Dimethyl Ether from Waste Gas Under Zero Emission Property
Johann Gruber-Schmidt*
Technical CEO, Australia
Submission: February 06, 2017; Published: August 30, 2017
*Corresponding author: Johann Gruber-Schmidt, Theresiengasse 47,A1180 Vienna, Australia, Tel: 004306781224352; Fax: 004312761980; Email: johann.gruber-schmidt@chello.at
How to cite this article: Johann G. Dimethyl Ether from Waste Gas Under Zero Emission Property. Agri Res & Tech: Open Access J. 2017; 11(2): 555807. DOI: 10.19080/ARTOAJ.2017.11.555807
Plant-Microbe Symbioses
Zero emission is the realization of carbon dioxide sinks, by converting carbon dioxide (CO2) to another component. We do not understand under zero emission storing carbon dioxide in wholes, the ocean, or in tanks. If we compare with a biological plant, we have to learn that the biological plant is converting carbon dioxide(CO2) and water(H2O) to biomass and oxygen based on solar energy. The process is divided into two parts, the light cycle and the dark cycle, optimized to the day and night cycle on earth (or in mechanistic interpretation: on the rotation of the earth around the axis). The aim of this photosynthesis process is to produce biomass during the growth process, oxygen is a waste product. Transferring the conversion of water and carbon dioxide to energy, we need hydrogen and as a waste product oxygen. If we now turn over to technical processes which include up scaling we need a fuel which can be produced from biogenic mass and gases and additional from water and carbon dioxide. In this article we focus on dimethyl ether (DME). To enable this process we need at least electric energy provided by the volatile wind and solar energy and by nuclear power.
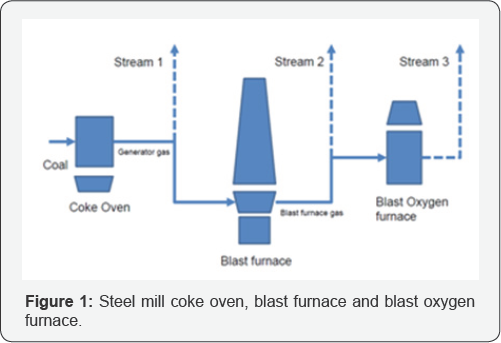
DME, known as dimethyl ether, is the simplest ether [1]. Dimethyl ether is liquid at room temperature T=25 °C and a pressure p=6bar. The molecular mass MZ = 46.0684g/mol, the formation enthalpy at T=298 °C Hf=271.2kJ/mol, the critical Tc = 467 °K(194 °C) and the critical pressure pc = 36bar. Dimethyl ether has a density at T=25 °C and p=6 bar ρ=650kg/m3, and a caloric heat value Hu= 8,3kWh/L. Dimethyl ether can be used instead of liquid propane g as ( LPG, Hu = 6,6 kWh/L )) or low pressure biogas (LPB, Hu = 5,0kWh/Nm3). If dimethyl ether is used instead of fossil Diesel, dimethyl ether has to be prepared according to the ASTM 7905 / 14 A/B and ISO 16 681: 2013 as fuel used in heavy trucks of VOLVO, trucks of FORD or heavy trucks of MACK TRUCK (Figure 1).
Pathways to DME
One pathway of the conversion of landfill gas and biogas to dimethyl ether is the conversion of the biogenic gases with an ATR (auto thermal reactor) to a synthetic gas mainly consisting of carbon monoxide(CO) and hydrogen(H2). The auto thermal reaction is based on CH4 + O2 =>CO2 + H2O + Q(1). The other reactions are based on carbon dioxide(CO2) 2CH4 + O2 + CO2 => 3H2 + 3CO + H2O(1) ( H2 : CO = 1: 1 ) and on steam (H2O) 4CH4 + O2 + H2O => 10H2 + 4 CO(2) (H2:CO=2.5:1). If O2 is in a substoichiometricconcentration a conversion to a hydrogen rich and carbon dioxide rich gas is gained CH4+O2+CO2 => 2H2+2CO2(3).( H2:CO2 = 1:1 ).
Another pathway of the conversion of landfill gas and biogas to dimethyl ether is the conversion of the biogenic gases with dry reforming to a synthetic gas mainly consisting of carbon monoxide (CO) and hydrogen (H2). Under dry reforming we understand CH4+CO2 => 2CO +2H2 (1) and CO2 => CO + 1/2 O2 (2) . In the most cases a gas mixture CH4 :CO2 is used as 1:1 at a temperature of T=600 °C up to 800 °C. Catalysts used are based on γ-Al2O3+Rh reaching a conversion of CH4 up to 85.5%. Additional reactions take place: C + H2O => CO + H2 (3) and C + CO2 => 2CO (4). It is watched that dry reforming leads to a production of carbon C. Therefore we have to add steam (H2O) to activate the WGS (water gas shift reaction). If we add steam(H2O) the RWGS (reverse water gas shift reaction) takes place CO + H2O => CO2 + 4H2(5). For the thermal conversion heat is can be produced by ceramic resistance heat generation with electric energy. Much more difficult is the controlling of the reaction of dry reforming with the production of heat by partial conversion of CH4 + O2=> CO2 + H2O(6). This leads to the needed heat Q (kJ/ mol) and the needed steam (H2O) supporting the WGS (water gas reaction) [2,3].
Beside the thermal catalytic conversion dry reforming with micro wave reforming was tested in a batch reactor. The reaction CO2 + CH4 =>2CO + 2H2 (ΔH =+247,35kJ/mol) was activated by f ~ 2450MHz with a power of P=80W at a operation time τ=4min and a reached conversion rate of 96,5%. The syngas composition was given 46vol% CO, 40vol% H2, C+CxHy 2vol% and H2O 12vol%. For the micro wave application we need electric energy P = 160W ele.
Steel mill flare gas conversion to DME
An interesting application is the conversion of flare gases in steel mills. We have the generator gas from the coke oven 4,81 CO vol%, 14,04 N2 vol%, 53,36 H2 vol%, 1,59 CO2 vol% 23,63 CH4vol, 0,72 O2 col%, 1,85 CxHyvol%, with an caloric heat value Hu= 15,46kJ/Nm3, the blast furnace gas oven 22,55 CO vol%, 48,59 N2vol%, 5,56 H2vol%, 22,3 CO2vol% 0 CH4vol, 0 O2 col% , 0 CxHy vol% with a caloric heat value Hu = 3,04kJ/Nm3 and the blast oxygen furnace gas oven 45,74 CO vol%, 1,93 N2vol%, 0,63 H2vol%, 20,82 CO2vol% 0 CH4 vol, 30,88 O2 col% , 0 CxHyvol% with an heating caloric value Hu = 5,12 kJ/Nm3. The steel mill uses coal as fuel, which is converted to coke in the coke oven. The gas from the coke oven is called generator gas, which will be recycled and therecycled gas (stream 1) can be used for conversion to dimethyl ether, the blast furnace gas is recycled and the recycled gas (stream 2) can be used, and the blast oxygen furnace gas is recycled and the recycled gas (stream 3) can be used again. The volume flows of the three streams coke oven gas, blast furnace gas and blast oxygen furnace gas are given by stream 1 Vol = 2452Nm3/h, stream 2 Vol = 27069Nm3/h and stream 3 Vol = 44235 Nm3/h. Stream 1 and 2 are continuous, stream 3 are in batch [4-6] (Table 1).
This simple example shows an application in heavy industry steel mill. In normal the streams 1,2 and 3 are flared. We ask now: can we use the streams in producing dimethyl ether? Which additional gases will be needed? Which energy is needed to produce dimethyl ether?The sum of the three streams is given 4,81 CO vol%, 14,04 N2vol%, 53,36 H2vol%, 1,59 CO2vol%23,63 CH4vol, 0,72 O2 col% , 1,85 CxHyvol%, with an caloric heat value Hu= 15,46kJ/Nm3. Separating with PSA ( pressure swing adsorption ) nitrogen and oxygen from the sum of gas streams leads to 4,81 CO vol%, 0 N2vol%, 53,36 H2vol%, 1,59 CO2vol% 23,63 CH4vol, 0 O2 col% , 1,85 CxHyvol%, with an caloric heat value Hu= 15,46 kJ/Nm3. Applying dry reforming CH4 + CO2 => 2CO + 2H2 (ΔH298 =+247,35kJ/mol)(1) in combination with the direct conversion reaction(synthesis) 3CO + 3H2 => CH3OCH3 +CO2 (ΔH298 =-90,475kJ/mol)(3) and the sum reaction 3CH4+CO2 => 2CH3OCH3(ΔH298 =75,809kJ/mol). But if we look at the gas composition we have methane(CH4), carbon dioxide(CO2), carbon monoxide(CO) and hydrogen(H2). Besides the dry reforming we have to add the water gas shift reaction (WGS) CO + H2O => CO2 + H2 (Δ H298 =-41,154kJ/mol) and the in combination with the methanol reaction CO2 + 3H2=> CO3OH + H2O (Δ H298 =-49,321kJ/mol) [7,8].
Applying steam reforming CH4+2H2O => CO2 + 4H2 (ΔH = +165,063kJ/mol) (1), and the conversion reaction CO2 + 3H2=> CH3OH + H2O (ΔH298 =-286,27kJ/mol) (2) and the sum reaction CH4+H2O => H2 + CH3OH (ΔH298 =-95,4kJ/ mol). Applying steam reforming as partial oxidation CH4+H2O => CO + 3H2 (ΔH298 =+206,205kJ/mol) (1), CO + H2O => CO2+ H2 (ΔH298 =-41,154kJ/ mol) (2) and the conversion reaction CO2 + 3H2 => CH3OH + H2O (ΔH298 =-49,321kJ/mol) (3) and the sum reaction CH4+H2O => H2 + CH3OH (ΔH298 =-286,27kJ/ mol).
The result of these two pathways show, that applying dry reforming or steam reforming in combination with the direct conversion to dimethyl ether (CH3OCH3) by carbon monoxide(CO) and hydrogen (H2) and the methanol reaction, we can only reduce the carbon dioxide concentration about 44%. In both cases we have the possibility to generate the needed energy by fossil methane(CH4) or by electric energy. This result is not surprising and the conclusion also is: to replace the fossil methane (CH4) by using electric driven electrolysis producing hydrogen (H2) and oxygen(O2) demands electric energy cheaper than coal. This constraint cannot be gained by renewable volatile energy (wind and or solar) only. One possibility again is to add regenerative nuclear power supporting the renewable volatile energy
Biogas conversion to DME
A second application is the conversion of biogas from Castor de oiled cake(DOC) in biogas plants. The properties of Castor de oiled cake is given by 0,4 - 0,5m3/kg TS, 70% methane(CH4), 0,9 TS / FM [2]. If we take 1t FM we get 400m3 Biogas consisting of 280m3 methane (CH4) and 120m3 carbon dioxide (CO2). Applying the process of dry reforming to the conversion of the biogas we need a molar fraction CH4: CO2 of 1:1. We have to add 160m3 carbon dioxide, so that we watch that castor de oiled cake(DOC) has the additional property to be a real carbon dioxide sink.
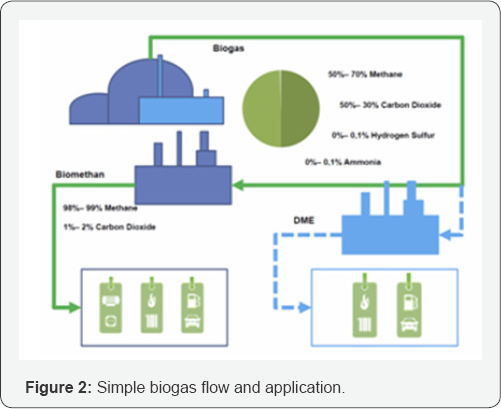
In the practical application in a biogas plant, we have to mix the Castor DOC with an additional substrate as cattle dung or orange peels. The mixture is optimal for 2g/L orange peel and 4g/L Castor DOC producing 1140ml/l biogas with 70% methane and 25% carbon dioxide and 5% carbon monoxide. These experimental results are in correlation with the theoretical biomethane potential (BMP) [9]. As shown in the Figure 2 we analyze the potential of conversion of biogas (methane and carbon dioxide to dimethyl ether
The reactions for dry reforming (prepared and adapted biogas as a mixture of methane and carbon dioxide)are given CH4 + CO2 => 2CO + 2H2(1), and CO + H2O => CO2 + H2(2) and CO + 2H2 => CH3OH with the sum reaction CH4 + H2O => CH3OH + H2 (ΔH298 = +115,3kJ/mol). Because of the existence of H2 we need energy for the conversion to methanol (CH3OH). If we compare with the direct conversion of carbon monoxide (CO) and hydrogen (H2) to dimethyl ether (CH3-O-CH3), we have the following reactions 3CH4+3CO2 => 6CO+ 6H2(1), 6CO + 6H2 => 2CH3-O-CH3+2CO2(2) and the sum reaction 3CH4+CO2 => 2 CH3- O-CH3 (ΔH298 =+75,8kJ/mol). This result shows again: realizing zero emission needs electric energy if we want to avoid fossil methane. One possibility again is regenerative nuclear power.
If we have the following example simple correlation 1m3 biogas ( 50 vol% CH4, 50 vol% CO2 ) lead to 1m3 carbon monoxide (CO) and 1m3 hydrogen (H2). 1m3 carbon monoxide (CO) and 1m3 hydrogen (H2) lead to 1l dimethyl ether. The electric energy needed to produce dimethyl ether P = 2 kW ele. This leads to the relation 1l dimethyl ether needs P = 2kW ele und Q = 1kW th.
Castor DOC is used under co fermentation with cattle manure dung in the mixture 2kg/L cattle manure dung and 4kg/L Castor DOC leading to 1,14 Nm3 biogas ( 68% - 72% methane ). Castor DOC has 84,87% dry mass and cattle manure dung has 81,07% dry mass so that we measure 334,97 L/kg dry mass. If we convert dry mass to fresh mass we get 1 t fresh mass (cattle manure dung, Castor DOC) leads to 290m3 biogas ( 70% methane). A typical biogas plant in India converts about 700t/d Castor de oiled cake(DOC), which leads to a volume flow of 8458 Nm3/h Biogas with 70% methane, 5920,86 Nm3/h methane(CH4) and 2537,5Nm3/h carbon dioxide(CO2). The strength of the carbon dioxide sink is then theoretically given by 3833,334Nm3/h, if we want to have the molar mixture of CH4:CO2 by 1:1.
The conversion of the advanced biogas from Castor de oiled cake to methanol needs energy. If we use an auto thermal reactor based on carbon dioxide(CO2) 2CH4+ O2 + CO2 => 3CO + 3H2 + H2O (ΔH298 =- 30,75kJ/mol) (exothermal reaction) or we use an auto thermal reactor based on steam (H2O) 4CH4 +O2 + 2H2O => 10H2 + 4CO (ΔH298 =+341,16kJ/mol) an endothermic reaction. If we take the exothermal auto thermal reaction in combination with 3CO +3H2 => CH3-O-CH3 + CO2 (ΔH298 =-333,125kJ/mol) it is carbon dioxide neutral and therefore no carbon dioxide(CO2) sink and therefore not useful for zero emission. If we take the endothermic auto thermal reaction in combination CO + 2H2 => CH3OH (AH298 =-94,475kJ/mol) (2) needs the endothermic reaction CO2 + H2 => CO + H2O (AH298 =41,145kJ/mol) (3) to become a weak carbon dioxide sink. From methanol (CH3OH) we can dehydrate dimethyl ether 2CH3OH => CH3-O-CH3 + H2O (AH298 =-111,03kJ/mol). The energy for the endothermic auto thermal reaction again can be gained as electric energy by nuclear power.
Conclusion
These two examples show that for realizing zero emission, we also need electric energy.The application in conversion of the flare gas streams 1,2,3 in a steel mill to dimethyl ether shows that at least 44% of the generated carbon dioxide can be reduced. The application of the conversion of biogas from castor DOC shows, that the application of exothermic auto thermal reforming is in the best case carbon dioxide neutral. If we ask for a carbon dioxide sink we need additional energy. The application in conversion of the flare gas to steel mill can reduce carbon dioxide if we take hydrogen from the recycled generator gas and replace it with methane, or we use additional electric energy to split water by electrolysis or supply the steam reforming of methane to hydrogen. Either we use methane or we use electric energy to realize zero emission.
The application in conversion of biogas from Castor DOC show that using the endothermic auto thermal reforming additional energy is needed to realize a carbon dioxide sink. Again we can use methane or we use electric energy to realize zero emission. We can conclude zero emission and dimethyl ether in combination demands external energy most common as electric energy. One possibility to generate electric energy can be done by nuclear power.
References
- NIST Chemistry Book (2016) Database 69.
- Amon T (2015) Gasausbeute in landwirtschaftlichen Biogasanlagen KTBL.
- Görisch U (2015) Biogasanlagen, Ulmer Publ.
- Späth J (2015) Analyse des Methanolmarktes.
- Höhlein B, Jülich FZ (2008) Methanol als Energieträger.
- Scheltes DG (2008) Efficient Utilisation of steel Mill By Products for High Temperature Heating Process Pittsburg.
- Worrell E (2010) Energy Efficiency Improvement and Cost Saving Opportunities for the U.S. Iron and Steel Industry, LBNL - 4779E.
- Vijay VK, Chandra R, Subbarao PMV (2008) Production of Methane through Anaerobic Digestation, Abu Dabi.
- Thiagarajne J, Rividhya PKS , Rajasakeran E (2013) A Review of Thermo chemical Energy Conversion Process of Non edible Seed Cakes, J.o. Bio Sciences 4(2): 7-15.






























