals may be produced by the cell to allow survival under drought. Actinomyces which can tolerate drier conditions than fungi can protect themselves from drought by synthesizing the amino acid proline. Regional climate factors exert major influences on distributions of microbial communities by determining temperature and soil water availability along topographic gradients [27]. Brockett et al. [27] found that soil water availability was an important determinant of microbial community composition, and fungal: bacterial biomass ratios decreased with increased soil water saturation at regional scales. Water is not essential transport medium for subtracts, it is also an important participant in hydrolysis process. However, excess soil water content results in limited O2 diffusion because O2 in water is much lower (about 104 times) than in air which will reduce the activity of aerobic MOs [28] but could increase the activities of anaerobes. Changes in soil redox status influence the pool of electron acceptors available to MOs, resulting in different MOs being adapted to static or dynamic redox conditions [29]. Our ability to prediction restoration of microbial functions such as water quality improvement via denitrification can be enhanced through understanding the relative influence of landscape compared to local factors on soil microbial communities. Higher contributions of gram positive bacteria were found in wetter soils, whereas higher contributions of gram-negative bacteria and fungi were observed in drier soils [30].
Temperature directly affects the activity of the soil MOs by determining the rate of physiological activity such as enzyme activity and indirectly by affecting physico-chemical properties such as diffusion and solubility of nutrients, mineral weathering and evaporation rates and so on. Within defined limits biological activity increases with increasing soil temperature. Organisms have a specific range of temperature at which their biological activity operates. For common soil organisms the temperature range at which they can be active ranges from about 0 to 60°C although no single species is likely to be active throughout the entire range. This temperature range is largely determined by the temperature at which soil can reach although organisms may be able to survive outside the soils temperature range or produce survival structures to allow them to survive under adverse conditions. It was once thought that the highest temperature that could be survived by MOs was around 40°C but this has been reduced to around 121°C. Within an organisms temperature range, there is a temperature optimum at which biological function performs better. Beyond this value, cellular processes do not work so efficiently and as the temperature increases away from their upper limit there are irreversible changes to the cell properties leading to cell death. Those organisms which have a high temperature limit would tend not to be so active at the lower extreme compared to organisms more suited to the lower limit.
The mechanisms behind temperature adaptations of soil microbes could be physiological adaptations of single species [31] or species shifts within the microbial community. Soil respiration is dependent on soil temperature and moisture and may increase or decrease as a result of changes in precipitation and increased atmospheric temperatures. Many ecosystems experience dramatic inter-and intra-annual variability in precipitation and temperature [32]. Such fluctuations in climate, from year-toyear or season-to-season, could have significant effects on soil microbial communities, directly via effects on soil moisture and temperature. For example, AMF (Arbuscular Mycorrhizal Fungi) composition has been shown to vary seasonally, with distinct differences between winter and summer AMF communities [33]. Different microbial groups have distinct optimal temperature ranges for growth and activity. Increase temperature can affect the composition of the microbial community, which in some cases could reduce the release of soil organic carbon owing to the loss of acclimatized microbial groups [34]. Changing soil temperature will likely alter microbial mediated denitrification and denitrification dynamics in soil environment due to shift of nitrifiers and denitrifiers population. Thus, microbial growth and activity generally decreases in winter, due to the decreased temperature. Extremely high temperatures, in general, are deleterious for many MOs. Indeed, some species may survive such adverse conditions by entering in to inactive forms, which may resist high temperatures. However, such expected seasonal dynamics may change in specific soil ecosystems, for example, in tundra soils, microbial biomass is at its maximum in late winter time when temperature is low [35]. The optimum average temperature for survival of microbes is just above 20°C while the higher limit is around 50°C [36].
Light
In soil, light directly affects those organisms on or just below the surface and indirectly by heating the soil surface. Phototrophs, such as plants, algae and cyanobacteria, use the energy from sunlight to synthesis carbohydrates. In plants, some of this material (photosynthate) finds its way into the soil biota via the roots and from leaf fall. With algae and cyanobacteria, the organic matter is directly inputted into soil either from release of material from the cell or when the cell dies. Parts of the light spectrum are more damaging to organisms than others. Ultraviolet light can damage DNA which induces mutations in the organisms resulting in death of the organism or biochemical changes through for example changes of enzymes and metabolic pathways. Photochemical degradation will lead to structural changes which organic molecules making them more or less easy to be degraded. Soils harbour enormous microbial diversity. The total fresh weight mass of organisms below temperate grassland can exceed 45 tonnes ha-1, equalling or exceeding above-ground biomass [37]. Bacteria are present in greatest numbers, with archaea 10-fold less. Estimates of the number of species of bacteria g-1 of soil range from 2000 to 18000. Fungi, however, often contribute the largest part of the total microbial biomass in soils. The soil environment is very complex and provides diverse microbial habitats. Soils vary greatly depending on climate, organisms, land form and parent material.
Soil organic carbon
Humus is the organic residue in the soil resulting from decomposition of plant and animal residues in soil, or it is thehighly complex organic residual matter in soil which is not readily degraded by MOs, or it is the soft brown/dark coloured amorphous substance composed of residual organic matter along with dead MOs. Soil flora and fauna plays a great role in improving the soil texture, nutrient and crop productivity. Bacteria on decomposing plant tissue secrets polysaccharides and other organic glue. Sticky sugar-protein called glomalin secreted by mycorrhizae possess cementing properties which helps to hold the soil particles together. Also decomposition of organic component by the bacteria increases the soil porosity which increases the infiltration capacity, thus protects the soil from the erosion [38]. Tropical soils show higher emission rates of CO2 as compared to the soils of temperate regions because of higher and longer thermal regimes where rate of organic matter decomposition is much higher due to enhancement of microbial activities. An increase in temperature in a high-latitude ecosystem resulted in an up to 50% decrease in bacterial and fungal abundance and soil respiration, suggesting that increased temperature does not always lead to enhanced C loss to the atmosphere. Sometimes perturbations in the soil environment could lead to community shifts and altered metabolic activity in MOs involved in soil nutrient cycling, and to increasing or decreasing survival and virulence of soil mediated pathogenic MOs like Salmonella typhimurium. Schindlbacher et al. [39] reported soil warming did not affect microbial biomass, nor did warming affect the abundances of most microbial groups. Warming significantly enhanced microbial metabolic activity in terms of soil respiration per amount of microbial biomass C. In summary, the 4°C increase in soil temperature during the snowfree season had no influence on microbial community composition and biomass but strongly increased microbial metabolic activity and hence reduced C use efficiency.
Vegetation /Cultivation
The crops rotation where plants predominate that are succulent and rich in proteins and sugars such as legumes release nutrients more quickly, whereas plants that are more fibrous, such as grasses and cereals, release nutrients more slowly, but promote more stable organic matter [40]. Systems that increase inputs of C and N through the inclusion of legumes or fibrous rooted crops in the crop rotation, may increase microbial populations and activities. Azospirillum is micro aerobic that fixes the N2 in association with roots of grasses. Inoculation of Azospirillum to the grass crops have positive hormonal effect on roots and plant growth [41]. Gram-positive and Gram-negative bacteria showed different patterns in substrate preference. Gram-positive bacteria were found to be dominant in soils with low substrate availability and in deeper soil layers [42], while Gram-negative bacteria were found to dominate soils with high availability of easily decomposable substrate [43]. Studies have shown varying results with regards to N immobilization in reduced-tillage systems. When farmers first convert to minimum or no-till cropping, they often encounter lower N availability for the first several years because of reduced mineralizable N. SOM (and N) accumulates under no-till, however, and a new equilibrium is established in which mineralized N and microbial biomass C are higher than under intensive tillage [44]. Anaerobic bacteria, such as clostridia, dominate in no-tilled soil as well as anaerobic methanogenic archaea, which were detected only in the no-tillage system. Microbial diversity was higher in plots in which only cereals (oat and maize) were grown [45]. In no-tillage conditions, the nutrient releasing is progressive, since the residues remain on the soil surface and therefore decomposition is slower. The crop rotation and tillage management together, determine the soil C:N ratio, which has significant effect on microbial composition [46], crop residue decomposition and nutrient cycling.
Transgenic crops holds great promise for improving agricultural output, but the potential unwanted effects of transgenic crops on soil microbial diversity is still not fully understood. The majority of studies addressing the risks of transgenic crop cultivation have addressed only aboveground effects. Mandal et al. (2015) observed that, soil enzyme activities and microbial population counts of cellulose decomposers, phosphate solubilizers and N2 fixers were found higher under Bt cotton than non-Bt cotton system and found higher activities of beneficial microbes and diversity under Bt-cotton than non Bt cotton cropping system might be due to greater rhizodeposition, leaf fall and root biomass accumulation serving as source of bio-energy for native microbes. In recent studied by Vinod Kumar et al. [47] reported that populations of effective rhizobia in plough layers (0-15cm depth) in soybean and cereal based cropping systems in Central India. Population of rhizobia was greater at maximum vegetative growth stage of crops. The presence of the host legume stimulated the rhizobial population by 25 fold (average most probable number 1262 cells g-1 soil in soybean -based cropping systems as compared to 50 cells g-1 in cereal-based system). There were 206 rhizobial cells g-1 soil in summer at sowing and re-bounded after rainy season at maximum vegetative growth stage to 2743 cells g-1 soil (by 13.3 fold) in soybean based rotations.
Bacterial and fungal population in Indian soils
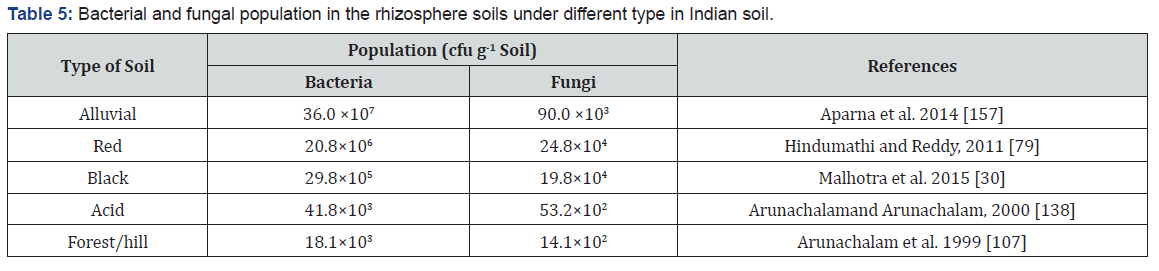
Soil microbes require what all other living organisms require a source of C and energy, adequate moisture and aeration, essential inorganic nutrients and trace elements necessary to build cells. Ideally, the soil should contain about 50% water filled pore space, allowing for a good balance of air and water filled pores for growth and activity of aerobic microbes. The basic plant nutrients cycle highlights the central role of soil organic matter. Cycling of many plant nutrients, especially N, P, S and B, closely follows parts of the C cycle. Plant residues and manure from animals fed forage, grain and other plant-derived foods are returned to the soil. This organic matter pool of C compounds becomes food for bacteria, fungi and other decomposers. Abiotic soil factors, including availability of nutrient, reaction, topsoil depth, water content, aeration, temperature and management practices, may affect the structure and activity of soil microbial communities [48-50]. Contrarily, low microbial population in forest hill and acid soil may be because addition of plant remains and wide C:N ratio and low cation (particularly Ca2+) content in the soil.
It is evident from the data that there is a wide range of soil type variation in bacterial and fungal population were studied by various researchers are given in (Table 5). Alluvial soil showed maximum abundance of bacterial population counts (36.0×107cfu g-1 soil) followed by red soil (20.8×106cfu g-1 soil), while minimum under forest /hill and acid soils (18.1×103 and 41.8×103cfu g-1 soil, respectively) in rhizosphere soils. Whereas, red and black soils showed higher fungal population (24.8×104 and 19.8×104cfu g-1 soil, respectively) as compared to acid and forest /hill soils. It is evident from the data that there is increase in population counts with improving soil aeration and optimum soil pH. Biological nitrogen fixation (BNF) is the major way to introduce N into arable land ecosystems and balance the C:N ration in the soil. BNF in deserts is mediated mainly by some heterotrophic bacteria, associative bacteria, cyanobacteria, actinorhizal plants and legumes. Several symbiotic systems of legumes which are tolerant to extreme conditions of salinity, alkalinity, acidity, drought, fertilizer, metal toxicity, etc., were identified. In fact, the existence of Rhizobium-tree legume symbioses, which are able to fix appreciable amount of N2 under arid conditions, is fascinating. Soil acidity is a significant problem facing agricultural production in many areas of the world and limits legume productivity [51,52[. Most leguminous plants require a neutral or slightly acidic soil for growth, especially when they depend on symbiotic N2 fixation [51,53]. It has been recently reported [54,55] that pasture and grain legumes acidify soil to a greater extent and that the legume species differ in their capacity to produce acids. Legumes and their rhizobia exhibit varied responses to acidity. Soil salinity and acidity are usually accompanied by mineral toxicity (specific ion toxicity), nutrient deficiency and nutrient disorder. Salt damage to non-halophytic plants grown in nutrient solution is often due to the effect of ion imbalance (disorder) rather than the osmotic potential [56]. In India, alluvial and black soils are commonly noticed normal soil pH and suitable for legume/oil seed production and these soils maintain a narrow C:N ratio as compared to acid/ forest soil. In this might due to alluvial and red soil more support bacterial and fungal population.
Microbisols
Mishra (2015) proposed a soil group called Microbisols, which represents a soil type usually characterized by effective group of microorganisms. Regulating ecosystem services control the processes of water flow, energy transfer, nutrient uptake and release, carbon transfer, and chemical processing, as well as services provided by creating and maintaining environments for diverse plant, animal, and microbial communities. The role of MOs in the genesis and maintenance of soil structure has recently been reviewed [57]. Our aim here is to emphasize the importance of this process in the rhizosphere. It has been demonstrated that there are more water-stable aggregates in the rhizosphere than in the non-rhizosphere soil [58]. Since the number of polysaccharide producing MOs is characteristically higher in the rhizosphere, it can be assumed that soil stabilization around the root can, at least to some extent, be due to ‘the rhizosphere microflora. In tropical soils, where most of the microbial population is concentrated in the root zone, it would be worthwhile to elucidate the relative importance of the root itself and that of associated MOs in soil structure stabilization. Such investigations should not be restricted to free-living MOs, (such as Asotojacter sp., Beijerinekia indica or Lipomgees starkeyi, which are well own polysaccharide producers), but should be extended to corrhizae which were reported to be involved in sand aggregation and’ dune stabilization in colder climates [59]. In contrast to this beneficial activity, MOs can be harmful in two ways: by decomposing the aggregating compounds originating from plants or microorganisms; and by coating soil particles with water-repellent films [60].
Soil health is worth quantifying because soils and their biota provide ecosystems functions that benefit humans. These ecosystem services can be of considerable value (Costanza et al., 1997) and include soil functions of storing and releasing water, decomposing plant and animal residues, transforming and recycling nutrients, sequestering and detoxifying organic toxicants, and promoting plant health by suppressing plant-pathogenic microbes and phytophagous fauna. It is often possible and desirable to measure soil function directly. Direct measurements of some soil functions may be too expensive (for example, direct measurements of nutrient transformations) or require observations across too much time (for example, the capacity of a soil to supply water for plant growth during a drought maybe observable only during rare drought years). In such cases, rather than measuring the soil function directly, it may be preferable to measure surrogates or proxies that are well correlated with the soil function. Soil organisms meet this criterion, because the abundance and diversity of soil organisms often are well correlated with many beneficial soil functions [61]. Indicators Relationship to soil condition and Rationale as a priority measurement Microbial biomass C and N Microbial catalytic potential and respiratory for C and N Potentially mineralizable N (anaerobic incubation) Soil productivity and N supplying potential Dehydrogenase activity, Soil respiration, water content and temperature Microbial activity measure At the beginning of any soil-monitoring program it may be possible to identify a suitable reference site where the actual values of SMB approach the attainable SMB value for that ecosystem (i.e. determined by localised climate, soil type, and land use). This is often the case for land restoration projects where either the characteristics of the site before disturbance have been well documented and/or a parallel undisturbed system can be identified [62, 63].
In the case of forest rehabilitation (e.g. after mining), the natural forest (under equilibrium for current climatic conditions) surrounding the rehabilitated areas sets the attainable value and, hence, the target for the recovery of the mined and rehabilitated soils.
MOs vs functions in soil
Living organisms both plant and animal types constitute an important component of soil. Though these organisms form only a fraction (less than 1%) of the total soil mass, but they play important role in supporting plant communities on the earth surface. Soil microbes, bacteria, archaea, and fungi play diverse and often critical roles in these ecosystem services (Figure 2). The vast metabolic diversity of soil microbes means their activities drive or contribute to the cycling of all major elements (e.g. C, N, P), and this cycling affects the structure and the functions of soil ecosystems as well as the ability of soils to provide services to people. While studying the scope and importance of soil microbiology, soil-plant-animal ecosystem as such must be taken into account. Although it may be useful to examine physical, chemical and biological aspects of soil quality individually, soil should be viewed as an integrated system. For example, physical and chemical properties are shaped by biological activity, and biological activity is enhanced or limited by chemical and physical condition. A healthy soil is ‘biologically active’ containing a wide diversity of MOs. Relevant biological properties include soil organic matter content, microbial biomass, respiratory activity, N mineralization, soil enzymes, soil fauna and population of suppressive organisms. For a good working definition of soil biological health and ease of analysis, biologically active fractions of SOM (soil organic matter) and biochemical attributes of soil have proven more useful. Therefore, the scope and importance of soil microbiology, can be understood in better way by studying aspects like:
a) Soil microbes and plant growth
b) Soil microorganisms and soil structure
c) Organic matter decomposition and humus formation
d) Biogeochemical cycling of elements
e) Soil microorganisms as bio-control agents
f) Degradation of pesticides
g) Biodegradation of hydrocarbons
Different bacterial genera are vital components of soils. They are involved in various biotic activities of the soil ecosystem to make it dynamic for nutrient transformation and sustainable for crop production [64]. They stimulate plant growth through mobilizing nutrients in soils, producing numerous plant growth regulators, protecting plants from phytopathogens by controlling or inhibiting them, improving soil structure and bioremediating the polluted soils by sequestering toxic heavy metal species and degrading xenobiotic compounds (like pesticides) [65,66]. Indeed, the bacteria lodging around/in the plant roots (rhizobacteria) are more versatile in transforming, mobilizing/ solubilizing the nutrients compared to those from bulk soils [67]. Therefore, the rhizobacteria are the dominant deriving forces in recycling the soil nutrients and consequently, they are crucial for soil fertility [68].
Currently, the biological approaches for improving crop production are gaining strong status among agronomists and environmentalists following integrated plant nutrient management system. In this context, there is an ongoing rigorous research worldwide with greater impetus to explore a wide range of rhizobacteria possessing novel traits like heavy metal detoxifying potentials [66], pesticide degradation/tolerance [69], salinity tolerance [70], biological control of phyto-pathogens and insects [88] along with the normal plant growth promoting properties such as, phytohormone [70,71], siderophore [72], 1-aminocyclopropane-1-carboxylate, HCN, and ammonia production, nitrogenase activity [62,73] phosphate solubilization [65] etc.
Although, the mechanisms of rhizobacteria-mediated plant growth promotion are not completely identified, the so called plant growth promoting rhizobacteria however, have been reported to exhibit the above mentioned properties to expedite the plant growth and development [74]. The present review is an effort to elucidate the concept of rhizobacteria in the current scenario and their underlying mechanisms of plant growth promotion with recent updates. The latest paradigms of a wide range of applications of these beneficial rhizobacteria in different agro-ecosystems have been presented explicitly to garner broad perspective regarding their functioning and applicability.
Plant growth promoting rhizobacteria
Plant growth promoting rhizobacteria (PGPR) are the soil bacteria inhabiting around /on the root surface and are directly or indirectly involved in promoting plant growth and development via production and secretion of various regulatory chemicals in the vicinity of rhizosphere. Generally, PGPR function in three different ways: synthesizing particular compounds for the plants, facilitating the uptake of certain nutrients from the soil, and lessening or preventing the plants from diseases. The PGPR, are characterized by the following inherent distinctiveness’s:a) they must be proficient to colonize the root surface
a) they must be proficient to colonize the root surface
b) they must survive, multiply and compete with other microbiota, at least for the time needed to express their plant growth promotion /protection activities, and
c) they must promote plant growth [75].
The growth promoting ability of cyanobacteria was shown even for upland crops like tomato and PGPR action of increasing its vitamin C content [76] at IARI, followed by Rao (1975) who showed indole acetic acid (IAA) and gibberellin production by Azotobacter and significant improvement of Vitamin C content in tomato upon inoculation. Alternatively, Somers et al. [77] classified PGPR based on their functional activities as
a) biofertilizers (increasing the availability of nutrients to plant),
b) phyto stimulators (plant growth promotion, generally through phytohormones),
c) rhizoremediators (degrading organic pollutants) and
Furthermore, in most studied cases, a single PGPR will often reveal multiple modes of action including biological control [79]. In general, these can be separated into extracellular (ePGPR), existing in the rhizosphere, on the rhizoplane, or in the spaces between cells of the root cortex, and intracellular (iPGPR), which exist inside root cells, generally in specialized nodular structures [80]. Some examples of ePGPR are like, Agrobacterium, Arthrobacter, Azotobacter, Azospirillum, Bacillus, Burkholderia, Caulobacter, Chromobacterium, Erwinia, Flavobacterium, Micrococcous, Pseudomonas and Serratiaetc (30). Similarly, some examples of the iPGPR are Allorhizobium, Azorhizobium, Bradyrhizobium, Mesorhizobium and Rhizobium of the family Rhizobiaceae. Most of rhizobacteria belonging to this group are Gram-negative rods with a lower proportion being Gram-positive rods, cocci or pleomorphic [81]. Moreover, numerous actinomycetes are also one of the major components of rhizosphere microbial communities displaying marvelous plant growth beneficial traits [81]. Among them, Micromonospora sp., Streptomyces sp., Streptosporangium sp., and Thermobifida sp., which have shown an enormous potential as biocontrol agents against different root fungal pathogens, are worthy of mention [81].
Mechanisms of plant growth promotion
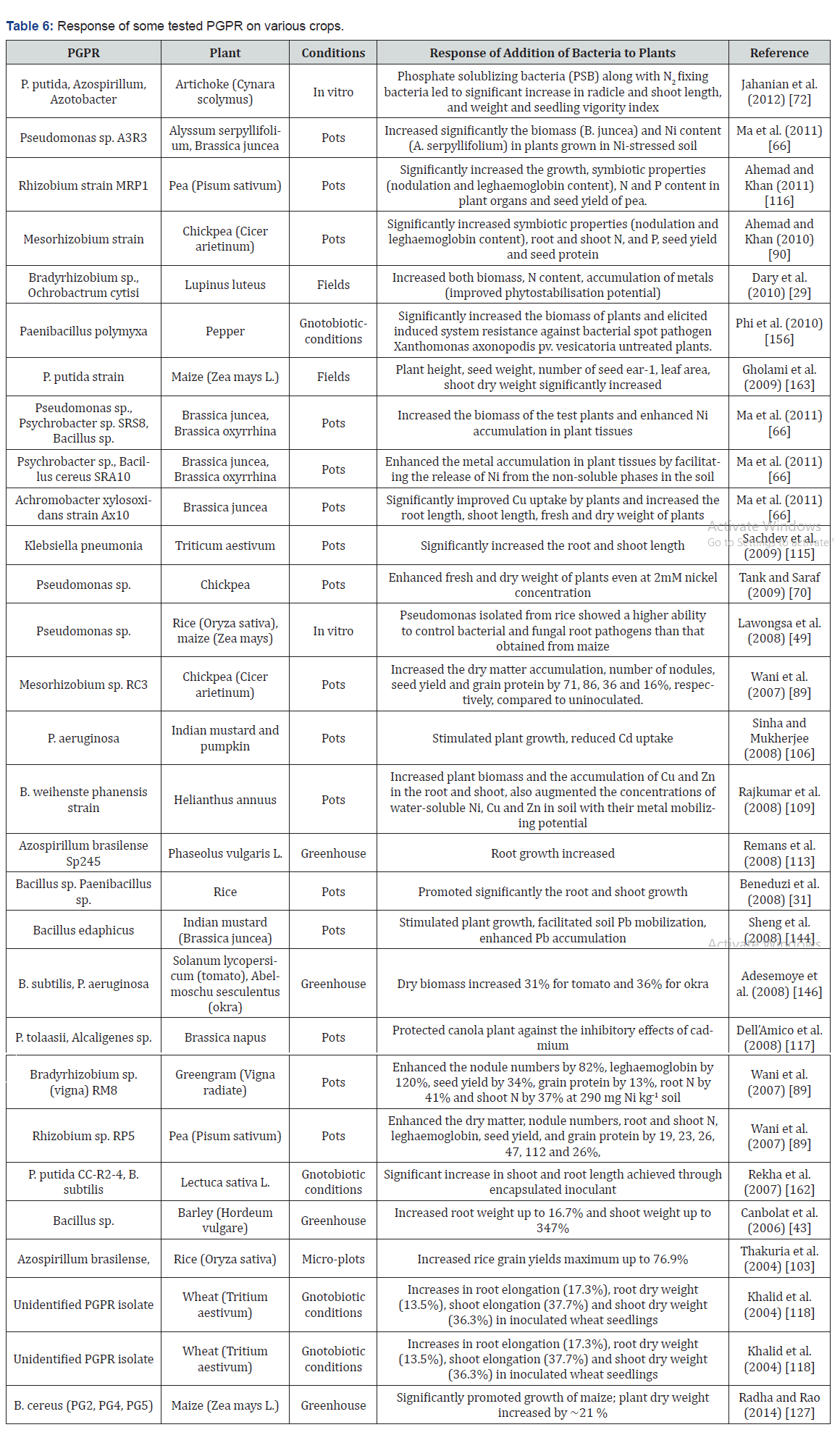
According to Kloepper and Schroth [82], PGPR mediated plant growth promotion occurs by the alteration of the whole microbial community in rhizosphere niche through the production of various substances [82]. Generally, PGPR promote plant growth directly by either facilitating resource acquisition (N, P and essential minerals) or modulating plant hormone levels (Table 6), or indirectly by decreasing the inhibitory effects of various pathogens on plant growth and development in the forms of biocontrol agents [68].
Direct mechanisms
Nitrogen fixation:
Nitrogen (N) is the most vital nutrient for plant growth and productivity. Although, there is about 78% N2 in the atmosphere, it is unavailable to the growing plants. The atmospheric N2 is converted into plant-utilizable forms by BNF which changes nitrogen to ammonia by N2 fixing MOs using a complex enzyme system known as nitrogenase [26]. In fact, BNF accounts for approximately two-thirds (2/3) of the N2 fixed globally, while the rest of the N is industrially synthesized by the Haber-Bosch process (Rubio and Ludden, 2008). BNF occurs, generally at mild temperatures, by N2 fixing MOs, which are widely distributed in nature (Raymond et al. 2004). Furthermore, BNF represents an economically beneficial and environmentally sound alternative to chemical fertilizers [83].
N2 fixing organisms are generally categorized as
a) symbiotic N2 fixing bacteria including members of the family rhizobiaceae which forms symbiosis with leguminous plants (e.g. rhizobia) [84] and non-leguminous trees (e.g. Frankia) and
b) non-symbiotic (free living, associative and endophytes) N2 fixing forms such as cyanobacteria (Anabaena, Nostoc), Azospirillum, Azotobacter, Glucono acetobacter diazotrophicus and Azocarus etc. [81].
However, non-symbiotic N2 fixing bacteria provide only a small amount of the fixed N2 that the bacterially-associated host plant requires [68]. Leguminous plants and rhizobia communicate through the gene expression by reciprocally transmitting signals for the activation of the symbiotic genes in two partners. A type of phenolic called flavonoids, are released by host plants into the rhizosphere. Flavonoids act as a chemo-attractant for the bacteria to the plants, and eventually colonies of rhizobia to the root hairs. Flavonoid signal activities expression of nodulation (nod) genes. Symbiotic biological N2 fixing rhizobia within the rhizobiaceae family (a-proteobacteria) infect and establish symbiotic relationship with the roots of leguminous plants. The establishment of the symbiosis involves a complex interplay between host and symbiont [85] resulting in the formation of the nodules wherein the rhizobia colonize as intracellular symbiont.
The genes for N2 fixation, called nif genes are found in both symbiotic and free living systems [26]. Nitrogenase (nif) genes include structural genes, genes involved in activation of the Fe protein, iron molybdenum cofactor biosynthesis, electron donation, and regulatory genes required for the synthesis and function of the enzyme. Since N2 fixation is a very energy demanding process, requiring at least 16mol of ATP for each. The process of N2 fixation is carried out by a complex enzyme, the nitrogenase complex [26]. Dinitrogenase reductase provides electrons with high reducing power while dinitrogenase uses these electrons to reduce N2 to NH3. Based on the metal cofactor three different N fixing systems have been identified
a) Mo-nitrogenase,
b) V-nitrogenase and
c) Fe-nitrogenase.
Structurally, N2-fixing system varies among different bacterial genera. Most biological N2 fixation is carried out by the activity of the molybdenum nitrogenase, which is found in all diazotrophs [86]. The biochemical mechanism of N2 fixation can be written in simplified form as follows: nitrogenase. The above mechanism indicates that N2-fixing systems can thrive in soils poor in N. that they are a source of proteins, and that they provide N for soil fertility. Adenosine triphosphate (ATP) is the source of energy necessary for the cleavage and reduction of N2 into ammonia. In rhizobia, for instance, ATP results from oxidative degradation of sugars and related molecules. These sugars are manufactured by the host-plant during photosynthesis and transferred to the nodules. In general, for each gram of N2 fixed by Rhizobium, the plant fixes 1-20gC through photosynthesis. This is an indication that symbiotic N2 fixation requires additional energy which, in nitrate-fed plants, can be used to produce more photosynthates (products of photosynthesis). The extra energy cost of N2 fixation can, however safely be carried by most field-grown legumes with little or no loss of production.
Fixation of atmospheric N is essential because of the reasons:
a. Fixed N2 is lost through the process of N cycle through denitrification.
b. Demand for fixed N2 by the biosphere always exceeds its availability.
c. The amount of N2 fixed chemically and lightning process is very less (i.e. 0.5%) as compared to biologically fixed N2
d. Nitrogenous fertilizers contribute only 25% of the total world requirement while biological N2 fixation contributes about 60% of the earth’s fixed N2
e. Manufacture of nitrogenous fertilizers by “Haber” process is costly and time consuming.
f. The numbers of soil MOs carry out the process of biological N2 fixation at normal atmospheric pressure (1 atmosphere) and temp (around 20°C).
Two groups of microorganisms are involved in the process of BNF.
A. Non-symbiotic (free living) and B. Symbiotic (associative)
Non-symbiotic (free living):
Depending upon the presence or absence of oxygen, non-symbiotic N2 fixation prokaryotic organisms may be aerobic heterotrophs (Azotobacter, Pseudomonas, Achromobacter) or aerobic autotrophs (Nostoc, Anabena, Calothrix, BGA) and anaerobic heterotrophs (Clostridium, Kelbsiella. Desulfovibrio) or anaerobic Autotrophs (Chlorobium, Chromnatium, Rhodospirillum, Meihanobacteriumetc). Desulphovibrio is chemotrophic N2 fixing bacterium. Rhodopseudomonas, Rhodospirillum and Chromatium are N2 fixing photoautotrophic bacteria. These bacteria add up to 10-25kg of N ha-1 annum-1. Many free living blue-green algae (cyanobacteria) perform N2 fixation, e.g., Anabaena, Nustoc, Aulosira, Cylmdrospermum, Trichodesmium. These are also important ecologically as they live in waterlogged soils where denitrifing bacteria can be active.
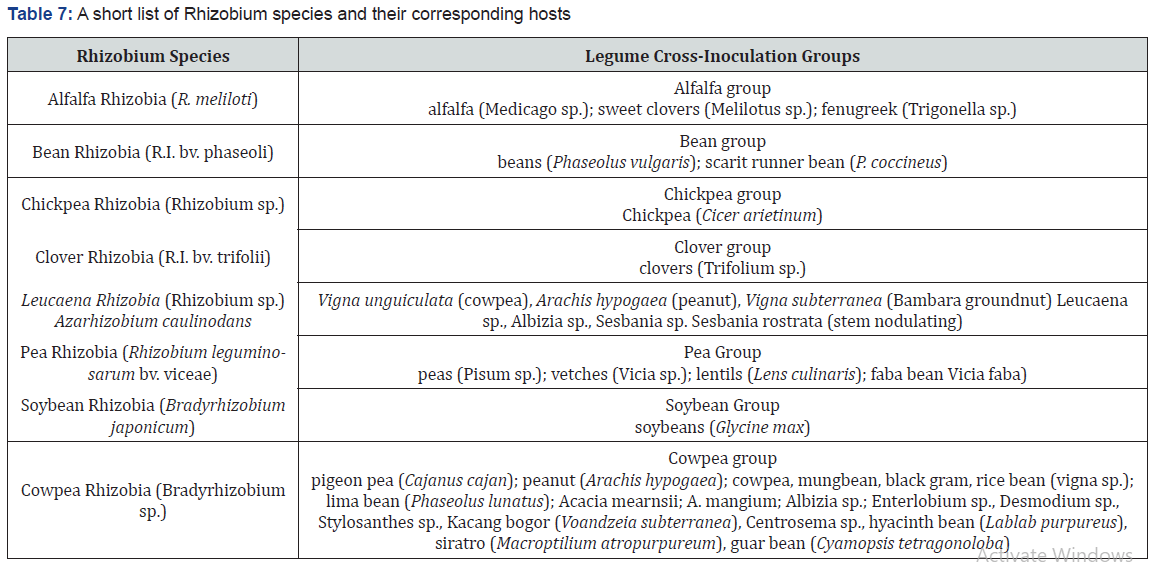
Symbiotic (associative):
Rhizobium is aerobic, gram negative N2 fixing bacterial symbionts of Papilionaceous roots. Sesbania rostrata has Rhizobium in root nodules and Aerorhizobium in stem nodules (table 7). Frankia is symbiont in root nodules of many non-leguminous plants like Casuarina and Alnus. Xanthomonas and Mycobacterium occur as symbiont in the leaves of some members of the families Rubiaceae and Myrsinaceae. Several species of Rhizobium live in the soil but are unable to fix N2 by themselves. They do so only as symbiont in the association of roots of legumes.
Rawat et al. [87] conducted a long-term field experiment for 8 years on a Vertisol in central India to assess quantitatively the direct and residual N effects of soybean inoculation with Bradyrhizobium and wheat inoculation with Azotobacter in a soybean-wheat rotation. Inoculation of soybean increased grain yield by 10.1% (180kg ha-1), but the increase in wheat yields with inoculation was only marginal (5.6%; 278kg ha-1). Residual and direct effects of Rhizobium and Azotobacter inoculants caused a fertilizer N credit of 30 kg ha-1 in wheat. Application of fertilizers or microbial inoculation favoured the proliferation of rhizobia in crop rhizosphere due to better plant growth. Additional N uptake by inoculation was 14.9kg N ha-1 by soybean and 20.9kg N ha-1 by wheat crop, and a gain of +38.0kg N ha-1 year-1 to the 0-15 cm soil layer was measured after harvest of wheat.
Phosphate solubilization:
P, the second important plant growth-limiting nutrient after N, is abundantly available in soils in both organic and inorganic forms [74]. Despite of large reservoir of P, the amount of available forms to plants is generally low. This low availability of phosphorous to plants is because the majority of soil P is found in insoluble forms, while the plants absorb it only in two soluble forms, the monobasic (H2PO4-) and the diabasic (H2PO4-) ions [81]. The insoluble P is present as an inorganic mineral such as apatite or as one of several organic forms including inositol phosphate (soil phytate), phosphomon esters, and phosphotriesters [68]. To overcome the P deficiency in soils, there are frequent applications of phosphatic fertilizers in agricultural fields. Plants absorb fewer amounts of applied phosphatic fertilizers and the rest is rapidly converted into insoluble complexes in the soil [88]. But regular application of phosphatic fertilizers is not only costly but is also environmentally undesirable. This has led to search for an ecologically safe and economically reasonable option for improving crop production in low P soils. In this context, organisms coupled with phosphate solubilizing activity, often termed as phosphate solubilizing microorganisms (PSM), may provide the available forms of P to the plants and hence a viable substitute to chemical phosphatic fertilizers [89]. Of the various PSM (s) inhabiting the rhizosphere, phosphate-solubilizing bacteria (PSB) are considered as promising biofertilizers since they can supply plants with P from sources otherwise poorly available by various mechanisms [90]. Bacterial genera like Azotobacter, Bacillus, Beijerinckia, Burkholderia, Enterobacter, Erwinia, Flavobacterium, Microbacterium, Pseudomonas, Rhizobium and Serratia are reported as the most significant phosphate solubilizing bacteria [81] (Figure 3).
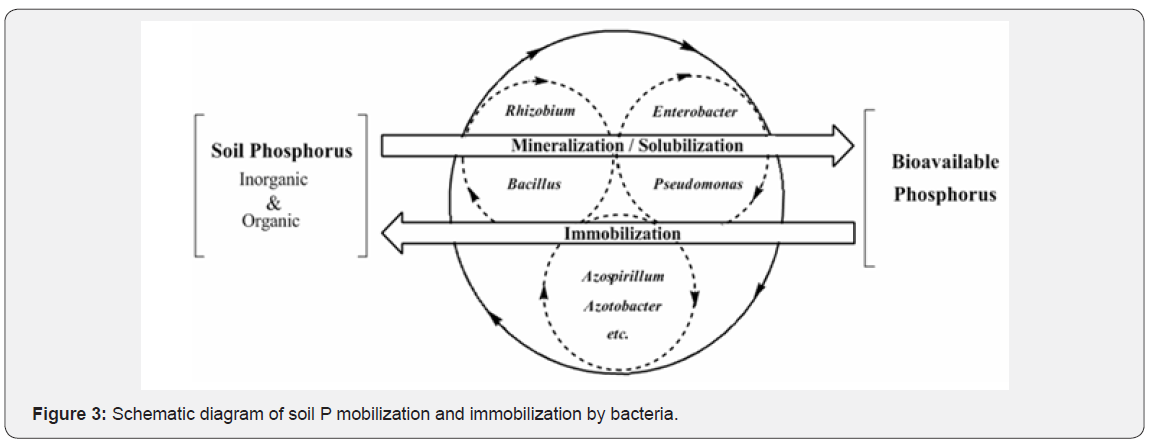
The process of P-solubilization commences with the decrease in the pH of the medium suggesting the role of organic acids in the P-solubilization mechanism. Nineteen P-solubilizing P. fluorescent strains belonging to P. fluorescens, P. poae, P. trivialis and Pseudomonas sp. produced gluconic acid, oxalic acid, 2-ketogluconic acid, lactic acid, succinic acid, formic acid, citric acid and malic acid in the culture filtrates during the solubilization of various rock phosphates [91,92]. The strains differed quantitatively and qualitatively in the production of organic acids during solubilization of phosphate from the substrates. P. corrugata produced gluconic and 2-ketogluconic acid during the growth at lower temperature [93]. Vyas et al. [94] reported the detection of gluconic, citric and isocitric acids during the tricalcium phosphate solubilization by Rahnella sp.
The production of organic acids especially gluconic acid seems to be the most frequent agent of mineral phosphate solubilization by bacteria such as Pseudomonas sp., Erwinia herbicola, Pseudomonas cepacia and Burkholderia cepacia [95]. Another organic acid identified in strains with phosphatesolubilizing ability is 2-ketogluconic acid, which is present in Rhizobium leguminosarum [96], R. meliloti [97], and other unidentified soil bacteria [98]. Conversely, the mineralization of organic P occurs through the synthesis of a variety of different phosphatases, catalyzing the hydrolysis of phosphoric esters [68]. Besides providing P to the plants, the PSB also augment the growth of plants by stimulating the efficiency of BNF, enhancing the availability of other trace elements by synthesizing important plant growth promoting substances [99].
Siderophore production:
Siderophores are low molecular weight bio-molecules secreted by MOs in response to iron starvation for acquisition of iron from insoluble forms by mineralization and sequestration. Although some siderophores are known to chelate other ions, their specificity and avidity for iron is the most consistent feature. Siderophores produced by rhizosphere inhabitants has been studied well and it has been reported that ability to produce siderophores not only improve rhizosphere colonization of producer strain but also play an important role in iron nutrition of plant and antagonism against phytopathogens. Bacteria are the most abundant MOs in the rhizosphere; therefore, they are highly important for the plant physiology, particularly in terms of competitiveness against plant pathogens and the mineral supply [100]. The “rhizosphere” term was originally introduced by Hiltner [101] to illustrate the particular zone of soil surrounding plant roots in which microbe populations are stimulated by root exudates. Rhizobacteria produce metal-chelating agents that possess the ability to transport several heavy metals [26], indicating that heavy metals may be directly affect siderophore regulation. Consequently, heavy metal ions may reduce the free siderophore concentration in the milieu due to complex formation. This process interferes with siderophore complexation with Fe and thus decreases the soluble Fe concentration. Several siderophore-producing bacteria associated with plants, such as Brassica juncea and Aquilegia bertolonii, have been isolated from metal-contaminated soils [102,103]. In addition, Idris et al. [103] have characterized the indigenous bacteria associated with the nickel hyper accumulator Thlaspi goesingense using cultivation and cultivation-independent techniques and found that the majority of bacterial strains were able to produce siderophores. In both gram-negative and grampositive rhizobacteria, Fe3+ in Fe3+-siderophore complex on bacterial membrane is reduced to Fe2+ which is further released into the cell from the siderophore via a gating mechanism linking the inner and outer membranes. During this reduction process, the siderophore may be destroyed/recycled [104,105]. Thus, siderophores act as solubilizing agents for iron from minerals or organic compounds under conditions of iron limitation [106]. Not only iron, siderophores also form stable complexes with other heavy metals that are of environmental concern, such as Al, Cd, Cu, Ga, Fe, Pb and Zn, as well as with radionuclides including U and Np [107,108]. Binding of the siderophore to a metal increases the soluble metal concentration [109]. Hence, bacterial siderophores help to alleviate the stresses imposed on plants by high soil levels of heavy metals. Plants assimilate Fe3+ from bacterial siderophores by means of different mechanisms, for instance, chelate and release of Fe3+, the direct uptake of siderophore-Fe complexes, or by a ligand exchange reaction [110]. Numerous studies of the plant growth promotion vis-a-vis siderophore mediated Fe-uptake as a result of siderophore producing rhizobacterial inoculations have been reported [109]. For example, Crowley and Kraemer [83] revealed a siderophore mediated iron transport system in oat plants and inferred that siderophores produced by rhizosphere MOs deliver iron to oat, which has mechanisms for using Fesiderophore complexes under iron-limited conditions.
Phytohormone production:
It is now well established that there are two sources of phytohormones naturally available for the plants: endogenous production by the plant tissues, and exogenous production by associated MOs, including numerous soil bacteria and fungi (reviewed by Costacurta and Vanderleyden, [111] and Patten and Glick, [112]. Microbial synthesis of the phytohormone auxin (indole-3-aceticacid/IAA) has been known for a longtime. It is reported that 80% of MOs isolated from the rhizosphere of various crops possess the ability to synthesize and release auxins as secondary metabolites [112]. Generally, IAA secreted by rhizobacteria interferes with the many plant developmental processes because the endogenous pool of plant IAA may be altered by the acquisition of IAA that has been secreted by soil bacteria [68,113]. Evidently, IAA also acts as a reciprocal signaling molecule affecting gene expression in several MOs. Consequently, IAA plays important role in rhizobacteria-plant interactions [114]. Moreover, down-regulation of IAA as signaling is associated with the plant defense mechanisms against a number of phytopathogenic bacteria as evidenced in enhanced susceptibility of plants to the bacterial pathogen by exogenous application of IAA or IAA produced by the pathogen [114]. IAA has been implicated in virtually every aspect of plant growth and development, as well as defense responses. This diversity of function is reflected by the extraordinary complexity of IAA biosynthetic, transport and signaling pathways [115]. Generally, IAA affects plant cell division, extension, and differentiation; stimulates seed and tuber germination; increases the rate of xylem and root development; controls processes of vegetative growth; initiates lateral and adventitious root formation; mediates responses to light, gravity and florescence; affects photosynthesis, pigment formation, biosynthesis of various metabolites, and resistance to stressful conditions. IAA produced by rhizobacteria likely, interfere the above physiological processes of plants by changing the plant auxin pool. Moreover, bacterial IAA increases root surface area and length, and thereby provides the plant greater access to soil nutrients. Also, rhizobacterial IAA loosens plant cell walls and as a result facilitates an increasing amount of root exudation that provides additional nutrients to support the growth of rhizosphere bacteria [68]. Thus, rhizobacterial IAA is identified as an effect or molecule in plant–microbe interactions, both in pathogenesis and phyto-stimulation [114]. An important molecule that alters the level of IAA synthesisis the amino acid tryptophan, identified as the main precursor for IAA and thus plays a role in modulating the level of IAA biosynthesis [90]. Strangely, tryptophan stimulates IAA production while, anthranilate, a precursor for tryptophan, reduces IAA synthesis. Starting with tryptophan, at least five different pathways have been described for the synthesis of IAA, and most pathways show similarity tothose described in plants, although some intermediates can differ [114]:
a) IAA formation via indole-3-pyruvic acidand indole-3- acetic aldehyde is found in a majority of bacteria like, Erwinia herbicola; saprophytic species of the genera Agrobacterium and Pseudomonas; certain representatives of Bradyrhizobium, Rhizobium, Azospirillum, Klebsiella, and Enterobacter,
b) The conversion of tryptophan into indole-3-acetic aldehyde may involve an alternative pathway in which tryptamine is formed as in pseudomonads and azospirilla and
c) IAA biosynthesis via indole-3-acetamide formation isreported for phytopathogenic bacteria Agrobacterium tumefaciens, P. syringae, and E. herbicola; saprophytic pseudomonads like (e.g. P. putida and P. fluorescens)
d) IAA biosynthesis that involves tryptophan conversion into indole-3-acetonitrile is found in the cyanobacterium (Synechocystis sp.) and
e) the tryptophan-independent pathway, more common in plants, is also found in azospirilla and cyanobacteria. Most Rhizobium species have been shown to produce IAA [116].
Since, IAA is involved in multiple processes including cell division, differentiation and vascular bundle formation, these three processes are also essential for nodule formation. Hence, it seems likely that auxin levels in the host legume plants are necessary for nodule formation [68, 113]. It is also reported that the inoculation with R. leguminosarum bv. viciae wherein the IAA biosynthetic pathway had been introduced, produced potential N2 fixing root nodules containing up to 60-fold more IAA than nodules formed by the wild-type counterpart in Vicia hirsute [117]. Environmental stress factors which modulate the IAA biosynthesis in different bacteria include acidic pH, osmotic and matrix stress and C limitation [113].
1-Aminocyclopropane-1-carboxylate (ACC) deaminase:
Generally, ethylene is an essential metabolite for the normal growth and development of plants [118]. This plant growth hormone is produced endogenously by approximately all plants and is also produced by different biotic and abiotic processes in soils and is important in inducing multifarious physiological changes in plants. Apart from being a plant growth regulator, ethylene has also been established as a stress or mone (119). Under stress conditions like those generated by salinity, drought, water logging, heavy metals and pathogenicity, the endogenous level of ethylene is significantly increased which negatively affects the overall plant growth. For instance, the high concentration of ethylene induces defoliation and other cellular processes that may lead to reduced crop performance [119,120]. Plant growth promoting rhizobacteria which possess the enzyme, 1-aminocyclopropane- 1-carboxylate (ACC) deaminase, facilitate plant growth and development by decreasing ethylene levels, inducing salt tolerance and reducing drought stress in plants [121]. Currently, bacterial strains exhibiting ACC deaminase activity have been identified in a wide range of genera such as Acinetobacter, Achromobacter, Agrobacterium, Alcaligenes, Azospirillum, Bacillus, Burkholderia, Enterobacter, Pseudomonas, Ralstonia, Serratia and Rhizobium etc. [122]. Such rhizobacteria take up the ethylene precursor ACC and convert it into 2-oxobutanoate and NH3 [123]. Several forms of stress are relieved by ACC deaminase producers, such as effects of phytopathogenic MOs (viruses, bacteria, and fungi etc.), and resistance to stress from polyaromatic hydrocarbons, heavy metals, radiation, wounding, insect predation, high salt concentration, draft, extremes of temperature, high light intensity and flooding [68,124]. As a result, the major noticeable effects of seed/root inoculation with ACC deaminase-producing rhizobacteria are the plant root elongation, promotion of shoot growth, and enhancement in rhizobial nodulation and N, P and K uptake as well as mycorrhizal colonization in various crops [68].
Indirect mechanisms
The application of MOs to control diseases, which is a form of biological control, is an eco-friendly approach [124]. The major indirect mechanism of plant growth promotion in rhizobacteria is through acting as bio-control agents [113]. In general, competition for nutrients, niche exclusion, induced systemic resistance and antifungal metabolites production are the chiefmodes of bio-control activity in PGPR [124]. Many rhizobacteria have been reported to produce antifungal metabolites like, HCN, phenazines, pyrrolnitrin, 2, 4-diacetylphloroglucinol, pyoluteorin, viscosinamide and tensin [120]. Interaction of some rhizobacteria with the plant roots can result in plant resistance against some pathogenic bacteria, fungi and viruses. This phenomenon is called induced systemic resistance [124]. Moreover, ISR involves jasmonate and ethylene signaling within the plant and these hormones stimulate the host plant’s defense responses against a variety of plant pathogens [68]. Many individual bacterial components induce ISR, such as lipopolysaccharides (LPS), flagella, siderophores, cyclic lipopeptides 2, 4-diacetylphloroglucinol, homoserine lactones, and volatiles like, acetoin and 2, 3-butanediol [124].
Applications of PGPR as multifunctional agents:
The effect of PGPR in crop productivity varies under laboratory, greenhouse and field trials. Because, soil is an unpredictable environment and an intended result is sometimes difficult to achieve. Climatic variations also have a large impact on the effectiveness of PGPR but sometimes unfavorable growth conditions in the field are to be expected as normal functioning of agriculture [90]. Plant growth promoting traits do not work independently of each other but additively as it was suggested in the “additive hypothesis,” that multiple mechanisms, such as phosphate solubilization, dinitrogen fixation, ACC deaminase and antifungal activity, IAA and siderophore biosynthesis etc. are responsible for the plant growth promotion and increased yield [125]. Under both natural agroecological niches and controlled soil environments, significant increase in yields of different crop plants has been observed following PGPR applications. Due to the existing reluctance worldwide to embrace foods produced by genetically modified plants, PGPR may be advantageous as a means of promoting plant growth. The wide scale application of PGPR may decrease the global dependence on agricultural chemicals.
Soil microbes and soil structure:
From an agricultural point of view, a well-structured soil has low bulk density, is well aerated, absorbs rainfall and irrigation water rapidly, and is easily penetrated by plant roots and soil animals. The growth of fine plant roots and fungal hyphae knits soil particles and small aggregates together into larger units called macroaggregates. Soil organisms play important role in soil aggregation. Constituents of soil are viz. organic matter, polysaccharides, lignins and gums, synthesized by soil microbes play important role in cementing/ binding of soil particles. Further, cells and mycelial strands of fungi and actinomycetes, Vormicasts from earthworm is also found to play important role in soil aggregation. Different soil MOs, having soil aggregation/soil binding properties are graded in the order as fungi > actinomycetes > gum producing bacteria>yeasts. examples are: fungi like Rhizopus, Mucor, Chaetomium, Fusarium, Cladasporium, Rhizoctonia, Aspergillus, Trichoderma and bacteria like Azofobacler, Rhizobium Bacillus and Xanlhomonas. Grasses have a greater effect than other plants on stable aggregate production because a large proportion of plant biomass is maintained underground in the root system. Production of polysaccharide gums by rhizosphere MOs and plant roots enhances the formation and stabilization of macroaggregates because the materials act as glues to bind particles together. Roberson et al. [126] found improved aggregate stability in cover cropped soils as compared to conventional soils. Cover crops with a higher C:N ratio (e.g., mixtures containing grasses) may promote greater polysaccharide production and thus greater aggregate stability than cover crops with lower C:N ratio (e.g., legumes).
Soil microbes and organic matter decomposition and humus formation:
C cycling is the continuous transformation of organic and inorganic C compounds by plants and micro and macro-organisms between the soil, plants and the atmosphere. Decomposition of organic matter is largely a biological process that occurs naturally. Its speed is determined by three major factors: soil organisms, the physical environment and the quality of the organic matter. In the decomposition process, different products are released: CO2, energy, water, plant nutrients and resynthesized organic C compounds. Different organic residues contain different organic compounds. There is great variation in the rate of decomposition of organic residues. Sugars, starches and simple proteins are very rapidly decomposed. On the other hand Fats, waxes and lignins are very slowly decomposed. Hemicellulose, celluloses and protein are intermediate. Even though the composition may vary the end products are more or less the same (Figure 4).
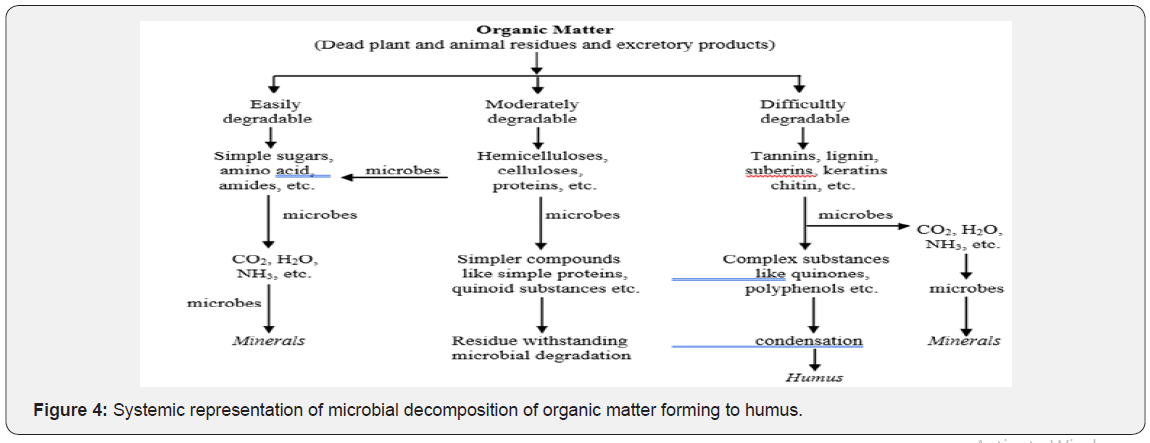
The continual addition of decaying plant residues to the soil surface contributes to the biological activity and the C cycling process in the soil. Breakdown of soil organic matter and root growth and decay also contribute to these processes. Successive decomposition of dead material and modified organic matter results in the formation of a more complex organic matter called humus. This process is called humification. The organic matter serves not only as a source of food for MOs but also supplies energy for the vital processes of metabolism that are characteristics of living beings. MOs such as fungi, actinomycetes, bacteria, protozoa etc. and macro organisms such as earthworms, termites, insects etc. plays important role in the process of decomposition of organic matter and release of plant nutrients in soil. Thus, organic matter added to the soil is converted by oxidative decomposition to simpler nutrients/substances for plant growth and the residue is transformed into humus. Organic matter/substancesinclude cellulose, lignins and proteins (in cell wall of plants), glycogen (animal tissues), proteins and fats (plants, animals). Cellulose is degraded by bacteria, especially those of genus Cytophaga and other genera (Bacillus, Pseudomonas, Cellulomonas, and Vibrio Achromobacter) and fungal genera (Aspergillus, Penicilliun, Trichoderma, Chactomium and Curvularia). Lignins and proteins are partially digested by fungi, protozoa and nematodes. Proteins are degraded to individual amino acids mainly by fungi, actinomycetes and Clostridium. Under anaerobic conditions of waterlogged soils, methane are main C containing product which is produced by the bacterial genera (strict anaerobes) Methanococcus, Methanobacterium and Methanosardna.
Biogeochemical cycling of elements:
All living organisms depend on the supply of essential elements from the earth. Since the earth is a closed system with a finite supply of essential elements such as hydrogen (H), oxygen (O), carbon (C), nitrogen (N), sulfur (S) and P. Soil fertility can be maintained when nutrients are efficiently recycled through the soil food web and soil-plant-animal system. Microbes are critical in the process of breaking down and transforming dead organic material into forms that can be reused by other organisms. The biogeochemical process through which organic compounds are broken down to inorganic compounds or their constituent elements is known “Mineralization”, or microbial conversion of complex organic compounds into simple inorganic compounds & their constituent elements is known as mineralization. Sahu et al. [127] reported that prolonged water-logging induced severe stress on microbial processes. However, post-drainage, there was significant improvement in microbial populations, N mineralization, soil enzymes and soil fertility, and also demonstrated that the benefits of rain-water conservation in Vertisols for C sequestration and N accretion and improvement of soil biological quality. Soil microbes plays important role in the biochemical cycling of elements in the biosphere where the essential elements (C, P, S, N, Fe, etc.) undergo chemical transformations. Through the process of mineralization organic C, N, P, S, Fe, etc. are made available for reuse by plants. Earlier a field study by Vinod Kumar et al. [128] who showed significantly positive effect of inoculation with soybean-rhizobia on wheat yield (grain and straw) was noticed. Maximum grain and straw yield (5043 and 7704kg ha-1 respectively) was achieved by application of recommended dose of chemical fertilizers (NPK) along with soybean-rhizobia seed inoculation, it gave ~20 and ~8% higher yields respectively as compared to fertilized and uninoculated plots (4211 and 7127kg ha-1 respectively) while minimum by unfertilized uninoculated (2560 and 5037kg ha-1).
Soil microbes as bio-control agents:
Bio-control of plant diseases involves the use of an organism to inhibit the pathogen and reduce disease (129). There are many definitions for biological control; however, the basic idea involves a strategy for reducing disease incidence or severity by direct or indirect manipulation of MOs [22, 128]. Consequently, understanding the mechanisms of biological control of plant diseases through the interactions between bio-control agent and pathogen may allow manipulating the soil environment to create conditions conducive for successful bio-control or to improve bio-control strategies [130, 131]. Bio-control microbes that are applied to seeds or soil prior to planting may colonize the spermosphere and/or rhizosphere of seedlings and thus may be present at or near infection courts of soilborne pathogens. Bio-control agents may participate in a variety of trophic and nontrophic interaction mechanisms including production of antifungal compounds, hyper-parasitism of pathogens, stimulation of host plant defenses, or competitive colonization of spermosphere and rhizosphere substrates. However, soils are both densely populated and highly competitive habitats, and poor competitive ability of introduced fungal agents in soil communities, leading to unsuccessful establishment and activity, has often been observed [132].
Several eco-friendly bio-formulations of microbial origin are used in agriculture for the effective management of plant diseases, insect pests, weeds etc. eg: Trichoderma sp. and Gleocladium sp. are used for biological control of seed and soil borne diseases. Fungal genera Entomophthora, Beauveria, Metarrhizium and protozoa Maltesiagrandis, Malamebalocustiae, etc are used in the management of insect pests. Nuclear polyhydrosis virus (NPV) is used for the control of Heliothis/American boll worm. Bacteria like Bacillus thuringiensis, Pseudomonas are used in cotton against Angular leaf spot and boll worms.
Degradation of pesticides /pollutants in soil by MOs:
Pesticide degradation is the process by which a pesticide is transformed into a benign substance that is environmentally compatible with the site to which it was applied. Globally, an estimated 1 to 2.5 million tons of active pesticide ingredients are used each year, mainly in agriculture. Pesticides reaching the soil in significant quantities have direct effect on soil microbiological aspects, which in turn influence plant growth. Some of the most important effects caused by pesticides are:
a) alterations hi ecological balance of the soil microflora,
b) continued application of large quantities of pesticides may cause everlasting changes in the soil microflora,
c) adverse effect on soil fertility and crop productivity,
d) inhibition of N2-fixing soil MOs such as Rhizobium, Azotobacter, etc. and cellulolytic and PSM,
e) suppression of nitrifying bacteria, Nitrosomonas and Nitrobacter by soil fumigants ethylene bromide, telone and vapam have also been reported,
f) alterations in N balance of the soil,
g) interference with ammonification in soil,
h) adverse effect on mycorrhizal symbioses in plants and nodulation in legumes and
i) alterations in the rhizosphere microflora, both quantitatively and qualitatively.
The pesticides /chemicals reaching in the soil are acted upon by several physical, chemical and biological forces exerted by microbes in the soil and they are degraded into non-toxic substances. This process of degradation of pesticides and conversion into non-toxic compounds by MOs is known as “biodegradation”. For example, bacterial genera like Pseudomonas, Clostridium, Bacillus, Thiobacillus, Achromobacter etc. and fungal genera like Trichoderma, Penicillium, Aspergillus, Rhizopus, and Fusarium are playing an important role in the biodegradation of the toxic chemicals /pesticides in soil. Trichoderma sp. also have shown considerable potential for bioremediation activity in soil, especially for hydrocarbon pollutants, and many of the same ecological constraints on efficacy may apply in bioremediation systems. Trichoderma sp. and especially T. harzianum can degrade a number of soil pollutants; for example, Katayama and Matsumura [133] reported on the ability of T. harzianum to degrade organochlorine pesticides, particularly endosulfan.
a) Strategies for bioremediation: For the successful biodegradation /bioremediation of a given contaminant following strategies are needed.
b) Passive/intrinsic Bioremediation: It is the natural bioremediation of contaminant by tile indigenous MOs and the rate of degradation is very slow.
c) Biostimulation: Practice of addition of N and P to stimulate indigenous MOs in soil.
d) Bioventing: Process /way of bio-stimulation by which gases stimulants like oxygen and methane are added or forced into soil to stimulate microbial activity.
e) Bioaugmentation: It is the inoculation /introduction of MOs in the contaminated site /soil to facilitate biodegradation.
f) Composting: Piles of contaminated soils are constructed and treated with aerobic thermophilic MOs to degrade contaminants. Periodic physical mixing and moistening of piles are done to stimulate microbial activity.
g) Phytoremediation: Can be achieved directly by planting plants which hyper accumulate heavy metals or indirectly by plants stimulating MOs in the rhizosphere.
h) Bioremediation: Process of detoxification of toxic / undesirable chemicals /contaminants in the soil and other environment by using MOs.
i) Mineralization: Complete conversion of an organic contaminant into inorganic constituent by a species or group of MOs.
For successful biodegradation of pesticide/pollutants in soil, following aspects must be taken into consideration.
a) Organisms must have necessary catabolic activity required for degradation of contaminant at fast rate to bring down the concentration of contaminant,
b) the target contaminant must be bioavailability,
c) soil conditions must be congenial for microbial/plant growth and enzymatic activity and
d) cost of bioremediation must be less than other technologies of removal of contaminants.
Biodegradation of hydrocarbons
Natural hydrocarbons in soil like waxes, paraffin’s, oils etc. and petroleum-based products are the major source of energy for industry and daily life. Leaks and accidental spills occur regularly during the exploration, production, refining, transport and storage of petroleum and petroleum products. The amount of natural crude oil seepage was estimated to be 600,000 metric tons year-1 with a range of uncertainty of 200,000 metric tons year- 1 [134]. Release of hydrocarbons into the environment whether accidentally or due to human activities is a main cause of water and soil pollution [135]. Soil contamination with hydrocarbons causes extensive damage of local system since accumulation of pollutants in animals and plant tissue may cause death or mutations [136]. The technology commonly used for the soil remediation includes mechanical, burying, evaporation, dispersion, and washing. However, these technologies are expensive and can lead to incomplete decomposition of contaminants.
The process of bioremediation, defined as the use of MOs to detoxify or remove pollutants owing to their diverse metabolic capabilities is an evolving method for the removal and degradation of many environmental pollutants including the products of petroleum industry [137]. In addition, bioremediation technology is believed to be non-invasive and relatively cost-effective [138]. Biodegradation by natural populations of MOs represents one of the primary mechanisms by which petroleum and other hydrocarbon pollutants can be removed from the environment [139] and is cheaper than other remediation technologies [140]. The extensive biodegradation of alkyl aromatics in marine sediments which occurred prior to detectable biodegradation of n-alkane profile of the crude oil and the MOs, namely, Arthrobacter, Burkholderia, Mycobacterium, Pseudomonas, Sphingomonas and Rhodococcus were found to be involved for alkyl aromatic degradation. Microbial degradation of petroleum hydrocarbons in a polluted tropical stream in Lagos, Nigeria was reported by Adebusoye et al. [141]. Nine bacterial strains, namely, Pseudomonas fluorescens, P. aeruginosa, Bacillus subtilis, Bacillus sp., Alcaligenes sp., Flavobacterium sp., Micrococcus roseus and Corynebacterium sp. were isolated from the polluted stream which could degrade crude oil.
Management strategies of microbial diversity in soil
Soil organisms are important elements for preserved ecosystem biodiversity and services thus assess functional and structural biodiversity in arable soils is interest. Main threats to soil biodiversity occurred by mechanical impacts (soil compaction, soil tillage) and chemical stress (plant protection measures) in agricultural management [142]. Soil microbial communities are often difficult to fully characterize, mainly because of their immense phenotypic and genotypic diversity, heterogeneity and crypticity. With respect to the latter, bacterial populations in soil top layers can group to more than 109 cells g-1 soil [143]. Scientific interest has long focused on the structure of microbial communities in the rhizosphere, assessed by cultivation-based studies. These studies have shown that the microbial diversity in the rhizosphere is often extensive and that there are distinct differences in bacterial community structures between non rhizosphere and rhizosphere soil. Recently, several studies on different plant species in different locations, using a range of cultivation-based and molecular methods, indicated that plant type is indeed a major factor influencing the structure of microbial communities. In conformity with earlier knowledge, several recent studies have shown that soil management practices, such as crop rotation, tillage, fertilizer, compost, manure, or pesticide applications and irrigation greatly affect soil microbial population [144]. During the colonization of plant roots by soil bacteria, MOs from the bulk soil undergo selective enrichment in the plant rhizosphere in response to different root exudate components. However, cultivation based methods are very selective as only a small proportion of soil MOs can be cultured [145]. Following agricultural management practices can improve the diversity of beneficial soil microbial communities in different crop rhizospheres/soil:
Cereal-legumes crop rotation:
There is a hypothesis that the plant species cultivated is a major determinant of the soil microbial population since plants provide nutrients. Plant roots secrete a wide variety of organic compounds to attract MOs, including sugar, ethylene, amino acids, organic acids, vitamins and enzymes. MOs respond differently to the compounds released by plants, therefore exudates of different plants select different microorganisms [146,147]. Legume supported farming systems have multiple impacts on the sustainability of farming in terms of resource use and effects on the environment. Increases in population size and diversity of decomposer invertebrates such as earthworms and collembola have been noted under perennial forage legumes [148,149]. Hydrogen gas, as a by-product of BNF, supports the growth of hydrogen-fixing bacteria in the rhizosphere of the legume nodule which again support populations of soil fauna [150]. Increases in populations of collembolan feeding on soil bacteria as well as earthworm and nematode populations have been reported in H2-treated soils [151].
Pulses are known to improve the microbial environment in the soils. They are known to release a part of unused nitrate fixed through symbiotic N2 fixation to the soil. Also low molecular weight organic compounds are released to the soil as exudates. Rhizobium-soybean symbiosis is of comparable importance to other Rhizobium-cereal associations with respect to N economy to the succeeding crop. These increases in microbial activity which in turn influence mineralization and immobilization of nutrients like N, P and S depending upon the environment. The maximal value of microbial biomass N (7mg N g-1 soil) was observed in soybeansorghum cropping system when no N has been applied to post rainy season sorghum [152]. Niewiadomska et al. [152] observed the highest numbers of ammonifying MOs under maize during the phase of emergence in a crop rotation with wheat. AMF mobilize P and other minerals from the soil and exchange these nutrients for C with their plant hosts. Significant cultivar variability in the response to AM fungi has been measured in faba bean, pea, alfalfa, corn, wheat, peach palm and pearl millet [153].
Green manuring/cover crops:
One of the major effects of a cover crop on the soil community is the increased input of organic matter contributed by cover crop residues. The large sources of C and energy for microbial communities that could be provided by cash crop residues are often removed from agricultural systems by current management practices. When a green manure crop is incorporated into the soil, microbes multiply to break down the fresh plant material, resulting in a rapid increase of MOs population by 2-6 folds. While the cover crop is alive and growing, the roots release a range of molecules (e.g., sugars, amino acids etc.) during the growing period [154], sugars, dead cells, mucilage and other materials into the soil, although the amount of these substances is too small to directly improve the soil fertility, they can directly influence the community composition and biomass of soil MOs [155]. If the cover crop is mowed, the cut portions of the plant provide habitat, shade and food at the soil surface. If the cover crop is incorporated into the soil, all parts of the plant enter the organic matter pool and become a food source. An increased microbial activity and diversity induced by preceding crops such as canola, rapeseed and barley [156]. Organic manures contain high amount of organic matter which improve soil moisture retention, aeration which improves soil microbiological quality [157]; and associated enzyme activities.
Integrated nutrient management approaches:
Manuring and fertilizer application also have a significant impact on the species diversity of bacteria and fungi. They cause significant changes in the microbial populations which are largely mediated through changes in soil pH. Application of nitrogenous fertilizers like ammonium sulphate increases the fungal population whereas FYM (farm yard manure) and NPK application increased the population of fungi, bacteria and actinomycetes [158]. Annual application of FYM in a 40year long term experimental site (LTFE, Jabalpur) in soybean-wheat rotation improved the rhizobial numbers on an average by 1.5fold over chemical fertilized site and 2.5fold over unfertilized. Increased crop growth by chemical fertilizers also stimulated rhizobial populations by 1.9fold over unfertilized soybean-wheat cropping. The proportion of slow-growing soybean bradyrhizobia was lower in soybean based rotations (38%) as compared to cereal based rotations (64%), [159]. The development of the large and active microbial community that results from organic inputs is desirable because of the multiple roles it plays in soil fertility. Large microbial populations may also support a greater diversity in higher trophic levels (e.g., soil fauna) and thus increase the potential for the soil to sustain crop growth in the face of perturbations such as drought stress. Inoculation with biological additives containing MOs may result in short-term, but probably minor and not cost-effective, stimulation of processes regulating soil fertility. Any benefit of inoculation, however, will almost always be short-lived because of the inability of the foreign organisms to compete with the locals. The foundation of a healthy and thriving microbial community is good nutrition and protection from major stresses. Maurya et al. [160] found that maximum diversity of microbial population density of both Azotobacter and Azospirillum MOs are present in Agro-forestry based crop rotation. More numbers of all groups of analyzed cultivable MOs were observed in organic agriculture fields in comparison to conventional fields, e.g., the number of bacteria had increased by 70%, actinobacteria by 290%, cultivable filamentous fungi by 110%, yeasts and maltose fermenting bacteria by 190% [72].
Application of biofertilizers /bacterial inoculant:
A formulation containing one or more beneficial bacterial strains (or species) in an easy to use and economical carrier material either organic, inorganic, or synthesized from defined molecules. The inoculant is the means of bacterial transport from the factory to the living plant. The immediate response to soil inoculation with associative, non-symbiotic PGPB (but also for rhizobia) varies considerably depending on the bacteria, plant species, soil type, inoculant density and environmental conditions. In general, shortly after the bacteria are introduced into the soil, the bacterial population declines progressively [161]. This phenomenon (together with bacterial biomass production and the physiological state of the bacteria in the inoculant, discussed below) may prevent the buildup of a sufficiently large PGPB population in the rhizosphere to obtain the intended plant response. The inoculated bacteria sometimes cannot find an empty niche in the soil for survival except in sterilized soil, a condition which does not exist in large-scale agriculture. They must compete with the often better-adapted native microflora and withstand predation by protozoans. A major role of inoculant formulation is to provide a more suitable microenvironment (even temporarily) to prevent the rapid decline of introduced bacteria in the soil. Recent year number of biofertilizers commercially uses in agriculture (Table 8) for improving MOs population in the rhizosphere towards improve crop growth and yield.
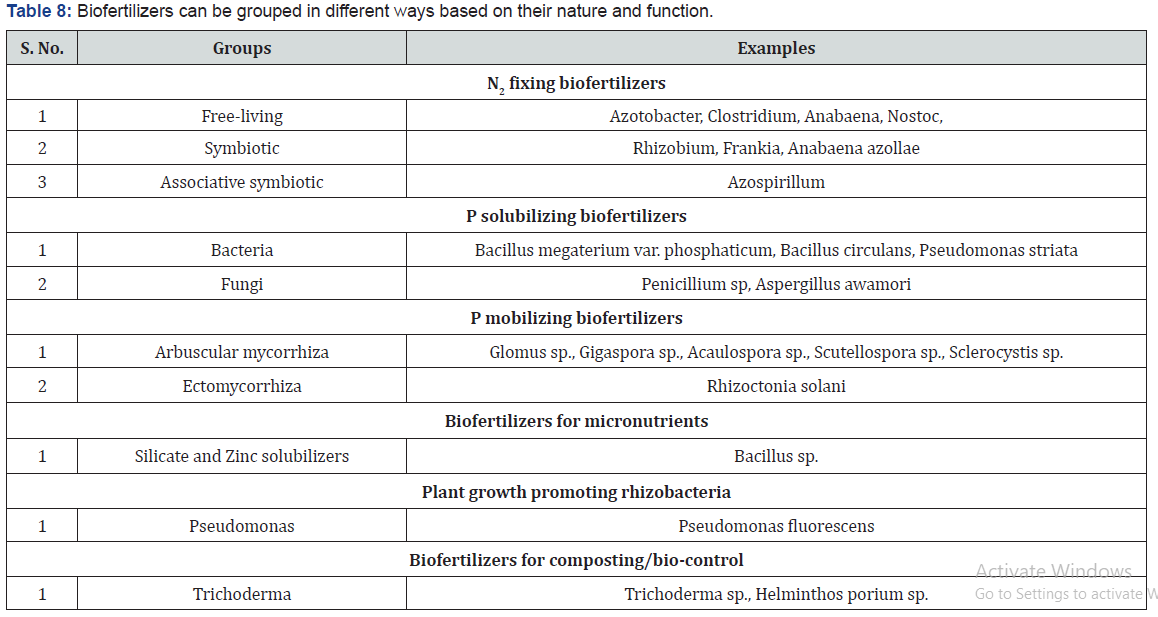
Soil manipulation (tillage):
Microbes need regular supplies of active SOM (soil organic matter) in the soil to survive in the soil. Long-term no-tilled soils have significantly greater levels of microbes, more active C, more SOM, and more stored C than conventional tilled soils. A majority of the microbes in the soil exist under starvation conditions and thus they tend to be in a dormant state, especially in tilled soils. The bacterial activity increases the loss of C as CO2, and triggers explosions of bacterial predators such as protozoa. A single tillage event is generally inconsequential to MOs, but repeated tillage eventually reduces the amount of soil organic matter that fuels the soil food web. The mechanical action of tillage can kill individual organisms and tends to temporarily reduce populations of fungi, earthworms, nematodes, and arthropods. Conventional tillage consisting of plowing or disking can induce water and nutrient losses, and then soil degradation with low organic matter content and a fragile physical structure [130]. However, conservation tillage, particularly no tillage, had positive effects on soil properties. Tillage and residue management affect not only soil properties but also soil microbial community. No-tillage with residue application was proved to increase the soil microbial community [162]. In many cases, both bacteria and fungi were more abundant under no-tillage than conventional tillage [163]. In no-tillage systems, fungi domination was frequently found and the residue was mainly decomposed by the fungal community [162]. Bacteria were generally considered to be the predominant decomposers of incorporated crop residues under conventional tillage [163]. However, more information is necessary to understand the effect of different tillage system combined with different residue quantity on soil microbial community.
Conclusion
Soil microorganisms play vital roles in many soil processes. Above information showed that the direct and interactive impacts of soil environment (ecology), management practices (soil temperature, moisture, organic carbon etc.) as well as soil types will likely reshape MOs soil communities and also their functional activities. It also appears that trying to change soil microbial populations in a meaningful way remains a difficult challenge for those seeking to do so by adding microbial preparations or novel soil amendments. Soil microbial populations demonstrate a high degree of resilience, and changes in the community will most likely be accomplished through greater attention to altering the soil environment to be a more favorable habitat. Legume rotation in cropping system and organic C source designate that significant changes occur in the diversity of important MOs involved in nutrient transformations, phytochrom production, plant disease control and growth promotion in response to various soil managing practices which are part of intensive/sustainable agriculture. Therefore, there is need to understand the aspects of microbial diversity in order to soil health and sustain the soil productivity on a long term basis.
References
- Krasilnikov NA (1958) Soil microorganisms and higher plants (English translation, 1961). Office of Technical Services, US Dept. Commer, Washington.
- Slankis V (1973) Hormonal relationships in mycorhizal development. In: Marks GC, Koslowski TT (Eds.): Ectomycorhizae. Academic Press, New York and London p. 232-291.
- Bossio DA, Scow KM (1995) Impact of carbon and flooding on the metabolic diversity of microbial communities in soils. Appl Environ Microbiology 61: 4043-4050.
- Winding A, Hund-Rinke, K Rutgers M (2005) The use of microorganisms in ecological soil classification and assessment concepts, Ecotox. Environ Safe 62(2): 230-248.
- Finegan B (1984) Forest succession. Nature 312: 109-114.
- Fierer N, Nemergut D, Knight R, Craine JM (2010) Changes through time: integrating microorganisms into the study of succession. Res Microbiol 161(8): 635-642.
- Johnson EA, Miyanishi K (2008) Testing the assumptions of chronosequences in succession. Ecol Lett 11(5): 419-431.
- Addison J, Trofymow J, Marshall V (2003) Abundance, species diversity, and community structure of Collembola in successional coastal temperate forests on Vancouver Island, Canada. Appl Soil Ecology 24(3): 233-246.
- Vitousek PM, Turner DR, Kitayama K (1995) Foliar nutrients during long-term soil development in Hawaiian montane rain forest. Ecology 76(3): 712-720.
- DeLuca T, Nilsson MC, Zackrisson O (2002) Nitrogen mineralization and phenol accumulation along a fire chronosequence in northern Sweden. Oecologia 133(2): 206-214.
- Grunzweig JM, Sparrow SD, Yakir D, Stuart Chapin F (2004) Impact of Agricultural Land‐use Change on Carbon Storage in Boreal Alaska. Global Change Biology 10(4): 452-472.
- Ranjard L, Richaume AS (2001) Quantitative and qualitative microscale distribution of bacteria in soil. Res Microbiology 152(8): 707-716.
- Vyas P, Robin J, Sharma KC, Rahi P, Gulati A (2010) Cold-adapted and rhizosphere competent strain of Rahnella sp. with broad-spectrum plant growth-promotion potential. J Microbiol Biotechnology 20(12): 1724-1734.
- Ekelund F, Ronn R, Christensen S (2001) Distribution with depth of protozoa, bacteria and fungi in soil profiles from three Danish forest sites. Soil Biol Biochemistry 33(4-5): 475-481.
- Bardgett RD, Leemans DK, Cook R, Hobbs PJ (1997) Seasonality of the soil biota of grazed and ungrazed hill grasslands. Soil Biol Biochemistry 29(8): 1285-1294.
- Kim J, Rees DC (1994) Nitrogenase and biological nitrogen fixation. Biochemistry 33(2): 389-397.
- Madsen E (1995) Impacts of agricultural practices on subsurface microbial ecology. Advances in Agronomy 54: 1-67.
- Richter D, Markewitz D (1995) How deep is soil? Bio Science 45(9): 600-609.
- Fierer N, Schimel JP, Holden PA (2003) Variation in microbial community composition through two soil depth profile. Soil Biology and Biochemistry 35(1): 167-176.
- Ajwa HA, Rice CW, Sotomayor D (1998) Carbon and nitrogen mineralization in tallgrass prairie and agricultural soil profiles. Soil Science Society of America Journal 62(4): 942-951.
- Trumbore S (2000) Age of soil organic matter and soil respiration: radiocarbon constraints on belowground C dynamics. Ecological Applications 10(2): 399-411.
- Chowdhury N, Marschner P, Burns RG (2011) Soil microbial activity and community composition: impact of changes in matric and osmotic potential. Soil Biology and Biochemistry 43(6): 1229-1236.
- Rietz DN, Haynes RJ (2003) Effects of irrigation induced salinity and sodicity on soil microbial activity. Soil Biology & Biochemistry 35(6): 845- 854.
- Sardinha M, Muller T, Schmeisky H, Joergensen RG (2003) Microbial performance in soils along a salinity gradient under acidic conditions. Applied Soil Ecology 23(3): 237-244.
- Jiang H, Dong H, Yu B, Liu X, Li Y, et al. (2007) Microbial response to salinity change in Lake Chaka, a hypersaline lake on Tibetan plateau. Environ Microbiol 9(10): 2603-2621.
- Hofte M, Dong Q, Kourambos S, Krishnapillai V, Sherratt D, et al. (1994) The sss gene product, which affects pyoverdin production in Pseudomonas aeruginosa 7NSK2, is a site-specific recombinase. Mol Microbiol 14(5): 1011-1020.
- Brockett BFT, Prescott SJ, Grayston SJ (2012) Soil moisture is the major factor influencing microbial community structure and enzyme activities across seven biogeoclimatic zones in western Canada. Soil Biol Biochemistry 44(1): 9-20.
- Skopp J, Jawson MD, Doran JW (1990) Steady-stste aerobic microbial activity as a function of soil-water content. Soil Science Soc of America Journal 54(6): 1619-1625.
- DeAngelis KM, Silver WL, Thompson AW, Firestone MK (2010) Microbial communities acclimate to recurring changes in soil redox potential status. Environ Microbiol 12(12): 3137-3149.
- Malhotra J, Aparna K, Dua K, Sangwan N, Trimurtulu N, et al. (2015) Microbial and genetic ecology of tropical Vertisols under intensive chemical farming. Environ Monit Assess 187(1): 2-17.
- Malcolm GM, Lopez-Gutierrez JC, Koide RT, Eissenstat DM (2008) Acclimation to temperature and temperature sensitivity of metabolism by ectomycorrhizal fungi. Global Change Biology 14(5): 1169-1180.
- Craine JM, Nippert JB, Elmore AJ, Skibbe AM, Hutchinson SL, et al. (2012) Timing of climate variability and grassland productivity, PNAS 109(9): 401-3405.
- Dumbrell AJ, Ashton PD, Aziz N, Feng G, Nelson M, et al. (2011) Distinct seasonal assemblages of Arbuscular mycorrhizal fungi revealed by massively parallel pyrosequencing. New Phytol 190(3): 794-804.
- Li WKW, Dickie PM (1987) Temperature characteristics of photosynthetic and heterotrophic activities: seasonal variation in temperate microbial plankton. Applied and Environ Microbiology 53(10): 2282-2295.
- Schadt CW, Martin AP, Lipson DA, Schmidt SK (2003) Seasonal dynamics of previously unknown fungal lineages in tundra soils. Science 301(5638): 1359-1361.
- Vannier G (1994) The thermo biological limits of some freezing intolerant insects -The super cooling and thermo stupor points. Acta Oecologica 15: 31-42.
- Ritz K, McHugh M, Harris J (2003) Biological diversity and function in soils: contemporary perspectives and implications in relation to the formulation of effective indicators. OECD Expert Meeting on Soil Erosion and Soil Biodiversity Indicators, Rome p. 1-11.
- Watson GW, Kelsey P (2006) The impact of soil compaction on soil aeration and fine root density of Quercus palustris. Urban Forestry & Urban Greening 4(2): 69-74.
- Schindlbacher A, Rodler A, Kuffner M, Kitzler B, Sessitsch A, et al. (2011) Experimental warming effects on the microbial community of a temperate mountain forest soil. Soil Biol Biochemistry 43(7): 1417-1425.
- Sarrantonio M, Gallandt E (2003) The role of cover crops in North American cropping systems. J Crop Production 8(1-2): 53-74.
- Dastager SG, Deepa CK, Pandey A (2010) Isolation and characterization of novel plant growth promoting Micrococcus sp NII-0909 and its interaction with cowpea. Plant Physio Biochem 48(12): 987-992.
- Fierer N, Schimel JP, Holden PA (2003) Variation in microbial community composition through two soil depth profile. Soil Biology and Biochemistry 35(1): 167-176.
- Kramer C, Gleixner G (2006) Variable use of plant- and soil-derived carbon by microorganisms in agricultural soils. Soil Biology and Biochemistry 38(11): 3267-3278.
- Simard RR, Angers DA, Lapierre C (1994) Soil organic matter quality as influenced by tillage, lime and phosphorus. Bio Fertil Soils 18(1): 13-18.
- Quadros PD, Zhalnina K, Davis-Richardson A, Fagen JR, Drew J, et al. (2012) The Effect of Tillage System and Crop Rotation on Soil Microbial Diversity and Composition in a Subtropical Acrisol. Diversity 4(4): 375-395.
- Fierer N, Strickland MS, Liptzin D, Bradford MA, Cleveland CC (2009) Global patterns in belowground communities. Ecol. Letter 12(11): 1-2.
- Vinod Kumar, Rawat AK, Rao DLN, Mitra NG, Naik KR, et al. (2016) Ecology of soybean-rhizobia in Vertisols of Madhya Pradesh, population in different cropping systems and influence of inoculating preceding wheat crop. Ph.D. thesis, JNKVVV, Jabalpur, (MP) India.
- Griffiths RI, Whiteley AS, O’Donnell AG, Bailey MJ (2003) Influence of depth and sampling time on bacterial community structure in an upland grassland soil. FEMS Microbiology Ecology 43(1): 35-43.
- Lauber CL, Strickland MS, Bradford MA, Fierer N (2008) The influence of soil properties on the structure of bacterial and fungal communities across land-use types. Soil Biology and Biochemistry 40(9): 2407-2415.
- Andrew DR, Fitak RR, Munguia-Vega A, Racolta A, Martinson VG, et al. (2012) Abiotic factors shape microbial diversity in Sonoran desert soils. Appl and Environ Microbiology 78(21): 7527-7537.
- Bordeleau LM, Prevost D (1994) Nodulation and nitrogen fixation in extreme environments. Plant Soil 161(1): 115-124.
- Correa OS, Barneix AJ (1997) Cellular mechanisms of pH tolerance in Rhizobium loti. World J Microbiol Biotechnology 13(32): 153-157.
- Brockwell J, Pilka A, Holliday RA (1991) Soil pH is a major determinant of the numbers of naturally-occurring Rhizobium meliloti in non-cultivated soils of New South Wales. Aust J Exp Agriculture 31: 211-219.
- McLay CDA, Barton L, Tang C (1997) Acidification potential often grain legume species grown in nutrient solution. Aust J Agric Research 48: 1025-1032.
- Tang C, Barton L, Raphael C (1998) Pasture legume species differ in their capacity to acidify soil. Aust J Agric Research 49(1): 53-58.
- Wyn Jones RG (1985) Salt tolerance in plants, In Chemistry in Britain. British Association for the Advancement of Science, London, United Kingdom p. 454-459.
- Hepper CM (1975) Extracellular polysaccharides of soil bacteria. In: N. Walker (Ed.): Soil Microbiology. Butterworths, London p. 93-110.
- Harris RF, Chesters G, Allen ON, Attoe OJ (1964) Mechanisms involved in soil aggregate stabilization by fungi and bacteria. Soc Amr Proc 28(4): 529-532.
- Koske AE, Sutton JC, Sheppard BR (1975) Ecology of Endogone in - Lake Huron sand dunes. Can J Botany 53: 87-93.
- Bond RD (1964) The influence of the microflora on the physical properties of soils. II. Field studies on water repellent sands. Austr J Soil Research 2(1): 123-131.
- Pankhurst C, Doube BM, Gupta VVSR (Eds.) (1997) Biological Indicators of Soil Health. CAB
- White PS, Walker JL (1997) Approximating nature’s variation: selecting and using reference information in restoration ecology. Restoration Ecology 5(4): 338-349.
- Hobbs RJ, Harris JA (2001) Restoration ecology: repairing the earth’s ecosystems in the new millenium. Restoration Ecology 9(2): 239-246.
- Chandler D, Davidson G, Grant WP, Greaves J, Tatchell GM (2008) Microbial bio-pesticides for integrated crop management: an assessment of environmental and regulatory sustainability. Trends Food Sci Technology 19(5): 275-283.
- Ahemad M, Malik A (2012) Bioaccumulation of heavy metals by zinc resistant bacteria isolated from agricultural soils irrigated with waste water. Bacteriology Journal 2(1): 12-21.
- Ma Y, Rajkumar M, Luo Y, Freitas H (2011) Inoculation of endophytic bacteria on host and non-host plants-effects on plant growth and Ni uptake. J Hazard Mater 195: 230-237.
- Hayat R, Ali S, Amara U, Khalid R, Ahmed I (2010) Soil beneficial bacteria and their role in plant growth promotion: a review. Ann Microbiology 60(4): 579-598.
- Glick BR (2012) Plant growth-promoting bacteria: Mechanisms and applications. Hindawi Publishing Corporation, Scientifica.
- Ahemad M, Khan MS (2012) Evaluation of plant growth promoting activities of rhizobacterium Pseudomonas putida under herbicide-stress. Ann Microbiology 62(4): 1531-1540.
- Tank N, Saraf M (2010) Salinity-resistant plant growth promoting rhizobacteria ameliorates sodium chloride stress on tomato plants. J Plant Interaction 5(1): 51-58.
- Ahemad M, Khan MS (2012) Ecological assessment of biotoxicity of pesticides towards plant growth promoting activities of pea (Pisum sativum)-specific Rhizobium sp. strain MRP1. Emirates J Food Agriculture 24(4): 334-343.
- Jahanian A, Chaichi MR, Rezaei K, Rezayazdi K, Khavazi K (2012) The effect of plant growth promoting rhizobacteria (PGPR) on germination and primary growth of artichoke (Cynara scolymus). Int J Agric Crop Science 4: 923-929.
- Khan AG (2005) Role of soil microbes in the rhizospheres of plants growing on trace metal contaminated soils in phytoremediation. J Trace Elem Med Biol 18(4): 355-364.
- Khan MS, Zaidi A, Wani PA, Oves M (2009) Role of plant growth promoting rhizobacteria in the remediation of metal contaminated soils. Environ Chem Letter 7(1): 1-19.
- Kloepper JW (1994) Plant growth-promoting rhizobacteria (other systems). In: Okon Y (Ed.), Azospirillum/Plant Associations. CRC Press Boca Raton FL USA p. 111-118.
- Aiyer RS, Venkataraman GS, Sundara Rao WVB (1964) Effect of N fixing blue green algae and Azotobacter chroococcum on vitamin C content of tomato fruits. Science and Culture 30: 556-557.
- Somers E, Vanderleyden J, Srinivasan M (2004) Rhizosphere bacterial signalling: a love parade beneath our feet. Crit Rev Microbiology 30(4): 205-240.
- Antoun H, Prevost D (2005) Ecology of plant growth promoting rhizobacteria. In: Siddiqui, ZA. (Ed.) PGPR: biocontrol and biofertilization Springer Dordrecht p. 1-38.
- Kloepper JW (2003) A review of mechanisms for plant growth promotion by PGPR In: Reddy MS, Anandaraj M, Eapen SJ, Sarma YR and Kloepper JW (Eds.), Abstracts and Short Papers. 6th International PGPR Workshop, 5-10 October 2003, Indian Institute of Spices Research, Calicut, India p. 81-92.
- Figueiredo MVB, Seldin L, Araujo FF, Mariano RLR (2011) Plant growth promoting rhizobacteria: fundamentals and applications. In: Maheshwari DK (Ed.), Plant growth and health promoting bacteria. Springer-Verlag, Berlin, Heidelberg p. 21-42.
- Bhattacharyya PN, Jha DK (2012) Plant growth-promoting rhizobacteria (PGPR): emergence in agriculture. World J Microbiol Biotechnology 28(4): 1327-1350.
- Kloepper JW, Schroth MN (1981) Relationship of in vitro antibiosis of plant growth promoting rhizobacteria to plant growth and the displacement of root microflora. Phytopathology 71(10): 1020-1024.
- Ladha JK, de Bruijn FJ, Malik KA (1997) Introduction: assessing opportunities for nitrogen fixation in rice-a frontier project. Plant Soil 194(1-2): 1-10.
- Zahran HH (2001) Rhizobia from wild legumes: diversity, taxonomy, ecology, nitrogen fixation and biotechnology. J Biotechnology 91(2-3): 143-153.
- Giordano W, Hirsch AM (2004) The expression of MaEXP1, a Melilotus alba expansin gene, is upregulated during the sweet clover-Sinorhizobium meliloti interaction. Mol Plant Microbe Interact 17(6): 613-22.
- Bishop PE, Jorerger RD (1990) Genetics and molecular biology of an alternative nitrogen fixation system. Plant Mol Biology 41: 109-125.
- Rawat AK, Rao DLN, Sahu RK (2013) Effect of soybean inoculation with Bradyrhizobium and wheat inoculation with Azotobacter on their productivity and N turnover in a Vertisol. Archives of Agronomy and Soil Science 59(11): 1559-1571.
- McKenzie RH, Roberts TL (1990) Soil and fertilizers phosphorus update. In: Proceedings of Alberta Soil Science Workshop Proceedings, Feb. 20-22, Edmonton, Alberta p. 84-104.
- Khan MS, Zaidi A, Wani PA (2006) Role of phosphate solubilizing microorganisms in sustainable agriculture-a review. Agron Sustain Development 27(1): 29-43.
- Zaidi A, Khan MS, Ahemad M, Oves M (2009) Plant growth promotion by phosphate solubilizing bacteria. Acta Microbiol Immunol Hung 56(3): 263-284.
- Gulati A, Rahi P, Vyas P (2008) Characterization of phosphate-solubilizing fluorescent pseudomonads from rhizosphere of seabuckthorn growing in cold deserts of Himalayas. Curr Microbiology 56(1): 73-79.
- Vyas P, Gulati A (2009) Organic acid production in vitro and plant growth promotion in maize under controlled environment by phosphate-solubilizing Pseudomonas fluorescent. BMC Microbiology 9: 174-181.
- Trivedi P, Sa T (2008) Pseudomonas corrugata (NRRL B-30409) mutants increased phosphate solubilization, organic acid production and plant growth at lower temperatures. Current Microbiology 56(2): 140-144.
- Vyas P, Robin J, Sharma KC, Rahi P, Gulati A (2010) Cold-adapted and rhizosphere competent strain of Rahnella sp. with broad-spectrum plant growth-promotion potential. J Microbiol Biotechnology 20(12): 1724-1734.
- Rodriguez H, Han Y, Lei XG (1999) Cloning, sequencing and expression of an Escherichia. coli acid phopshatase/phytase gene (app A2) isolated from pig colon. Biochem Biophys Res Community 257(1): 117-123.
- Halder AK, Mishra AK, Bhattacharya P, Chakrabarthy PK (1990) Solubilization of rock phosphate by Rhizobium and Bradyrhizobium. J Gen Applied Microbiology 36(2): 81-92.
- Halder AK, Chakrabarty PK (1993) Solubilization of inorganic phosphate by Rhizobium. Folia Microbiology 38(4): 325-330.
- Duff RB, Webley DM (1959) 2-Ketogluconic acid as a natural chelator produced by soil bacteria. Chem Ind p. 1376-1377.
- Suman A, Shasany AK, Singh M, Shahi HN, Gaur A, et al. (2001) Molecular assessment of diversity among endophytic diazotrophs isolated from subtropical Indian sugarcane. World J Microbiol Biotechnology 17(1): 39-45.
- Antoun H, Kloepper JW (2001) Plant growth promoting rhizo- bacteria. In: Brenner S, Miller JF (eds) Encyclopedia of genetics. Academic Press London pp. 1477-1480.
- Hiltner L (1904) Uber neuere Erfahrungen und Probleme auf dem Gebiete der Bodenbakteriologie unter besonderer Beru cksichtigung der Grundungung und Brache. Arb Dtsch Landwirtsch Ges 98: 59-78.
- Katayama A, Matsumura F (1993) Degradation of organochlorine pesticides, particularly endosulfan, by Trichoderma harzianum. Environmental Toxicology and Chemistry 12(6): 1059-1065.
- Idris R, Trifonova R, Puschenreiter M, Wenzel WW, Sessitsch A (2004) Bacterial communities associated with flowering plants of the Ni hyper accumulator Thlaspi goesingense. Applied Environ Microbiology 70(5): 2667-2677.
- Rajkumar M, Ae N, Prasad MNV, Freitas H (2010) Potential of siderophore-producing bacteria for improving heavy metal phytoextraction. Trends Biotechnology 28(3): 142-149.
- Mishra, B.B. (2015) Soil Classification: Issues and Opportunities for Indian System, The 26th Dr. S.P. Raychaudhuri Memorial Lecture, 19th October, 2015, NBSS & LUP, Nagpur. Journal of the Indian Society of Soil Science, 63(Supplement), p. 41-52.
- Indiragandhi P, Anandham R, Madhaiyan M, Sa TM (2008) Characterization of plant growth-promoting traits of bacteria isolated from larval guts of diamondback moth Plutella xylostella (Lepidoptera: Plutellidae). Curr Microbiology 56(4): 327-333.
- Neubauer U, Furrer G, Kayser A, Schulin R (2000) Siderophores, NTA and citrate: potential soil amendments to enhance heavy metal mobility in phytoremediation. Int J Phytoremediation 2: 353-368.
- Kiss T, Farkas E (1998) Metal-binding ability of desferrioxamine B.J. Inclusion Phenom Mol Recognit Chemistry 32: 385-403.
- Rajkumar M, Ae N, Prasad MNV, Freitas H (2010) Potential of siderophore-producing bacteria for improving heavy metal phytoextraction. Trends Biotechnology 28(3): 142-149.
- Schmidt W (1999) Mechanisms and regulation of reduction-based iron uptake in plants. New Phytology 141(1): 1-26.
- Costacurta A, Vanderleyden J (1995) Synthesis of phytohormones by plant-associated bacteria. Crit Rev Microbiology 21(1): 1-18.
- Patten CL, Glick BR (1996) Bacterial biosynthesis of indole-3- acetic acid. Canad J Microbiology 42(3): 207-220.
- Spaepen S, Vanderleyden J, Remans R (2007) Indole- 3-acetic acid in microbial and microorganism-plant signaling. FEMS Microbiol Review 31(4): 425-448.
- Spaepen S, Vanderleyden J (2011) Auxin and plant-microbe interactions. Cold Spring Harb Perspect Biol 3(4).
- Santner A, Calderon-Villalobos LIA, Estelle M (2009) Plant hormones are versatile chemical regulators of plant growth. Nature Chem Biology 5(5): 301-307.
- Ahemad M, Khan MS (2011) Effect of tebuconazole-tolerant and plant growth promoting Rhizobium isolate MRP1 on pea-Rhizobium symbiosis. Science Horticulture 129(2): 266-272.
- Camerini S, Senatore B, Lonardo E, Imperlini E, Bianco C, et al. (2008) Introduction of a novel pathway for IAA biosynthesis to rhizobia alters vetch root nodule development. Arch Microbiology 190(1): 67-77.
- Khalid A, Akhtar MJ, Mahmood MH, Arshad M (2006) Effect of substrate-dependent microbial ethylene production on plant growth. Microbiology 75(2): 231-236.
- Saleem M, Arshad M, Hussain S, Bhatti AS (2007) Perspective of plant growth promoting rhizobacteria (PGPR) containing ACC deaminase in stress agriculture. J Indian Microbiol Biotechnology 34(10): 635-648.
- Maurya BR, Kumar A. Raghubanshi R, Singh V (2012) Diversity of Azotobacter and Azospirrillum in rhizosphere crop rotations in eastern Uttar Pradesh. Res Journal of Microbiology 7(2): 123-130.
- Nadeem SM, Zahir ZA, Naveed M, Arshad M (2007) Preliminary investigations on inducing salt tolerance in maize through inoculation with rhizobacteria containing ACC deaminase activity. Canadian J Microbiology 53(10): 1141-1149.
- Kang BG, Kim WT, Yun HS, Chang SC (2010) Use of plant growth-promoting rhizobacteria to control stress responses of plant roots. Plant Biotechnol Report 4(3): 179-183.
- Nadeem SM, Zahir ZA, Naveed M, Arshad M (2007) Preliminary investigations on inducing salt tolerance in maize through inoculation with rhizobacteria containing ACC deaminase activity. Canadian J Microbiology 53(10): 1141-1149.
- Lugtenberg B, Kamilova F (2009) Plant-growth-promoting rhizobacteria. Ann Rev Microbiology 63: 541-556.
- Bashan Y, Holguin G (1997) Azospirillum-plant relationships: Environmental and physiological advances (1990–1996). Can J Microbiology 43(2): 103-121.
- Roberson EB, Sarig S, Firestone MK (1991) Cover crop management of polysaccharide-mediated aggregation in an orchard soil. Soil Science Soc of America Journal 55(3): 734-739.
- Sahu RK, Rawat AK, Rao DLN (2014) Traditional rainwater management system (‘Haveli’) in Vertisols of central India improves carbon sequestration and biological soil fertility. Agriculture Ecosystems and Environment 200: 94-101.
- Vannier G (1994) The thermo biological limits of some freezing intolerant insects -The super cooling and thermo stupor points. Acta Oecologica 15: 31-42.
- Cook RJ, Baker KF (1983) The Nature and Practice of Biological Control of Plant Pathogens. American Phytopathological Society St Paul MN.
- Fravel DR (1988) Role of antibiosis in the biocontrol of plant diseases. Annu Rev Phytopathology 26: 75-91.
- Handelsman JO, Parke JL (1989) Mechanisms in biocontrol of soil borne plant pathogens. In: Plant-Microbe Interactions. Molecular and Genetic Perspectives 3: 27-61.
- Van Veen JA, Van Overbeek LS, van Elsas JD (1997) Fate and activity of microorganisms introduced into soil. Microbiology and Molecular Biology Reviews 61(2): 121-135.
- Katayama A, Matsumura F (1993) Degradation of organochlorine pesticides, particularly endosulfan, by Trichoderma harzianum. Environmental Toxicology and Chemistry 12(6): 1059-1065.
- Kvenvolden KA, Cooper CK (2003) Natural seepage of crude oil into the marine environment. Geo-Marine Letters 23(3-4): 140-146.
- Holliger C, Gaspard S, Glod G, Heijman C, Schumacher W, et al. (1997) Contaminated environments in the subsurface and bioremediation: organic contaminants. FEMS Microbiol Rev 20(3-4): 517-523.
- Alvarez PJJ, Vogel TM (1991) Substrate interactions of benzene, toluene, and para-xylene during microbial degradation by pure cultures and mixed culture aquifer slurries. Applied and Environmental Microbiology 57(10): 2981-2985.
- Medina-Bellver JI, Marín P, Delgado A, Rodríguez-Sánchez A, et al. (2005) Evidence for in situ crude oil biodegradation after the Prestige oil spill. Environmental Microbiology 7(6): 773-779.
- April TM, Foght JM, Currah RS (2000) Hydrocarbon-degrading filamentous fungi isolated from flare pit soils in northern and western Canada. Can J Microbiol 46(1): 38-49.
- Ulrici W (2000) Contaminant soil areas, different countries and contaminant monitoring of contaminants, in Environmental Process II. Soil Decontamination Biotechnology 11: 5-42.
- Leahy JG, Colwell RR (1990) Microbial degradation of hydrocarbons in the environment. Microbiological Reviews 54(3): 305-315.
- Adebusoye SA, Ilori MO, Amund OO, Teniola OD, Olatope SO (2007) Microbial degradation of petroleum hydrocarbons in a polluted tropical stream. World J of Microbi and Biotechnology 23(8): 1149-1159.
- Mediene S, Valantin-Morison M, Sarthou JP, Tordonnet S de, Gosme M, et al. (2011). Agro ecosystem management and biotic interactions: a review. Agron Sustain Development 31(3): 491-514.
- Torsvik V, Ovreas L (2002) Microbial diversity and function in soil: from genes to ecosystems. Current Opin Microbiology 5(3): 240-45.
- Sheng XF, Jiang CY, He LY (2008) Characterization of plant growth-promoting Bacillus edaphicus NBT and its effect on lead uptake by Indian mustard in a lead-amended soil. Can J Microbiology 54(5): 417-422.
- Torsvik V, Gosoyr J, Daae FL (1990) High diversity in DNA of soil bacteria. Applied Environ Microbiology 56(3): 782-787.
- Garbeva P, Van Elsas JD (2008) Rhizosphere microbial community and its response to plant species and soil history. Plant Soil 302(1-2): 19-32.
- Berg G, Opelt K (2006) The rhizosphere effect on bacteria antagonistic toward the pathogenic fungus Verticillium differs depending on plant species and site. FEMS Microbiology Ecology 56(2): 250-261.
- Eisenhauer N, Milcu A, Sabais ACW, Bessler H, Weigelt A, et al. (2009) Plant community impacts on the structure of earthworm communities depend on season and change with time. Soil Biology and Biochemistry 41(12): 2430-2443.
- Sabais ACW, Scheu S, Eisenhauer N (2011) Plant species richness drives the density and diversity of Collembola in temperate grassland. Acta Oecologica 37(3): 195-202.
- Kopke U, Nemecek T (2010) Ecological services of faba bean. Field Crops Research 115(3): 217-233.
- Dong Z, Layzell DB (2001) H2 oxidation, O2 uptake and CO2 fixation in hydrogen treated soils. Plant and Soil 229(1): 1-12.
- Niewiadomska A, Klama J, Wolna-Maruwka A, Sulewska H (2010) Effect of manure application on the development dynamics of proteolytic and ammonification bacteria under maize (Zea mays L.) cropping. Acta Scientiarum Polonorum Agriculture 9: 11-20.
- Clement CR, Habte M (1995) Genotypic variation in vesicular-arbuscular mycorrhizal dependence of the pejibaye palm. Journal of Plant Nutrition 18: 1907-1916.
- Borner H (1960) Liberation of organic substances from higher plants and their role in the soil sickness problem. Botanical Review 26(3): 393-424.
- Ladygina N, Hedlund K (2010) Plant species influence microbial diversity and carbon allocation in the rhizospere. Soil Biology & Biochemistry 42(2): 162-168.
- Larkin RP, Griffin TS, Honeycut CW (2010) Rotation and cover crop effects on soil-borne potato diseases, tuber yield and soil microbial community. Plant Disease 94(12): 1491-1502.
- Aparna K, Pasha MA, Rao DLN, Krishnaraj PU (2014) Organic amendments as ecosystem engineers: microbial, biochemical and genomic evidence of soil health improvement in a tropical arid zone field site. Ecological Engineering 71: 268-277.
- Sharma N, Srivastava LL, Mishra B (1983) Studies on microbial changes in soil as a result of continuous application of fertilizers, farmyard manure and lime. J Indian Soc Soil Science 31: 202-206.
- Van Bruggen AHC, Semenov AM (2000) In search of biological indicators for soil health and disease suppression. Applied Soil Ecology 15(1): 13-24.
- Mandal A, Thakur JK, Sahu A, Manna MC, Patra AK (2015) Impact of transgenic cotton on microbial activity and diversity under Vertisols of Central India. The 2nd Int. Conference on Bio-resource and Stress Management during 7-10th Jan 2015 at PJTSAU, Rajendranagar, Hyderabad, India p. 168.
- Bashan Y, Levanony H (1988) Adsorption of the rhizosphere bacterium Azospirillum brasilense Cd to soil, sand and peat particles. J. Gen. Microbiology 134: 1811-1820.
- Gouaerts B, Mezzalama M, Unno Y, Sayre KD, Luna-Guido M, et al. (2007) Influence of tillage, residue management, and crop rotation on soil microbial biomass and catabolic diversity. Applied Soil Ecology 37(1-2): 18-30.
- Helgason BL, Walley FL, Germida JJ (2009) Fungal and bacterial abundance in long-term no-till and intensive-till soils of the northern great plains. Soil Science Soc of America Journal 73(1): 120-127.






























