Participatory Potato (Solanum tuberosum L.) Bacterial Wilt (Ralstoniasolanacearum (E.F.Smith.)) Management in the Central Highlands of Ethiopia
Bekele Kassa*
Ethiopian Institute of Agricultural Research, Holetta Agricultural Research Center, Ethiopia
Submission: March 08, 2017; Published: August 29, 2017
*Corresponding author: Bekele Kassa, Ethiopian Institute of Agricultural Research, Holetta Agricultural Research Center, Ethiopia, Email: bkassa74@yahoo.com
How to cite this article: Bekele K. Participatory Potato (Solanum tuberosum L.) Bacterial Wilt (Ralstoniasolanacearum (E.F.Smith.)) Management in the Central Highlands of Ethiopia. Agri Res & Tech: Open Access J. 2017; 11(1): 555801. DOI: 10.19080/ARTOAJ.2017.11.555802
Abstract
In the major potato growing areas of the central highlands of Ethiopia, experiments on Effective microorganism (EM), Bio-agents as soil amendment, seed cycle and crop rotation using farmers' field school (FFS) approach was conducted between 2009 and 2014. In the soil amendment activity the treatments were improved management package (full fertilizer rate, recommended spacing and improved variety (Jalene); soil amended with full rate of EM-fortified compost+improved management; Soil amended with % EM- fortified compost and % fertilizer of the recommended rate for the area+improved management packages; Soil amended with % EM - fortified compost and % fertilizer of the recommended rate for the area + % EM spray on the foliage weekly+improved management packages; seed tube treated/socked in EM at a dilution of 1:1000v/vl for 30 minutes and farmers practice (1/2 of the recommended fertilizer to the area using local variety). In the rotation trial, one-season rotation and two-season rotation experiments were conducted on fields which had medium infection. In the seed cycle experiment, three improved potato varieties namely: Jalene (CIP-384321.19), Guden (CIP-386423.13) and Awash (CIP-378501.3) and two local varieties, 'Nechabeba' and 'Agazer' were tested for four consecutive years. In the soil amended with EM fortified compost experiment, improved variety had the lowest 21% and 47% mean disease incidence and percent latent infection compared to the local variety which had 25% and 53% respectively. Plots amended with % EM-fortified compost, % fertilizer of the recommended rate for the area and % EM spray had the lowest percentage of latently infected tubers, 26.7% and 30.0%, on the improved and local variety respectively compared to the control which had 50.0% and 76.8% but the incidence of the disease was not statically significant. In the seed cycle experiment, in the 1st season, the local variety had significantly (p<0.05) higher 20.93% BW incidence than the improved variety which had 3.98% whereas in the 4th cycle it increased by 75% and 50% respectively. Under wear potato production scenario, in the 1st cycle, the yield loss was estimated to 4 and 7% in improved and local varieties but in the 4th cycle it has been increased to 21% and 32% respectively. If the produce is for seed purpose, then the estimated loss was increased from 5.34% to 28.6% and from 20.78 to 67.34% from the 1st to the 4th on improved and local varieties respectively. A One season rotation of potato with cabbage, beans, and carrot significantly (P<0.05) reduced the incidence of the disease and had 15.4, 22.1 and 27.3% respectively compared to 45.2% in the control and resulted in negative and significant, Y= -0.2588x +29.767 (R2=0.7946) and Y= -0.177x+28.019 (R2=0.4597), correlation of mean tuber yield over disease incidence. Similarly, the two-season rotation had significant (p<0.05) negative correlation Y=-0.177x+28.019 (R2=0.4597) tuber yield over disease incidence and significantly (P<0.05) increased marketable tuber yield compared to the mono crop potato. The result of these research activities suggested that FFS through session supported extension approach was found to be effective to generate and disseminate bacterial wilt management technologies. Soil amendment with fortified compost helped to reduce the incidence of the disease. Sources of seed and seed revolving year after year was found to be the major factor for high tuber yield loss and the main path for the dissemination of the causative agent of wilt. One and two season rotation with beans and cabbage reduced wilt incidence, latent infection in tubers and increased tuber yield.
Keywords: Potato; Ralstoniasolanscearum; FFS; EM-fortified compost; Seed cycle; Rotation; Tuber yield loss
Introduction
Tremendous growths over last decade in area planted to potato (Solanumtuberosum) in Sub-Saharan Africa [1] likewise in Ethiopia, the area under potato increased three fold [2]. But the yield per unit area was static and national mean yield is about 8 t ha-1 [3] which is very low compared with production statistics in the globe. The low yield is attributed to a number of biotic and abiotic factors in addition to poor crop management. Bacterial wilt (BW), caused by , is the second most important yield-reducing biotic factor in the country after late blight [2]. The wide adaptation of the pathogen strain and occurrence of the disease with high incidence makes the crop under risk [4]. Since BW cannot be controlled by chemicals, persists in the soil for a long time and disseminated as latent in seed tubers it is increasingly becoming a major threat to potato production. Dereje et al. [5] reported 16 to 45% tuber yield loss. In Ethiopia, BW is caused by race 3, biovar 2-A of R. solanacearum [6]. This race has a narrow host range and can be successfully controlled by integrated disease management (IDM). Bekele and Abebe [7], Berrios & Rubirigi [8], Van der Zaag [9] reported that BW free seed [10-12], crop rotation [13] disease-free soil and French et al. [14], Tusiime et al. [15] less-susceptible varieties are important components of IDM to reduce BW. Integrating the above mentioned components with sanitation in a given potato production field help to significantly to reduce and even eradicate the disease as reported in Australia [16] and Peru [17]. The successful control of BW in Shashemene and in large in the country may, however, be more difficult than we anticipate because of small and very fragmented landholdings that limit crop rotation, the major contributing factor, and because of lack of knowledge about the pathogen behavior [18] which could be improved by using the most effective extension approach, FFS, that was suggested specially to transfer knowledge intensive technologies such as integrated pest management (IPM). Ortez et al. [19] and Bekele et al. [20], demonstrated how session supported FFS has been effective in promoting late blight control strategies for reasonable number of farmers in a season [19,20].
Since bacterial wilt is also soil borne, soil amendment with organic and inorganic materials help to improve soil health and plant health. Innumerable research findings suggested that the use of farm yard manure and inorganic fertilizer (NPK) in the potato production system can reduce the incidence of the disease. Messiha [21] reported soil amendment reduce 50% in survival period of the pathogen in Egyptian and Dutch soils. According to APNAN [22] and Higa [23], fortified compost with effective micro organism (EM) improved the quality and productivity of crops. Research reports and reviews by Myint & Rana [24] indicated that application of EM found most effective as bio control agents against crop diseases. Moreover, irrespective to the temperature of the area, because of the presence of anaerobic microorganisms consisting mainly of lactic acid bacteria, purple bacteria and yeast, the presences of EM accelerates composting and take significantly shorter period compared to composting without EM. According Yamada & Xu [25], organic fertilizer inoculated with EM improved the growth of the crop and reduced the incidence of soil borne diseases. EM also has an advantage of low ecological pollution, conserves the environment, sustainable and easy for application.
Seed tubers are the major path of dissemination for BW. Tubers harvested from polluted soils are potentially infected and convey the pathogen latently and hence tubers with latent infection are taking the largest share in dissemination of the disease. According to Elphinstone & Aley [26] seed insure healthy harvests and is the measure components of integrated BW management [26]. However, disease free seeds developed in research centers have not been made use of by farmers because of lack of agencies that could multiply and supply healthy certified seeds to the farming community. Hence, all potato farmers in the country often save and plant seed from their own fields year after year or buy the smallest sized tubers and latently infested tubers from local markets practices exposed farmers to disease buildup in the field and to get low yield. Bekele & Abebe [7], Ephrem et al. [26], Habtamu et al. [27] reported that the main seed source for farmers are tubers retained from their previous harvest and/ or purchased from local market that resulted in increasing the incidence of the disease and declining tuber yield.
Since bacterial wilt is also soil borne, rotation with non-host crops to BW were suggested as a management approaches to reduce the incidence of the diseases. Several research reports indicated that crop rotation helps to significantly reduce the disease [9,16,20,28,29]; Verma & Shekhawat [10], Gunadi et al. [11]. According to Verma & Shekhawat [10] five-year rotation that included wheat, lupine and maize) and Bekele & Berga [20] three years rotation in seek plots with wheat, beans and maize reduced wilt and increased yield. Higher potato yields were also obtained under crop rotation in the presence of bacterial wilt in Kenya [30]. In the south east Ethiopia, where the activities were undertaken, a diversity of crops: pulses, cereals, vegetables, root crops and sugarcane are grown. However, these crops are sometimes rotated with potato but often potato comes successively for two or three seasons which may probably contributed to the high level of bacterial wilt incidence in the area. Therefore, the study was undertaken to increase farmer's knowledge on bacterial wilt through FFS extension approach and to generate participatory BW management technologies using one and two-season rotations with beans and vegetables. To control BW, several experiments were conducted in Shashemenewereda between 2009 and 2014. These included testing various sets of IDM as well as identifying IDM components. The purpose of the experiments reported in this paper were
a. To facilitate farmers to acquire knowledge in the processes of BW technologies development
b. To quantify the effects of EM fortified compost as an amendments
c. Seed cycle and
d. Crop rotation on BW incidence and tuber yields
Material and Methods
The studies were conducted in Shashemene District, East Arsi, on farmer's field from 2009 to 2014. The district was selected in consultation with respective Agricultural offices as they are the major potato production belt and is hot spot for the disease. Since the disease is disseminated through seed, the seed produced in this area believed to be the main source of the inoculum for the country. Shashemene is located on latitude 7° 12' North and 38° 06' East longitude having an elevation range of 1700 to 2100 meters above sea level (masl) with annual rain fall of 1200mm. It has 12 °C and 27 °C mean of an annual minimum and maximum temperature with suitable l soil for potato production. All activities reported here are executed with participation of volunteer farmers organized under FFS to develop BW control technologies in their own farm.
Soil amendments
The experiment was conducted on on-farm in shashemen district in three kabeles namely Faje sole, Hursosimbo and Idela burka using improved (CIP-386423.13) and local (Nechabeba) variety. The treatments were:
i. Improved management package (full fertilizer rate, recommended spacing and improver variety (IP);
ii. Soil amended with full rate of EM-fortified compost, recommended spacing and improver variety (IP+EM);
iii. Soil amended with % EM - fortified compost and % fertilizer of the recommended rate for the area plus % EM spray on the foliage (1/2EMCFEMS);
iv. Soil amended with % EM- fortified compost and % fertilize (1/2EMCF); 5) seed tube treated/socked in EM at a dilution of 1:1000v/vl for 30 minutes (EMTT) and
v. Farmers practice (1/2 fertilizer (1/2FC). The design was RCBD in 2x6 factorial arrangements where variety being factor A and the six treatments were considered as factor B. The data were subjected to analysis of variances using SAS software.
Seed cycle
This study was conducted for four successive seasons (2009B, 2010A, 2010B and 2011A) using a participatory approach with farmers. An improved variety, Jalene (CIP-384321.19), Guden (CIP-386423.13) and Awash (CIP-378501.3) which are less susceptible to BW and local varieties (Nechabeba and Agazer) relatively susceptible to the disease were used. The plot size was 36m2, planted in a spacing of 75cm x 30cm between rows and plants respectively. Hilling at planting, rouging of volunteer potatoes and post emergence cultivation were exercised by farmers. The experiment was laid out in a randomized complete block design, where each farmer's field (farm) was considered as a replication within the season.; the number of fields per season were 5 but, one of the experimental field was not handled properly by the group hence it was excluded.
Crop rotation
Rotation experiments were conducted between 2011 to 2014 seasons on on-farm permanent plots at Edola Burka, FagiFole and HursoSimbo, in the vicinity of Shashemene district. In the first season, preceding the experiments, the fields were planted to a BW-susceptible local potato variety “Nechabeba” in order to increase the inoculum amount in the soil. In addition during harvest diseased plants were chopped, spread uniformity and incorporated to the soil all over the field. Both one-season and two season rotation trials were conducted in a field that exhibited an initial mean BW incidence of 30.5%. Crops that are dominating the cropping system in the area were considered in the rotation trial. The one season rotation trial was carried out in year 2011, 2012 and 2013 and the two season's rotation experiment was done in 2011, 2012, 2013 and 2014 main seasons. Plots were laid out in a randomized complete block design with four replications in the plot size of 6m x 6m. The improved potato variety Guden (CIP-386423.13) was planted before and after revolving crops in 75cm x 30cm spacing between rows and plants whereas for Cabbage, and Beans were 75 x 30cm and 40 x 10cm respectively and Carrot seeds were drilled in 20cm separated furrows. For potato fertilizers was applied at planting in the form of urea and di-ammonium phosphate (DAP) at a rate of 165 kg and 295kg but for rotation crops the rate was 100kg (DAP). Late blight, the major disease of potato under main rainy season, was controlled with one spray of Ridomil MZ 63.2% WP and two sprays of Dothane M-45 80% WP starting from the first visible symptom of the disease at a rate of 3kgha-1 and 2.5kgha-1 respectively. The treatments for both trials are shown below
Treatments
(Table 1)
Result and Discussion
Soil amendment
The result exemplify statistically significant (P<0.05) differences among varieties in common scab (Streptomyces sp.) incidence. Over all disease mean infection on the improved variety and local variety was 1.9 and 3.4 in 1-5 scale respectively. This Implies that tuber seed treatment with EM had more effect on the improved variety compared to on local variety. Though the soil in the area where the trial was conducted is known to be with high wilt infection and soil inoculum in the experimental plots was up graded by incorporating BW infected plants, mean disease incidence was not surpass 23% 9 (Table 2). The incidence of the disease was not significantly differed among varieties, treatments and variety by treatment interaction. However, over all mean disease incidence was slightly higher on the improved variety (21%) compared to the local variety (2 5%) Among treatments, %2 EM-fortified compost, %2 fertilizer of the recommended rate and % EM spray had the lowest (18.8%) and (19.3%) incidence on the compared to the control treatment which had 25.5 and 33.0% respectively. The above indicated treatment reduced percent latent infection by 50% and 63% over the control in improved and local variety and improved tuber yield (Table 3). Messiha [21] reported a 50% reduction in survival period of the pathogen in Egyptian and Dutch soils. Our results agreed with APNAN 1995 and Higa [22], who reported that, EM fortified compost reduced the incidence of wilt. Research reports and reviews [24] also indicated EM as bio control agents for bacterial wilt disease.
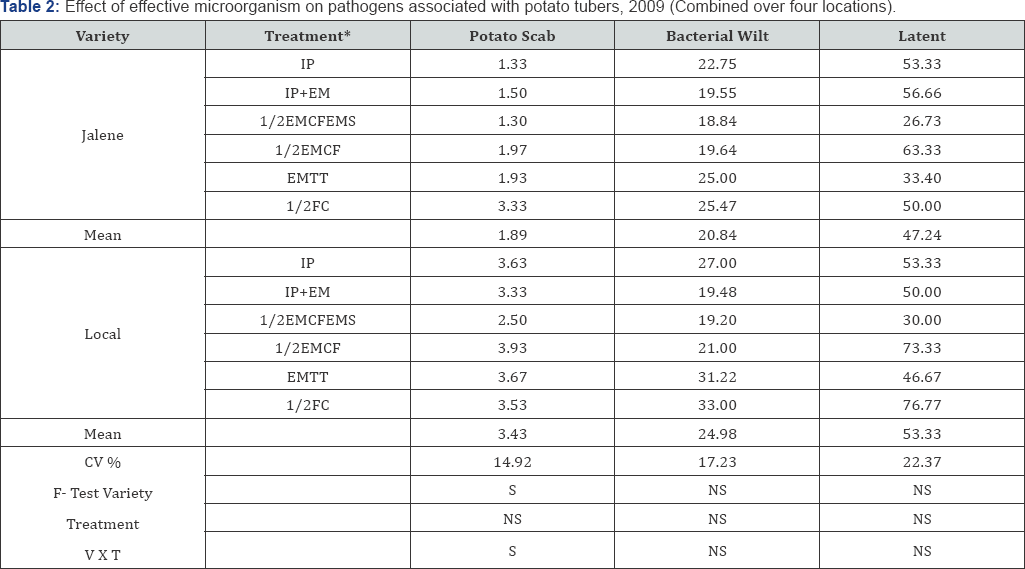
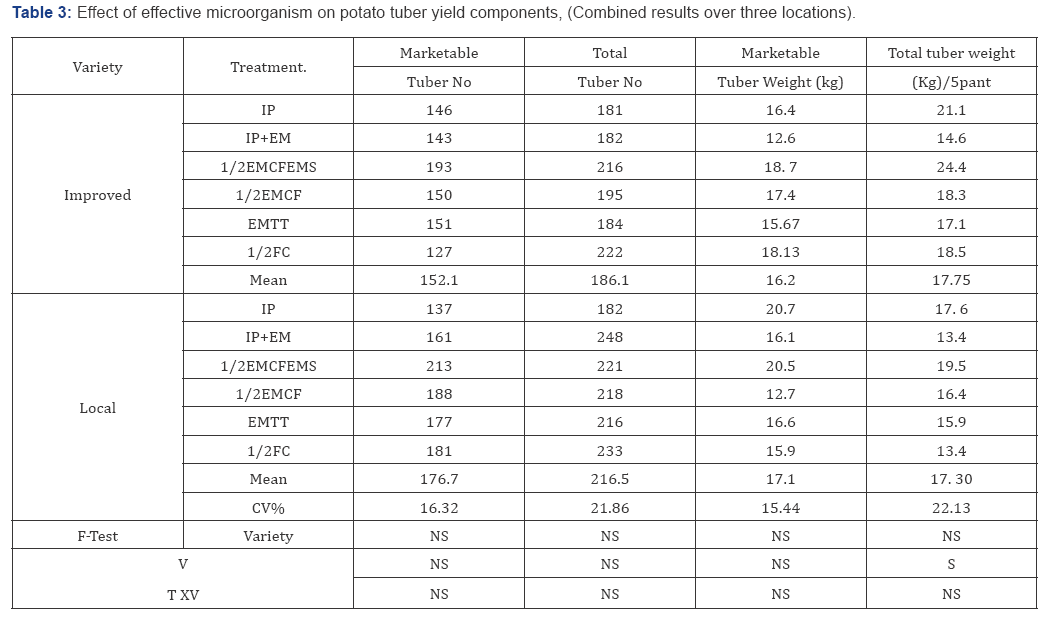
*IP=improved management package (recommended full fertilizer rate and spacing); IP+EM=soil amended with full rate of EM-fortified compost, recommended spacing; % EMCFEMS=Soil amended with % EM-fortified compost and % fertilizer of the recommended rate for the area, % EM spray and recommended spacing; % EMCF=Soil amended with % EM fortified compost; EMTT-seed tuber treated/socked in EM at a dilution of 1:1000 for 30 minutes and % FC=Farmers practice (^fertilizer).
Though differences between varieties, treatments and variety by treatment interaction in latent infection was statistically non-significant however, the incidence of the disease on variety Jalene had slightly lower (47.24%) compared to the local variety which had 53.33%. Among treatments the 4th treatment using improved variety had the lowest latent infection (26.73%) and the highest being (76.7%) on local variety treated with EM before planting. Mean percent latent infections on the improved and local varieties was in the range of 33.45 to 63.35 and 46.7 to 73.3 respectively but the rest of the treatments had in the range of 30.0% to 63.3%.
Seed cycle
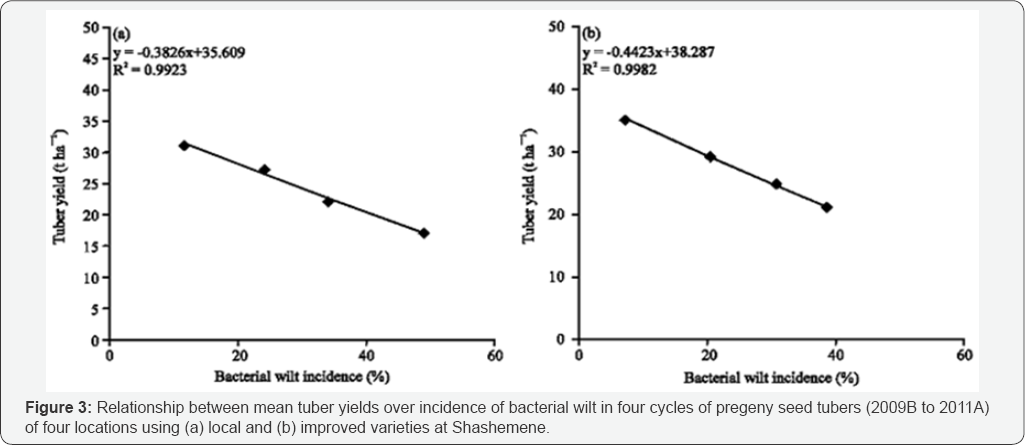
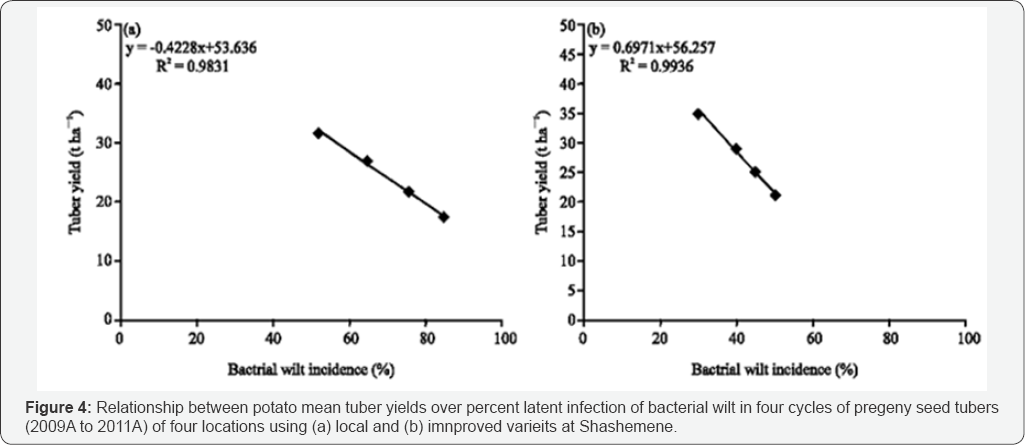
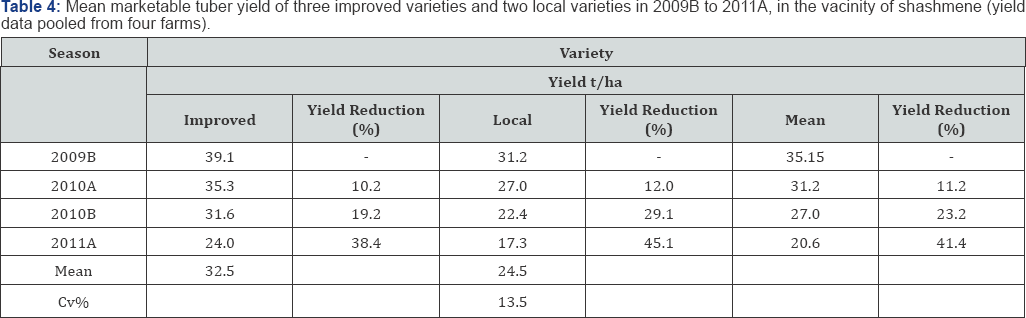
Seed tuber cycle significantly (p<0.05) affected the progress of the disease on the local relatively susceptible varieties as compared to the less susceptible improved verities. The incidence of the disease increased from 4% to 26% and 21 to 53% from the 1St cycle to the 4th cycle in local and improved variety respectively (Table ....). Hayward [31] and Moslem Khani et al. [32] reported that the pathogen is mainly transmitted through tuber seed and survived in the soil. Tubers harvested from polluted soils are potentially infected and convey the pathogen latently and hence tubers with latent infection are taking the largest share in dissemination of the pathogen. The relationship mean seed tuber over percent latent infection in tuber is presented in Figure 1 & 2. The relationship of mean tuber yield of varieties over latent infection demonstrated strong and significant (P<0.001) relationship, y=-0.4228x + 53.636 (R2=0.9831) and y=-0.6971x + 56.257 (R2=0.9936) with local and improved varieties respectively. Seed cycle of seed tubers also significantly and negatively affect marketable and total tuber yield. Mean marketable tuber yield reduced 10.2, 19.2 and 38.4% on the improved varieties in the 2nd, 3rd and 4th seed cycle while the corresponding percentage figure of mean tuber on the local variety were 12.0,29.0 and 45.0%. However, over all mean yield have been reduced by 11.0, 23.0 and 41.0% respectively. The relationship between mean tuber yield over incidence of bacterial wilt in the four season seed cycle (2009B to 2011A) had negative and strong relationship y= -0.3826x + 35.609 (R2=0.9923) and y=-0.4423x + 38.287 (R2=0.9982) over local and improved varieties. Under ware potato production, in 1st cycle, the yield loss was 4 and 7% on improved and local varieties whereas; in the 4th cycle it increased to 21 and 32% respectively (Figure 3a,3b). When potato was produced for seed the loss increased from 5 to 21% and from 29 to 67% from the 1st to the 4th cycle on improved and local variety respectively (Figure 4 (ab)) (Table 4). A huge tuber yield loss due to infected seed was reported by Hayward [31]. On the other hand, successful control of BW in Burundi [8] and Rwanda [9] have reported by suing clean seed of a less susceptible variety [14,15], and suggested its greater potential to dramatically reduce wilt. However, as reported by Bekele & Abebe [7], the current supply of clean seed is far below the demand, and as a result, most farmers use their own seed or seed of unknown origin bought from markets.
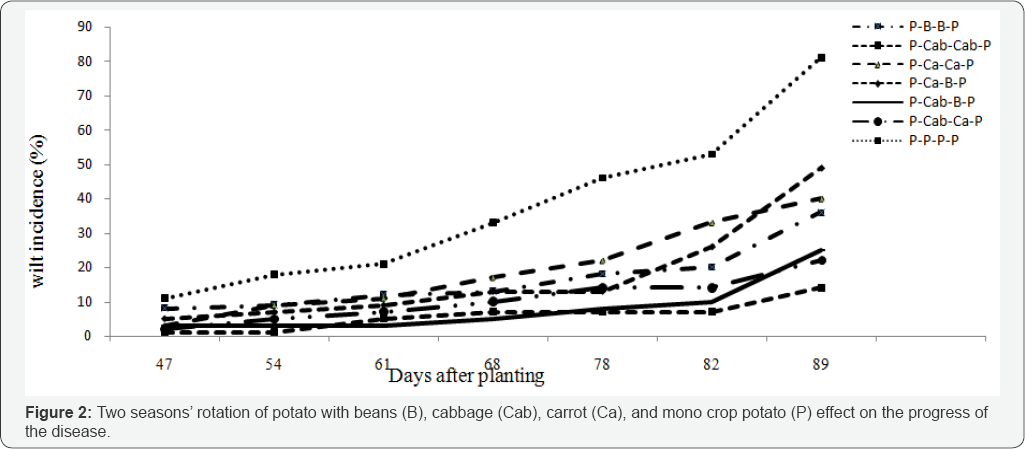
Rotation
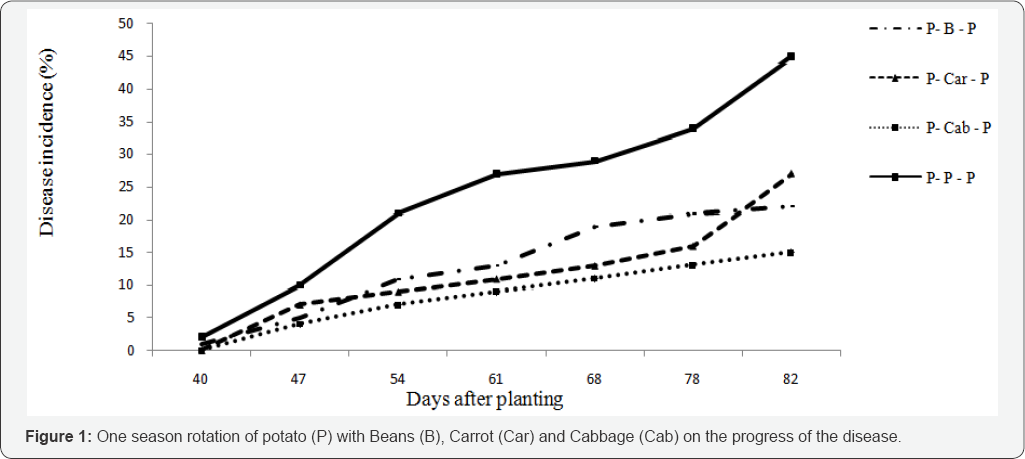
One season rotation: one season rotation had no significant effect on the onset of the disease. The symptom of the disease was recorded with in the same week in all treatments but, the disease appeared slightly earlier on the control treatment. Mean disease incidence and total and marketable tuber yield in one season rotation with beans, carrot and cabbage significantly (p<0.05) differ from the control (Table 5) this result corroborates the findings of Verma & Shekhawat [10], who reported that rotation reduce wilt incidence significantly compared to a mono crop. The highest yields were obtained following rotations with beans, which allowed the highest incidence of BW. This could be attributed to the contribution of the pulse crop to soil fertility. Similarly, the high yields after shallow rooted crops, such as carrots and cabbage could be that these crops do not exploit soil nutrients at the lower soil depths. The lowest incidence of BW occurred after rotations with cabbage (15.4%), beans (22.1%) and carrot (27.3%) compared to the control treatment (45.2%). Total and marketable potato yields were also significantly higher after these crops. However, significantly (p<0.05) highest tuber yield was obtained following rotation with beans, which allowed the highest incidence of BW. This could be attributed to the free nutrient (N) fixed from the air that improved the fertility of the soil. Similarly, the high yields after shallow rooted crops, such as cabbage and carrots, could be that these crops do not exploit soil nutrients at the lower soil depths of 15-30cm, where the potato is most active. Gunadi et al. [11] also reported that potato yields following rotations with carrots were higher than rotations with crops that have longer roots. This suggests that the nutrient exploitation of crops selected for rotation with potatoes should be considered (Table 5).
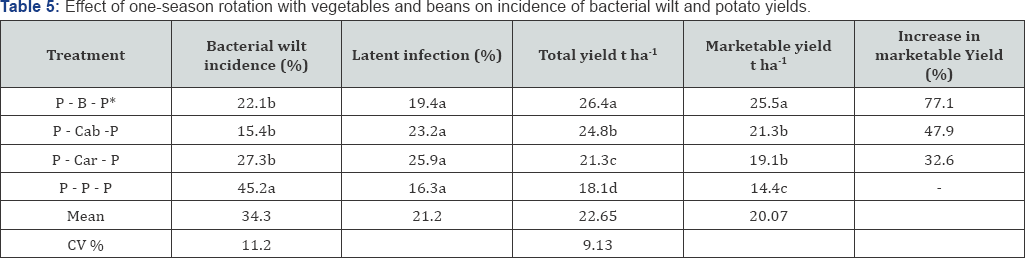
*P-B-P = Potato-Beans-potato; P-Cab-P = Potato-Cabbage-Potato; P-Car-P = Potato-Carrot-Potato and P-P-P = Potato-Potato-Potato; Means followed by the same letter in a column are not significantly different at 5 % probability level.
Cabbage in the system reduced BW incidence by 43.5% and 77.3% over rotation with beans and carrot respectively. This result was agreed with the findings of Lemga et al. [12] in which beans helped to reduce BW when used as a rotational crop. However, since the pathogen can survive in soil for years [31] one season rotation may not be sufficient to significantly suppress the development of the pathogen but still the rotation crop may evolve in the change of physical and chemical properties of the soil which determine conduciveness for the disease development as stated by Jan et al. Percent latent infection in tubers did not vary significantly among treatments. However, it was found slightly higher in rotation treatments compared to mono crop potato. This result was not surprising because, the largest proportions of infected tubers in susceptible variety are often rot with low inoculum load and discarded during harvest. It should be noted that the suppression of field symptom by the rotation crop may not warrant reducing a potential danger of latently spreading of BW. As it was reported by Berga, less susceptible potato varieties to bacterial wilt have the potential to be latently infected and promote the spread of BW if they used as a seed. However, this technology demonstrated its significant role to wear potato production systems, since latent infection do not affect the market
Two-season rotation
Days to onset of BW were not affected by the different rotation treatments. However, after onset of wilt at 47 days after planting, disease progress with the mono crop control was faster, and total incidence at 78 days after planting was the highest (81.5%), differing significantly (P<0.05) when compared to the incidences under rotations, which were all below 46.4 %. Two season rotation effect on the incidence of the disease and tuber yield is presented in Table 6. Mean BW incidence was as high as 40.3 % with the range of 14.0% to 81.1% (Table 6). The treatments have shown significant differences among each other and to the control treatment. The lowest and statically significant (p<0.05) incidence of the disease was recorded when cabbage followed cabbage or when it was considered in the rotation before beans and carrot. Whereas, carrot-beans, carrot -carrot and beans-beans had significantly highest disease incidence compared to the cabbage based rotation system but reduced the disease significantly compared to the mono crop potato.
Reduction in the incidence of bacterial wilt was greatest where cabbage-cabbage was planted in the consecutive seasons followed by two different crops like cabbage-carrot and cabbage- beans were planted in succession (Table 6). Bang and Wiles reported that in a two-season maize sweet potato rotation, wilt incidence was reduced by 32%, compared to 24% in a sweet potato-sweet potato rotation, whereas the mono crop check had a wilt incidence of 88%. In our study, unlike the previous report the progress of wilt with time, as well as total wilt incidence were lowest with cabbage-cabbage (14.0%) followed by cabbage-beans (21.9%). This showed that in two season rotation one crop spp. like cabbage can serve the purpose by planting consecutively. However, two different crops like cabbage-beans and cabbage-carrot which has shallow root planted successively in the rotation had also lowest disease incidence compared to the control. The highest tuber yield was obtained in beans- beans (29.3ha-1) followed by cabbage-beans (27.4ha-1) cabbage- cabbage (20.7ha-1) cabbage-carrot (20.7ha-1) and carrot-beans significantly (p<0.05) higher compared to the mono crop potato. This was happened probably all this rotation crops are shall rooted compared to potato which is more active in nutrient uptake at the depth of 15-30cm. Gunadi et al. [11] reported that potato yields following rotations with carrots were higher than rotations with crops that have longer roots, such as maize, and tomatoes. This suggests that the nutrient exploitation of crops selected for rotation with potatoes should be considered. Generally, vegetables and beans which have a reasonable share in the cropping system in the area are very suitable for rotation to reduce the incidence of the disease. However cabbage, which is the second most important horticultural crop in Shashemene should get priority in rotation, since it has more contribution to reduce the inoculum load of the causative agent of BW in the soil.
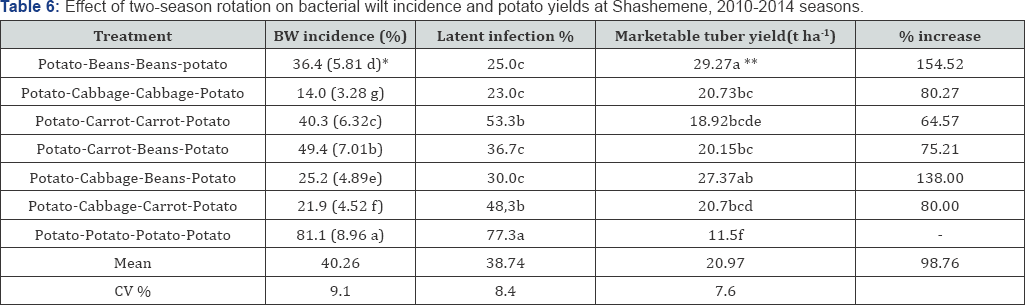
*Square root values.
**Means followed by same letters in columns are not significantly different at 5% probability level
Conclusion and recommendation
The studies reported here demonstrate that BW can be control if the technology is developed on farmers fields in participatory manner. Because BW management is largely depends and needs knowledge about the pathogen. The lesson learned from this research was that, the participated farmers have changed their views about the disease. They were able to differentiate the symptom, acquire knowledge on lifecycle and means of dissemination of the causative agent. Results from soil amendment trial, EM fortified compost accelerate the decomposition of the material used for composting and showed living organisms and substances produced or released from EM inhibited the reproduction rate of the pathogen in question as it was reflected on the incidence of the disease. However, the mode of action is not evident. Farther intensive investigations into potential beneficial uses may lead to the adaptation of the EM technology as a component in an integrated bacterial wilt management.
Research results in seed cycle study indicated that how seed could be degenerate through time due to R. solanascearum and attribute to huge tuber yield loss. It also manifested how disease free seed is important to improve potato tuber yield per unit area. The research study was also suggested that wear potato farmers, where the disease is a threat, should not retain seed from their produce for more than two cycles. Farmers in low and mid altitude areas and having similar agro-ecologies with Shashemene should use potato seeds produced in the high elevation potato fields where the weather condition is not conducive for the development of the pathogen so that the dissemination of the pathogen as latent could be reduced and at the same time can minimize the yield loss what they are exercising. The research result of this work call for an immediate need to develop and materialize seed conspiracy at national and regional level.
One and/or two seasons rotating potato with cabbage, beans and carrot reduced the disease incidence in moderately R. solanascearum infected soils. The technology should be considered as part of an integrated bacterial wilt management where the disease is a threat and cabbage, beans and carrot exists in the given cropping system. We strongly suggest that for areas with BW problem, crop rotation be considered as an important component of an integrated control option to get significant impact in pathogen reduction in the soil and concurrently to reduce the amount of latent infection in tubers which has vital role in the dissemination of the disease to new un polluted areas by the pathogen.
Acknowledgement
This study was conducted with the support of the Ethiopian Institute of Agricultural research. The author is indebted to W/ ro Tiruwerk among for data collection.
References
- Scott GJ, Rosegrant MW, Ringler C (2000) Global projections for root and tuber crops in the year 2020. Food Policy 25: 561-597.
- Bekele K, Eshetu B (2008) Potato Disease Management. Root and Tuber Crops the Untapped Resources. In: Gebremedhin W, Endale G, et al. (Eds.), Ethiopian Institte of Agricultural Research, Ethiopia, pp. 79-95.
- FAO STAT (2008) Potato world. Production consumption. International year of the Potato. USA.
- Bekele K (1996) Incidence and Distribution of Major Potato Diseases in 1993 & 1994 off-Season in Central Ethiopia 1993. In: Abstract of the 4th Annual Conference of the Crop Protection Society of Ethiopia, (CPSE) Ethiopia p. 15.
- Derje G, Gebremedhin W, Bekele K (2008) Bacterial wilt of Potato: An emerging Threat to Ethiopia Potato Industry. Regional Potato Workshop. 11-12 March 2008. Holetta Agricultural Research Center, Ehtopenia.
- Yaynu H (1989) Characteristics of isolates of Pseudomonas solanacearum in Ethiopia. Ethiopian Journal of Agricultural Science 11: 7-13.
- Bekele K, Abebe C (2013) Seed Tuber Cycle and Latent Infection for the Spread of Potato Bacterial Wilt Ralstonia solanascearum (Smith) a major threat for Seed Production in Ethiopia. Asian Journal of Plant Pathology 7(2): 74-83.
- Berrios DE, Rubirigi A (1993) Integrated control of bacterial wilt in seed production by the Burundi national Center for International Agricultural achieved in Kabale through the combined use of clean seed, crop rotation, uncon-ecology and control of bacterial wilt caused by Pseudomonas solanacearum. North Carolina State University Raleigh.
- Vander ZP (1986) Potato production under Pseudomonas solanascearum conditions; Sources and management of planting materials. In: Persely GJ (Eds.), Bacterial wit disease in Asia and South Pasfic. ACIAR Proceedings 13, Canberra, Australia, p. 84-88.
- Verma RK, Shekhawat GS (1991) Effect of crop rotation and chemical soil treatment on bacterial wilt of potato. Agricultural Science, USA p. 134-135.
- Gunadi N, Chujoy E, Kusmana M, Surviani I, Gunawan OS, et al. (1998) Effect of crop rotation patterns on Ralstoniasolanacearum population in the soil. Potato reseach in Indonesia. Results in working paper series, CIP/RIV pp. 56-61.
- Lemaga B, Kakuhenzire R, Kassa Be, Ewell PT, Prious S (2005) Integrated control of potato bacterial wilt in Eastern Africa: The experience of Highlands initiative. In: Allen C, Prior P, et al. (Eds.), Wilt Disease and the Ralstonia solanascarum Spicies Complex. APS press, St. Paul, Minnesota USA, pp. 510.
- Priou S, Gutarra L, Fernandez H, Aley P (1999) Sensitive detection of Ralstonia solanacearum in latently Research. Australia p. 284-288.
- French ER, Anguiz R, Aley P (1997) The usefulness of potato resistance to Ralstonia (Pseudomonas) solanacearum for the integrated control of bacterial wilt. In: Prior P, Allenand C, et al. (Eds.), Bacterial wilt diseases. Springer, USA pp. 381-385.
- Tusiime G, Adipala E, Opio F, Bhagsari AS (1996) Occurrence of Pseudomonas solanacearumlatent infection in potato tubers and weeds in highland Uganda. African Journal of Plant Protection 6:108-118.
- Lloyd AB (1976) Bacterial wilt in cold temperature climate of Australia. In: Planning conference and workshop on infected potato tubers and soil by post-enrichment ELISA. Impact on a changing world: CIP Program Report 1997-1998. International Potato Center, Peru, p. 111122.
- French ER (1994) Strategies for integrated control of bacterial wilt of potatoes. In: Hayward AC, Hertman GL (Eds.), Bacterial wilt: The disease and its causative agent, Pseudomonas solanacearum. CAB International, UK pp. 288.
- Bekele K (2016) Participatory Farm-level Innovation: Potato (Solanumtuberosum L.). Bacterial Wilt (Ralstonia solanacearum (E.F.Smith.) Management in the Central Highlands of Ethiopia. EJAS.
- Ortez O, Fano H, Winters P, Thiele G, UNda VJ, et al. (1999) Understanding farmers response to late blight (LB): Evidence from the base line study in Peru, Bolivia, Ecuador and Uganda. In: Hartman GL, Hayward AC (Eds.), Proceedings of the global initiative on late blight, A threat to global food security. Crissman potato program, Bacterial wilt, ACIAR Proceedings No. 45. Australian.
- Bekele K, Lemaga B (2001) Effect of preceding crop, variety and post emergence cultivation (hilling) on the incidence of bacterial wilt. Can J Microbial 29: 433-440.
- Messiha NAS (2006) Bacterial Wilt of Potato (Ralstoniasolanascearum Race 3, Biovar 2): Disease Management, Pathogen Survival and Possible Eradication. Wageningen Universiteit, Netherlands pp. 151.
- APNAN, Higa (1995) EM application manual for APNAN countries. ASIA-Pacific National Agriculture Network.
- Higa T (1994) Effective microorganisms: A new dimension for nature farming. In: Parr JF, Hornick SB, et al. (Eds.), Proceedings of the Second International Conference on Kyusei Nature Farming. US department of Agriculture, Washingeton, USA, p. 20-22.
- Myint L, Mukhaarachchi RSK (2006) Development of Biological control of Ralstoniasolanacearum through antagonistic Microbial Population. International Journal of Agriculture & Biology 8 (5): 657-660,
- Yamada K, Xu H (2001) Properties of Application of Organic Fertilizer Inoculated with Effective Microorganisms. Journal of Crop Protection 3(1): 255-268.
- Elphinstone JG, Aley P (1992) Integrated control of bacterial wilt of potato in the warm tropics of Peru. In: Hartman GL, Hayward AC (Eds.), Bacterial wilt Proceedings No 45. Korea, pp. 276-283.
- Habtamu K, Alemayehu C, Bekele K, Pananjay GBG, Tiwari k (2012) Evaluation of different potato variety and fungicide combinations for the management of potato late blight (Phytophthora infestans) in southern Ethiopia. International Journal of Life Sciences 1(2): 8-15.
- Kloos JP, Fernandez BB, Tumapon AS, Villanueva L (1991) Effects of crop rotation on incidence of potato bacterial wilt caused by Psuedomonassolanacearum E.F. Smith. Asian Potato Journal 2: 1-3.
- Kishore V, Sunaina V, Shekhawat GS (1994) Relative effectiveness of various crop rotations on incidence of potato bacterial wilt. In:Shekawat GS, Khurana SMP, et al. (Eds.), Proceedings of Potato: Present and future. Indian Potato Association. India, pp.184-187.
- Barton D, Smith J, Murimi ZK (1997) Socio-economic inputs to 'biological control of bacterial wilt diseases of potato in Kenya'. ODA RNRRS Crop protection: R66292 (NR International: ZA0085), p. 22.
- Hayward AC (1991) Biology and epidemiology of bacterial wilt caused by Pseudomonassolanascearum. Annu Rev Phytopathol 29: 65-87.
- Granada GA, Sequeira L (1983) Survival of Pseudomonas solanascearum in soil, rhizosphere and plant roots. Can J Microbiol 29: 433-440.






























