The Effect of Removing Mineral Fertilizers from the Roots on Plant Productivity
Akhtyamova GA and Chikov VI*
Kazan Institute of Biochemistry and Biophysics, FRC Kazan Scientific Center of RAS, Russia
Submission: February 13, 2019; Published: February 22, 2019
*Corresponding author: Chikov VI, Kazan Institute of Biochemistry and Biophysics, FRC Kazan Scientific Center of RAS, ul. Lobachevskogo 2/31, Kazan, Tatarstan, Russia
How to cite this article: Akhtyamova GA and Chikov VI. The Effect of Removing Mineral Fertilizers from the Roots on Plant Productivity. Agri Res& Tech: Open Access J. 2019; 20(1): 556115. DOI: 10.19080/ARTOAJ.2019.20.556115
Abstract
As is known, in the course of selection many new highly productive breeds were created in the 20th century. But this process resulted in cultivated plants’ root system reduction, that’s why they became non-resistant to unfavorable environment factors (primarily drought). It was explained by the fact, that mostly favourable conditions were created to plants and there was no need for them to develop extensive root system. At the same time, it’s quite evident that drought resistance, for example, is determined, first of all, by the ability of water-conducting roots to to reach water going deep into the soil. It is worth to note that to produce1 g of dry matter, a plant in the process of photosynthesis will have to spend hundred - thousand-fold water volume. The only way out is the formation of an extensive water-conducting root system and prevention of a break between a plant roots and water going deep into the soil.
Keywords: Soil; Plant roots; Environment factors; Photosynthetic products; Fertilizers
Introduction
Our research of different drought-hardiness varieties of barley showed that in the early stages of ontogenesis resistant plants have twice the size of the root system [1]. Experimental variation of photosynthetic products mass (removal of part of the leaves) or inhibition of their export from the leaves (increased nitrate level) have revealed that this process can be regulated. Further experiments with the level of mineral nutrition in the soil and washed sand showed the possibility of increasing root growth by 1.5-2 times [2]. An additional effect (up to three-fold) of root growth stimulation is obtained by spraying plants with complex compounds of copper and zinc (ammonia) [3].Our research of different drought-hardiness varieties of barley showed that in the early stages of ontogenesis resistant plants have twice the size of the root system [1]. Experimental variation of photosynthetic products mass (removal of part of the leaves) or inhibition of their export from the leaves (increased nitrate level) have revealed that this process can be regulated. Further experiments with the level of mineral nutrition in the soil and washed sand showed the possibility of increasing root growth by 1.5-2 times [2]. An additional effect (up to three-fold) of root growth stimulation is obtained by spraying plants with complex compounds of copper and zinc (ammonia) [3].
Special test, revealed that when barley plants were sown in boxes with grey forest soil (size 60x60x60cm), it was enough to place the seeds in a sowing furrow of 5 x 5cm to increase their root-coverage almost twice Data reported in this work showed that separation of seed and fertilizer flows during sowing is an important condition for the development of water-conducting root system. This report presents the results of first full-scale tests, in which an attempt was made to separate the flows on a farm scale.
Methods
The tests were carried out on industrial seeding of Nur-R3 barley variety. Since an appropriate seeder with the separation of seed and fertilizer flows for crops has not yet been available, we decided to use a standard seeder СЗС-3,6, but modified it as follows (Figure 1).
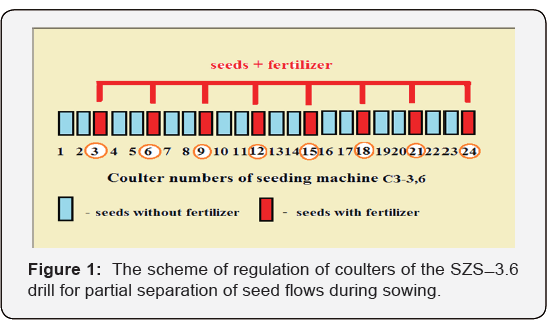
This seeder has 24 openers, each supplies seeds from one box, and mineral fertilizers from the other through corrugated plastic tubes. Both flows can be regulated and even completely blocked by latches (in the case of fertilizers). Therefore, we left one opener unchanged (where both seeds and fertilizers are supplied), in two next openers the flow of fertilizers was blocked by special latches. As a result, eight openers of the seeder worked in a regular mode. In these openers seeds were sown together with fertilizers, and the remaining sixteen ones (two after each regular one) sowed only seeds. As a result, the fertilizers per planting area unit decreased three-fold, while the total number of seeds sown remained unchanged.
Thus, each opener without fertilizers was located near an opener with fertilizers. Therefore, the initial stage of development of seeds sown in sixteen openers was in a fertilizer-free environment. But later on these plants could get the required quantity of mineral nutrition from the next row. Thus, the efficiency of fertilizers use was to increase.
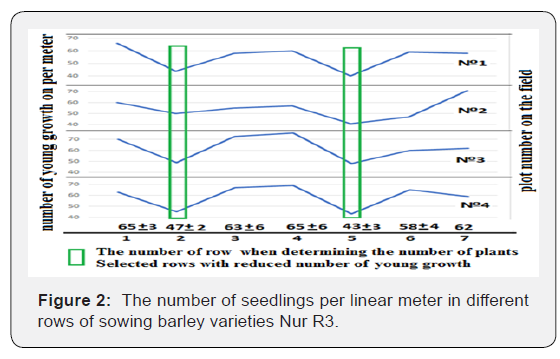
Due to the cold weather, spring sowing was done only on May 9, which is a week later than usual. Because of the cold, the emergence was late, too. On May 30, 2018, we have tested the number of sprouts in different seeding rows. Since it was impossible to determine in the field, which row of plants refers to which opener (with or without fertilizers), we adopted the following method. We selected a levelled area in the field with uniform shoots. Then a meter ruler was placed along one row and the number of sprouts per linear meter was calculated. After that, the number of plants per linear meter in the next row was calculated. And so, moving a ruler across the rows of sowing to the right (each time on one row), the number of plants in seveneight rows was calculated. We revealed that a row with a lower number of shoots was found every two rows. Next, moving in the sowing along the rows ahead, the next levelled area of the field was selected and this procedure was repeated. Thus, four curves were obtained, in each of which (regardless of the sampling area) a row with a decreased number of shoots was found at regular intervals of two rows (Figure 2).
On June 5, 2018, plants were sprayed with solution of ammonium compounds (10-4М concentration) at a levelled planting area (3 x 7m). Then, at the end of tillering on June 16, samples of plants from different roes were collected (including those sprayed with ammonia) to assess the degree of tilling capacity, mass of roots and shoots. The sampling procedure was the same, moving from the original row to the right, sequentially, one after the other, collected samples from each next row. Samples of control plants (untreated with ammonia) were sampled from the same rows as the samples of plants processed by ammonium compounds. Thereat the total number of plants in each of the options in the test was 12-15 (for fertilized options) and 18-24 for the unfertilized ones. The tables show the average statistical data with a standard error.
Results
The calculation of the number of plants per meter in the neighboring rows showed explicit dependence on availability of fertilizers in the soil near the seeds (Figure 2). Where there were fertilizers, sprouts were in a smaller quantity. At some plots, the number of plants per meter was calculated for more than seven rows. However, the pattern of decrease in the number of shoots repeated in every two rows. These facts confirm the depressing effect of mineral fertilizers on germination of seeds. Therefore, by dividing the flows of seeds and fertilizers at sowing, it is possible to save expensive seed material.
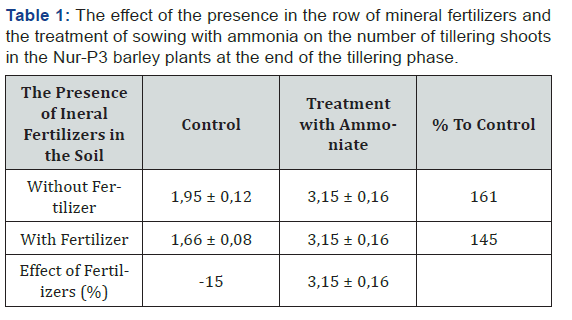
The subsequent analysis of morphometric indices of test barley showed their reliable dependence both on presence of fertilizers in soil, and on treatment of plants by complex ammonium compounds. These effects had an impact on sprout formation (Table 1). Fertilizers reduced tilling capacity both in the control plants (without ammonium treatment), and in the treated ones. It should be noted that the effect of ammonium compounds on tilling capacity manifested to a greater extent in the absence of mineral fertilizers. This shows some antagonism effects of fertilizers and ammonia on the metabolism of plants.
Our previous research [4,5] of the effect of mineral fertilizers (particularly nitrates) and ammonia on photosynthesis allow to assume that they effect mainly through the change of photosynthetic metabolism and transport of the main final product of photosynthesis - saccharose from leaves. The change of extracellular (cell-wall) invertase activity is also involved in this mechanism. When ammonia increase the Ph of the extracellular aquatic environment of leaves, invertase decreases its activity, since this enzyme is active only in the acid medium. Sucrose is not hydrolyzed. The absence of saccharose hydrolysis contributes to its successful load into the phloem vessels and export from the leaf. In addition, there is a decrease in osmolality of extracellular aquatic environment, since due to sucrose hydrolysis to molecules are formed of a one (glucose + fructose). The reduction of osmolality near the guard cells opens them and thus increases the flow of carbon dioxide into the leaf (and hence its photosynthetic digestion).
The study of photosynthetic metabolism direction [5] showed its dependence on the activity of photosynthesis products use by the acceptors, which depends on the level of nitrate nutrition [6,7]. But in terms of a lower nitrate feeding, when the use of carbohydrate products of photosynthesis in synthetic processes of new tissues is complicated, sugars can be used only for synthesis of cellular walls cellulose. The latter is only possible for the synthesis of the roots cell walls, as these cells are highly vacuolated and cellulose is the basis of their dry weight.
Hence, it follows that to increase the roots mass it is required to sow crops in the unfertilized soil (primarily refers to nitrates). But at the same time fertilizers should be near for the following development of the above-ground organs of the plant (including the economically important ones). By the time the waterconducting roots are well-developed, the feeding roots will reach the nearby mineral fertilizers and they will be able to produce rich yield. The symbiosis of plants with soil microorganisms is of great importance. The above mentioned process created the wellknown chernozemic (black) soils. But mineral fertilizers suppress microorganism activity. Therefore, it is believed that in the course of cultural farming for the last 100 years, half of the organic matter of our black soils were lost [8].
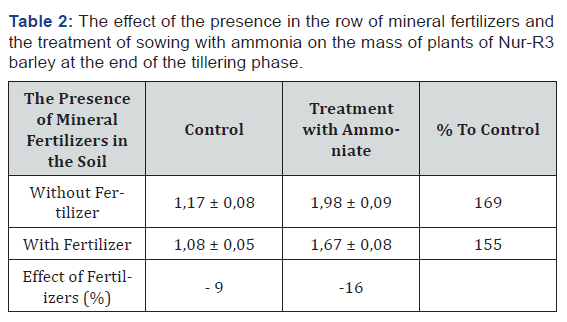
Similar effects of these factors were found in the analysis of dry mass of plants (Table 2). However, in this case, the effect of ammonia with present mineral fertilizers was less significant. This allows us to make a conclusion about the possibility of further intensification of root formation process after studying details of this mechanism.
Conclusion
The tests clearly signify the need to divide the flows of seeds and fertilizers in the sowing process. This will significantly reduce the use of mineral fertilizers in agriculture and increase their efficiency. The plants root system free of high level of nitrates in the zone of their activity will be more actively interacting with the soil microflora, which would supply the required amount of nitrogen due to microbiological absorption of air nitrogen.
Finally, mankind should adopt agricultural practices which will allow to intensify development of cultivated plants root system to a degree which will gradually exclude the use of mineral fertilizers completely. Assessment of the evolutionary development of nature over the last billion years makes it quite obvious that it is perfectly possible. After all, our black soils rich in nutrients (for plants) were formed by nature without any mineral fertilizers. And it is our task to live in harmony with nature. The increased root mass will serve the basis for accumulation of soil organic mass which, in due course, will increase its fertility. It requires the efforts of agricultural researchers and scientists to search the ways to maximize the root mass. It is worth to note that symbiotic microorganisms work in drought, too. One may have probably observed, that if to dig out a plant from soil in drought, it is possible to see roots covered with a 2-3cm layer of soil balls. While around is dehydrated medium. These are the free symbiote microbes. They absorb nitrogen of air getting water and sugar from a plant. This process is controlled by the plant. The plant gives microorganisms as many sugars as it needs nitrogen. Microorganisms also protect the plant from water loss, as they isolate a wet root from excessive water loss. Lower rain-wash of mineral fertilizers from soil will reduce their transfer to rivers, which would improve the ecological situation in nearshore.
Since the development and serial production of a new seeder which would separate the flows of seed and fertilizer in the course of sowing will take time and will require additional pilot testing, the first step in the development of this technology, perhaps, in the next vegetation year will be an extended testing, and then application of the above technology using the old seeder in probable drought areas (and not only).
References
- Blokhin VI, Tagirov MS, Chikov VI (2016) Influence of mineral nutrition on tillering process of different morphological biotypes of spring barley. Niva Tatarstana 2(3): 40-42.
- Chikov VI, Akhtyamova GA, Batasheva SN, Diurbin DS, Tagirov MSH, et al. (2017) Rooting in early stages of ontogenesis of different barley morphobiotype. Niva Tatarstana 3(4): 50-52.
- Chikov VI (2002) The European Bureau of the World Intellectual Property Organization, N WO 02/092538 A2. 21.11.
- Chikov VI, Bakirova GG, Avvakumova NY, Belova LA, Zaripova LM (2001) Apoplastic transport of 14C-photosynthates measured under drought and nitrogen supply. Biologia Plantarum 44(4): 517-521.
- Chikov VI, Bakirova GG, Batasheva SN, Sergeeva AA (2006) The influence of ammoniates on 14CO2 assymilation in flax. Biologia Plantarum 50(4): 749-751.
- Batasheva S, Abdrakhimov F, Bakirova G, Isaeva E, Chikov V (2007) Effect of nitrates supplied with the transpiration flow on assimilate translocation. Rus J Plant Physiology 54(3): 373-380.
- Batasheva S, Abdrakhimov F, Bakirova G, Isaeva E, Chikov V (2010) Effects of sodium nitroprusside, the nicric oxyde donor, on photosynthesis and ultrastructure of common flax leaf blades. Russian Journal of Plant Physiology 57(3): 376-381.
- Kovda VA, Pachepsky PA (1989) Soil resources of the USSR, their use and restoration. Pushchino, pp. 35.






























