Geochemical Weathering Patterns in Relation to Lithology and Land Use in the Shrink Swell Soils of Yavatmal District, Maharashtra, India
Bhaskar BP*, Satyavati PLA, Anantwar SG and Gopal Tiwari
ICAR-National Bureau of Soil Survey and Land Use Planning, India
Submission: October 24, 2018,Published: February 06, 2019
*Corresponding author: Bhaskar BP, Principal Scientist, ICAR-National Bureau of Soil Survey and Land Use Planning, Amravati road, Nagpur-440033, Maharashtra, India
How to cite this article: Bhaskar B, Satyavati P, Anantwar S, Gopal T. Geochemical Weathering Patterns in Relation to Lithology and Land Use in the Shrink Swell Soils of Yavatmal District, Maharashtra, India. Agri Res& Tech: Open Access J. 2019; 19(5): 556107. DOI: 10.19080/ARTOAJ.2019.19.556107
Abstract
Seven shrink-swell soils;
a. On Late Paleozoic to Mesozoic fluvial sedimentary sequence of Wardha valley [Chanoda-limestone (P1), Wani-sandstone (P2), Nagdhari-basalt (P3)].
b. On Deccan basalt sequence of Upper Cretaceous to Paleocene [Selodi (P3), Loni (P5)].
c. On [Dhanki under irrigation (P6)].
d. On Archean group gneiss Kharbhi (P7)
Were studied for geochemical variations in genetic horizons with weathering indexes and elemental ratio’s. These shrink-swell soils have mean of 23.53±2.22% Si, 8.82±1.18% Al, 6.2±1.74% Fe and 0.81±0.31% Ti with least variability of Si and Al in Ap, Bw and Cr horizons but moderately variable in slickensided horizons. The pedotransfer functions showed that Al is powerful predictor of clay, Ca for CaCO3 and bulk density with Al, Fe and Ti whereas negative relation of COLE (coefficient of linear expansion) with molar ratio’s of Fe, Ca, Si and Ti and saturated hydraulic conductivity (K sat) with molar ratio of mMg and mTi. The positive curvilinear relationship of Fine clay with CALMAG index was due to generation of exchangeable Ca and Mg from fine clay and that of Mg being an integral part of smectite. The chemical weathering patterns in soils as assessed by molar ratio’s of mobile elements with Ti showed lowering of mSi to mTi ratio in the genetic horizons of irrigated Dhanki (P6) and enrichment of carbonates in slickensided horizons of Selodi (P3), Loni (P5) and Kharbhi (P7).
Keywords: Soil geochemistry; Vertisols; Molar ratio’s; Mass balance; Yavatmal
Abbreviations: ICP-AES: Inductively Coupled Plasma Atomic Emission Spectroscopy; MAP: Mean Annual Precipitation; CV: Coefficient of Variation; CEC: Cation Exchange Capacity
Introduction
The Vertisols and associated soils in India occupy 73 million hectares, out of which 38% (28 million hectares) are Vertisols, 37% Vertic Inceptisols and 21% Entisols [1]. The fine textured black Vertisols dominate in the basins [2,3] with abundant smectite [4]. The elemental composition in relation to particle size in Vertisols of Gujarat showed that coarse silt contains 4% of CaO and Na2O but decreased to less than 1% in fine silt fraction and have molar ratio of Al and Al+Fe more than 0.5 indicating the presence of montmorillonite-bedillite group [5]. Chemical changes in soils during weathering were quantified in several ways including the normalized value of element (or oxide) using their parent rock concentrations or immobile elements [6], ratio of elements to immobile elements [7,8], measurement and calculation of loss or gain of weight (or volume) based on immobile element [9] and chemical weathering indices [10]. These weathering indices were used for assessing weathering patterns of red and black soils of Purna valley [11].
The chemical weathering in shrink-swell soils was assessed through loss of major elements (mobile) with respect to an immobile element (Ti as stable index element) [12,13]. In subsequent studies, Nordt and Driese [14] was used CALMAG index to derive reliable mean annual precipitation (MAP) for Vertisols with standard error of ±108mm. In contrast to the abundant literature on basaltic soils in many countries, the geochemical knowledge of Vertisols and associated soils occurring on different geological formations along Penganga and Wardha valleys in Yavatmal district, Maharashtra is remarkably limited. These soils on uplands are extensively used for rainfed cotton cultivation and in canal irrigated areas for sugarcane cultivation. They provide scope for understanding the changes in weathering patterns of Vertisols and vertic intergrades under altered moisture regimes and in identifying geochemical indicators of long-term effects of irrigation in the region. The present study on geochemical properties of shrink-swell soils details chemical weathering patterns through weathering indices and estimated losses and gains of elements during pedochemical processes.
Materials and Methods
Elemental analysis
Seven representative Vertisols and associated pedons (P1 to P7) in cotton and sugarcane growing Yavatmal district were selected for geochemical characterization (Figure 1). The Chanoda series on limestone (P1), Wani series on sandstone (P2), Dhanki series on basalt (P6), and Kharbhi series on granite gneiss (P7) were classified as Typic Haplusterts; Selodi series on basalt (P3) as Sodic Haplusterts (sugarcane)*; Loni series (P5) as Chromic Haplusterts and Nagdhari series (P4) as Fluventic Haplustepts [15]. These profiles have variety of B horizons reflecting subsurface horizons like cambic (Bw) or calcic (Bk) and slickensided zones (Bss). The fine earth fraction (<2mm) of each horizon samples were used for elemental analysis by ICP-AES – Teledyne Leeman Labs, USA (Model Prodigy High dispersion ICP) in the chemical laboratory of Soil Resource Studies. The macroelements were analyzed. Schematic geochemical patterns in shrink-swell soils are presented in Figure 2.

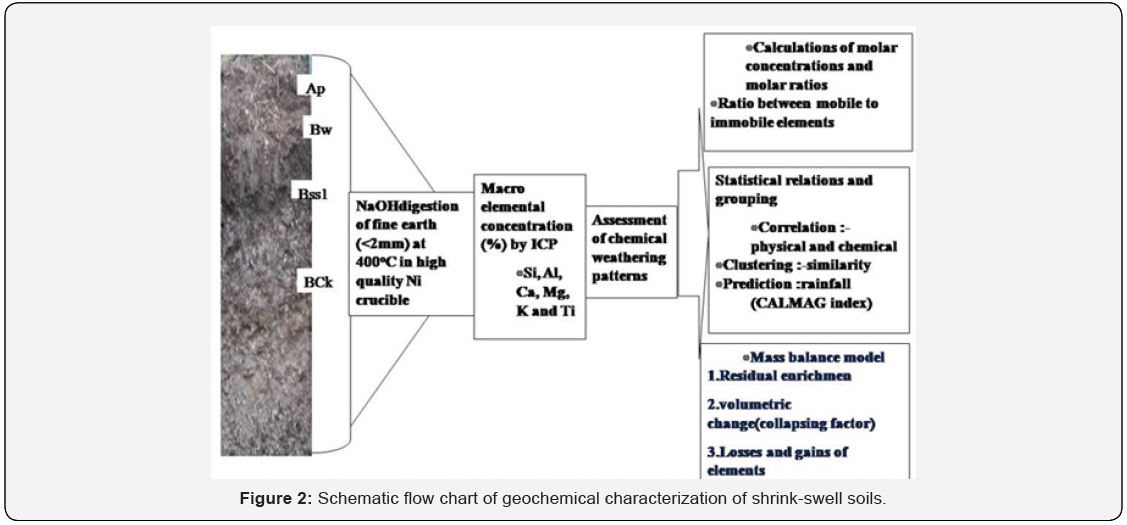
Molar ratios
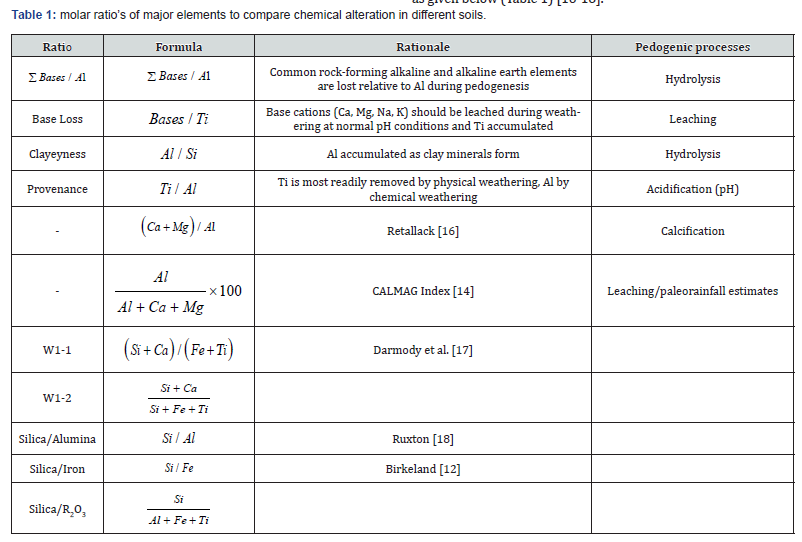
For each ratio, the percent of major elements were first divided by molecular weight to facilitate calculation of molar ratios. The weathering indices were calculated by molar ratio’s of major elements to compare chemical alteration in different soils as given below (Table 1) [16-18].
Mass balance
A geochemical mass balance calculations were made assuming Ti as immobile element as per the formulae of Brimhall et al. [19].
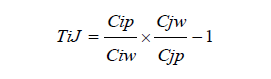
Where C represents concentration with subscript i for immobile element, j for element of interest, w for weathered material and p for parent rock. i, j=-1, then the element is completely depleted during weathering.
Results
Elemental composition
The insoluble elemental constituents in well drained environments include Si, Al, Fe and Ti whereas soluble constituents include Ca, Mg, K and Na. Among insoluble elements Si is dominant with mean of 23.9±3.5%, minimum of 13.09 in Bss3 horizon (P7) maximum of 36.01% in Bss horizon (P6, Table 2). The profile distribution of Si is irregular in Chanoda (P1) and Wani (P2) with values of 16.63% in Bw (P2) to 26.52% in Bss horizons (P1). The Al is 4.82% in Bss4 horizons (P1) to 10.7 % in Bw2 horizon of P4 with profile distribution pattern of irregular trends in soils of sedimentary sequence of Wardha valley (P1 and P2), decreasing trends in P7 and increasing concentrations of Al in Bw horizons of P4 (10.2 to 10.7%). The Fe is less than 5% in the profiles of P1 and P2 as compared to other soils. The Fe distribution pattern is irregular in all soils except in Nagdhari (P4) where as gradational decrease in Fe from 7.76% to 5.46%. In general, Ti is less than 1% P1, P2 and P3 with irregular depth trends.
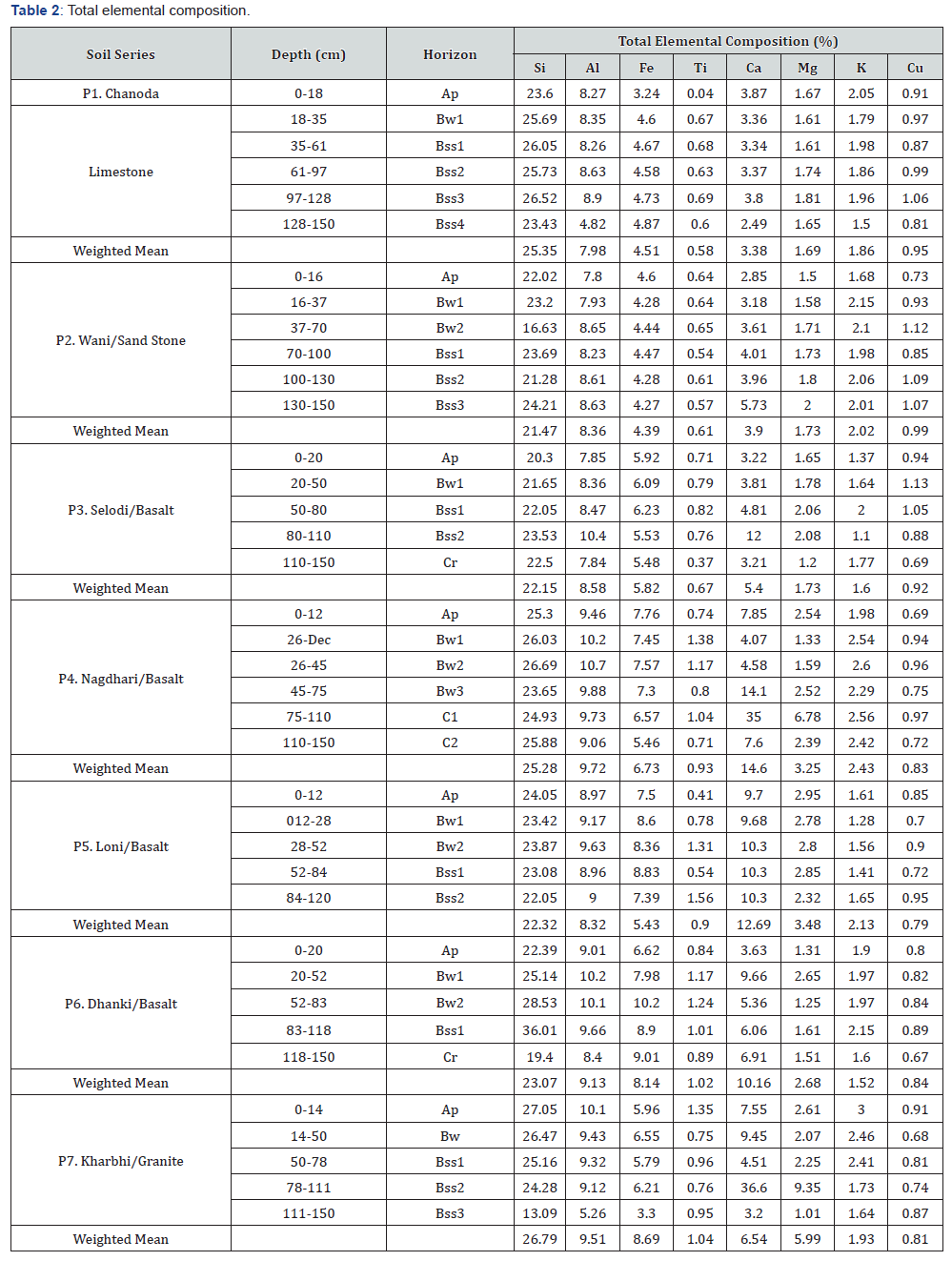
Among soluble constituents, Ca is dominant ranging from 2 to 37% but reaches maximum in Bss horizons of P3. The Mg content is more than 2%in P4 and P7 but reaches to 9.35% in P7 where Ca content is 36.6%. In Nagdhari soil (P4), the Mg distribution shows gradational increase from 1.33% (Bw) to 6.58% (C horizon) with an increment of Ca more than 35%. The increment of Ca and Mg concentrations is in agreement with increase of CaCO3 content and appearance of lime nodules with strong effervescence in the profiles. This kind of distribution pattern of Ca and Mg strongly supports the addition of soluble constituents [20,21]. Generally, the K content is more than 1.5% but exceeding more than 2% in the profiles of P4 and P7. The Cu content is less than 1% with irregular trends in all soils but exceeding 1% in some slickensided horizons of P1, P2 and P3.
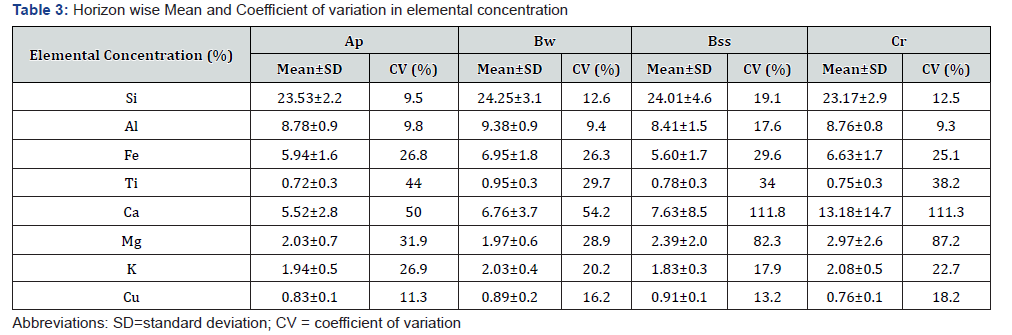
Horizon wise mean and coefficient of variation of elemental composition
The percent concentration of insoluble constituents such as Si, Al, Fe and Ti is high in Bw horizons with mean of 24.25±3.05% for Si, 9.38±0.88% for Al, 6.95±1.83% for Fe and 0.95±0.28% for Ti (Table 3). Among these elemental groups, the coefficient of variation is 19.11% for Si, 17.59% for Al in Bss horizons where as 44.02% for Fe and 49.95% for Ti in Ap horizons. It is observed that Cr horizons have high the concentration of soluble constituents with mean 13.18±14.67% for Ca, 2.97±2.59% for Mg, 2.08±0.47% for K. The slickensided B horizons have mean 7.63±8.52% for Ca and 2.39±1.96% for Mg. The coefficient of variation is 82 to 111% for these elements. The Bss horizons have high mean concentration of Cu (0.91±0.12%) with CV of 13.23%.
Weathering ratios
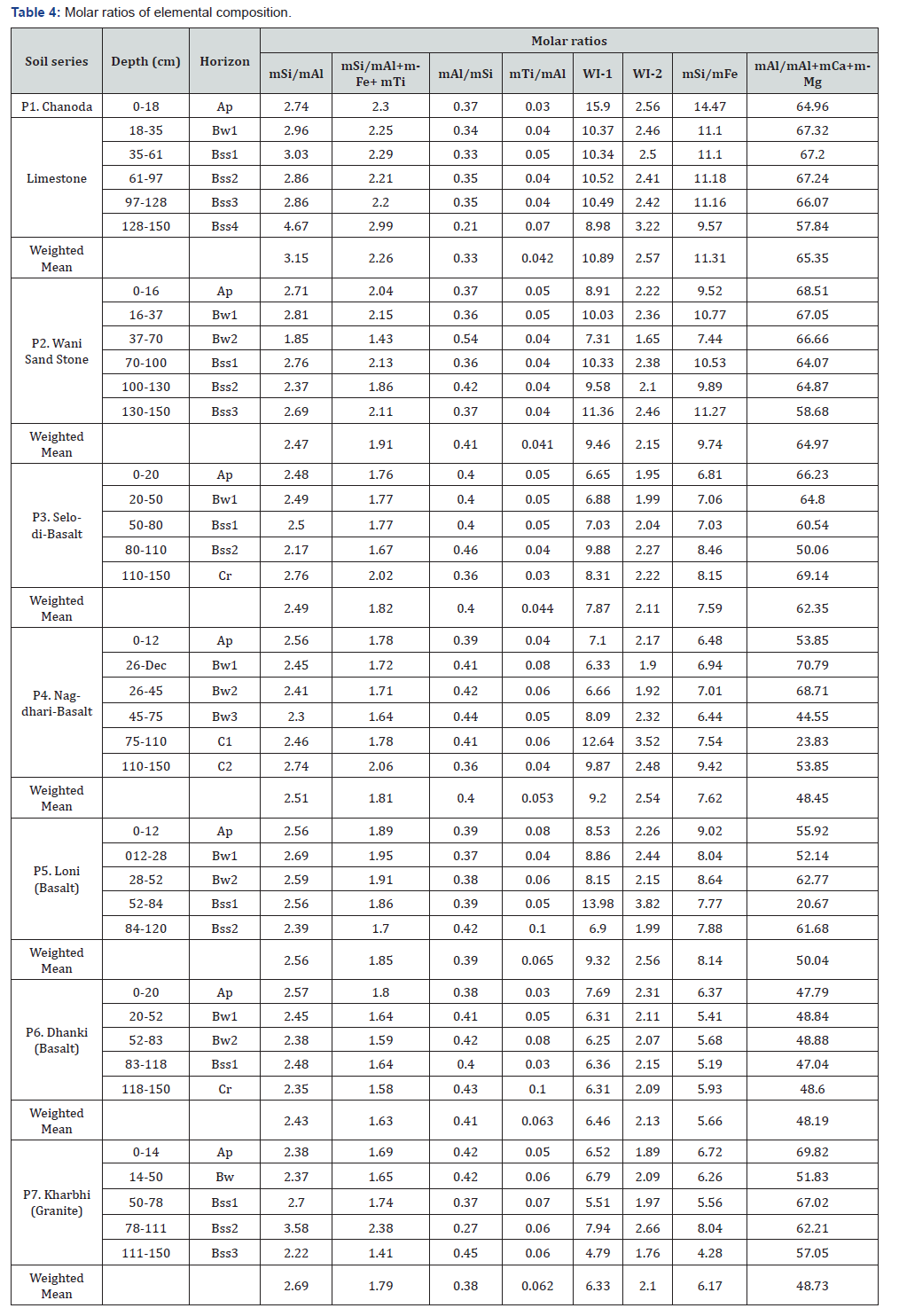
The molar ratios of Si, Al, Fe and Ti are frequently used in homogeneous weathering system of shrink-swell soils. The reason for such criteria is to minimize the effects of down profile variations from parent material composition. The mSi/mAl(Sa) and mSi/(mAl + mFe + mTi) - (Saf) ratio’s vary from 1.85 to 4.67 with a mean of 2.62±0.44. This ratio is less than 3 in majority of horizons due sequence of soil age but with constraints of time dependence of soil composition change. The Sa and Saf in these soils are in agreement with basaltic soils as found in Hainan island, China [22]. The clayness (mAl / mSi) varies from 0.21 (Bss4, P1) to 0.54 (Bw2, P2) with uniform distribution in case of Selodi (P3), Nagdhari (P4), Loni (P5) and Dhanki (P6). These values are in agreement with values reported in Vertisols [14]. There is strong positive curvilinear relationship between total Al and clay as expressed in regression equation: Clay (%) = 8E- 05 (Al%)3 -0.014 (Al%)2 + 0.665 (Al%) with R2 (coefficient of determination) value of 0.53** (significant at 1% level). The relation of Al with Ti is expressed in regression equation as: Al (%) = 0.026 (Ti%)2 – 0.060 (Ti%) with R2 of 0.435* (significant at 5% level). This relationship indicates that Ti is also behaved similarly moving either as fine grained Ti rich mineral (Fe and Ti oxides) or adsorbed as Ti on clays. This observation is in agreement with the observations reported by Young and Nesbitt [23]. These soils with CEC > 50cmol (+) kg-1 and 50% clay content indicate the presence of stratified aluminosilicates [24].
The depth wise variations of mSi/mTi ratio shows variable rate of aluminosilicate weathering but lower values in case of irrigated Selodi (P3) with values of 46 to 52 in Ap to Bss horizons as against upland soils having weighted mean of 67.13 (P1) and range of 65.49 to 69.87. The depth function of mFe/mTi is similar to that of mSi/mTi with values of 6.9 (P1) to 14.1 (Bss1; P5) indicating the co-migration of Fe along with Si and Ti (Table 4). The mSi/mFe ratio is more than 10 in Chanoda (P1) but shows increasing trends from 6.81 to 8.46 in Selodi (P3). The bases to R2O3 are less than 1 except for Bss horizons where this ratio exceed 1. Similar depth trends of this ratio reported in Vertisol topo-sequence of Turkey [25]. The reasons verified for enrichment of bases in these soils is due to regional climate (semi-arid hot with distinct seasonal variations in rainfall that favour shrink-swell phenomenon) coupled with high Ca and Mg presence and formation of Fe rich trioctohedral smectite [26]. These soils have Si to sesquioxide ratio less than 2 with mean 1.91±0.32. The CALMAG index (mAl/mAl + mCa + mMg) varies from 47 to 68 with a mean of 57±12.03% and CV of 20.9%. The weathering indices -1 (mSi + mCa/mFe + mTi) has a mean of 8.87±2.33 and CV of 26.24%, whereas weathering indices-2 (mSi + mCa/mAl + mFe + mTi). Varies from 1.65 to 3.53 with mean of 2.33±0.44 (Table 4). The weathering ratios generally indicate moderate weathering of shrink-swell soils in the region.
Chemical weathering patterns
The chemical weathering in shrink-swell soils is assessed through loss of major elements (mobile) with respect to an immobile element that is unlikely to experience loss in solution (Ti as stable index elements [12,13]. These shrink swell soils have mCa to mTi ratio less than 10 in Chanoda (P1) and Dhanki (P6) with weighted mean 6.02 (P1) to 7.56 (P2) but Wani (P2) has ratio of 11.92 in Bss3 horizon with weighted mean of 7.74. The gradational increase of this ratio with depth in P2 (from 5.24 to 11.92) and P3 (from 5.42 to 18.75) clearly indicates enrichment of calcium. The depth function of this ratio is irregular indicating differential mobility of calcium in P4 and P7.
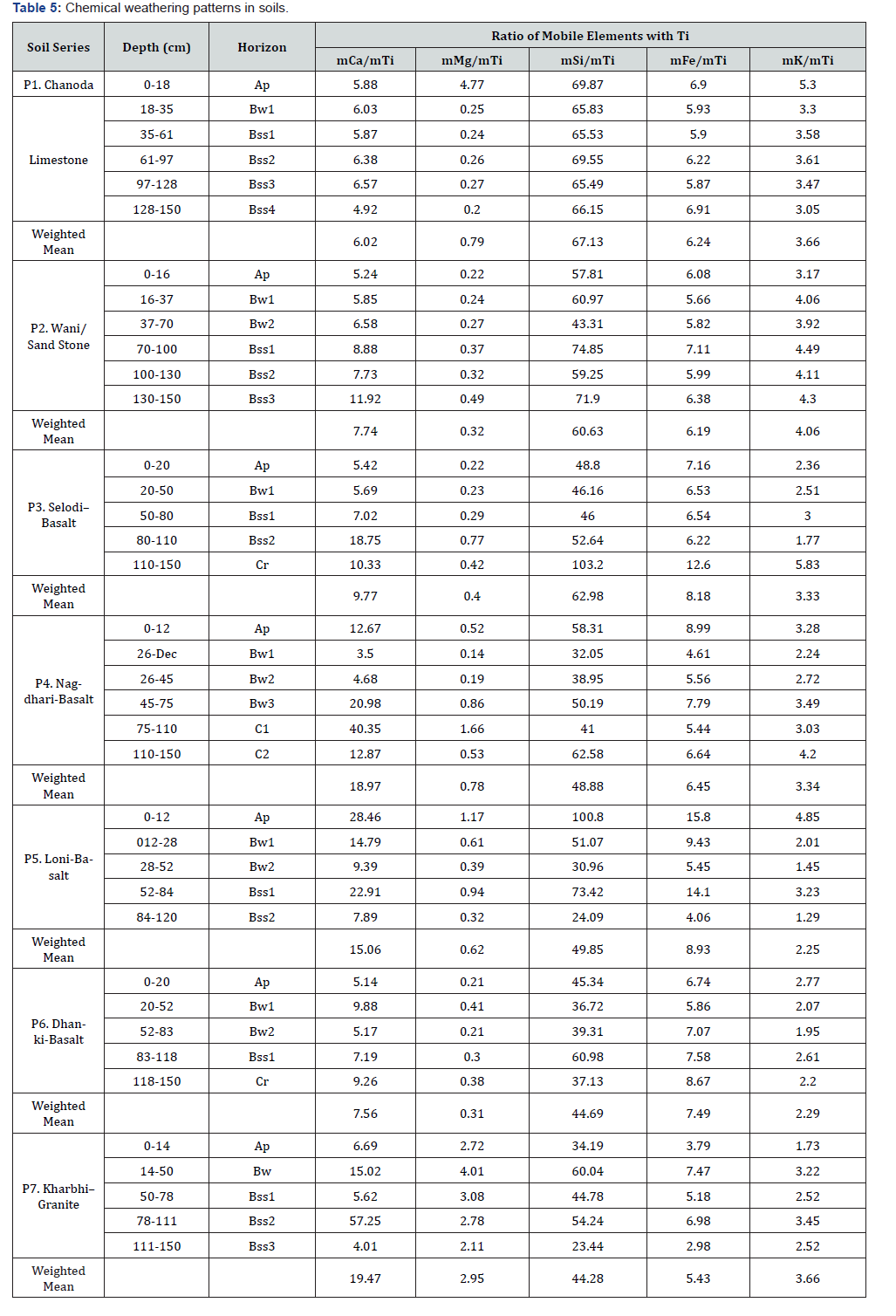
The mMg/mTi ratio is more than 0.3 in majority of horizons except in Kharbhi (P7) where this ratio is more than 2 with weighted mean 2.95 (Table 5). The mSi / mTi ratio shows distinct variations between the soils and horizons indicating variable rate of aluminosilicate mineral weathering. The Irrigated soils (P4, P5, P6 and P7) have weighted mean of mSi / mTi ratio less than 50 in control section (25 - 100cm). The mFe / mTi ratio varies from 2.98 in Bss3 (P7) to 14.1 in Bss1 (P5) with weighted mean of 8.93 in Loni (P5) and 8.18 in Selodi (P3). The depth functions of this ratio is similar to that of mSi / mTi with its maxima 6.9 in P1, 7.11 in Bss of P2 and 14.1 in Bss1 of P5. The mK / mTi ratio is varied from 1.45 in P5 to 4.49 in P2. These soils are ranked on the basis of weighted means of this ratio in the ascending order as: Wani (P2, 4.06) > Chanoda (P1) ≈ Kharbhi (P7, 3.66) > Selodi (P3) ≈ Nagdhari (P4, 3.33) > Dhanki (P6, 2.29) > Loni (P5, 2.25).
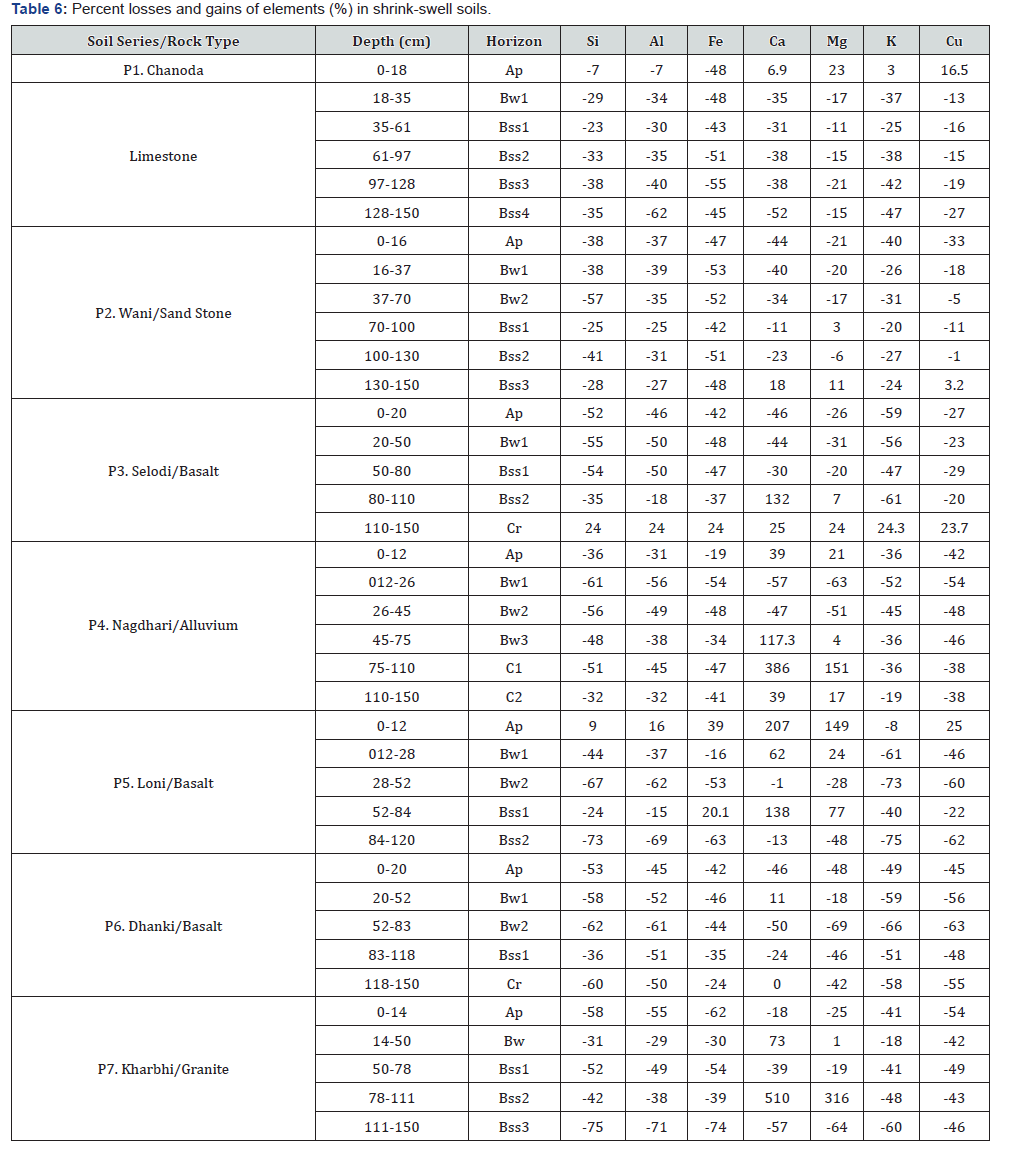
Losses and gains of elements
The Chanoda (P1) on limestone shows net loss of 7% in Si, Al and 48% of Fe but little gains of Ca (6.9%), Mg (23%), K (3%) and Cu (16.5%) in Ap horizon but net loss of 23 to 38% Si, 30 to 62 % Al, 43 to 55% Fe, 31 to 52% Ca, 11 to 21% Mg, 25 to 47% K and 13 to 27% Cu in Bw and Bss horizons (Table 6). The Wani (P2) shows loss of 25 to 57% of Si, Al, Fe but shows decreasing the rate of loss of Ca from 44% in Ap to 11% in Bss1, 21% to 3% for Mg and 47 to 20% for K. Little gain of Ca (18%), Mg (11%) and 3.2% of Cu in Bss3 is recorded. The Selodi (P3) and Nagdhari (P4) shows more than 50% loss of Si, K and Al, 37 to 48% of Fe, Ca and 20 to 31% of Mg, Cu in genetic B horizons with considerable gains of Ca and Mg in slickensided Bss3 horizons. The Loni (P5) shows surface enrichment of Si, Al, Fe, Ca, Mg and Cu but loss of 8% of K in Ap horizons whereas loss of all elements in succeeding genetic B horizons. In Dhanki (P6), there is a loss of all elements but 11% gain of Ca in Bw horizon. The Bss3 horizon in Kharbhi (P7) have 63 to 73% loss of Si, Al, Fe, K and Cu and gain of 510% of Ca and 316% of Mg in Bss2 horizon.
Discussion
Vertisols and associated vertic intergrades are easily distinguished by high amounts of shrink-swell clays, minimal horizon differentiation due to pedoturbation, prolonged wide cracks in dry seasons, very sticky and plastic consistency when wet and have unique subsurface slickensides reflecting vertization process [27]. Geochemical analyses revealed that the main elemental constituent in these studied profiles are Si (22 to 27%), Al (8 to 9.7%) and Fe (4.4 to 8.7%), Ti (0.58 to 1.04%), K (1.52 to 2.43%) and alkali-earth elements (3.4 to 14.6% Ca and 1.69-5.99% Mg). In general, the more geochemically mobile total elements (Na, Mg, K, and Ca) will decrease with weathering grade as they are released from host minerals and leached from the parent rock. Less mobile and immobile oxides such as Si, Al, P, Mn, Fe and Ti will remain behind [28]. Molecular weathering ratios reveal the changing chemical proportions as a result of processes or properties like clayeyness (Al/Si), calcification (Ca + Mg)/Al), base to alumina ratio (Ca + Mg + Na + K/Al). The molar ratio of the Si-to Al + Fe Ti is uniform with slight depth variation but its values >2 in Chanoda (P1) and some horizons of Wani (P2). This index confirmed the predominance of silica and the uniformity of their chemical composition. Moreover, Si/Al ratio is varied from 2.27 to 2.94 with Si remaining clearly more abundant than Al. The clayeness (mAl/mSi) varies from 0.21 to 0.54 with uniform distribution. Al is good predictor of total clay content (R2=0.53*) due to its concentration in clay fraction as part of 2:1 phyllosicate structure. The influence of carbonate weakens this relationship because carbonates dilute Al content in relation to clay and also due to the tendency of lowering of fine clay with increase of carbonate content. The very rich alkaline and alkali-earth elements in the profiles are probably related to the low topographic position and the strongly contrasted climate which limits leaching rate and moderates lixiviation [29]. Base enrichment in these soils is in agreement with lowering of the CALMAG index in soil control section (25 to 100cm) and is used as calcification factor to construct its relation with CaCO3 as calcification factor = 0.019 (CaCO3)3 -0.007 (CaCO3)2 -3.69 (CaCO3) + 69.02 (R2=0.26*).
The CALMAG was used as predictor of mean annual rainfall [14] with the regression equation as mean annual rainfall (MAP) = 22.89*CALMAG - 435.8. This regression equation was used with the CALMAG index to approximate the propable rainfall of 587mm (Nagdhari, P4-North West) to 1749mm (Chanoda, South East) as against the current rainfall gradient of 498mm to 928mm. The molar ratio of bases to Al is more than 1 indicating high degree of alkalinity [16]. The very high Si/Al ratio was in agreement with the predominance of 2/1 clay minerals representing typical bisiallitisation process [10]. The increase of Fe and Mg in slickensided zones are indicative of presence of an iron rich smectite as confirmed with molar ratio of Fe to Fe + Mg exceeding 0.5 [5]. The distinct chemical weathering patterns showed lowering of molar ratios of Si to Ti (44 to 48, loss of plagioclase) under irrigated conditions and enrichment of carbonates with higher Ca to Ti ratios in upland soils (15 to 19). Ti is an appropriate choice of immobile index element mainly because of evidence of detrial influx of silt sized Zr grains and their subsequent translocation within the profile [30]. These soils are characterized by net removal of 35 to 75% of Si, 30 o 60% of Al, Fe, K and Cu and net gains of Ca of 60 to 120% and of Mg content of 20 to 60% in lower parts of the profiles (Table 6) due to its accumulation and in-situ crystallization. The net loss of redox sensitive elements Fe and Cu is significant with strong co-variation and lowering of ɛh conditions developing during the times of soil saturation. The inflections in the transport of elements as noted at the interface of slickensides to bedrock (critical depth of vadose zone) and degree to which the evaporation exceeds precipitation (dominant in semiarid hot conditions). The mass transfer of individual elements is greater within the soil. The calculated losses of Si, Al, Fe and K (negative values) due to chemical weathering and mechanical mixing that obliterate the vertical patterns of mass change through homogenization. This is true to say here that elemental chemistry is having relation with physical processes in genesis and evolution of soils having constant thickness of horizons on local landscapes. The effect of clay formation (clayeyness, mAl/mSi) on soil water movement is the source of difference in the rate of mass loss between the soils and within the horizons. The mean annual rainfall in the region (700 to >1000mm) with its concentration more in valleys account for higher rates of weathering in more stable environment. The residence time of soil saturation increases and favors the accumulation of aluminous phases due to very slow to slow saturated hydraulic conductivity [31]. It is true to say that at lower elevations having thick clay soil cover impedes water flow through soil column and slowly reach chemical saturation control that accelerate silicate weathering rates [32]. That is the reason why the chemical weathering patterns assessed through molar ratios of Ca/Ti and Si/Ti showed distinct variations in Selodi (P3), Loni (P5) and Dhanki (P6) under irrigated tracts as compared to other soils under rainfed.
Acknowledgement
Authors wish to express thanks to Dipak Sarkar, former Director for his encouragement and support to bring out this manuscript for publication. We wish to express thanks to Sri. S.V. Bobde and Sri. S.S. Gaikwad for their support during field survey in Yavatmal district.
Conflict of Interest
There is no conflict of interest as this project is fully done under my guidance as institute approved research project.
References
- Murthy RS (1981) Distribution and properties of vertisols and associated soils. Proceedings of the seminar on improving the management of Indian deep black soils. pp. 9-16.
- Krishnan MS (1982) Geology of India and Burma. (6th edn), CBS Publishers and Distributors.
- Deshpande GC (1998) Deccan Traps. In: Geology of Maharashtra, Geol Soc India. pp. 129-162.
- Bharghava GP, Bhattacharjee JC (1982) Morphology, genesis and classification of salt affected soils. In Review of Soil Research in India, Part-II, International Congress of Soil Science, Indian Society of Soil Science, New Delhi, pp. 508-528.
- Satyavati PLA, Raja P, Ray SK, Anantwar SG, Bhaskar BP (2012) Elemental compostion and Minerology of silt and clay fractions of cracking clay soils of semiarid and arid parts of Gujarat, India. Clay Research 76: 518-527.
- Minarik L, Absolon K, Kollnerova Z, Klecka M (1983) Chemical change of granite during its weathering. In: Augustithis SS (ed.), Leaching and Diffusion in Rocks and their weathering products, Theophrastus Publications. S.A. Athens, pp. 293-306.
- Chesworth W, Dejou J, Larroque P (1981) The weathering of basalt and relative mobilities of the major elements in Belbex, France. Geoch Cosmochim Acta 45(7): 1235-1243.
- Guan PNG, Sun M, Tan, (2001) Weathering indices for rhyolitic tuff and granite in Hong Kong. Engineering Geology 59(1-2): 147-159.
- Huston DL (1993) The effect alteration and metamorphism on wallrocks to the Balcooma and Dry river south volcanic-hosted massive sulphide deposites, Queensland, Australia. Jour Geochem Expl 48(3): 277-307.
- Price JR, Velbel MA (2003) Chemical weathering indices applied to weathering profiles developed on heterogeneous felsic metamorphic parent rocks. Chem Geol 202(3-4): 397-416.
- Raja P, Bhaskar BP, Malpe M, Anantwar SG, Tapaswi PM (2010) Geochemistry of salt affected soils in Purna valley, Maharashtra. Their Palaeoclimatic implications. Jour Appl Geoch 12(3): 491-501.
- Birkeland P (1999) Soils and Geomorphology. Oxford University Press, New York, USA, pp. 448.
- Muhs DR, Bettis EA, Been J, McGeehin J (2001) Impact of climate and parent material on chemical weathering mid-continental North America during last glaciation. Science Society of America Journal 65(6): 1761-1777.
- Nordt LC, Driese SG (2010) New weathering index improves paleorainfall estimates from Vertisols. Geology 38(5): 407-410.
- Soil Survey Staff (2014) Keys to Soil Taxonomy. United States Department of Agriculture, Natural Resource Conservation Service, (10th edn). pp. 1-325.
- Retallack G (1998) Core concepts of paleopedology. Quaternary International 51/52: 203-212.
- Darmody RG, Thorn CE, Allen CE (2005) Chemical weathering and boulder mantles, Kärkevagge, Swedish Lapland. Geomorphology 67: 159-170.
- Ruxton BP (1968) Measures of the degree of chemical weathering of rocks. Journal of Geology 76(5): 518–527.
- Brimhall GH, Lewis CJ, Ford C, Bratt J, Taylor G, et al. (1991) Quantitative geochemical approach to pedogenesis: importance of parent material reduction, volumetric expansion and eolian influx in lateralization. Geoderma 51: 51-91.
- Bratley SL, White AF (2009) Approaches to modeling weathered regolith. Rev Mineral Geochem 70(1): 435-484.
- Bhaskar BP, Dipak Sarkar, Bhattacharyya T, Satyavati PLA, Bobde SV, et al. (2015) Geochemical characterization of shrink-swell soils in Yavatmal district, Maharashtra. Journal of Applied Geochemistry 17(1): 98-108.
- Zhang GL, Pan J, Huang CM, Tong Gong Z (2007) Geochemical features of a soil chronosequence developed on basalt in Hainan Island, China. Revista Mexicana de Cinecias Geologicas 24(2): 261-269.
- Young GM, Nesbitt HW (1998) Processes controlling the distribution of Ti and Al in weathering profiles of siliclastic sediments and sedimentary rocks. Journal of sedimentary Research 68(3): 448-455.
- Pal DK, Deshpande SB (1987) Characteristics and genesis of minerals in some benchmark vertisols of India. Pedologie 37: 259-275.
- Dengiz O, Saglam MH, Ozaytekin H, Bagkhan O (2013) Weathering rates and some physico –chemical characteristics of soils developed on a calcic toposequences, Carpathian Jour Earth Environ 8(2): 13-24.
- Decarreau A, Bonnin D, Badaut-Trauth D, Couty R, Kaiser P (1987) Synthesis and crystallogenesis of ferric smectite by evolution of Si-Fe co-precipitates in oxidizing conditions. Clay Minerals 22(2): 207-223.
- Mermut AR, Padmanabham E, Eswaran H, Dasog GS (1996) Pedogenesis. Developments in Soil Science 24: 43-61.
- Arikan F, Ulusay R, Aydin N (2007) Characterization of weathered acidic and a weathering classification based on a rating system. Bull Eng Geol Environ 66: 415-430.
- Aydinalp C (2010) Some important properties and classification of vertisols under Mediterranean climate. Afr J Agricult Res 5(6): 449-452.
- Driese SG, Mora CI, Stiles CA, Joeckel RM, Nordt LC (2000) Mass-balance reconstruction of a modern Vertisol: implications for interpreting the geochemistry and burial alteration of Paleo-Vertisols. Geoderma 95(3-4): 179-204.
- Chadwick OA, Brimhall GHJr, Hendricks DM (1990) From black to gray box- a mass balance interpretation of pedogenesis. Geomorphology 3(3-4): 369-390.
- White AE, Bullen TD, Schultz MS, Blum AE, Huntington TG, et al. (2001) Differential rates of feldspar weathering in granitic regoliths. Geochemica et Cosmochimica Acta 65(6): 847-869.






























