Determination of the Appropriate Doses of Promising Botanical Powders against Maize Weevil, Sitophilus zeamais Mots (Coleoptera: Curculionidae) on Maize Grain
Tadele Shiberu* and Mulugeta Negeri
Department of Plant Sciences, Ambo University, Ethiopia
Submission: May 01, 2017; Published: May 24, 2017
*Corresponding author: Tadele Shiberu, Department of Plant Sciences, College of Agriculture and Veterinary Sciences, Ambo University, Ethiopia, Email: tshiberu@yahoo.com
How to cite this article: Tadele S ,Mulugeta N. Determination of the Appropriate Doses of Promising Botanical Powders against Maize Weevil, Sitophilus zeamais Mots (Coleoptera: Curculionidae) on Maize Grain. Agri Res & Tech: Open Access J. 2017; 6(5): 555698. DOI: 10.19080/ARTOAJ.2017.06.555698.
Abstract
The present laboratory study was carried out in the entomological laboratory the department of plant science, Ambo University. The experiment was carried out using randomized complete block design with three replications to evaluate the efficacy of four locally available botanicals. The botanicals evaluated were: Neem (Azadirachta indica) leaf and seed, hop bush (Dodonae angustifolia) fresh leaf, Pyrethrum (Chrysanthemum cinerariaefolium) flower and Lomen grass (Cymbopogon citratus) leaf powders at three different doses (3, 4 and 5gm)/100g of grain maize against maize weevil, Sitophilus zeamais Motsch. Their effectiveness was determined using different parameter such as grain damage and weight losses. The results revealed that all test materials at 5g doses exhibited mortality action against maize weevil while at 3g doses it gave the lowest percentage efficacy. Powdered leaves and seeds of neem and pyrethrum flower at 4 and 5gm were showed statistical significant (P<0.01) differences while powdered leaves of lemon grass at the similar rates were observed with moderately effectiveness and fresh leaves of hop bush gave the lowest mortality rate within 28 days of exposure in all tested doses. The result showed that the lower number of grains damaged in maize grains treated with botanical powders and the grain weight loss was also found low as compared to the untreated control. These findings suggest that botanical powders exert better mortality effect and hence reduced maize grain damage. It was also revealed that despite the high seed damage recorded in all botanical products, the S. zeamays numbers were relatively higher in untreated control while no grain seed damage was observed in standard check which was treated with Actellic dust. Maize grains treated with botanicals, indicated that insect reproduction and development were impaired in all botanical pesticides. However, all the doses tested for their insecticidal efficacy had an effect on the percentage weevil mortality and was found to be directly proportional to the amount of powder used. Therefore, The present study was suggested that Pyrethrum flower, neem leaf and seed powder can be used as good alternatives to chemical insecticides against S. zeamays due to their higher high mortality, lower grain damage and lower maize weight losses recorded as compared to untreated and synthetic insecticides.
Keywords: Botanicals; Powder; Mortality; Maize weevil; Sitophilus zeamais; Grain damage Actellic dust
Introduction
Maize is the third most important cereal crop following wheat and rice. It is also, one of the most popular crop plants all over the world, grown in over 140 million hectares [1]. In Ethiopia, Maize is one of the major staples consumed food second place in contribution which 17% of land and production, 205,472.37 ton in year of 2011/12 (CSA, 2012).
Dankyi et al. [2] reported maize is an important source of carbohydrate and forms about 90-95% of the total calories intake of the coastal Savanna people. Insect pest damage to stored grains results in major economic losses in Ethiopia where subsistence grain production supports the livelihood of majority of the population, grain loss caused by storage pests such as the maize weevil, Sitophilus zeamais (Mots.) threatens food security. Reduction of insect damage in stored grains is a serious problem in developing countries in the tropics due to favorable climatic conditions and poor storage structures [3].
Damage of maize grain begins from the field just before harvest and the insects are carried into the store where the population builds up rapidly [4]. However, during storage, maize seeds are very susceptible to attack by maize weevils among of these S. zeamais is very serious. Grain damaged by insect pests in storage often result in significant losses including loss of viability. Post harvest storage of maize is highly constrained by insect pest of maize weevil. Yield losses due to S. zeamais reported from farmers interviews, storage loss of maize due to weevil was estimated to be about 25 to 30% in the western part of Ethiopia [5].
There are however major problem to the use of chemical insecticides including human risk, cost of procurement, availability of insecticides on the market, development of resistant strains and residue in the food crops which has led scientists to investigating plant products as alternatives [6]. Moreover, there is a need to determine an appropriate dose of some botanicals to effectively control storage insect pests. Therefore, this study was carried out to determine the potential efficacy of different plant products at different doses in powder form to use in the management of S. zeamais, under storage conditions.
Materials and Methods
Rearing of maize weevils
The study was carried out at Ambo University plant science department under the laboratory of Entomology, having an altitude of 2287 meter above sea level, latitude of 08°7'0'' North and longitude of 38°7'0''East. The mean maximum and minimum temperature of the laboratory was 24.2±2°C and 16.8±2°C, respectively, during the study period.
The insects were reared on whole maize in 5 liters plastic jars after being treated for mites [7]. Whole grains of local variety bought from local market were disinfested in an oven at 600C for 1 hour [8] before using them as a substrate for insect rearing. The moisture content of maize grain was adjusted to 12-13%. After three weeks of oviposition, the parent weevils were sieved out after oviposition. Later the grain were kept in the incubator for adult emergence while the emerging generation of same age insects re-cultured at 24°C±2, 65±5% relative humidity (R.H.). The F1 generation was used for the experiment.
Collection and preparation of materials
Leaves and seeds of neem, Azadirachta indica were collected from Melka Werer Agricultural Research center, flower of Pyrethrum, Chrysanthemum cinerariaefolium was collected from Kulumsa Agricultural research center, leaves of hop bush, Dodonae angustifolia and lomen grass, Cymbopogon citrates were collected from Ambo areas.
All the plant materials were dried under shade, in a well- ventilated area in the Entomological Laboratory of Plant Science Department, before grinding into fine powders using hand machine blender and sieved using 80|im Laboratory sieve. The Actellic dust 2% insecticide was purchased from private limited chemical company. The powders were tested individually for their potential in protecting maize grains during storage.
Treatment application
The experiment was laid out in Randomized Compete Block Design (RCBD) with three replications. Twenty pairs of S. zeamais were introduced into the plastic jar containing 100g grain maize in 0.5 liter plastic jars at 3, 4 and 5g% (w/w), while in control treatment there was no any botanicals added and in the standard check Actellic 2% dust was added in 0.1g/100g of maize grain. Each botanical was weighed and added to the maize grain in each jar and shaken well for uniform coating. The jars were covered with muslin cloth and secured with rubber bands as a ventilated lid. The treated grains in the jar were kept for about 28 days and mortality rate assessments were performed regularly every 1, 7, 14, 21 and 28 days after exposure of botanical powders.
Seed germination
The effect of treatments and their interactions on seed germination and viability was examined after 28 days of grain storage period. Seed germination was tested using 50 randomly picked seeds from undamaged grains after separation of damaged and undamaged grains in each jar according to the methods described in Haines [9]. The 50 grain sub-samples were germinated on moistened filter paper (Whatman No. 1) in petri dishes arranged in a RCBD with four replicates. The experiment was maintained under laboratory conditions. The number of germinated seedlings from each petridish was counted and recorded after 7 days. The percent germination was computed according to the methods of Zibokere [10] as follows:
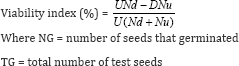
Data collection
Mortality rate were assessed for damage arising from natural insect infestations after 1, 7, 14, 21 and 28 days of treatment application. The assessment periods selected based on an earlier report by Dobie [11], Girma et al. [12]. Dead weevils were removed and counted during each assessment. Damage assessment was carried out on treated and untreated grains by taking samples of 50 grains from each jar. Each treatment was separated into undamaged and insect-damaged grains. The number of grains in each treatment was counted, weighed and the percent weight loss (percent insect damage) of maize grains in storage was computed according to the methods described in Haines (1991) as follows:
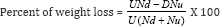
Where U = weight of undamaged grains, D = weight of insect- damaged grains,
Nu = number of undamaged grains Nd = number of insect- damaged grains.
Data analysis
The Analysis of Variance (ANOVA) was performed using Statistical Analysis Software (SAS, 2003), Data on % cumulative mortality as least significant difference (LSD) test was used to determine the differences among the means for different treatments, grain damage and yield loss parameters.
Results and Discussion
The results revealed that significant (P<0.05) differences among the treatments at all the doses tested for their insecticidal efficacy and showed an effect on the percentage weevil mortality and was found to be directly proportional to the amount of powder used. However, lower mortality was observed within one day after the exposure of weevils to botanical powders at all treatments.
A very lower amount of mortality resulted within 28 days when the weevils were exposed to the lowest dose (3g) of the botanical powders. Both leaf and seed powder of A. indica gave the efficacy of 86.33 and 83.33% at the dose of (5g) against maize weevils after 7th days of exposure, respectively. Similarly, after 14th days of application the efficacy of the neem seed was showed similar results, while neem leaves gave 100% mortality. The other botanicals indicated moderate results at the highest dose after 14th day exposure except C. cinerariaefolium was showed 92.33% mortality rate and it gave 100% mortality rate within 21 days of exposure. Whereas Cymbopogon citrates gave a moderate percent at 5g dose 62.22% mortality when compared with other botanical treatments and it was showed significantly (P<0.01) different results from that of untreated check. Dodonae angustifolia leaf powder gave less mortality rate even at 5g dose application within 28 days of exposure.
Results confirm that botanicals can be used to protect stored maize grains against S. zeamays infestations. The use of botanical products and other available materials to protect stored maize grains have been reported by other workers [13,14]. Lale [6] had advanced that oil products from the neem plant are particularly more effective against insects. Sitophilus zeamays lays eggs inside the maize grains therefore the present study revealed that the use of different botanical powders could be applied in the control of S. zeamays. In this study the live maize weevils S. zeamays showed least in maize grains that were treated with pyrethrum flower and neem leaf powder as compared with other plant materials. Marilei et al. [15], also reported that 40g of corn treated with 6g of the extracts from leaves and seeds extracts from neem can be considered as a viable alternative for controlling the S. zeamais in stored corn.
The results of the current study was confirmed the previous work of Wanyika et al. [16], that they have reported C. cinerariaefolium, affected the survival rate of the adult weevils in treatments (5.0 and 7.5g) with a mortality rate ranging from 76.66-100%. It is also supported the findings of Maribet & Aurea [17], who found that lower mortality of adult maize weevils from corn grains treated with C. citrates within 24 days after insect introduction.
Endersby & Morgan [18] observed that A. indica derivations are most effective as feeding poisons for nymphs or larvae of phytophagous insects. Hence, mortality rate of maize storage weevils, S. zeamays that caused by some botanicals as reported in the present study is most likely due to these inherent properties.
Moreover, the results indicated that all doses gave more than 30% deaths of weevils were dead after 28 days of exposure to the botanicals powder (Table 1). All tested botanicals performed well in the reduction of maize weevil during maize storage as compared to the untreated control.
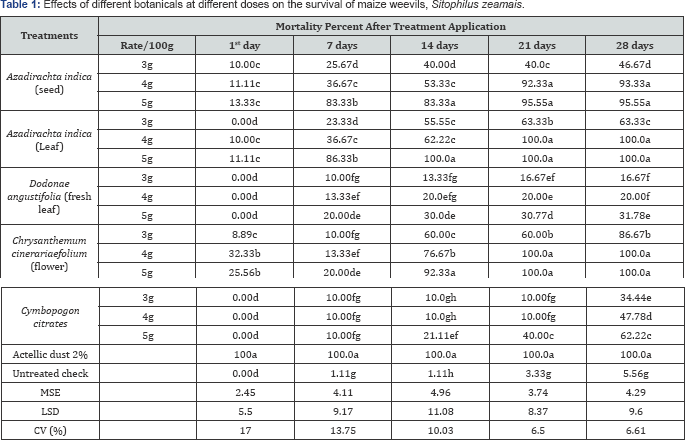
Note: Means with the same letter are not significantly different.
Grain damage and weight losses
The grain damaged and weight losses concerned from the maize grains treated with neem seed and pyrethrum flower powders, the lowest values were recorded at the dose of 5g.Similar numbers of weigh losses of maize grains were relatively recorded in all the treatments. This study suggests that neem leaf, neem seed and pyrethrum flower powder can be used as good alternatives to manage maize weevil S. zeamays from maize storage (Table 2) [19-21].
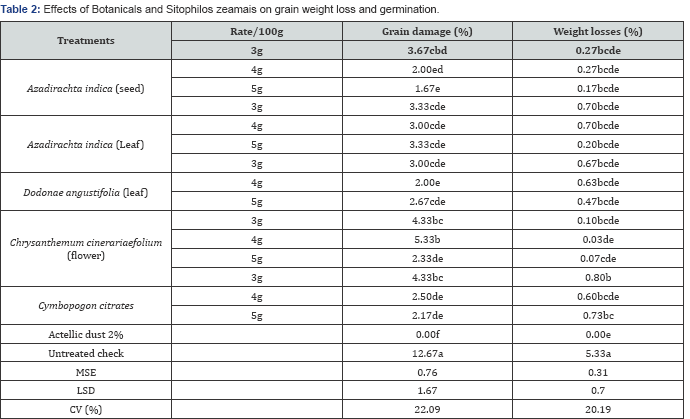
Note: Means with the same letter are not significantly different.
Conclusion
In general, this study has revealed the plant powders at 5g dosage had significant effects on mortality rate against S. zeamais within 28 days of exposure on maize grains. In particular, Azadirachta indica (seed), Azadirachta indica (leaf) and Chrysanthemum cinerariaefolium (flower) were observed at the dosage of 5g with good performance after 7 to 21 days of applications and gave of 83.33 to 100% of mortality and maize grain damage and weight losses. Finally, it is recommended that neem seed and leaf, and pyrethrum flowers were effective when used as at 4g powder/100g of maize grain against maize weevil.It is also economically important and environmentally safe.
References
- Zaidi G, Singh SKF (2005) Morphology and growth of maize-IITA/ CIMMYT Research, Guide 12, IITA. Ibadan, Nigeria, pp. 15-18.
- Dankyi AA., Anchirinah VM, Apau AO (1995) Profitability and impact of extension test plots on maize production. Newsletter, pp. 1-3.
- Bekele AJ, Obeng-Ofori D, Hassanali A (1997) Evaluation of Ocimum kenyense (Ayobangira) as source of repellents, toxicants and protectants in storage against three major stored product insect pests. J Appl Ent 121(1-5): 169-173.
- Adedire CO., Lajide L (2003) Ability of extracts often tropical plant species to protect maize against infestation by the maize weevil, Sitophilus zeamais during storage. Nigeria Journal of Experimental Biology 4(2): 175-179.
- Emana G (1998) Use of botanical plants in the control of stored maize grain insect pests in Ethiopia. In: Maize Production Technology for the Future: Challenges and Opportunities. pp. 105-108.
- Lale NES (2000) Stored Product Entomology and Acarology in tropical Africa. Molepublication, Maiduguri, Nigeria, pp. 204.
- Udo IO (2000) Efficacy of Candlewood, Zanthoxylum xanthoxyloides Lam. For the control of three stored product pests. University of Ghana, Legon, Accra, Ghana, pp. 81.
- Asmanizar DA., Idris AB (2008) Effect of selected plant extract on mortality of adult Sitophilus zeamais Motschulsky (Coleoptera: Curculionidae), pest of stored rice grains. Malaysian Applied Biology 37(2): 41-46.
- Haines CP (1991) Insects and Arachnids of tropical stored products: their biology and identification. A Training Manual. (2nd edn), Natural Resources Institute, ISTA, India, pp. 117.
- Zibokere DS (1994) Insecticidal potency of red pepper (Capsicum annuum) on pulse beetle (Callosobruchusmaculatus) infesting cowpea (Vigna unguiculata) seeds during storage 64(10): 727-728.
- Dobie P (1974) The susceptibility of different type of maize to post harvest infestation by Sitophilus zeamais and Sitotroga cerelella and the importance of this factor at the small scale farm level CIMMYT No. 10. pp. 98-113.
- Girma D, Tadele T, Abreham T (2008) Management of the maize weevil Sitophilus zeamais Motsch, (Coleoptera: Curculionidae) using Botanical insecticides on three Maize genotypes. Pest Management journal of Ethiopia 12: 49-56.
- Lale NES (1995) An overview of the use of plant products in the management of stored product coleoptera in the products. Post Harvest News Inform 6: 69N-75N.
- Keita MS, Vincent C, Schmith JP, Ramaswany S, Belanger A (2000) Effect of various essential oil on Callosobruchus maculatus (F.) (Coleoptera: Bruchidae). J Stored Prod Res 36(4): 335-364.
- Marilei NS, Alberto CV, Augusto TAP, Heron OSL (2010) Leaves, stems and seeds powder of neem (Azadirachta indica A. Juss.) used as repellent of Sitophilus zeamais (Coleoptera: Curculionidae) in stored corn. Brazilian Journal of Food Research 1(1).
- Wanyika HN, Kareru PG, Keriko JM, Gachanja AN, Kenji NJ, et al. (2009) Contact toxicity of some fixed plant oils and stabilized natural pyrethrum extracts against adult maize weevils (Sitophilus zeamais Motsch.). African Journal of Pharmacy and Pharmacology 3(2): 066069.
- Maribet LP, Aurea CR (2008) Insecticidal action of five plants against maize weevil, Sitophilus zeamais motsch. (Coleoptera: Curculionidae). KMITL Sci Tech J 8(1): 24-38.
- Endersby NM and Morgan WC (1991) Alternatives to synthetic chemical insecticides for use in crucifer crops. Biol Agri and Hort 8(1): 33-52.
- CSA (2012) Report of Federal Democratic Republic of Ethiopia, Statistical Report on Socio-Economic Characteristics of the Population in Agricultu ral Households, Land Use, Area and Production of Crops. Addis Ababa, Ethiopia.
- Gomez KA, Gomez AA (1984) Statistical procedures for Agricultural Research. (2nd edn), John Wiley and Sons, Inc., New York, USA.
- 2 SAS Institute (2003) The SAS System version 9.1 for windows. SAS Institute, Cary, NC, USA.






























