Activated Carbons-Promising Materials-Hydrogen Perspective
Rajalakshmi N and Ramesh T*
International Advanced Research centre for powder Metallurgy and New materials ARCI, India
Submission: May 24, 2017; Published: August 22, 2017
*Corresponding author: Ramesh T, International Advanced Research centre for powder Metallurgy and New materials ARCI, centre for fuel cell technology, IITM Research park 6, Kanagam road, Taramani, Chennai 600 113, India, Email: rajalakshmi@arci.res.in
How to cite this article: Rajalakshmi N, Ramesh T. Activated Carbons-Promising Materials-Hydrogen Perspective. Agri Res & Tech: Open Access J. 2017; 10(1): 555776. DOI: 10.19080/ARTOAJ.2017.10.555776
Introduction
Hydrogen is an important fuel in Proton Exchange Membrane Fuel Cell (PEMFC). The actual challenge for using hydrogen as an energy carrier consists in the development of a hydrogen storage system that operates at moderate temperature and pressure, minimizes weight and volume, while maintaining a reasonable cost. Reversible on-board methods such as cryogenic hydrogen storage, metal hydrides and high surface area sorbents are being explored, but are not as commercial-ready as compressed hydrogen. These methods employ systems that bind hydrogen at relatively low energies and allow for convenient refueling times of less than 5 minutes. U.S Department of Energy (DoE) has fixed the target of 6wt% hydrogen that any medium like gas, liquid, solid etc. that can store at ambient conditions for practical applications. Each technology has got its own advantages and disadvantages and none of them meet the criteria.
Activated carbon finds interest as storage medium for hydrogen in molecular form due to its high surface area and porosity. They also found to absorb by both physisorption and chemisorption methods. Activated carbon can be produced from various raw materials, that include shells of plants, the stones of fruits, woody materials, asphalt, metal carbides, carbon blacks, scrap waste deposits from sewage, polymer scraps etc., Activated carbons produced from coconut shells have been shown to have high volumes of micropores, making them the most commonly used raw material for applications where high adsorption capacity is needed. Sawdust and other woody scrap materials also contain strongly developed microporous structures which are good for adsorption from the gas phase.
Producing activated carbon from olive, plum, apricot, and peach stones yields highly homogenous adsorbents with significant hardness, Activated Carbon materials were cost effective and low density as compared to Metal hydrides, Alloys and other materials. Activated carbon with high surface area and microporosity is suitable for storage hydrogen at ambient condition. We are trying to synthesize activated carbons from agricultural wastes, which are abundant and cost effective. Choosing the correct precursor for the right application is very important because variation of precursor materials allows for controlling the carbons pore structure.

At CFCT we have synthesized many activated carbons from various raw materials like corn cob, coffee seeds, papaya seeds, lemon wastes, tamarind seeds, jute fibres, cotton fibres etc., The activated carbons need to be processed for porosity either by chemical or physical methods. Our study on carbon covers the following materials and is shown in the following Figure 1.
The various activation treatments to increase the surface area varies from acid, alkali treatment, gas phase activation etc., Each treatment has their own advantages in terms of surface groups, porosity, yield, economics etc., Inorganic acids mainly changes the surface chemistry of the carbons and alters their specific surface area and porosity. Phosphoric acid can cause a high surface area and high degree of porosity. The hydroxide groups can be increased by H2O2 treatment and If the solution pH is fixed at 2.5 or 11.5, the hydroxide groups decrease with the increase in oxygen groups like carbonyl and carboxyl. Nitric acid treatment increases the quantity of acidic surface functional containing N-O bonds. Carbons with well-developed meso- and microporous structure have been produced by ZnCl2 incorporation.
KOH activation successfully increased active carbon surface area and pore volume. Ammonium salts, borates, calcium oxide, ferric and ferrous compounds, manganese dioxide, nickel salts, hydrochloric acid, nitric acid and sulfuric acid have also been used for activation. Polymers and chelating agents have been selected to modify activated carbons. The density of positive surface charges on the carbons can be increased by cationic polymers. All the raw materials were processed to make it activated carbon and they were tailored for the following parameters as they are very important for hydrogen storage.
- Surface area
- Porosity (Volume and Pore size distribution)
- Density
- Thermal and electrical conductivity
- Surface chemistry (Oxygen groups)
- Shapes and forms (Powder, pellet, monolith) (Table 1) (Figure 2).


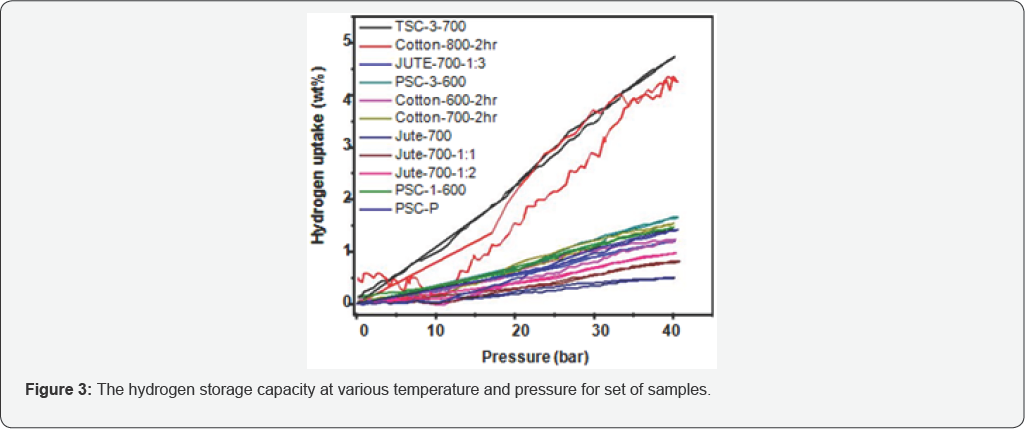
The hydrogen storage capacity is measured at various temperatures and pressures for a set of samples and the hydrogen absorption capacity at RT is shown in the following Figure 3.
Conclusion
To conclude, we have studied many activated carbons from agricultural wastes and found that a few of them are potential candidates for hydrogen storage. All these materials respond to various activation treatments, thereby enhancing the high surface area from 300m2/g to 2700m2/g. The presence of heteroatoms in the agricultural resources may be the reason for increased hydrogen absorption capacity.






























