Effect of Whip Smut Disease on the Quantitative and Qualitative Parameters of Sugarcane Varieties/Lines
Sarmad Mansoor*, M Aslamkhan, Nasir Ahmed Khan and Irfan Rasool Nasir
Department of Plant Pathology, University of Agriculture, Pakistan
Submission: September 09, 2016; Published: September 19, 2016
*Corresponding author: Sarmad Mansoor, Department of Plant Pathology, University of Agriculture, Faisalabad, Muhammad Umer and Brothers Distributor Pakistan Tobacco Company, Tanusa Sharif Dist D.G Khan, Pakistan.
How to cite this article: Sarmad M, M Aslamkhan, Nasir A K, Irfan R N. Effect of Whip Smut Disease on the Quantitative and Qualitative Parameters of Sugarcane Varieties/Lines. Agri Res & Tech: Open Access J. 2016; 2(3): 555588. DOI: 10.19080/ARTOAJ.2016.01.555588
Abstract
Whip smut caused by (Ustilagoscitaminea) is an important fungal disease which is widely distributed all over the world causing huge losses in sugarcane crop. Sugarcane crop basically required humid and hot climate for its development which is also favorable for the different diseases in sugarcane. U.scitaminea the pathogen of whip smut of sugarcane causes great damage in sugarcane crop quantitatively and qualitatively. It reduces cane height, Cane girth/diameter, brix, Pol. Reading, purity and Sugar recovery. Seven promising varieties/lines were taken. (S2003-Us-618, S2008-M-34, S2008-AUS-133, S2003-US-127, S2003-US-704, S2008-Fd-19, S2008-AUS-87). Qualitative analysis showed that the U.scitaminea significantly decreases the cane height, cane girth, percentage brix, polarity, purity, and sugar recovery in all these seven varieties/lines but in case of CCS showed the non-significant results.
Keywords: Sugarcane; Whip smut disease
Abbreviations: AARI: Ayub Agriculture Research Institute; SRI: Sugarcane Research Institute; LSD: Least Significant Difference; CCS: Commercial Cane Sugar
Introduction
Sugarcane (Saccharum officinarum L.) (Punjabi: Ganna, Urdu: Naishkar, Kamad) belongs to family Poaceae and crop is grown under 30 º south to 30 º north latitude with climatic conditions ranging from sub-tropical to tropical regions [1]. In Pakistan, Sugarcane is cultivated on a range of one million hectare. Sugarcane growing zones of Pakistan fall between 24˚ N latitude in Sindh to 34˚ N latitude in KPK. As for as the Sugarcane industry of Pakistan is concerned, it is the second largest agro based industry consisting of 81 sugar mills having annual crushing capacity of over 6.1 million tons. As the production of sugar is a seasonal activity so the sugar mills on an average work for only 150 days but it provides the sugar to general public throughout the year. The most important thing is that the sugar industry shares of Pakistan economy about 1.9% of GDP. Sugar recovery is very important for the production of sugar. So sugar mills and research centers concentrate to evolve new varieties with high sugar recovery and healthy cane production [2].
Pakistan is at 5th position in area and at 11th position in production among prominent sugarcane producing countries of all over the world. Punjab contributes round about 16% of total area of sugarcane production. Domestic normal cane yield (47 tons/hectare) is far below than the current potential. In the production of the cane yield Sindh with (57 tons/hectare) is leading province followed by KPK (45 tons/hectare) and Punjab (47 tons/hectare). Sowing of sugarcane is done in September and February [3]. Sugarcane is a perennial monoculture crop which is harvested after 12-18 months, allowing systemic pathogens to propagate and increase from one ratoon to the next. Thirdly, poor practice with respect to crop rotation in South Africa and inadequate periods between replanting, after older crops are removed, enables the spread of pathogens [4]. The most destructive fungal diseases that reduce 30-70% sugarcane production include brown rust caused by Pucciniamelanocephala; whip smut caused by Ustilagoscitaminae and red rot caused by Colletotrichumfalcatum [5].
Fungi are the most prevalent pest of sugarcane crop. Over 100 fungi were found to cause diseases in sugarcane all over the world [6]. It has been reported that the yield losses vary from 70.7% to 75.3% due to different types of disease causing pathogens [7].The susceptible varieties show significant losses due to poor management practices, secondary infection and intensive cultivation. The most suitable and economical process to control the disease is the use of resistant varieties. The resistant germ plasm of sugarcane plays a leading role for assessment of resistant varieties through breeding program [8].
The objective of my present research to study the influence of whip smut disease on the quantitative and qualitative parameters of sugarcane varieties/lines.
Materials and Methods
Seven (7) varieties/ lines were grown in field area of Ayub Agriculture Research Institute (AARI), Sugarcane Research Institute (SRI) and Faisalabad during 2015-2016. Plantation of sugarcane varieties/lines was done in 15 February 2015 in three meter long plot under RCBD design with three replications / repeats at sugarcane experimental area, Sugarcane Research Institute (SRI), Faisalabad in clay loam soil. The plot size was kept as 2.4 m width and 3 m length [9].
The varieties/lines were inoculated under natural field condition and artificially. The varieties/lines were grouped separately under two categories such as inoculated and uninoculated canes. Data was recorded after the harvesting of crop. Cane height, cane girth/diameter, brix, Pol. Reading, purity, CCS and Sugar recovery was recorded from these two categories and the yield losses were compared between the un inoculated and inoculated sugarcane varieties/lines.
Data Analysis
The data collected were analyzed statistically for analysis of variance and LSD test to discriminate the superiority of the means of different varieties as suggested by [10].
Results
Seven varieties/lines were evaluated to check the response of whip smut disease on the quantitative and qualitative parameters of sugarcane varieties/lines. These varieties/lines were grouped under two categories (uninoculated and inoculated).
To determine the yield losses in term of cane height, girth/ diameter seven (7) uninoculated and seven (7) inoculated canes of all these seven varieties/lines were compared with each other by calculating the means value. Yield losses were also determine in juice quality parameters (Brix, Pol. Reading, Purity, CCS and Sugar recovery). Cane height, cane girth/diameter showed the significant results. In term of juice quality parameters brix, polarity, purity and sugar recovery showed the significant results but CCS showed the non-significant results.
Cane height and cane girth
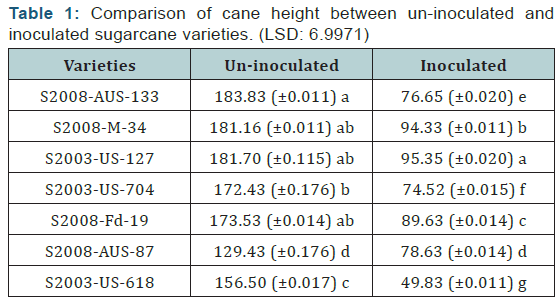
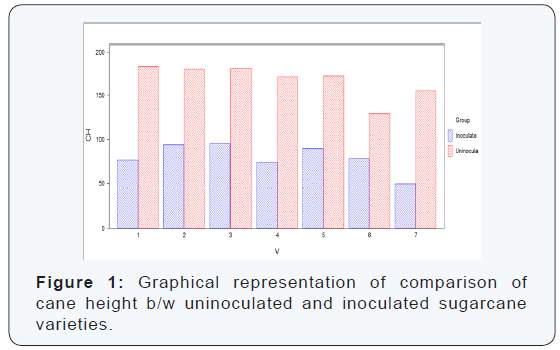
Seven varieties/ lines were grouped separately under two categories such as inoculated and uninoculated canes. From each varieties/ lines seven canes were selected. Height of each cane was measured with the help of centimeter tape. Girth or diameter of each cane was measured by vernier caliper in millimeters (mm) and then converted into centimeter (cm) by dividing it by 10. We can compare both uninoculated and inoculated sugarcane varieties. The pathogen significantly reduced the cane height clearly in the inoculated sugarcane varieties as compare to the uninoculated varieties. The difference between uninoculated and inoculated sugarcane varieties for cane height was significant.
The maximum cane height was recorded in the variety S2008- AUS-133 which was (183.83 cm) which is uninoculated variety, while the minimum cane height was recorded in the variety S2003-US-618 (49.83 cm) which was inoculated variety as shown in the (Table 1) (Figure 1).
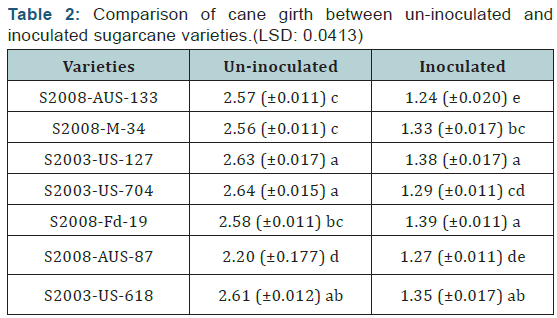
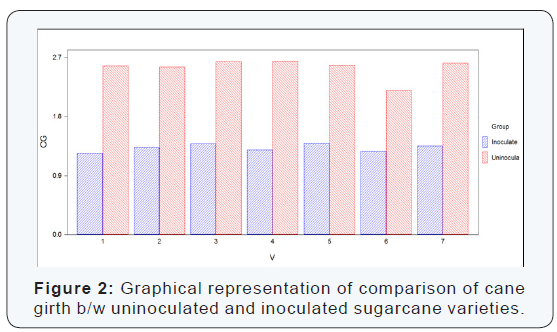
The cane girth can be seen that had been reduced considerably in case of inoculated cane. The mean values were calculated to conclude the reduction in cane girth. The reduction in cane girth was determined by comparing the mean values of cane girth of uninoculated and inoculated sugarcane varieties. The difference between uninoculated and inoculated sugarcane varieties for cane girth was significant. The maximum cane girth was recorded in the variety S2003-US-704which was uninoculated variety and girth was (2.64 cm), and the minimum cane girth was recorded in the variety S2008-AUS-133 which was inoculated variety and the girth was recorded (1.24 cm) as shown in (Table 2) (Figure 2).
Brix
Brix indicates the percent of cane sugar (sucrose) by weight (grams per 100 milliliter of water) in a solution or juice. First of all cane juice was extracted. The extracted juice was transferred to a 500ml metallic cylinder for brix determination. Brix was recorded through brix hydrometer calibrated at 24ºC. Through experimental evidence, it was noted that the brix of uninoculated are considerably greater than inoculated cane.
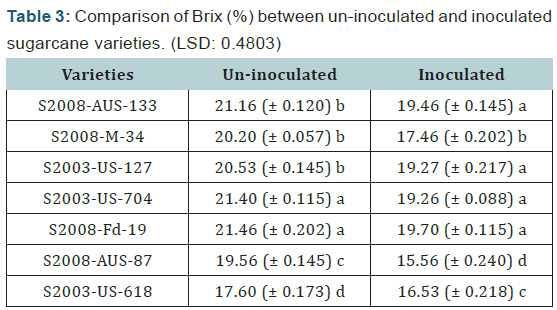
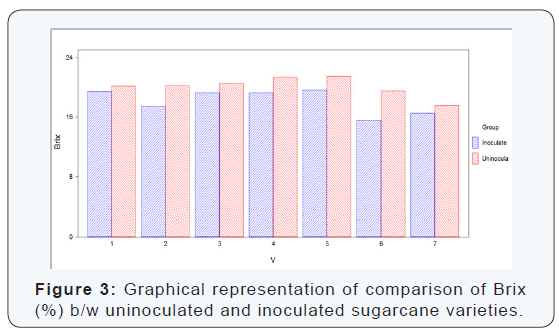
Seven varieties were selected to determine the brix percentage loss. From each variety/line seven canes were selected for extraction of juice to determine the juice quality losses. Maximum brix was recorded in variety S2008-Fd-19 (uninoculated) and minimum brix was observed in S2008- AUS-87 (inoculated) as shown below in (Table 3) (Figure 3). The difference between uninoculated and inoculated sugarcane varieties for brix (%) was significant.
Polarity reading
Polarity reading is the measurement of the angle of rotation of dextrorotatory substances. Polarity works on the principle that under certain standard conditions i.e. standard tube length, standard solution concentration and standard room temperature when a polarized light is passed through that sugar solution, then substances present rotate light at a definite angle. The measurement of that angle of rotation is polarity reading. From each variety/line seven canes were selected for extraction of juice to determine the juice quality losses. 4-5 g of dry lead sub acetate was mixed into 100ml of extracted juice. The juice was filtered into a volumetric flask through a filter paper and injected into 200mm Polari-meter tube to record the polarity reading.
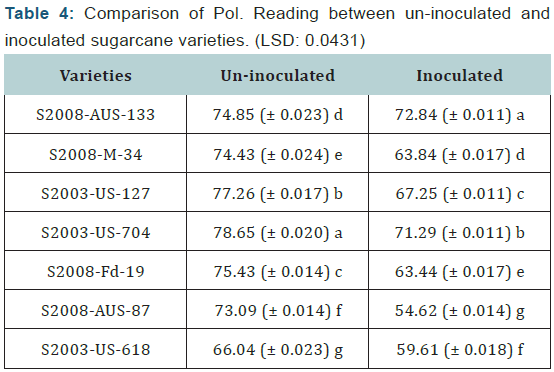
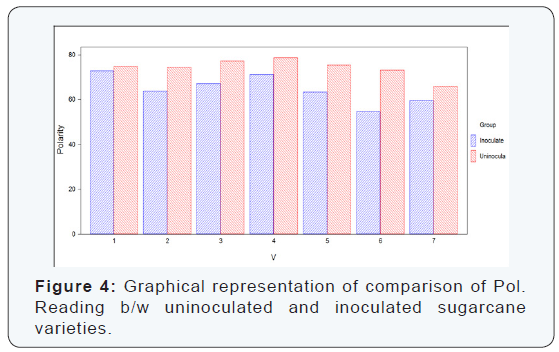
Statistical values of polarity reading that were taken by polarimeter in seven sugarcane varieties which were divided in two groups (uninoculated and inoculated). It is concluded from the calculation of polarity reading of the both uninoculated and inoculated sugarcane varieties that the maximum polarity reading was recorded in uninoculated varieties as compared to inoculated sugarcane varieties. The maximum polarity reading was recorded in the variety S2003-US-704 (78.65%) which was uninoculated variety, while the minimum polarity reading was recorded in the variety S2008-AUS-87 (54.62%) which was inoculated variety. The difference between uninoculated and inoculated sugarcane varieties for polarity reading was significant as shown (Table 4) (Figure 4).
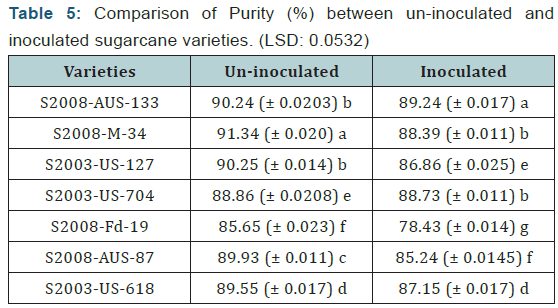
Purity
Purity is the percentage of Sucrose in the total solids in a sample. It was calculated by the following formula:
Apparent purity (%) = Polarity × 100/Brix
Purity for both uninoculated and inoculated canes was recorded. Following table shows the recorded purity of seven sugarcane varieties of healthy and diseased sample.
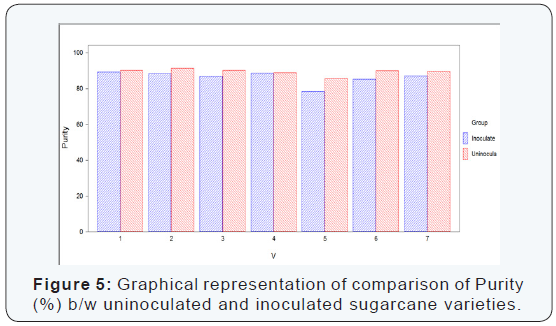
Numerical values of purity (%) that were calculated by the above given formula in seven sugarcane varieties which were divided in two groups (uninoculated and inoculated). It is concluded from the calculation of purity (%) of the both uninoculated and inoculated sugarcane varieties that the maximum purity (%) was recorded in uninoculated varieties as compared to inoculated sugarcane varieties. From each variety/line seven canes were selected for extraction of juice to determine the juice quality losses.
The maximum purity (%) was recorded in the variety S2008-M-34 (91.34%) which was uninoculated variety while the minimum purity (%) was recorded in the variety S2008-Fd-19 (78.43%) which was inoculated variety. The difference between uninoculated and inoculated sugarcane varieties for polarity reading was significant as shown (Table 5) (Figure 5).
Commercial Cane Sugar (CCS)
That percentage by weight of a quantity of cane which would be recovered as pure sucrose is known as C.C.S.
It was calculated by the Australian formula which is also known as Queen Land Formula:
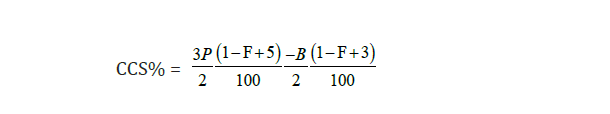
*P = Pol. Percentage Reading, *B = Brix Percentage and *F = Fiber Percentage
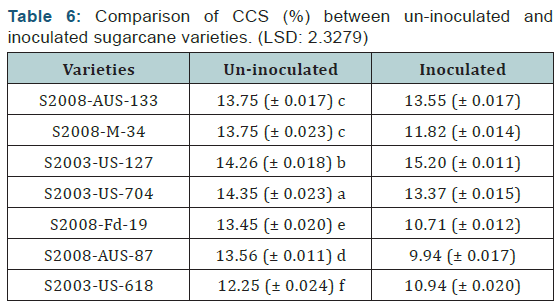
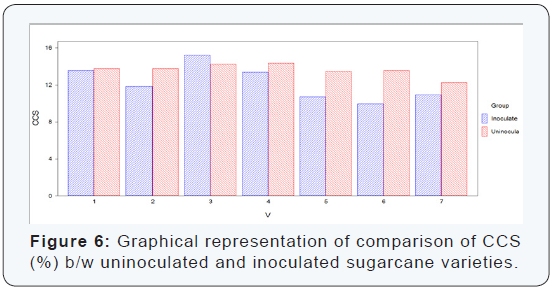
Statistical values of CCS (%) that were calculated by formula in seven sugarcane varieties which were divided in two groups (uninoculated and inoculated). The maximum CCS (%) was recorded in the inoculated variety S2003-US-127 (15.20%) while the minimum CCS (%) was recorded in the variety S2008- AUS-87 (9.94%) which was uninoculated variety. It is concluded from the calculation of CCS (%) of the both uninoculated and inoculated sugarcane varieties that the maximum CCS (%) was recorded in inoculated variety as compared to uninoculated sugarcane variety. The difference between uninoculated and inoculated sugarcane varieties for CCS (%) was non-significant as shown (Table 6) (Figure 6).
Sugar recovery
It is calculated by multiplying CCS with a constant factor0.94. Actually to convert brown sugar into white sugar; 6% white sugar is also lost so a constant factor is used.
It can be denoted by a formula:
Sugar Recovery (%) = C.C.S × 0.94
A comparison of sugar recovery between different uninoculated and inoculated varieties is shown in the table.
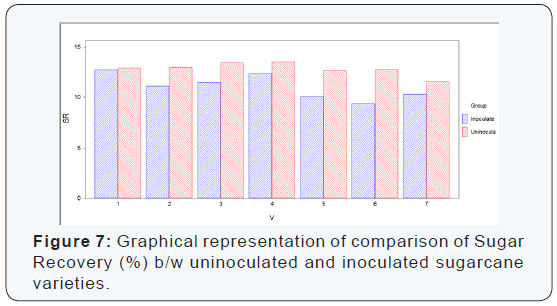
Numerical values of sugar recovery (%) that were calculated by formula, in seven sugarcane varieties which were divided in two groups (uninoculated and inoculated). It is concluded from the calculation of sugar recovery (%) of the both uninoculated and inoculated sugarcane varieties that the maximum sugar recovery (%) was recorded in uninoculated varieties as compared to inoculated sugarcane varieties. The maximum sugar recovery (%) was recorded in the variety S2003-US-704 (13.52%) which was uninoculated variety while the minimum sugar recovery (%) was recorded in the variety S2008-AUS-87 (9.35%) which was inoculated variety.
The difference between uninoculated and inoculated sugarcane varieties for sugar recovery was significant as shown (Table 7) (Figure 7). Significant and non-significant difference in parameters of juice quality was recorded. CCS was showed non-significant results between uninoculated and inoculated sugarcane varieties while brix, polarity, purity and sugar recovery showed significant results.
Discussion
The average reduction in case of cane height and cane girth was recorded as compared to uninoculated and inoculated sugarcane varieties. The maximum cane height was recorded in variety (S2008-AUS-133) which was uninoculated variety and the minimum height was recorded in the variety (S2003-US-618) which was inoculated variety. And the maximum cane girth was recorded in the variety (S2003-US-704) which was uninoculated variety and minimum girth was recorded in (S2008-AUS-133) which was inoculated variety.
The maximum reduction was recorded in cane height and cane girth was in the inoculated sugarcane varieties as compared to uninoculated sugarcane varieties. Various investigators had also been reported that the whip smut fungus has the potential to infect the thickness (girth) and height of the cane. However it was concluded that there was 69.2% cane losses in terms of height and girth when compared to uninoculated sugarcane varieties [11]. Our investigations also showed the same results that whip smut of sugarcane decreased the height and cane girth in different varieties significantly.
Furthermore, there was clear difference between uninoculated and inoculated sugarcane varieties with respect to brix, polarity, purity, CCS and sugar recovery. Brix of cane juice is believed to be the basic criteria to determine the juice quality greater value of brix indicates the high quality sugarcane juice. The standard value to brix is above 20 [12]. Whip smut of sugarcane also reduces the brix value and causes great damage to sugarcane crop. The average losses that were observed in case of brix were 10.36 %. Individually, the variety S2008-Fd-19 was infected considerably regarding brix %. Uninoculated sample of this variety showed the value of 21.46% brix whereas the inoculated sample of this variety give only 15.56% brix. This reduction is due to the infection by pathogen [13].
Polarity accounts the major role in juice quality. High amount of polarity shows good quality sugarcane juice. The standard value of polarity percentage range between 10.49-17.86% [14]. The pathogen Ustilagoscitaminea decreases the amount of polarity of the sugarcane juice. During the study, average reduction in Polarity of the sugarcane juice was observed up to 12.74% as compared to uninoculated sugarcane varieties. Viswanathan and Rao [15] reported the loss in polarity 7.4-38.7 %. Our experiment also showed the loss in polarity in this range.
In case of purity, standard value of purity is 80% or above [16]. Losses were greater in all inoculated sugarcane varieties. It means that the pathogen while invading the sugarcane plant results in the decrease in the purity of the cane juice. The average yield losses in all diseased canes were recorded 16.42% as compared to the uninoculated sugarcane varieties where as Viswanathan and Rao [15] Reported the loss in purity (%) from 0.5-8.3 % which are far less from our findings but it is confirmed that whip smut pathogen reduces the purity percentage considerably.
Standard value of CCS is 10-15% [17]. Yield losses were also observed in case of Commercial Cane Sugar (CCS) were 10.35%. Jaroenthai [13] reported the losses in CCS up to 7-13 % which is less as compared to the varieties/lines we tested. It is confirmed that losses in cane height, girth, brix, polarity, purity, CCS and sugar recovery are due to whip smut of sugarcane but different levels of losses reveals that there is difference in disease incidence in sugarcane clones [18].
Conclusion
U.scitaminea the pathogen of whip smut of sugarcane causes great damage in sugarcane crop quantitatively and qualitatively. Qualitative analysis revealed that the whip smut disease of sugarcane greatly reduced cane height and cane girth (diameter), brix, polarity, purity, CCS and sugar recovery in all these seven promising varieties/lines (S2003-Us-618, S2008-M-34, S2008-AUS-133, S2003-US-127, S2003-US-704, S2008-Fd-19, S2008-AUS-87). Qualitative analysis showed that the U.scitaminea significantly decreases the cane height, cane girth, percentage brix, polarity, purity, and sugar recovery in all these seven varieties/lines but in case of CCS showed the nonsignificant results.
Acknowledgment
This research is collaboration between the University of Agriculture Faisalabad, Pakistan and Ayyub Agriculture Research Institute (AARI) Faisalabad, Pakistan. The author gratefully acknowledged the support of his supervisor Prof. Dr. Muhammad Aslam Khan and Co- supervisor Dr. Muhammad Abdul Shakoor for their innovative ideas to complete this research.
References
- Rao, Ramana TC, Bhagyalakshmi KV, Rao JT (1979) Indian atlas of sugarcane. Sugarcane Breeding Institute, Coimbatore, India.
- Shaukat MI (2009) A comprehensive studies on sugarcane. University of Agriculture, Faisalabad, Pakistan.
- Chattha AA, Afzal M, Iqbal MA, Ahmad F, Chattha MU (2004) CPF-243, an early maturing, high yielding and high sugar variety 6: 25-27.
- Bailey RA (1995) Coping with disease problems. Proceedings of the South African Sugar Technologists‟ Association 69: 215-216.
- Khan HMWA, Chattha AA, Munir M, Zia A (2009) Evaluation of resistance in sugarcane promising lines against whip smut. Pak J Phytopathol 21(1): 92-93.
- Subhani MN, Chaudhry MA, Khaliq A, Muhammad F (2008) Efficacy of various fungicides against sugarcane red root (Colletotrichumfalcatum). Int J Agri Biol 10 (6): 725-727.
- Sandhu SA, Bhatti DS, Rattan BK (1969) Extent of losses caused by smut (Ustilagoscitaminea Syd.). Jour Res (PAU.) 6: 341-344.
- Begum F, Talukdar MI, Iqbal M (2007) Performance of various promising lines for resistance to sugarcane smut (Ustilago scitaminea Sydow). Pak Sugar J 22: 16-18.
- Bock KR (1964) Studies on sugarcane smut (Ustilagoscitaminea) in Kenya.Trans Brit mycol SOC 3: 403- 417.
- Gomez KA and Gomez AA (1984) Statistics for Agriculture Research (2nd edn.), John Willing and Sons, New York.
- Kirtikar, Verma HS (1962) A review on effect of sugarcane diseases on yield and juice qualities in Uttar Pradesh. Indian Sugar 12: 103-108.
- Mao LC, Xu YQ, Que F (2007) Maintaining the quality of sugarcane juice with blanching and ascorbic acid. Food Chemistry 104(2): 740-745.
- Jaroenthai K, Dongchan S, Anusonpornpurm S, Pliansinchai U (2007) Occurrence of Sugarcane Diseases in the Germplasm Collection at Mitr Phol Sugarcane Research Centre at Chaiyaphum, Thailand. Proc. Int. Soc. Sugar Cane Technol 26: 1040-1045.
- Blackburn FHB (1984) Sugarcane. Longman Inc. New York, USA, pp. 30-42.
- Viswanathan R, Rao GP (2011) Disease scenario and management of major sugarcane diseases in India. Sugar technol 13(4): 336-53.
- Clements FH (1980) Sugarcane crop logging and crop control, principles and practices.
- Robertson MJ, Muchow RC, Wood AW, Campbell JA (1996) Accumulation of reducing sugars by sugarcane: Effects of crop age, nitrogen and cultivar. Field Crops Res 49(1): 39-50.
- The University Press of Hawaii, Honolulu, USA, pp. 360-395.






























