Effective Management of Major Tomato Diseases in the Gangetic Plains of Eastern India through Integrated Approach
Asit Kumar Mandal1, Praveen Kumar Maurya2, Subrata Duttal and Arup Chattopadhyay1*
1All India Coordinated Research Project on Vegetable Crops, Directorate of Research, Bidhan Chandra Krishi Viswavidyalaya, India
2Department of Vegetable Crops, Faculty of Horticulture, Bidhan Chandra Krishi Viswavidyalaya, India
Submission: August 03, 2017; Published: August 28, 2017
*Corresponding author: Arup Chattopadhyay, All India Coordinated Research Project on Vegetable Crops, Directorate of Research, Bidhan Chandra Krishi Viswavidyalaya, Kalyani-741235, West Bengal, India, Email: chattopadhyay.arup@gmail.com
How to cite this article: Asit K M, Praveen K M, Subrata D, Arup C. Effective Management of Major Tomato Diseases in the Gangetic Plains of Eastern India through Integrated Approach. Agri Res & Tech: Open Access J. 2017; 10(5): 555796. DOI: 10.19080/ARTOAJ.2017.10.555796
Abstract
Huge losses of marketable fruit yield and high infestation with different diseases has rendered open field tomato production uneconomical, causing whole fields to be abandoned prior to harvest in the Gangetic plains of eastern India. Therefore, an integrated approach involving physical, biological and chemical module has been formulated to manage diseases like early blight, late blight, target leaf spot, Sclerotium collar rot, and tomato leaf curl virus under control. Our results showed that major diseases prevalent in this zone could effectively be controlled in a substantial manner over the years through integration of management practices adopted both in nursery field [Seed priming with Seed Pro (Bacillus subtilis, IIHR strain) @4g/kg of seed followed by soil application of Seed Pro @10g/kg of soil while filling of plug trays and soil drenching of Seed Pro @5% after seed germination followed by covering of nursery bed with 50-mesh nylon net] and main field [Border row planting (2 rows) of maize at least 30 days before transplanting of seedlings in the main field followed by seedling dip with 0.1 % (Carbendazim 12%+ Mancozeb 63% WP) at the time of transplanting and sequential spraying with Acephate 75% WP @1.5g/l on 10 days after transplanting (DAT), Fipronil 5% SC @1.5ml/l on 20 DAT, Copper hydroxide 77% WP (2.0g/l) on 25 DAT, imidacloprid 70% WG @2g/15l on 40 DAT, Fenamidone 10%+Mancozeb 50% WDG (0.25%) two to three times from 45 DAT at 10 days intervals]. Such approach could also be beneficial for prolong tomato production and high economic return. This technology could easily be recommended in areas of the tropics where early autumn or autumn season cultivation of tomato is hampered by major diseases.
Keywords: Tomato; Integrated disease management; AUDPC; PDI; ICBR
Abbreviations: DAT: Days After Transplanting; ToLCV: Tomato Leaf Curl Virus; PDI: Percent Disease Index; AUDPC: Area Under The Disease Progress Curve; ICBR: Incremental Cost Benefit Ratio
Introduction
Tomato (Solanum lycopersicum L.), one of the most popular vegetable crops in the world, shares a coveted position in India as fresh vegetable and also being used as a variety of processed products such as juice, ketchup, sauce, canned fruits, puree, paste, etc. There has been a gradual increase in area under tomato cultivation in India while the productivity has been fluctuating ranging from 14.70t/ha in 1991-92 to 21.20t/ha in 2013-14 [1]. The major limiting factors towards production of optimum yield are considerable biotic stresses caused by fungi, bacteria, viruses, viroids, nematodes and insect-pests [2-5] in existing varieties and hybrids. Outdoor production of tomato is seriously impaired due to increasing infections with evolving early blight (Alternaría solani), late blight (Phytophthora infestans) populations [6] and leaf curl virus (ToLCV) diseases [7] particularly in the Gangetic plains of eastern India. ToLCV has risen to alarming proportions in the plains of India and has become a limiting factor in tomato cultivation particularly during summer crop (February to May) in southern Indian states [8,9] and autumn crop (August to December) in northern plains [10-12] and both early-autumn and autumn-winter (September to February) in Eastern India, particularly in West Bengal [13,14] causing yield loss up to 100% in favourable condition [15]. Apart from the leaf symptoms that are known as early blight, Alternaria solani can cause less economically important symptoms on tomato, including collar rot (basal stem lesions at the seedling stage), stem lesions on the adult plant, and fruit rot [16]. Yield losses up to 79% from early blight damage have been reported from Canada, India, the United States, and Nigeria [17-20]. Collar rot can cause seedling losses of 20% to 40% in the field [19]. Similarly, late blight (Phytopthora. infestans) has worldwide distribution, but most severe epidemics occur in areas with frequent cool and moist weather. This plant pathogen is one of the most devastating organisms in human history, being responsible for the devastating Irish potato famine in the 1840s, and it is arguably the most important pathogen of tomatoes causing yield loss of 91.80% [21]. Target leaf spot of tomato, caused by Corynespora cassiicola, has been reported on both greenhouse and field tomatoes in over 25 countries [22]. Epidemiologically, target leaf spot has been shown to occur at temperature above 20 °C with severe disease occurring at 32 °C [23] . Disease is also promoted by high humidity and leaf wetness durations of 16-44 hours. Over the years, it has become one of the most serious foliar and fruit diseases of tomato in Florida [24] and has risen alarming in the Gangetic plains of West Bengal, India [25]. Sclerotium rolfsii is one of the important fungal pathogens that also cause collar rot resulting substantial yield losses (30%) in tomato [26].
Farmers used to manage leaf blight diseases with spray schedules utilizing two or more different fungicide groups or fungicide formulations containing two different chemical groups at least for 8-10 times in one growing season in order to limit the development of fungicide resistant strains of A. solani which have been reported overseas [27,28]. On the other hand, management of whitefly with the use of systemic insecticides at least for 10-12 times is a common practice among the tomato growers of eastern India.
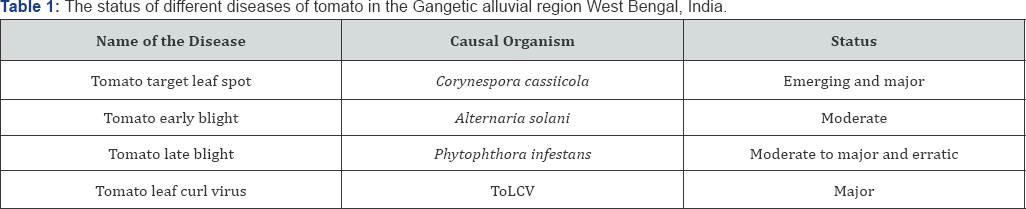
Exclusive reliance on fungicides/insecticides as a control strategy against these biotic stresses has resulted in several undesirable effects like pesticide pollution, resurgence of secondary pests, fungicide/insecticide resistance, elimination of beneficial fauna and different human health hazards. Resistance management is a key consideration for these biotic stresses in tomatoes. The most common methods of preventing resistance to fungicides include minimizing the number of applications per season of 'at-risk' products, using fungicides with diverse modes of action and applying them in alternation or as mixtures [29,31]. It has also been reported that the use of physical barrier can protect the crop against ToLCV disease [32]. It is reported that 50 mesh screens are indeed highly efficient in excluding whiteflies in nursery but these protection alone may not sufficiently protect against leaf curl disease since some whiteflies are still able to enter main field through gaps in entrances and on personnel. Alfalfa and maize can act as barrier crops in main field against the attack leaf hopper and beet curly top virus of tomato [33]. Managing the diseases through chemicals alone is not satisfactory in view of the environmental concerns and cost benefit ratio. Autumn winter tomato cultivation in the Gangetic alluvial region of West Bengal is facing a menace due to attack of several diseases which are listed in Table 1. Therefore, an attempt has been made to manage the important diseases of tomato through an integrated approach by combining barrier crop and economic use of bioagents and chemicals in the Gangetic plains of West Bengal which is regarded as the most promising tomato growing belts in India.
Materials and Methods
Experimental site and field growing
Field experiments were conducted during early autumn- winter season over three consecutive years (2014-15 to 2016-17) under All India Coordinated Research Projects on Vegetable Crops at the research farm of Bidhan Chandra Krishi Viswavidyalaya, Kalyani, West Bengal, India situated at 23.5° N latitude and 89.0° E longitude with an altitude of 9.75 m above the Mean Sea level.
Seeds of tomato hybrid (L-37, a susceptible hybrid against most of the diseases) were sown in well prepared nursery beds separately under low cost poly house covered with 200|im UV- stabilized polyethylene film. After germination six separately prepared seed beds were covered with 50-mesh nylon net and all the nursery management practices were followed in time without disturbing the insect proof net. Twenty-five days old separately treated seedlings were transplanted to the main field previously surrounded with 2 rows of maize sown 30 days before transplanting of tomato seedlings accommodating 25 plants in individual plot measuring 3mx3m during the 1st week of September each year. Plots were divided into six treatment combinations following Randomized Block Design with four replications. All the tomato plants were staked with bamboo sticks and the cultural operations as scheduled for its cultivation were followed in time [34].
Treatment combinations
Covering of nursery bed with nylon mosquito net with 50 mesh was the common in all treatments.
T1: Treatment with biological control
Nursery treatment:
a. Seed priming with Seed Pro (Bacillus subtilis, IIHR strain) @4g/kg of seed,
b. Soil application of Seed Pro @10g/kg of soil while bed preparation, and
c. Soil drenching with Seed Pro @5% after seed germination.
Main field treatment: Seedling dip with Seed Pro @5% and three sprays with Seed Pro @1.0% at 10 days interval starting from 45 days after transplanting (DAT).
T2: Treatment with fungicides
i. Nursery treatment: Seed treatment with Captan 50% WP (2g/kg)+drenching with Fosetyl Al 80% WP @0.1% immediately after germination+spray with Copper hydroxide 77% WP (2.0g/l) at 3-5 leaf stage.
ii. Main field treatment: Seedling dip with 0.1% (Carbendazim 12%+Mancozeb 63% WP)+spray with Copper hydroxide 77% WP (2.0g/l) on 25 DAT+spray with Fenamidone 10%+Mancozeb 50% WDG (0.25%) two to three times from 45 DAT at 10 days intervals.
T3: Treatment with Insecticides
Main field treatment: Spray with Acephate 75% WP @1.5g/l on 10 DAT+spray with Fipronil 5% SC @1.5ml/l on 20 DAT+spray with Imidacloprid 70% WG @2g/15l on 40 DAT.
T4: Treatment with fungicides and insecticides
i. Nursery treatment: Seed treatment with Captan 50% WP (2g/kg) + drenching with Fosetyl Al 80% WP @0.1% immediately after germination + spray with Copper hydroxide 77% WP (2.0g/l) at 3-5 leaf stage.
ii. Main field treatment: Seedling dip with 0.1% (Carbendazim 12% + Mancozeb 63% WP) + spray with Acephate 75% WP @1.5g/l on 10 DAT + spray with Fipronil 5% SC @1.5ml/l on 20 DAT + spray with Copper hydroxide 77% WP (2.0g/l) on 25 DAT + spray with imidacloprid 70% WG @2g/15l on 40 DAT + spray with Fenamidone 10% + Mancozeb 50% WDG (0.25%) two to three times from 45 DAT at 10 days intervals.
T5: Integrated management
i. Nursery treatment:
a. Seed priming with Seed Pro @4g/kg,
b. Soil application of Seed Pro @10g/kg of soil while potting, and
c. Soil drenching with Seed Pro @5% after seed germination.
ii. Main field treatment: Seedling dip with 0.1 % (Carbendazim 12%+Mancozeb 63% WP)+spray with Acephate 75% WP @1.5g/l on 10 DAT + spray with Fipronil 5% SC @1.5ml/l on 20 DAT+ spray with Copper hydroxide 77% WP (2.0g/l) on 25 DAT+spray with imidacloprid 70% WG @2g/15l on 40 DAT + spray with Fenamidone 10% + Mancozeb 50% WDG (0.25%) two to three times from 45 DAT at 10 days intervals.
T6: No spray (control)
Experimental data recording
Marketable fruits (excluding disease and insect damage fruits) of the periodical harvests from the individual plot were counted and weighed to express marketable fruit yield per plot (kg) and then it was converted to marketable fruit yield (quintal) in hectare. The number of pickings of marketable fruits was also counted in individual treatment.
The severity of different diseases (ToLCV, early blight, late blight, target leaf spot and Sclerotium collar rot) was recorded from all the individual plot by visual observation and based on different disease grading scales. Ten plants were selected for each treatment and target leaf spot and early blight disease severity was assessed from 40 days after transplanting (DAT) up to 100 DAT at 15 days intervals by using 0-5 scale [35], tomato late blight by 0-9 scale [36], and tomato leaf curl by using 0-5 scale [37]. Percent disease index (PDI) was calculated by using the following formula [38].
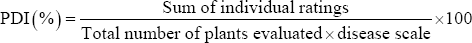
Relative spread of the disease was calculated among the different generations using the area under the disease progresscurve (AUDPC), following the standard method [39] as follows:
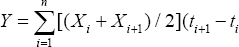
Y is the AUDPC, Xi is the disease incidence ofthe ith evaluation, and X i+1 is the disease incidence of the i + 1st evaluation,ti+1 - ti is the number of days between two evaluations.
Economic analysis
The incremental cost benefit ratio (ICBR) over the control was worked out considering the existing price of inputs, hired labour wages, interest on working capital for half life of total crop duration (5 months) @12.5% per year (as per the Commission for Agricultural Costs and Prices, Ministry of Agriculture and Farmers Welfare, Government of India), and the existing farm gate price of tomato fruits.
Statistical analysis
The data on marketable fruit yield (q/ha) collected from different treatment combinations were analyzed by the method of analysis of variance [40]. The percent data on the incidence of different diseases were transformed into their respective angular values before analysis. Data were analyzed using Statistical Analysis Systems software STPR.3
Results and Discussion
Effect of treatments on disease incidence
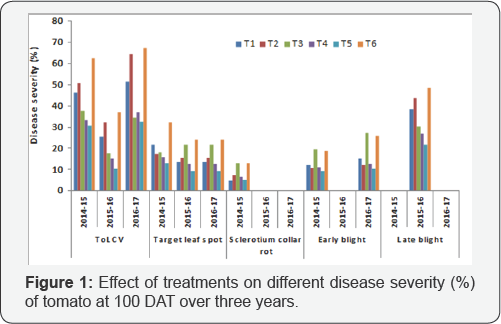
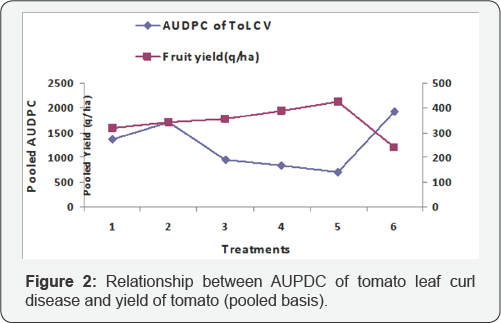
Different disease incidences like ToLCV, target leaf spot, Sclerotium collar rot, early blight, late blight were recorded at 40, 55, 70, 85 and 100 days after transplanting and fruit yield was calculated from cumulative harvest. The results of percent disease severity of different diseases at 100 days after transplanting (DAT) are presented in Table 2. In general all the treatment combinations had substantial positive effects on the reduction of percent disease incidence over control (Figure 1). Three years pooled data on the incidence of ToLCV disease are presented in Table 2. The relationship between pooled AUDPC and three years pooled yield data indicated that higher yield in T5 corresponds with lower AUDPC of ToLCV (Figure 2). The lowest disease incidence of 24.68% was recorded in plots receiving integrated management (T5) followed by the plots (T4) treated with fungicides and insecticides (28.52%). Among the management practices, the maximum reduction in ToLCV disease was found in integrated management T5(55.65%) followed by T4(48.75%) and T3(46.29%) over the control. Effective restriction in vector population at various growth stages of the crop is crucial for successful implementing viral disease control programs. Since the restricted initially at the seedling stage by covering of insect proof net followed by heating back from the barrier crop which was 90-100cm in height by this time. It has been reported that whiteflies have significantly higher wing beat frequencies than aphids, with a range from 166 to 224Hz. (i.e. times per sec.), while aphid wing beat frequencies ranged from 81 to 123 Hz [41]. This evidence clearly indicated that whiteflies are poor fliers and the maximum number of whiteflies concentrated at near ground level. This was further confirmed in a study on cotton field in Turkey that whiteflies could fly at a height of 60-80cm [42]. In this backdrop, the integrated management practices involving neonicotinoids (T5) could reduce whitefly populations substantially in tomato plots in advance of their mass migrations, if any that were thought to occur in the fall following systemic chemical protection of such tomato plots in sequential spraying. Imidacloprid is the first nicotinoid used to control whiteflies [43,44] in many crops. Our results agreed well with the observations of previous workers [45-47], who recommended neonicotinoid group of insecticides (thiomethoxam, imidacloprid and dinotefuron) to reduce whitefly populations in order to save tomato plants against leaf curl virus diseases. Nicotinoids are derived from naturally occurring nicotine compounds that block postsynaptic nicotinergic acetylcholine receptors [48,49]. They have low mammalian toxicity, minimum non target species effects and have a broad range of efficacy [50]. Nicotinoids are good at controlling phloem feeding insects because of their high water solubility and good residual activity, which makes them great systemic insecticides [51-53].
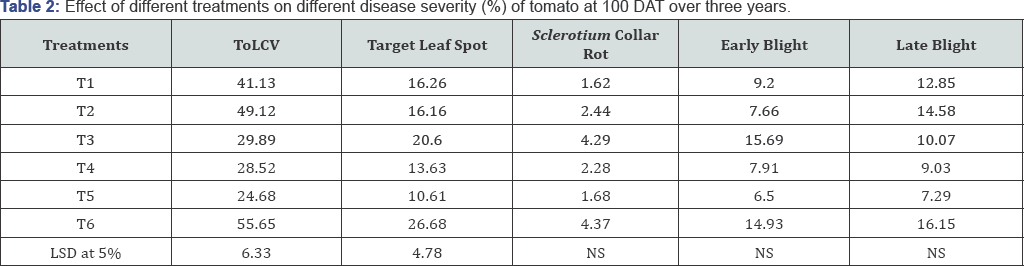
The pooled data on target leaf spot disease incidence revealed that the minimum incidence (10.61%) was also found in integrated management practices (T5) followed T4(13.63%) and T2 (16.16%). The reduction of disease incidence over control was found maximum in case of T5(60.23%) followed by T4(48.91%) and T2(39.43%). The treatment combinations involving fungicides (Fenamidone 10% + Mancozeb 50% WDG) having both systemic and contact action gave better reduction of disease over other treatment combinations. Our results agreed well with the earlier observations [22]. The application of bio-control agent, Seed Pro (Bacillus subtilis) (TJ and treatment with only insecticides (T3) did not substantially reduce target spot disease incidence with respect to control. The lack of efficacy in bio-control agent which is extremely safe to humans and the environment makes them unlikely candidate for use against target leaf spot disease in tomatoes as reported earlier [54].
The disease incidence of Sclerotium collar rot was recorded only in 2014-15 but no incidence was found in 2015-16 and 2016-17 (Figure 1). The minimum disease incidence (1.62%) and the maximum reduction (62.92%) of disease were found in treatment combinations involving bioagents (T1) followed by integrated management practices (T5). Substantial antagonistic activity of bio antagonistic against collar rot pathogen of tomato has also been observed [55]. It is reported that Bacillus subtilis controls Sclerotium rolfsii by 92% under greenhouse condition in peanut [56]. Bioagents secreted an array of chemically diverse antimicrobial secondary metabolites and hydrolytic enzymes such as proteases, cellulases, chitinases, lipases etc. which may have a possible role in enhancing the host growth and vigor, increasing antagonistic microbial activity and enabling them to resist the attack of this pathogen [57-59].
The disease incidence of early blight as influenced by different treatment combinations during 2014-15 and 201617 is presented in Figure 1. Data on disease incidence revealed that all treatment combinations reduced the disease intensity significantly as compared to control. The minimum disease incidence (6.50%) and the maximum disease reduction (56.46%) were recorded in integrated management practices (T5) followed by sole application of fungicides (T2) and combined application of fungicides and insecticides (T4). The above treatment combinations involved Mancozeb as one of the fungicides which was found to be effective for the management of early blight in tomato as reported by many workers [60-62]. Spray program based on only a protective fungicide is less effective than those that incorporated with systemic product in controlling early blight of potato [63]. Because the protective fungicide do not enter the leaf and a significant proportion of the products may be washed from the leaves by the heavy dew drops or rain fall; whereas, the systemic/translaminar fungicide is effective when applied both before and after infection, therefore timing is less critical than products with limited post-infection activity.
The disease incidence of late blight was recorded during 2015-16 due to favourable weather condition prevailing this year but no incidence was found in 2014-15 and 2016-17 (Figure 1). The minimum incidence of this disease was recorded in integrated management practices (T5) involving the fungicide combination of both systemic and contact action followed by T4 and the percent reduction of disease also followed the same trend. Earlier study also revealed that combination of fenamidone and mancozeb (Sectin) was found effective against late blight of tomato [64]. The use of sole bio-control agent was not effective against late blight of tomato as reported earlier [65].
Therefore, use of integrated approaches involving biocontrol agent application in nursery bed followed by seedling dip with fungicides and sequential spray combinations with both fungicides and insecticides could reduce the incidence of different diseases of tomato substantially in the Gangetic plains of West Bengal.
Effect of treatments on marketable fruit yield
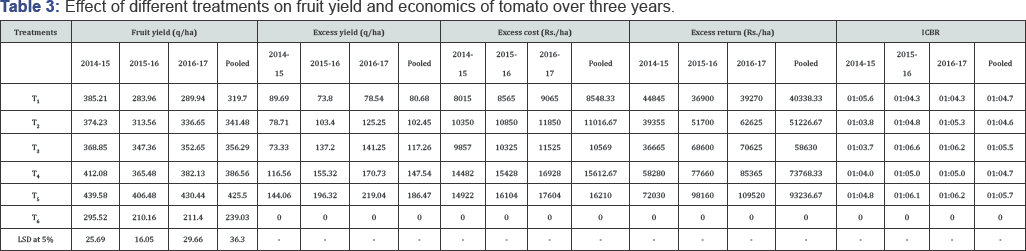
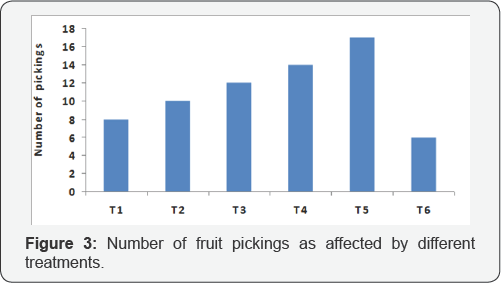
Marketable yield of each treatment was calculated after several pickings in three consecutive years and presented in Table 3. The pooled data showed that fruit yield was significantly more in all treatment combinations (319.70 to 425.50q/ha) compared to control (239.03q/ha) indicating the positive effects of different treatments on increase in yield of tomato. The maximum fruit yield (425.50q/ha) was obtained in plots receiving integrated management practices (T5) followed by T4 (386.56q/ha) and T3 (356.29q/ha). The percentage increase in marketable yield over control ranged from 33.74% (T1) to 78.01% (T5). The integrated management practices (T5) could significantly reduce the incidence of different diseases of tomato at right stages that result prolonged harvest of the crop (Figure 3), and ultimately increase the maximum tune of marketable fruit yield compared to other treatment combinations. On the other hand, susceptibility of the crop against different diseases in untreated control exhibited weak growth of the crop resulting very less number of pickings (Figure 3). Good control of tomato diseases like early blight [66], late blight [67], collar rot [68] and target leaf spot [22] with increased yield was obtained with combination products of fungicides and other integrated effects of biological factors. Our results also corroborated with earlier observations [69] who reported that the combined effect of the covering the seeding in the nursery with insect proof net and spraying with fungicide in the field resulted in significant increase in tomato yield, but the effect of the fungicide application alone gave insignificant increase in yield. On the other hand, sequential spraying of neonicotinoid group of insecticides in treatments T5 and T4 in the main field along with physical barriers in nursery and main field also reduced substantial vector (Bemisia tabaci) population which ultimately results less incidence of ToLCV disease and high marketable fruit yield. Our results find support with the observations of previous workers [45,70] who suggested prophylactic applications of imidacloprid is to be done to reduce early B. tabaci population and leaf curl virus outbreaks in tomato resulting high yield.
Economic analysis
The cost of cultivation was computed separately considering different treatment combination on the basis of existing prices of inputs, hired labour wages (Rs.225/- per man days), farm gate price of tomato fruit (Rs.5000/- per quintal), interest on working capital for half life period of total crop duration (5 months) (@12.5% per year) at the time of this study and presented in Table 3. It revealed that the cost of tomato production was highly influenced by different treatment combinations. To identify and judge the cost effectiveness of the respective treatments, incremental cost benefit ratio (ICBR) i.e. the ratio between change in return and change in cost over control treatment in absolute terms for the respective treatment combinations were computed subsequently.
From the three years pooled data it was observed that the maximum excess return (Rs.93.23667/-) was obtained with integrated management practices (T5) followed by combined treatment with fungicides and insecticides (T4) (73,768.33/-). The minimum excess return was recorded in T1 (Rs.40,338.33/- ). Similarly, the incremental cost benefit ratio was found to be the maximum in integrated management practices (T5) (1:5.71) followed by T3(1: 5.51) and the minimum ICBR was recorded in T2(1:4.61). This was happened due to high excess yield recorded in integrated management practices with minimum difference in excess cost involved as compared to other promising treatments. The comparison of ICBR among different management practices in tomato disease was also cited [71-75].
Conclusion
Therefore, it can be concluded from the present study that major tomato diseases prevalent in the Gangetic plains of eastern India, more particularly in West Bengal could effectively be reduced in a sustainable manner through integration of physical, biological and chemical management practices adopted both in nursery and main field. Farmers who find difficulty to grow tomato during early-autumn season of this zone to fetch high market price, could easily be grown the crop by adopting the technologies involving seed priming with Seed Pro @4g/ kg of seed followed by soil application of Seed Pro @10g/kg of soil while filling of plug trays and soil drenching of Seed Pro @5% after seed germination followed by covering with 50- mesh nylon net of nursery bed supplemented with border row planting (2 rows) of maize at least 30 days before transplanting of seedlings in the main field followed by seedling dip with 0. 1. (Carbendazim 12%+Mancozeb 63% WP) at the time of transplanting and sequential spraying with Acephate 75% WP @1.5g/l on 10 DAT, Fipronil 5% SC @1.5ml/l on 20 DAT, Copper hydroxide 77% WP (2.0g/l) on 25 DAT, imidacloprid 70% WG @2g/15l on 40 DAT, Fenamidone 10% + Mancozeb 50% WDG (0.25%) two to three times from 45 DAT at 10 days intervals for better management of tomato diseases, enhancement of fruit yield and assurance of high economic return. Mass adoption of this technology among the tomato growers could make a dent for increasing tomato productivity of the country as a whole.
References
- NHB (2015) Indian Horticulture Data Base. National Horticulture Board, Ministry of Agriculture and Farmers Welfare, India.
- Balanchard D (1992) A colour atlas of tomato diseases. Wolfe Publication Limited, Book House, London, p. 298.
- Gomaa AMI (2001) Pathological studies on early blight of tomato. Faculty of Agriculture, Cairo University, Egypt
- Abdel-Sayed MHF (2006) Pathological, physiological and molecular variations among isolates of Alternaría solani the causal of tomato early blight disease. Faculty of Agriculture, Cairo University, Egypt, p.181.
- Abada KA, Mostafa SH, Hillal MR (2008) Effect of some chemical salts on suppressing the infection by early blight disease of tomato. Egyptian Journal of Applied Sciences 23(20): 47-58.
- Horneburg B, Becker HC (2011) Selection for Phytophthora field resistance in the F2 generation of organic outdoor tomatoes. Euphytica 180: 357-367.
- Scholthof KBG, Adkins S, Czosnek H, Palukaitis P, Jacquot E, et al. (2011) Top 10 plant viruses in molecular plant pathology. Mol Plant Pathol 12(9): 938-954.
- Saikia AK, Muniyappa V (1989) Epidemiology and control of tomato leaf curl virus in southern India. Tropical Agriculture 66(4): 350-354.
- Sadashiva AT, Reddy M, Reddy K, Krishna M, Singh TH, et al. (2002) Breeding tomato (Lycopersicon esculentum Mill.) for combined resistance to bacterial wilt and tomato leaf curl virus. Proceedings of the International Conference on Vegetables India, pp. 125-133.
- Som MG (1973) Studies on inheritance of the resistance to leaf curl virus in tomato (Lycopersicon esculentum Mill.). Division of Vegetable Crops and Floriculture, IARI, India, p. 87.
- Mayee CD, Kanwar JS, Nandpuri KS (1974) The comparative performance of different genotypes of tomato vis-à-vis leaf curl and mosaic. Journal of Research 11: 362-64.
- Banerjee MK, Kalloo G (1987) Sources and inheritance of resistance to leaf curl virus in Lycopersicon. Theoretical and. Applied Genetics 73(5): 707-710.
- Nath S (2003) Breeding tomato (Lycopersicon esculentum Mill.) resistant to leaf curl virus. Bidhan Chandra Krishi Viswavidyalaya, India, p.107.
- Chattopadhyay A, Dutta S, Dutta P, Hazra P (2011) Studies on heterobeltiosis, combining ability and gene action in tomato (Solanum lycopersicum). International Journal of Plant Breeding 5(2): 88-93.
- Green SK, Kalloo G (1994) Leaf curl and yellowing viruses of pepper and tomato: an overview. Technical Bulletin 21, AVRDC, Taiwan.
- Walker JC (1952) Diseases of vegetable crops. Mc Graw Hill Company Inc. New York, USA, pp. 306-308.
- Basu PK (1974) Measuring early blight, its progress and influence on fruit losses in nine tomato cultivars. Canadian Plant Disease Survey 54: 45-51.
- Datar VV, Mayee CD (1981) Assessment of loss in tomato yield due to early blight. Indian Phytopathology 34: 191-195.
- Sherf AF, Macnab AA (1986) Vegetable diseases and their control. J Wiley and Sons, New York, USA, pp. 634-640.
- Gwary DM, Nahunnaro H (1998) Epiphytotics of early blight of tomatoes in Northeastern Nigeria. Crop Protection 17: 619-624.
- Byrne JM, Hausbeck MK, Latin RX (1997) Efficacy and economics of management strategies to anthracnose fruit rot in processing tomatoes in the midwest. Plant Disease 81: 1167-1172.
- Schlub RL, Smith LJ, Datnoff LE, Pernezny K (2009) An Overview of Target Spot of Tomato caused by Corynespora cassiicola. Acta Horticulturae (ISHS) 808: 25-28.
- Pernezny K, Datnoff LE, Rutherford B, Carroll A (2000) Relationship of temperature to growth, sporulation, and infection of tomato by the target spot fungus Corynespora cassiicola. 1999-2000 Report of TomatoResearch (Florida Tomato Committee).
- Pernezny K, Datnoff LE, Mueller T, Collins J (1996) Losses in fresh- market tomato production in Florida due to target spot and bacterial spot and the benefits of protectant fungicides. Plant Disease 80: 559563.
- Anonymous (2014) Annual Report, AICRP on Vegetable Crops. BCKV, Kalyani Centre, India.
- Thiribhuvanamala G, Rajeswar E, Doraiswamy S (1999) Inoculum levels Sclerotium rolfsii on the incidence of stem rot in tomato. Madras Agricultural Journal 86: 334.
- Pasche JS, Wharam CM, Gudmestad NC (2004) Shift in sensitivity of Alternaria solani in response to QI fungicides. Plant Disease 88: 181187.
- Rosenzweig N, Atallah ZK, Stevenson WR (2008) Evaluation of QI fungicide application strategies for managing fungicide resistance and potato early blight epidemics in Wisconsin. Plant Disease 92: 561-568.
- Rosenzweig N, Olaya G, Atallah ZK, Cleere S, Stanger C, et al. (2008) Monitoring and tracking changes in sensitivity to azoxystrobin fungicide in Alternaria solani in Wisconsin. Plant Disease 92: 555-560.
- Staub T (1991) Fungicide resistance: practical experience with anti-resistance strategies and the role of integrated use. Annu Rev Phytopathol 29: 421-442.
- Brent KJ, Hollomon DW (2007) 'Fungicide resistance in crop pathogensHow can it be managed? Fungicide Resistance Action Committee, Crop Life International: Brussels, Belgium.
- CropLife (2008) Fungicide Resistance Management Strategies. CropLife Australia Limited: Canberra ACT.
- Antignus Y, Nestel D, Cohen S, Lapidot M (2001) Ultraviolet-deficient greenhouse environment affects whitefly attraction and flight- behavior. Environmental Entomology 30: 394-399.
- Broadbent L (1964) Control of plant virus diseases. In: Corbett MK, Sister HD (Eds.), Plant virology. University of FIa- Press, pp. 330-336.
- Chattopadhyay A, Dutta S, Bhattacharya I, Karmakar K, Hazra P (2007) Technology for Vegetable Crop Production. All India coordinated research project on vegetable crops. Directorate of Research, West Bengal, India.
- Mayee CD, Datar VV (1986) Phytopathometry. Marathwad Agricultural University, Parabhani, p. 95.
- Malcolimson JF (1976) Assessment of field resistance to late blight (Phytophthora infestans) in potatoes. Transactions of the British Mycological Society 67: 321-325.
- Friedmann M, Lapidot M, Cohen S, Pilowsky M (1998) A novel source of resistance to tomato yellow leaf curl virus exhibiting a symptomless reaction to viral infection. Journal of the American Society for Horticultural Science 123: 1004-1007.
- Wheeler BEJ (1969) An Introduction to Plant Diseases. J Wiley and Sons Limited, Singapore.
- Shaner G, Finney RE (1977) The effect of nitrogen fertilization on the expression of slow mildewing resistance in knox wheat. Phytopathology 67: 1051-1056.
- Gomez KA, Gomez AA (1984) Statistical procedures for Agricultural Research. John Wiley Sons, Singapore, p. 63.
- Byrne DN (1987) An Examination of Whitefly Flight. College of Agriculture, University of Arizona, Vegetable Report, pp. 49-50.
- Atakan E, Canhilal R (2004) Evaluation of Yellow Sticky Traps at Various Heights for Monitoring Cotton Insect Pests. Journal of Agricultural and Urban Entomology 21(1): 15-24.
- Elbert A, Overbeck H, Iwaya H, Tsuboi S (1990) Imidacloprid, a novel systemic nitromethylene analogue insecticide for crop protection. In: Proceedings Brighton Crop Brighton, England, pp. 21-28.
- Mullins JW, Engle CE (1993) Imidacloprid (BAY NTN 33893): a novel chemistry for sweetpotato whitefly control in cotton. In: Herber DJ, Richter DA (Eds.), Proceedings, Symposium: Beltwide Cotton Conferences. National Cotton Council, Memphis, TN, pp. 719-720.
- Ahmed NE, Kanan HO, Sugimoto Y, Ma YQ, Inanaga S (2001) Effect of imidacloprid on incidence of tomato yellow leaf curl virus. Journal of Plant Diseases and Protection 85(1): 84-87.
- Cahill M, Denholm I, Byrne FJ, Devonshire AL (1996) Insecticide resistance in Bemisia tabaci -current status and implications for management. Brighton Crop Protection Conference: Pests and Diseases. British Crop Protection Council, UK, pp. 7S-80.
- Cahill M, Gorman K, Day S, Denholm J, Elbert A, et al. (1996) Baseline determination and detection of resistance to imidacloprid in Bemisia tabaci (Homoptera: Aleyrodidae). Bulletin of Entomological Research 86: 343-349.
- Cahill M, Jarvis W, Gorman K, Denholm J (1996) Resolution of baseline responses and documentation of resistance to buprofezin in Bemisia tabaci (Homoptera: Aleyrodidae). Bulletin of Entomological Research 86: 117-122.
- Polston JE, Anderson PK (1997) The emergence of whitefly-transmitted geminiviruses in tomato in the Western Hemisphere. Plant Disease 81: 1358-1369.
- Bai D, Lummis SCR, Leicht W, Breer H, Stelle DB (1991) Action of imidacloprid and a related nitromethylene on cholinergenic receptors of an identified insect motor neurone. Pesticide Science 33: 197-204.
- Liu MY, Casida JE (1993) High affinity binding of [3H] imidacloprid in the insect acetylcholine receptor. Pesticide Biochemistry and Physiology 46: 40-46.
- Woolweber D, Tietjen K (1999) Chloronicotinyl insecticides: a success of the new chemistry. In I. Yamamoto and J.E. Casdia (Eds.), Nicotinoid Insecticides and the Nicotinic Aceytlcholine Receptor. Springer, Tokyo, pp. 106-126.
- Kagabu S (1999) Studies on the synthesis and insecticidal activity of neonicotinoid compounds. Pesticide Science 21: 231-239.
- Yamada T, Takahashi H, Hatano R (1999) A novel insecticide, acetamiprid. In: Yamamoto I, Casida JE (Eds.), Nicotinoid Insecticides and the Nicotinic Acetylcholine Receptor. Springer, Tokyo, pp. 149-176.
- Maienfisch P, Huerlimann H, Rindlisbacher A, Gsell L, Dettwiler H, et al. (2001) The discovery of thiamethoxam: a second generation neonictinoid. Pesticide Science 57: 165-176.
- Pohronezny K, Wadill VH, Schuster DJ, Sonoda RM (1986) Integrated pest management for florida tomatoes. Plant Disease 70: 96-103.
- Mahato A, Biswas MK, Patra S (2017) Eco-Friendly Management of Collar Rot Disease of Tomato Caused by Sclerotium rolfsii (Sacc.). International Journal of Pure and Applied Bioscience 5(1): 513-520.
- Abd-Allah EF (2005) Effect of a Bacillus subtilis isolates a Southern blight (Sclerotium rolfsii) and lipid composition of peanut seeds. Phytopathology 33(5): 460-466.
- Srivastava R, Khalid A, Singh US, Sharma AK (2010) Evaluation of arbuscular mycorrhizal fungus, fluorescent Pseudomonas and Trichoderma harzianum formulation against Fusarium oxysporum f. sp. lycopersici for the management of tomato wilt. Biological Control 53: 24-31.
- Harman GE (2011) Multifunctional fungal plant symbionts: new tools to enhance plant growth and productivity. New Phytologist Commentry, Forum, pp. 647-649.
- Harman GE, Herrera-Estrella AH, Horwitz BA, Lorito M (2012) Special issue: Trichoderma - from Basic Biology to Biotechnology. Microbiology 158: 1-2.
- Sobolewski J, Robak J (2004) New products used for complex disease control on tomato growing in open field. Progressive Plant Protection 44: 1105-1107.
- Chourasiya PK, Lal AA, Simon S (2013) Effect of certain fungicides and botanicals against early blight of tomato caused by Alternaria solani (Ellis and Martin) under Allahabad Uttarpradesh, India conditions. International Journal of Agriculture Sciences Research 3: 151- 156.
- Sahu DK, Khare CP, Patel R (2013) Seasonal occurrence of tomato diseases and survey of early blight in major tomato growing regions of Raipur district. The Ecoscan 4: 153-157.
- Sahu DK, Khare CP, Singh HK, Thakur MP (2013) Evaluation of newer molecules for management of early blight of tomato in Chhattisgarh. The Bioscan 8: 1255-1259.
- Horsfield A, Wicks T, Davies K, Wilson D, Scott P (2010) Effect of fungicide use strategies on the control of early blight and potato yield. Australasian Plant Pathology 39(4): 368-375.
- Khadka R, Rawal BR, Marasaini M, Shrestha GK, Chapagain T, et al. (2016) Evaluation of different fungicides and clones against potato late blight (Phytophthora infestans) in western Nepal. Proceedings of the 5th International Conference on Agriculture, Environment and Biological Sciences (ICAEBS-16), pp. 71-74.
- Becktell MC, Daughtrey ML, Fry WE (2005) Epidemiology and management of petunia and tomato late blight in the greenhouse. Plant Disease 89: 1000-1008.
- Capriotti M, Marchi A, Coatti M, Manaresi M (2005) Cabriotop: the broad spectrum fungicide for the control of the main grapevine and tomato diseases. Informatore Fitopatologico 55: 38-45.
- Alexandrov V (2011) Efficacy of some fungicides against late blight of tomato. Bulgarian Journal of Agricultural Sciences 1(4): 465-469.
- Banyal DK, Mankotia V, Sugha SK (2008) Integrated Management of Tomato Collar Rot Caused by Sclerotium rolfsii. Journal of Mycology and Plant Pathology 38(2): 2008.
- Jamal MA (2012) Transfer of Integrated Pests and Disease Management of Tomato Crop to the Farmers Fields. Khartoum University, Sudan.
- Stansly PA, Conner JM (2000) Impact of insecticides on silverleaf whitefly and tomato yellow leaf curl virus (TYLCV) on staked tomato. Arthropod Management Tests 25: 173-174.
- Salim HA, Salman IS, Jasim BN (2016) IPM Approach for the Management of Wilt Disease caused by Fusarium oxysporum f. sp. lycopersici on Tomato (Lycopersicon esculentum). Journal of Experimental Biology and Agricultural Sciences, 4(VIS): 742-747.






