Application of EPS in Agriculture: an Important Natural Resource for Crop Improvement
Shraddha Awasthi1*, Pratap Srivastava2 and Pradeep Kumar Mishra2
1Department of chemistry, Banaras Hindu University, India
2Department of chemical engineering and technology, Banaras Hindu University, India
Submission: June 25, 2017; Published: July 03, 2017
*Corresponding author: Shraddha Awasthi, Department of chemistry, Banaras Hindu University, India, Tel: 7839114748; Email: shraddha.iitbhu@gmail.com
How to cite this article: Shraddha A, Pratap S, Pradeep KM. Application of EPS in Agriculture: an Important Natural Resource for Crop Improvement. Agri Res & Tech: Open Access J. 2017; 8(2): 555731. DOI: 10.19080/ARTOAJ.2017.08.555731
Abstract
This unprecedented population increase would require an increased crop production, as plant sources satisfy up to 80% of humans dietary needs. The exo-polysaccharides (EPS) secreted from bacteria has shown enormous potential in the improvement of soil properties, disease suppression and plant growth. Thus, it might play a potential role in the improvement of agricultural productivity; however, it is yet highly underestimated.
Main Text
The human population, which is increasing annually by 1.4%, is expected to reach 8.3 billion by 2025. This unprecedented population increase would require an increased crop production, as plant sources satisfy up to 80% of humans dietary needs [1,2]. In this regard, microorganisms in soil can play a critical role as it plays key role in the maintenance of soil function and productivity, in both natural and managed agricultural soils. Their key involvement in important belowground processes such as soil structure formation, decomposition of organic matter, toxin removal, and the cycling of carbon, nitrogen, phosphorus, and sulphur [3] suggests its potential importance in agricultural productivity. For example, microorganisms play crucial roles in suppression of soil-borne plant pathogens, and thus help in promotion of plant growth [4]. Therefore, future exploitation of such belowground interactions for improvement in agriculture would depend on a better understanding of the biology of plant- microbe interaction [5].
The exopolysaccharides (EPS) secreted from bacteria might plays a potential role in improvement of agricultural productivity, which is yet unexplored. EPS have ubiquitous nature of alginates [6,7], which is widely known for its industrial applications [8,9]. It is used in plant tissue culture to produce artificial seeds, immobilizing enzymes by entrapment, as food and wound dressing material. EPS secreted from bacteria plays a key role in encystment of artificial seeds, which protects against desiccation and predation by the protozoon's [10], phage attack [11], and also affect the penetration of anti-microbial agents [12] and toxic metals [13]. However, its application in agriculture with respect to its role in plant growth and activity is less explored.
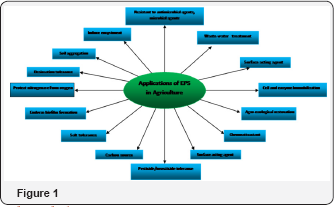
The exo-polysaccharides (EPS) secreted from bacteria has shown enormous effect on various soil properties and plant productivity. Some effects of EPS are mentioned in Figure 1. EPS possess unique water holding and cementing properties. Therefore, it play a vital role in the formation and stabilization of soil aggregates and regulation of nutrients and water flow across plant roots through biofilm formation [14,15]. Moreover, it helps to increase the uptake of nutrients by plant, and brings subsequent increase in plant's growth. Similarly, EPS protects nitrogenase against high O2 concentration, and participates in bacteria interaction with plants [16,17]. Bacterial EPS bind the Na+ ion in the root, through which the plant's Na+ accumulation decreases [18]. In that way, bacteria help to alleviate salt stress in plants. Sandhya et al. [19] reported that EPS produced by PGPR exhibit increased plant resistance to water stress. Co-inoculation of Phaseolus vulgaris L. with Rhizopus tropici and Paenibacillus polymyxa (which produces trehalose) has shown increased plant growth, N content, and nodulation under drought stress [20]. Plants treated with EPS-producing bacteria display increased resistance to water stress [21]. Alami et al. [22] observed a significant increase in root adhering soil to root tissue ratio in sunflower rhizosphere inoculated with the EPS-producing rhizobial strain YAS34 under drought conditions. Production of EPS by bacteria improved RAS permeability by increasing soil aggregation and maintaining higher water potential around the roots. Additionlly, the bacteria protects the seedlings from drought stress due to EPS secretion. Similar results were obtained with wheat plantlets, which was inoculated with Paenibacillus polymyxa Gouzou et al. [23] and Pantoea agglomerans [24] under salt stress. Hartel & Alexander [2 5] observed a significant correlation between the amount of EPS produced by cowpea, Bradyrhizobium strains and their desiccation tolerance. These finding indicate that it is possible to alleviate drought stress in the plants by increasing the population density of EPS-producing bacteria in the root zone. The EPS-producing Pseudomonas strain GAP-P45 acts as a plant growth promoting rhizobacteria and can alleviate the effect of drought stress in sunflower plants. It is attributed to improvement in soil structure and secretion of plant growth promoting substances. It indicates that the moisture sorption and colloidal stabilization properties of EPS are important, which could be potentially used for improved agro production. Moreover, studies on EPS should be considered in combination with other factors, such as bacterial spread along the root, physical properties of root adhering soil etc [26-31].
Acknowledgement
Shraddha Awasthi is highly thankful to MHRD for finantial assistance in the form of JRF and SRF.
References
- Mannion AM (1998) Future trends in agriculture: the role of agriculture. Outlook Agric 27: 219-224.
- Graham PH, Vance CP (2000) Nitrogen fixation in perspective: on overview of research and extension needs. Field Crops Res 65: 93-106.
- van Elsas JD, Trevors JY (1997) Modern soil microbiology. Marcel Dekker Inc., New York, USA.
- Doran JW, Sarrantonio M, Liebig MA (1996) Soil health and sustainability. Adv Agron 56: 2-54.
- Beringer JE (1986) Plant-microbe interactions. In: Silver S (Ed.), Biotechnology: potentials and limitations. Springer, Berlin, Germany, pp. 259-273.
- Fischer FG, Dorfel H (1955) Die Polyuronsauren Der Braunalgen (Kohlenhydrate Der Algen-I). Hoppe-Seylers Zeitschrift Fur Physiologische. Chemie 302(1-2): 186-203.
- Haug A, Larsen B, Smidsrod O (1967) Studies on sequence of uronic acid residues in alginic acid. Acta Chem Scand 21: 691-704.
- Rehm BH, Valla S (1997) Bacterial alginates: biosynthesis and applications. Appl Microbiol Biotechnol 48(3): 281-288.
- Hay SI, Okiro EA, Gething PW, Patil AP, Tatem AJ, et al. (2010) Estimating the Global Clinical Burden of Plasmodium falciparum malaria in 2007. PLoS Med 7(6): e1000290.
- Looijesteijn PJ, Trapet L, de Vries E, Abee T, Hugenholtz J (2001) Physiological function of exopolysaccharides produced by Lactococcus lactis. Int J Food Microbiol 64(1): 71-80.
- Sutherland M, Hendrik JJ, Vuuren V, Martha M (1994) Cloning, sequence and in vitro transcription/translation analysis of a 3.2-kb EcoRI- Hindlll fragment of Leuconostoc oenos bacteriophage L10, gene, 1994. Gene 148(1): 125-129.
- Costerton JW, Cheng KJ, Geesey GG, Ladd TI, Nickel JC, et al. (1987) Bacterial biofilms in nature and disease. Annu Rev Microbiol 41: 435464.
- Aleem A, Isar J, Malik A (2003) Impact of long-term application of industrial wastewater on the emergence of resistance traits in Azotobacter chroococcum isolated from rhizosphere soil. Bioresour Technol 86(1): 7-13.
- Roberson EB, Firestone MK (1992) Relationship between Desiccation and Exopolysaccharide Production in a Soil Pseudomonas sp. Appl Environ Microbiol 58(4): 1284-1291.
- Tisdall JM, Oadea JM (1982) Organic matter and water stable aggregates in soils. J Soil Sci 33: 141-163.
- Leigh JA, Coplin DL (1992) Exopolysaccharides in plant-bacterial interactions. Annu Rev Microbiol 46: 307-346.
- Mandal B, Mandal S, Csinos AS, Martinez N, Culbreath AK, et al. (2008) Biological and molecular analyses of the acibenzolar-S-methyl-induced systemic acquired resistance in flue-cured tobacco against Tomato spotted wilt virus. Phytopathology 98(2): 196-204.
- Ashraf M (2004) Photosynthetic capacity and ion accumulation in a medicinal plant henbane (Hyoscyamus niger L.) under salt stress.
- Sandhya V, Ali Sk Z, Reddy G, Venkateswarlu B (2009) Alleviation of drought stress effects in sunflower seedlings by exopolysaccharides producing Pseudomonas putida strain P45. Biology and Fertility of Soils 46(1): 17-26.
- Figueiredo MV, Burity HA, Martinez CR, Chanway C (2008) Alleviation of drought stress in the common bean Phaseolus vulgaris L. by coinoculation with Paenibacillus polymyxa and Rhizobium tropici. Appl Soil Ecol 4(2008): 182-188.
- Bensalim S, Nowak J, Asiedu SK (1998) A plant growth promoting rhizobacterium and temperature effects on performance of 18 clones of potato. American Journal of Potato Research 75(3): 145-152.
- Alami Y, Achouak W, Marol C, Heulin T (2000) Rhizosphere soil aggregation and plant growth promotion of sunflowers by exopolysaccharide producing Rhizobium sp. strain isolated from sunflower roots. Appl Environ Microbiol 66(8): 3393-3398.
- Gouzou L, Burtin G, Philippy R, Bartoli F (1993) Effect of inoculation with Bacillus polymyxa on soil aggregation in the wheat Rhizosphere: preliminary examination. Geoderma 56: 479-490.
- Amellal N, Burtin G, Bartoli F, Heulin T (1998) Colonization of wheat rhizosphere by EPS producing Pantoea agglomerans and its effect on soil aggregation. Appl Environ Microbiol 64(10): 3740-3747.
- Hartel PG, Alexander M (1986) Role of extracellular polysaccharide production and clays in the desiccation tolerance of cowpea Bradyrhizobia. Soil Sci Soc Am J 50(5): 1193-1198.
- Kerepesi I, Galiba G (2000) Osmotic and Salt Stress-Induced Alteration in Soluble Carbohydrate content in wheat seedlings. Crop Scie 40(2): 482-487.
- De Angelis PL (2012) Glycosaminogly can polysaccharide biosynthesis and production: Today and tomorrow. Appl Microbiol Biotechnol 94(2): 295-305.
- Lloret L, Barreto R, León R, Moreno S, Martínez-Salazar J, et al. (1996) Genetic analysis of the transcriptional arrangement of Azotobacter vinelandii alginate biosynthetic genes: identification of two independent promoters. Mol Microbiol 21(3): 449-457.
- Miller KJ, Wood JM (1996] Osmoadoptation by rhizosphere bacteria. Annu Rev Microbiol 50: 101-136.
- Mittler R (2006] Abiotic stress, the field environment and stress combination. Trends Plant Sci 11(1): 15-19.
- Patil SV, Patil CD, Salunke BK, Salunkhe RB, Bathe GA, et al. (2011] Studies on characterization of bioflocculant exopolysaccharide of Azotobacter indicus and its potential for wastewater treatment. Appl Biochem Biotechnol 163(4): 463-472.






























