The Effect of Humic Acid, Plant Growth Promoting Rhizobacteria and Seaweed on Essential Oil, Growth Parameters and Chlorophyll Content in Basil (Ocimum basilicum L.)
Hamidreza Bayat1* and Sergei Belopukhov2
1Department of Agronomy, Russian State Agrarian University, Russia
2Department of Chemistry, Russian State Agrarian University, Russia
Submission: January 18, 2019,Published: January 28, 2019
*Corresponding author: Hamidreza Bayat, Department of Agronomy. Russian State Agrarian University, Timiryazev Agricultural Academy, Russian Federation, Moscow, Russia
How to cite this article: Hamidreza B, Sergei B. The Effect of Humic Acid, Plant Growth Promoting Rhizobacteria and Seaweed on Essential Oil, Growth Parameters and Chlorophyll Content in Basil (Ocimum basilicum L.). Agri Res& Tech: Open Access J. 2019; 19(4): 556103. DOI: 10.19080/ARTOAJ.2019.19.556103
Abstract
Effect of Humic acid, plant growth promoting rhizobacteria and seaweed extract on growth parameters, essential oil and chlorophyll content of herb basil (Ocimum basilicum L.) evaluated. This experiment was done in the form of factorial in randomized complete block design (RCBD) in research field of Islamic Azad University, college of agriculture and natural resources branch of karaj, Mahdasht, Iran in 2017. The treatments were: A) Humic acid with three levels (a1 = nonuse, a2 = foliar application [3 lit/ha], a3= soil application [6lit/ha] , B) plant growth promoting rhizobacteria with two levels (b1= nonuse, b2= use [6lit/ha], C) seaweed extract (c1 = nonuse, c2 = soluble in irrigation water [1.5 lit/ha]).The results illustrated that consumption of Humic acid cause to increase in all studied traits. The highest amount of essential oil (1.1%), fresh weight (0.9kg/m2), dry weight (254.06gr/m2), chlorophyll spad (10.98), height (48.62cm), root length (22.09cm), shoot diameter (4.64mm) and leaf width (3.08cm) were observed at 6 lit/ha Humic acid. Also, results showed that application of seaweed extract lead to increase in fresh weight (0.89kg/m2), chlorophyll spad (11.54), height (47.87cm), root length (22.34cm), shoot diameter (4.44mm), and leaf width (3.02cm). Plant growth promoting rhizobacteria had no significant effect on evaluated traits except essential oil. Mean comparison of interaction effect between Humic acid levels, plant growth promoting rhizobacteria and seaweed extract showed that among the treatments, a3b2c2 (soil application of Humic acid 6lit/ha+ consumption of plant growth promoting rhizobacteria and seaweed) has the maximum amount of all evaluated traits. Overall, results showed that the effect of humic acid on studied traits was much more than plant growth promoting rhizobacteria and seaweed extract.
Keywords: Basil; Humic acid; Plant growth promoting rhizobacteria; Seaweed extract; Growth parameters
Abbreviations: HS: Humic Substances; PGPR: Plant Growth Promoting Rhizobacteria; RCBD: Randomized Complete Block Design; IAA: Indole Acetic Acid
Introduction
Basil has been used for many years as a medicinal plant in traditional medicine to treat headache, cough, parasitic diseases and disorders of the kidney. The external uses of the plant can refer to the use as a tourniquet in place of stings and applying oil directly on the skin to treat acne disease. This plant is also due to the active ingredients (essential oils), has an important role in industries, food, cosmetics that included in the insect repelling properties and anti-parasitic and antibacterial activity of this plant. Basil is an annual herb which is the economically most important species among other species of basil. Nowadays it is almost cultivated and utilized in all warm temperate regions as herb, spices and fresh vegetable [1].
Humic Acid is one of the most abundant organic constituents, which are present in soil and other environments such as streams, oceans and lakes. This substance is a principal component of humic substances and resulted from organic matter decomposition. The effect of Humic Acid on soils is undoubtedly the improvement of physical, chemical and biological soil properties according to other studies [2-4]. Humic Acid plays an extremely beneficial role in plant growth as a complementary agent in organic or chemical fertilizers. It has been shown that humic substances (HS) act as a positive regulator of plant growth and enhance root, leaf and shoot growth as well as fruit yield [5]. Furthermore, an enhancement in yield of various plant species such as vegetables, root crops, flowers and cereals by humic substances have been reported [6].
During the last centuries, seaweeds were used as fertilizer in farming land but nowadays it is also used in both agriculture and horticulture in Romans, Britain, France, Spain, Japan and China. Many searches have been done in recent years that have found wide application in modern agriculture for the use of marine macroalgae as fertilizer. It is reported that seaweeds contain all required trace elements and plant growth hormones and seaweed manure is rich in potassium and poor in nitrogen and phosphorus. A positive effect of several species of algae (Ascophyllum spp., Laminaria spp., Ecklonia sp.) on the growth, development and, consequently, yields of field crops have been proved so far. Seaweeds constitute a source of many substances, valuable from the point of view of plant physiology, which particularly help plants adapt to stressful conditions. Biologically active Algonac acids, polyphenols, free amino acids, and particularly natural plant phytohormones: auxins, cytokinins, intensify plant growth, increase the root weight, and induce photosynthetic processes and as a result improving plant development [7-11]. In some findings the authors particularly stress the importance of cytokines contained in seaweeds in creating plant resistance to diseases and nematodes [12-14]. Plant growth promoting rhizobacteria (PGPR) are groups of bacteria that actively colonize plant roots and are so useful for plant growth. The mechanism by which PGPR promote plant growth is still unknown, but it includes the ability to produce phytohormones, a symbiotic N fixation, synthesis of antibiotics, enzymes and fungicidal compounds. It has been stated that application of PGPR enhanced the growth and yield of several major crops [15].
Materials and Methods
This experiment was performed in a greenhouse of the Islamic Azad University of Karaj, faculty of Agriculture and Natural Resources, at Mahdasht, Karaj, Iran, located at 35.45º N and at 51.6º E and with the height of 1313 m from sea level. The experiment was designed as factorial in randomized complete block design (RCBD) with four replications per treatment. Prior to testing the depth between the surface and 30cm in the soil was randomly sampled and sent to the laboratory in order to determine the soil physical and chemical properties (Table 1).

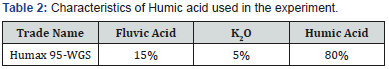
Experimental treatments were Humic Acid in three levels including {A1: nonuse (control), A2: Foliar application at 3 liters per hectare and A3: soil application at 6 liters per hectare}, PGPR treatment in two levels of {B1: nonuse and B2: consumption of 6 liters per hectare that PGPRs were including Azotobacter, Azospirillum and Pseudomonas}. In this experiment seaweed was used in two different levels {C1: nonuse, C2: the use of 1.5 liter per hectare in irrigation water}, the Humic acid was used in this experiment was 80% and named Humax 95-WGS (Table 2). All three bacteria used in this study were native of Iran and they were separated and purred in Water and Soil Institute in Iran. The cultivation was in the form of ditch and hill. In this research the distances between rows were 20cm and every plot had two rows of cultivation.
The seeds were mixed by PGPRs and then cultivated in every plot by the distance of 5cm in a row and buried in the soil in depth of 0.5 to 1cm. The irrigation process was done immediately after cultivation and after that it has done every four days. The use of Humic acid and seaweed was done in every 15 days. We had randomly chosen 4 plants in every row and tagged for data recording purpose. We aimed to measure traits and characteristics of essential oil, fresh weight, dry weight, chlorophyll spad [16], height, root length, shoot diameter and leaf width of basil. In order to measure dry weight samples were placed in oven for 48 hours in 70 ˚C. Moreover, Minolta SPAD-520 chlorophyll meter was employed to measure the leaf chlorophyll contents and selection of the plants for measurement was randomized. This experiment was repeated at least for 3 random plants in each plot.
Statistical analysis
One-way ANOVA was applied to analyze the obtained data using SAS software (Ver 9.1) [17]. For each source of treatment, the means of each trait were separated statistically using Duncan’s multiple range test, with significance defined as P≤0.05.
Essential oil
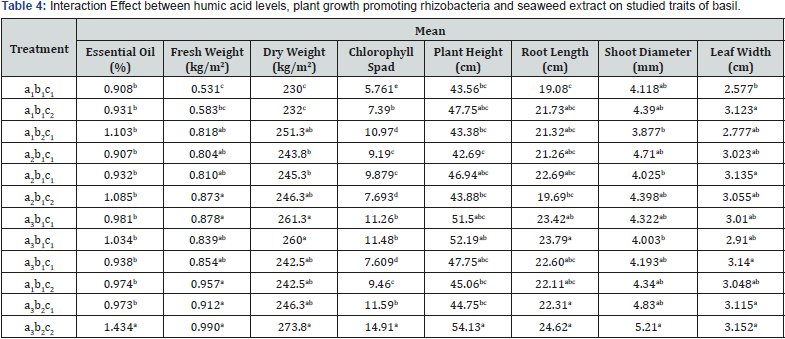
Table 3 shows that Humic acid and plant growth promoting rhizobacteria had significant effect on essential oil (P≤0.05). Essential oil ranged from 0.96 to 1.10%, and it increased as the Humic acid level was increased also. Also, consumption of plant growth promoting rhizobacteria increase essential oil at 1.96%. As indicate in (Table 3) there were no significant differences among seaweed extract treatment regarding to essential oil. The effect of interaction between Humic acid, Plant growth promoting rhizobacteria and seaweed extract on essential oil is shown in Table 4. High Humic acid level (6 lit/ha) and consumption of plant growth promoting rhizobacteria and seaweed extract had the highest essential oil of dill plants by 1.43%. Under organic condition essential oil of basil was two time as application of chemical manures [18]. There is reported that consumption of biologic fertilizer lead to increase in essential oil [19,20]. Besides, other researchers represented that organic fertilizers can raise amount of essential oil in medicinal plant [21,22].
Fresh and dry weight
Results in Table 3 indicate that Humic acid, plant growth promoting rhizobacteria and seaweed extract had significant effect on fresh weight (P≤0.01). Fresh weight increase at 6lit/ha with 0.9 (kg/m2) as compared to controls. The plants grown in plots received seaweed extract and plant growth promoting rhizobacteria had significantly more fresh weight with 0.89 and 0.87 (kg/m2) in compared to control respectively. As is seen in Table 3 humic acid led to increase in dry weight of basil (P≤0.01). Seaweed extract and plant growth promoting rhizobacteria had no significant effect on dry weight. The highest amount of dry weight was found at 6 lit/ ha Humic acid with 254.06 (gr/m2). According to the Table 4 high Humic acid level (6 lit/ha) and consumption of plant growth promoting rhizobacteria and seaweed extract had the highest fresh and dry weight of dill plants by 0.99 and 273.8(gr/m2) respectively. There are several reports regarding the action mechanisms of humic substances. These mechanisms divided into two groups:
A. Direct effect on the plants.
B. Indirect mechanisms through influencing the microorganisms in the soil, uptake of nutrient from the soil as well as physical properties of the soil.

Means with the same letter in a column are not significantly different according to DMRT at 1% level of probability.
H: Humic acid, PGPR: Plant growth promoting rhizobacteria.
ns, * and ** are not significant and significant at 5 and 1% level of probability, respectively.
Moreover, humic acid can act like a hormone and may exert its beneficial effects on the plant. Nevertheless, due to its instability in the soils it is assume that another growth regulator like IAA, might absorbed by hamates and as a result promotes the growth of the plant. These results are consistent with the results of other researchers [23-25]. Nelson [26] showed that PGPR has positive effects on plant growth. It has been also reported that PGPR promotes the growth and development of the basil plant through an increase in production of IAA, solubilization of phosphorus and resistance to pathogen and pests [26].
Spad
Means with the same letter in a column are not significantly different according to DMRT at 1% level of probability.
a: Humic acid {a1= control, a2= foliar application 3lit/ha, a3= soil application 6lit/ha}
b: Plant growth promoting rhizobacteria {b1= nonuse, b2= 6lit/ha}
c: Seaweed {c1= nonuse, c2= 1.5lit/ha}
As indicate in Table 3, Humic acid and seaweed extract had significant effect on chlorophyll spad (P≤0.01). consumption of Humic acid at 6lit/ha lead to increase chlorophyll spad by 10.98. Also, The plants grown in plots received seaweed extract had significantly more chlorophyll spad with 11.54 in compared to control (Table 3). According to the Table 4 high Humic acid level (6 lit/ha) and consumption of plant growth promoting rhizobacteria and seaweed extract had the highest chlorophyll spad of basil plants by 14.91. It seems that humic acid elevates the chlorophyll synthesis or arrests the degradation of the chlorophyll in basil plant and thereby affect the chlorophyll content [2]. In addition to this, seaweed extracts possess remarkable amount of hormones such as auxins, cytokines and betaines which can positively affect the synthesis of the chlorophyll in the leaves [27]. Our findings were in accordance with previous findings of Whap ham et al. [28], which observed that application of seaweed (Ascophyllum nodosum) increased chlorophyll of cucumber seedlings and tomato plant [28]. Also, seaweed extract spray enhanced the leaf chlorophyll in plants [7]. Recently Thirumaran et al. [29] on Cyamopsis tetragonoloba indicated that seaweed extract increased photosynthetic pigments such as chlorophyll a, b and total chlorophyll and carotenoids [29].
Plant height
Results of the measurements of plant height is given in Table 3. Plant height was significantly affected by Humic acid and seaweed extract (P≤0.01). Plant height increase at 6 lit/ha with 48.62(cm). Applications of seaweed extract led to rise in plant height with 47.87(cm). Plant growth promoting rhizobacteria had no significant effect on plant height (Table 3). The effect of interaction between Humic acid, Plant growth promoting rhizobacteria and seaweed extract on plant height is shown in Table 4. High Humic acid level (6 lit/ha) and consumption of Plant growth promoting rhizobacteria and seaweed extract had the highest essential oil of dill plants by 54.13(cm). The increases in plant height can be explained by the proposal that humic acid make the nutrient more accessible to the plant and due to important role of them in several aspects of plant bioactivities, the growth and development of the plant is promoted [30]. Moreover, humic acid enhances the soil porosity and significantly affects the development of the root which in turn can induce the shoot growth [31]. It has been also noted that humic substance has a remarkable impact on physical and chemical properties of the soil and consequently ameliorate the nutrient mineral uptake and aerial soil properties [32]. Meanwhile, an observed enhance in plant height in our study can be due to presence of the auxins in seaweed extract which can induce cell division and elongation and as a result enhance the shoot growth, leaf size and fresh and dry weigh [33]. Our findings are in agreement with the results of other researchers [34,35].
Root length, shoot diameter and leaf width
The data presented in Table 3 illustrated that the effect of humic acid application and seaweed extract on Root length, shoot diameter and leaf width was significant (P≤0.05). Plant growth promoting rhizobacteria had no significant effect on mention traits (Table 3). Consumption of Humic acid at 6lit/ha lead to increase in Root length, Shoot diameter and leaf width with 22.9 (cm), 4.64 (mm) and 3.08 (cm) respectively. The plants grown in plots received seaweed extract had significantly more root length, shoot diameter and leaf width 22.34 (cm), 4.44 (mm) and 3.02(cm) respectively. The effect of interaction between humic acid, plant growth promoting rhizobacteria and seaweed extract on root length, shoot diameter and leaf width is shown in Table 4. High Humic acid level (6 lit/ha) and consumption of plant growth promoting rhizobacteria and seaweed extract had the highest root length, shoot diameter and leaf width of dill plants by 24.62 (cm), 5.21 (mm) and 3.15 (cm) respectively. Application of the humic acid and seaweed remarkably enhanced the above traits. An increase in growth parameters of basil by application of the humic acid can be partially ascribed to the better absorption of the nutrient by plant. It has been stated that consumption of the humic acid enhanced the nitrogen uptake by the root of the olive plants [36]. According to the previous published reports, favorable effects of humic acid on plant growth and development is attributed to the its hormone-like mode of action which implicate in several aspects of plant metabolisms including cell respiration, photosynthesis, oxidative phosphorylation, protein synthesis, and various enzymatic reactions [37-39]. Albeit, it is assume that the mechanism of action of humic acid on plant growth is identical to that of plant hormones, but it is still not clear that humic acid possess hormone-like components [39] although it has been shown that humic acid might have these components [40]. Apart from humic acid, seaweed extracts also have cytokines and can influence various physiological activities such as chlorophyll synthesis which lead to an increase in photosynthesis and as a result induce the growth of the plant [41]. The presences of the macronutrients in seaweed extract, which have a significant role on the plant nutrition such as nitrogen, potassium and phosphorous and are necessary for growth, can also enhance the growth parameters of the plant [42].
Conclusion
This experiment has uncovered the physiological mechanism by which humic acid and seaweed extract can influence plant growth and essential oil positively and produce significant increases in measured traits. It is observed that humic acid and seaweed was more effective than plant growth promoting rhizobacteria on studied traits in basil plant. According to the above findings, the use of humic acid and seaweed extract because of less harmful disadvantage and positive effects on plant growth is an efficient approach to replace chemical fertilizers for sustainable cultivation of basil in Iran and other developing countries.
References
- Halliwell B, Gutteridge JC (1995) The definition and measurement of antioxidants in biological systems. Free Radic Biol Med 18(1): 125-126.
- Apak R, Güçlü K, Demirata B, Özyürek M, Çelik S, Bektaşoğlu B, et al. (2007) Comparative evaluation of various total antioxidant capacity assays applied to phenolic compounds with the CUPRAC assay. Molecules 12(7): 1496-1547.
- Tawaha K, Alali F, Gharaibeh M, Mohammad M, El-Elmat T (2007) Antioxidant activity and total phenolic content of selected Jordanian plant species. Food Chemistry 104(4): 1372-1378.
- Al-Mustafa A, Al-Thunaibat O (2008) Antioxidant activity of some Jordanian medicinal plants used traditionally for treatment of diabetes. Pak J Biol Sci 11(3): 351-368.
- Al-Dabbas M, Saleh M, Al-Ismail K (2015) Preservation methods impacted phenolic flavonoid and carotenoid contents and antioxidant activities of carrots (Daucus carota L.). Journal of Food Processing and Preservation 39(6): 1618-1625.
- Al-Eisawi D, Takruri H (1989) A checklist of wild edible plants in Jordan. Arab Gulf Journal of Scientific Research, Agricultural and Biological Sciences B7(1): 79-102.
- Tukan S, Takruri H, Al-Eisawi D (1998) The use of wild edible plants in the Jordanian diet. Int J Food Sci Nutr 49(3): 225-235.
- Al-Kurd R, Takruri H, Al-Sayyed H (2008) Tannin content of selected plants used in Jordan. Jordan Journal of Agricultural Sciences 4(3): 265-273.
- Al-Sayyed H, Takruri H, Shomaf M (2014) The effect of date palm fruit (Phoenix dactylifera L.) on 7, 12-dimethylbenz (α) anthracene (DMBA)- induced mammary cancer in rats. Research Opinions in Animal and Veterinary Sciences 4(1): 11-18.
- Luterotti S, Bicanic D, Pozgaj R (2006) New simple spectrophotometric assay of total carotenes in margarines. Anal Chim Acta 573-574: 466- 473.
- Agbor G, Vinson J, Donnelly P (2014) Folin-Ciocalteau reagent for polyphenolic assay Int J Food Sci Nutr Diet 3(8): 147-156.
- Pękal A, Pyrzynska K (2014) Evaluation of aluminium complexation reaction for flavonoid content assay. Food Analytical Methods 7(9): 1776-1782.
- Molyneux P (2003) The use of the stable radical diphenylpicrylhydrazyl (DPPH) for estimating antioxidant activity. Songklanakarin J Sci Technol 26(2): 211-219.
- Laerd Statistics (2018) Statistical tutorials and software guides.
- Alezandro M, Youn M, Lajolo F, Genovese M (2011) Commercial spices and industrial ingredients: evaluation of antioxidant capacity and flavonoids content for functional foods development. Ciência e Tecnologia de Alimentos 31(2): 527-533.
- Al-Juhaimi F, Ghafoor K (2011) Total phenols and antioxidant activities of leaf and stem extracts from coriander, mint and parsley grown in Saudi Arabia. Pak J Bot 43(4): 2235-2237.
- Mikami-Konishide I, Murakami S, Nakanishi K, Takahashi Y, Yamaguchi M, et al. (2013) Antioxidant Capacity and Polyphenol Content of Extracts from Crops Cultivated in Japan, and the Effect of Cultivation Environment. Food Science and Technological Research 19(1): 69-79.
- Cirlini M, Mena P, Tassotti M, Herrlinger K, Nieman K, Dall’Astam C, et al. (2016) Phenolic and Volatile Composition of a Dry Spearmint (Mentha spicata L.) Extract. Molecules 21(8): E1007-E1022.
- Li Z, Lee H, Liang X, Liang D, Wang Q, et al. (2018) Profiling of phenolic compounds and antioxidant activity of 12 cruciferous vegetables. Molecules 23(5): E1139.
- Amarowicz R, Narolewska O, Karamać M, Kosińska A, Weidner S (2008) Grapevine leaves as a source of natural antioxidants. Pol J Food Nutr Sci 58(1): 73-78.
- Katalinić V, Generalić I, Skroza D, Ljubenkov I, Teskera A, et al. (2009) Insight in the phenolic composition and antioxidative properties of Vitis vinifera leaves extracts. Croat J Food Sci Technol 1(2): 7-15.
- Khanam U, Oba S, Yanase E, Murakami Y (2012) Phenolic acids, flavonoids and total antioxidant capacity of selected leafy vegetables. Journal of Functional Foods 4(4): 979-987.
- Boligon A, Janovik V, Boligon A, Pivetta C, Pereira R, et al. (2013) HPLC analysis of polyphenolic compounds and antioxidant activity in Nasturtium officinale. International Journal of Food Properties 16(1): 61-69.
- Hassanen N, Eissa A, Hafez S, Mosa E (2015) Antioxidant and antimicrobial activity of celery (Apium graveolens) and coriander (Coriandrum sativum) herb and seed essential oils. Int J Curr Microbiol App Sci 4(3): 284-296.
- Liua X, Ardob S, Bunninga M, Parryb J, Zhoub K, et al. (2007) Total phenolic content and DPPH radical scavenging activity of lettuce (Lactuca sativa L.) grown in Colorado. LWT-Food Science and Technology 40(3): 552-557.
- Sorour M, Hassanen N, Ahmed M (2015) Natural antioxidant changes in fresh and dried celery (Apium graveolens). American Journal of Energy Engineering 3: 12-16.
- Zeb A (2015) Phenolic profile and antioxidant potential of wild watercress (Nasturtium officinale L.). Springerplus 4: 714-721.
- Arfa N, El-Desouky A, Bahlol H, Soliman A (2017) Utilization of Jew’s mallow stems as a natural source of dietary fiber in pan bread product. Annals of Agric Sci, Moshtohor 55(2): 333-342.
- Nour V, Trandafir I, Cosmulescu S (2017) Bioactive compounds, antioxidant activity and nutritional quality of different culinary aromatic herbs. Not Bot Horti Agrobo 45(1): 179-184.
- Qura’n S (2010) Ethnobotanical and ecological studies of wild edible plants in Jordan. Libyan Agriculture Research Center Journal International 1: 231-243.
- Kaur S, Mondal P (2014) Study of total phenolic and flavonoid content, antioxidant activity and antimicrobial properties of medicinal plants. Journal of Microbiology and Experimentation 1(1): 1-6.
- Pelltte P, Shadervian S (2013) Food composition tables for the use in the Middle East. (2nd edn), Beirut, Lebanon: American University of Beirut Press, USA, pp. 126.
- U.S. Department of Agriculture, Agricultural Research Service (2018) USDA Nutrient Database for Standard Reference, Release 18.
- Kumar A, Kumar V, Khan K, Kumar A (2017) Experimental investigation on drying of mint leaves (M. Pulegium) in solar tunnel dryer. Int J Pure App Biosci 5(2): 682-689.






























