Examination of Percentage Aluminium Saturattion As a Criterion for Liming Tropical Acid Soils of Nigeria
ME Ikiriko1, JAI Omueti2* and EY Thomas2
1Department of Crop and Soil Science, University of Port Harcourt, Nigeria
2Department of Agronomy, University of Ibadan, Nigeria
Submission: June 13, 2016; Published: June 23, 2016
*Corresponding author: JAI Omueti, Department of Agronomy, University of Ibadan, Oyo State, Nigeria, Email:omueti70@yahoo.com
How to cite this article: ME Ikiriko, JAI O, EY Thomas. Examination of Percentage Aluminium Saturattion As a Criterion for Liming Tropical Acid Soils of Nigeria. Agri Res & Tech: Open Access J. 2016; 1(5): 555574. DOI: 10.19080/ARTOAJ.2016.01.555574
Abstract
Percentage aluminium saturation as a criterion for liming acid soils in Nigeria was examined. Surface soil (0-15 cm) samples with extreme acidity (3.8-4.9), formed on coastal plain sands (Obio-Akpor and Egor) and sandstone (Nsukka), as part of the soils occurring over the sedimentary rocks in the humid region of southern Nigeria were collected to grow a hybrid maize (DMR White) in pots in green house for six weeks. Four rates ranging from 0 to 3 tons/ha of calcium oxide (CaO) were used as the lime, ranging from 0 to 3 tons/ha for six weeks in greenhouse. The treatments were arranged in a completely randomized design (CRD) with three replications. Soil chemical properties tested were significantly (P<0.05) influenced by increased rates of lime application. Soil pH increased from 4.6 in the unlimed soils to 6.7 in soils treated with 3 t/ha CaO. The exchangeable calcium increased from an average of 0.44 cmolkg-1 at a pH 4.6 and 3.96 cmolkg-1 at a pH 6.7. Liming depressed the uptake of manganese and zinc in all the soils but increased the uptake of calcium and phosphorus. The exchangeable aluminium and percentage aluminium saturation decreased with increasing rates of lime application. Available Phosphorus was low even with a reduction of soluble aluminium in the soil solution, indicating that for proper management of these soils, liming should be supplemented with adequate phosphorus fertilization. Increase in calcium uptake at 3 t/ha was not reflected in the dry matter yield of maize compared to 1 t/ha of lime thus, the 3 t/ha of lime can be regarded as fertilizer rather than as soil amendment. The percentage aluminium saturation was reduced from the mean value of (68.7 to ≤ 33.3)% at 1 t/ha of lime established to be the most suitable criteria for liming acid soils of Nigeria; hence, it is recommended for maize cultivation.
Keywords: Acid soil; Aluminium saturation; Coastal plain sands
Introduction
Soil acidity poses a major problem to crop production in the tropics as a result of inherent soil infertility characterised by high hydrogen and aluminium ions in the soil [1]. Most acid soils have been found to be low in fertility as a result of poor physical, chemical and biological properties. Crop production on such soils is seriously constrained, particularly in areas where adequate management practices have not been put in place [2].
Soil acidity is common in all regions where precipitation is high enough remove appreciable amounts of exchangeable bases from the surface layer of soils [3]. The soil of humid tropics is highly weathered and subjected to frequent and intensive rainfall. In Nigeria, acid soils occur in the southern zone, particularly in parts of Lagos, Ogun, Anambra, Imo, Cross River, Edo, Enugu, Akwa, Ibom, Delta and Rivers State.
The use of exchangeable aluminium as a criterion was first championed by Kamprath [4] and has been found to be reliable in the management of soils with problems associated with over liming the soil [4]. Lime is routinely used to raise soil pH and at present, so many criteria for applying it are used in Nigeria. Studies on Nigeria soils do not support either raising soil pH to neutral or the use of exchangeable Al as a criterion for liming Nigeria soils [5] and there is a dearth of information on the appropriate means of liming Nigerian acid soils especially in the study area. The study determined the percentage of aluminium saturation as a criterion for liming tropical acid soils of Nigeria and to contribute to our knowledge of liming acid soils.
Materials and Methods
Description of the study area
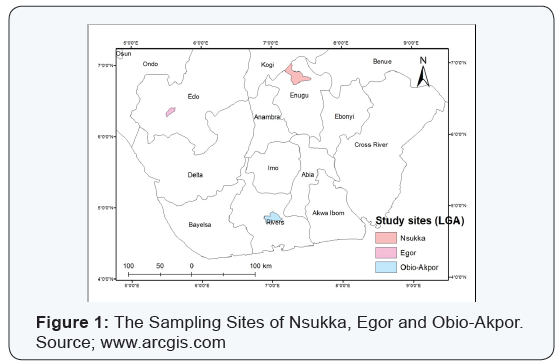
Twenty-one surface soil samples (0 - 15 cm) were collected from coastal plain acid sands of Egor and Obio-Akpor and sandstone of Nsukka soils occurring over sedimentary rocks in the humid region of Nigeria [6]. The vegetation largely consists of mangrove swamp in the coast through rainforest to derived savanna in the interior. The rainfall pattern is bimodal with the early rains (April - July) and late rains (August -October) with five months of dry season and a short dry period in August. Soil samples were air dried, crushed with mortar and pestle to break the clods and to enable the soil to pass through a 2 mm sieve for particle size distribution and routine analysis. For the determination of organic carbon and total nitrogen, the soil was further passed through 0.5 mm sieve (Figure 1).
Greenhouse trials
Three rates (1, 2 and 3 tons/ha) of lime with a control were laid out in a completely randomized design in three replicates. Four hundred grammes of 2 mm sieved soils were weighed into 700ml plastic water cups, which were perforated at the bottom and covered with cotton wool to allow for easy drainage and treated with the various rates of lime. The lime was thoroughly mixed with the soil mass and moistened to approximately field capacity and allowed to equilibrate for forty-eight hours. Nutrient status of the soil was determined at the beginning of the experiment and basal nutrients in the form of urea, muriate of potash and single superphosphate were supplied to augment the deficiency observed. Hybrid maize (D.M.R.white) was obtained from Eagle seed service office at Ibadan. Each pot was sown with four seeds and later thinned to two seedlings 2WAS. The plants were allowed to grown for forty-two 42 days, the top-growth was harvested and oven- dried at 60ºC for forty-eight hours and weighed.
Laboratory analysis for soil samples
Soil analysis was carried out before and after cropping. Particle size analysis was done using the Bouyoucos hydrometer method [7]. The pH of the soils was determined in 1N KCL using a 1:1 soil solution ratio with a glass electrode pH meter. Organic carbon was estimated by Walkely and Black method as described by Anderson [7]. Total N, available P were determined by Bray’s P-1 extraction and molybdenum blue method and the colour intensity is read on a spectrophotometer, exchangeable cations (Ca, Mg, K, and Na) and exchangeable acidity by 1 N KCL extraction were determined by methods largely described by Thomas [8]. ECEC was determined by summation of exchangeable bases (Ca, Mg, K, and Na) and exchangeable acidity. Free iron oxides was determined using the method described by Blakemore [9] and Percentage aluminium saturation was calculated using the below formula;
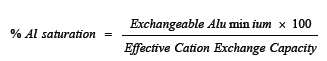
Statistical analysis
Data collected were subjected to analysis of variance (ANOVA) and treatment means were separated using the least significant difference at 5% probability level using Genstat statistical package version 8.0.
Results and Discussion
The general properties of the soil studied
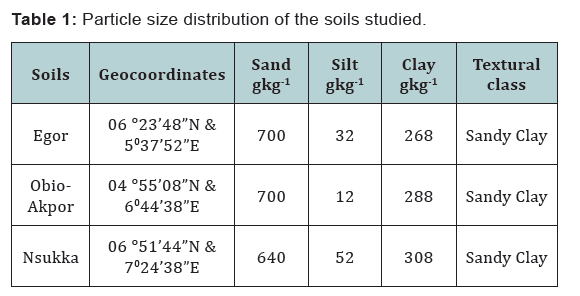
The Egor soil had the lowest base saturation of 25 % while Obio-Akpo and Nsukka have the highest with a close range of 34 and 35 % respectively. The particle size analysis shows dominance of sand fraction 64 to 70 % in the soils and the clay content varies from 26.8 to 30.8 % and they all fall into the same textural class which is sandy clay (Table 1). The clay content of all the soils are all in the range 20 to 430 gkg-1 as reported for tropical soils [10].
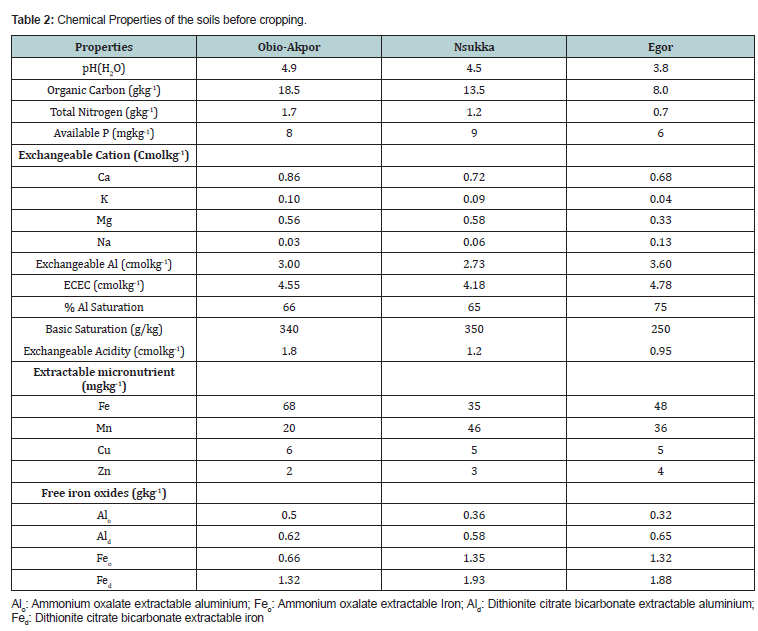
The exchangeable aluminium of the soils varied from (2.73 to 3.60) cmolkg-1 and percentage aluminium saturation ranged from 65 to 75% which are considered high [11] and thus, harmful to the growth of plants. The soils are generally low in available phosphorus and organic carbon. The mean effective cation exchange capacity was 4.18 cmolkg-1 for Nsukka, 4.55 cmolkg-1 for Obio-Akpor and 4.78 cmolkg-1 for Egor soils. The pH of the soils was strongly acidic ranging from 3.8 for Egor, 4.5 for Nsukka and 4.9 for Obio-Akpor soils (Table 2). The low nutrient status of the soil could be attributed to losses due to leaching resulting from high rainfall that characterised tropical soils [12].
The ammonium oxalate extractable Al ranged from 0.50 to 3.6 gkg-1 while the oxalate extractable iron varies from 0.66 to 1.35 gkg-1 for Obio-Akpor, Nsukka and Egor soils respectively. The dithionite citrate bicarbonate extractable Al also ranged from (0.58 to 0.65) gkg-1 and the dithionite citrate extractable iron varied from 1.32 to 1.93 gkg-1 for Obio-Akpor, Nsukka and Egor soils respectively (Table 2). The high amount of free iron oxides may be attributed to the low soil pH.
Soil chemical properties
The exchangeable Al3+ values varied from 2.00 to 2.8 cmolkg-1 for all the soils in the control without lime treatment and reduces as the lime rate increases. The lowest values of exchangeable aluminium were 0.6 cmolkg-1, 0.9 cmolkg-1 and 0.4 cmolkg-1 at 3 t ha1 for Obio-Akpor, Nsukka and Egor soils respectively. The exchangeable acidity values ranged from 2.1 to 3.2 cmolkg-1 for the soils in control without lime treatment and reduced drastically as lime rate increases. The lowest values of exchangeable acidity were 1.1 cmolkg-1, 1.2 cmolkg-1 and 1.0 cmolkg-1 at 3 t ha1 for Obio-Akpor, Nsukka and Egor soils respectively (Table 3). Both the exchangeable aluminium and acidity responded significantly (P<0.05) as the rates of lime application increases. This result is similar to the findings of Buni [13], who posited that the decrease in exchangeable aluminium may be due to the increased displacement of aluminium by calcium in the exchange site and by the subsequent precipitation of aluminium as aluminium hydroxide as the soil was limed. Also,an increase in soil pH resulted in precipitation of exchangeable Al3+ and insoluble aluminium hydroxides thus reducing concentration of aluminium in the soil solution [14].
The exchangeable Ca2+ increased from 0.38 cmolkg-1 for Obio- Akporsoils, 0.50 cmolkg-1 for Nsukka soils, and 0.45 cmolkg-1 for Egor soils, all from the control without lime treatment to 3.62 cmolkg-1, 4.50 cmolkg-1 to 4.02 cmolkg-1 all at 3 t ha1. Nsukka and Egor soils had the highest of exchangeable calcium followed by Obio-Akpor soils with the lowest value (Table 3). The different rates of application of lime significantly increase (P<0.05) exchangeable Ca2+ in all the soils. This increased in exchangeable Ca2+ could be attributed to calcium oxide used for the experiment, which added calcium to the soils. This increase in calcium uptake at 3 t/ha did not reflect in the dry matter yield as compare to the 1 t/ha of lime applied. Thus, the 3 t/ha of lime could be regarded as fertilizer rather than as soil amendment. This result is similar to the findings of Omueti [15], who reported that an increase in exchangeable calcium is probably due to liming.
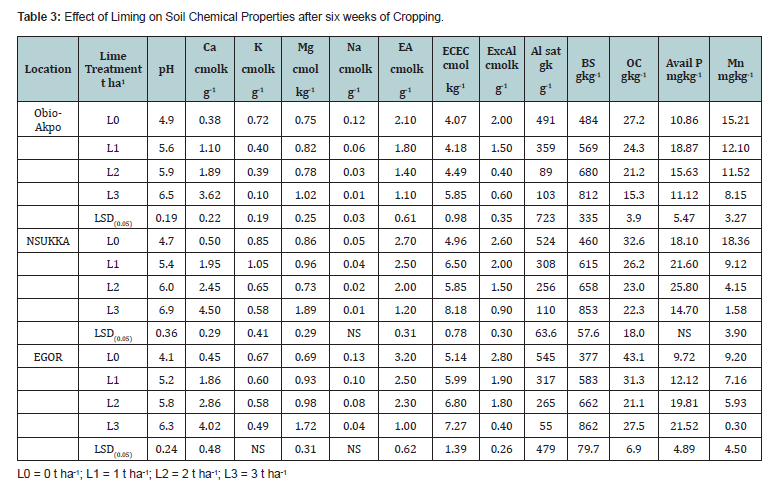
Exchangeable Mg2+ values for all the soils ranges from 0.69 to 0.86 cmolkg-1 in the control without lime treatment for Obio- Akpor, Nsukka and Egor soils respectively. The highest values of exchangeable Mg2+ varied from (1.02 to 1.89) cmolkg-1 and were obtained at 3 tons per ha of lime in all the soils (Table 3). The increase in exchangeable Mg2+ (P<0.05) may be due to the application of lime which increased the soil pH to 6.5 and above, a range in which more magnesium is found in soil solution.
The aluminium saturation values varied from 491 to 545 gkg-1 for all soils in the control without lime treatment and the lowest values were 89 gkg-1 at 2 t ha1 for Obio-Akpor soils, 110 gkg-1 for Nsukka and 55 gkg-1 for Egor soils both at 3 t ha1. Aluminium saturation decreased significantly (P<0.05) as the rate of liming application increases (Table 3). The decreased of percentage aluminium saturation could be attributed to the decrease of exchangeable aluminium by exchange with cations and also, the increase exchange capacity induced by the higher electrolyte concentrations as predicted by theory or constant potential surface [12]. The base saturation varies from 377 to 460 gkg-1 for all the soils in the control without lime treatment and the highest values were 812 gkg-1 for Obio-Akpor soils, 853 gkg-1 for Nsukka soils and 862 gkg-1 for Egor soils all at 3 t ha1 of lime. Base saturation increase significantly (P< 0.05) with increases rate of lime application (Table 3). This increase may be attributed to the dosage of lime applied which in turn supplied calcium to the soil which replaced the exchangeable aluminium from the exchange site.
The soil pH increased with increasing rate of lime irrespective of the rate of application. The soil pH values were significantly increased (P<0.05) with increasing rate of lime application in all location. Soil pH significantly increased from 4.1 to 4.9 in the control pots without lime application to 6.5, 6.9 and 6.3 at the rate of lime 3 tons ha-1 for Obio-Akpor, Nsukka and Egor respectively (Table 3). This responses of pH to lime has also been observed in tropical soils in several regions of the world [16]. The increased in soil pH is probably due to the increase in basic cations (Ca) and neutralization at the exchange complex [13].
Liming significantly (P<0.05) increases the available phosphorus for Obio-Akpor and Egor soils, although the increase were not consistent in all the soils. The highest values of available phosphorus ranged from 18.87 mgkg-1 at 1 tons per ha for Obio- Akpor, 25.8 mgkg-1 at 2 t ha1 for Nsukka and 21.52 mgkg-1 at 3 t/ha for Egor soils (Table 3). The available phosphorus in all the soils still falls within the sufficiency range. The increase of available phosphorus can be explained in terms of the activities of iron, aluminium and manganese at low pH. When the soil pH is low, the activities of iron, aluminium and manganese are significantly form complexes with the soluble forms of phosphorus thus fixing the element. With lime application, the pH is increased and the iron and aluminium which dominate the soils become less active, thus releasing the soluble phosphate ions in form of the diphosphates and monophosphate. This result is similar to Unagwu et al. [17], reduction in soil acidity leads to phosphorus availability.
Nutrient uptake
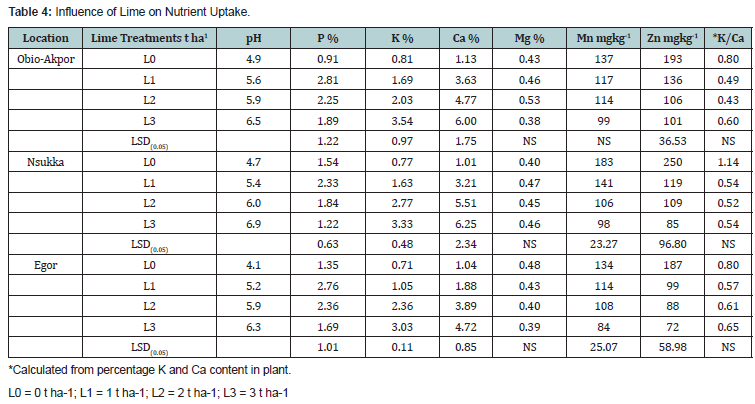
The uptake of available phosphorus from all the soils were statistically significant, the increase of phosphorus uptake was not consistent with the rates of lime of application (Table 4). The lowest uptake were observed at 3 t/ha corresponding to 1.22 to 1.89 mgkg-1 and the highest values were observed at 1 tons per ha corresponding to 2.89, 2.33 and 2.76 mgkg-1 for Obio-Akpor soils, Nsukka soils and Egor soils respectively. This result is similar to the findings of Amarasiri [18]. Maximum P uptake by plant occurred when soils were limed to pH between 5 and 6. Liming of ultisols and oxisols to pH values above 6 will increased fixation of phosphorus either through precipitation as calcium phosphates or through sorption by freshly precipitated iron and aluminium hydroxides in the limed soil [5].
Calcium uptake increased significantly (P<0.05) with increasing rate of liming application over the control, the lowest values were observed at 1 t ha1 corresponding to 1.88 to 3.63 cmolkg-1 and the maximum uptake were observed at 3 t ha1 with the responding values of 6.00, 6.25 and 4.72 cmolkg-1 for Obio- Akpor, Nsukka and Egor soils respectively (Table 4).
The total magnesium uptake for all soils were not significantly influenced with increasing rates of liming application over the control, the maximum values ranged from (0.43 to 0.58) cmolkg-1 and the minimum values were 0.38, 0.45 and 0.39 cmolkg-1 for Obio-Akpor, Nsukka and Egor soils respectively (Table 4). Total potassium uptake from all soils were significantly (P<0.05) influenced with increasing rates of lime application (Table 4). The maximum potassium uptake were 3.54 , 3.33 and 3.03 cmolkg-1, all at 3 tons per ha and the minimum uptake were 1.69, 1.63 and 1.05 cmolkg-1 all at 1 t ha1 for Obio-Akpor, Nsukka and Egor soils respectively.
The manganese uptake by maize plant from all soils decreased with increasing rates of lime application over the control but significant (P<0.05) for Nsukka and Egor soils respectively (Table 4). The highest uptake of manganese values (117,141 and114 mgkg-1) all at 1 t ha1 and the lowest values uptake of manganese were 99, 98 and 84 mgkg-1 all at 3 tons per ha Obio-Akpor, Nsukka and Egor soils respectively. The uptake of manganese by plant reduced drastically with increasing soil pH and rate of liming application. This result is in agreement with the findings of Juo [5].
Dry matter yield
The dry matter yield obtained from all soils increased significantly (P<0.05) with increasing rates of lime application over the unlimed soils, although the increase were not consistent among the soils. The dry matter yield values for the unlimed soils varied from 1.42, 1.64 and 1.14 g pot-1 and the highest values ranged from 4.14 to 4.58 g pot-1, and all were obtained for Obio- Akpor, Nsukka and Egor soils respectively (Table 5). The dry matter yield values obtained for unlimed soils were relatively low when compared to the limed soils. This could be attributed to the inherent acidic nature of the soils associated with aluminium and manganese toxicity of the soils, and low exchangeable calcium of the soils under study (Table 2). The maximum yield was obtained at an average soil pH of 5.4 for all the three soils and dropped when the soils were limed to an average soil pH of 6.5. This result indicates that over liming depresses the maize dry matter yield. According to Sanchez [19], the lower yield obtained at higher rates of lime is attributed to the nearly flat plateau. The yield values to deficiencies of micronutrients such as manganese and zinc which are induced by high rates of lime application. The deficiencies of Mn and Zn were not observed but suspected as their uptake were drastically depressed as the rates of lime increases. The increase in calcium uptake at 3 t ha1 of lime did not reflect in the dry matter yield of maize compared to the 1 t ha1 of lime applied (Table 5). Thus, the 3 t ha1 can be regarded as fertilizer rather than as soil amendment.
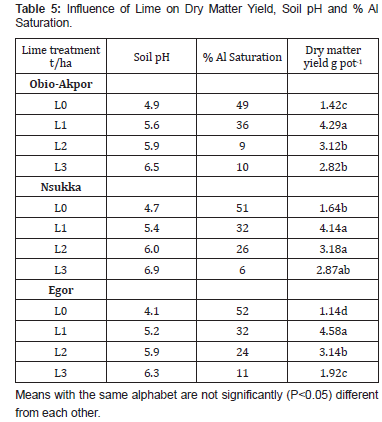
From the above discussion, the appropriate lime requirement for these soils is about 1 t ha1 having the mean yield of 4.34 g pot-1. This lime rate reduced the percentage aluminium saturation from the mean of 68.7% (Table 2) to 33.3% for all the soils (Table 5). This finding is in agreement with the conclusion of IITA [20-22], which stated that the critical level of exchangeable aluminium saturation for maize (Cv.TZPB) required for 90 percent maximum yield was about 35 percent. The lime rate of 1ton per ha was sufficient enough to maintained the exchangeable aluminium at a tolerant level for the variety of maize used. Hence, the removal of exchangeable aluminium or the reduction of soluble aluminium to about 33.3% of the effective cation exchange capacity is a more reliable basis for predicting lime requirement of tropical acid soils of Nigeria.
Conclusion and Recommendation
Soil acidity is often associated with aluminium toxicities and soil nutrient depletion, which results in poor agricultural production, experiment was carried out involving laboratory and greenhouse study, to examine the percentage aluminium saturation as a criterion for liming acid soils of the tropics, especially Nigerian soils, using quick lime as the liming material. In the laboratory experiment, lime reduced all acid parameters indicators (Exchangeable aluminium, exchangeable acidity, soil pH and percentage aluminium saturation) associated with the soils; and also, the availability of phosphorus was increased. Liming reduced the amount of free iron oxides in all the soils studied.
In the greenhouse studies, the 3 t ha1 of lime applied did not reflect in the dry matter yield of maize. The percentage aluminium saturation was reduced from a mean value of 68.7% to 33.3%, which corresponds to 1 t/ha of lime application, improved the dry matter yield of maize. The different rates of lime application increased the uptake of P, K, Ca and Mg, but the uptake of Zn, Mn and the K/Ca ratio in plant decreased as the rates of lime application increases.
In conclusion, the use of percentage aluminium saturation as the basis for calculating lime requirement is justified in terms of the improved yield response obtained, although the lime applied supplied calcium to the soil which was reflected in the effective cation exchange capacity of the soil. The following are therefore recommended:
- For efficient and prudent management of the soils study, lime should always be applied with sufficient or adequate phosphorus fertilizer application.
- The most suitable criterion for liming was found to be the reduction of percentage aluminium saturation in soil solution to ≤ 33.3% at 1 t ha-1 of lime. At this level, the soil pH will not be drastically increased.
References
- Beegle DB, Lingenfelter DD (1996) Soil Acidity and Aglime. Agronomy Facts 3. Penn State College of Agricultural Sciences. The Pennsylvania State University, University Park, PA, USA.
- He Z, Yang X, Baligar VC, Calvert DV (2003) Microbiological and BiochemicalIndexing Systems for Assessing Quality of Acid Soils. Adv Agron 78: 89-138.
- Conyers M (1986) The Relationship between Average Annual Rainfall and Exchangeable Aluminium in soils of Southern-Eastern New South Wales. Aust J Exp Agric 26: 587-590.
- Kamprath EJ (1971) Potential detrimental effects from liming highly weathered soils to neutrality. Soil Crop Sci Soc Fla Proc 31: 200-203.
- Juo ASR, Uzu FO (1977) Liming and Nutrient Interactons in two ultisolss from Southern Nigeria. Plant and Soil 47(2): 419-430.
- Ojanuga AG, Lekwa G, Akamigbo FRO (1981) Survey, Classification and Genesis ofAcid Sands. In: EJ Udo & RA Sobulo (Eds.), Acid Sands of Southern Nigeria, Soil Science Society of Nigeria, pp: 1-18.
- Anderson JM, Ingram JSI (1993) Tropical Soils Biology and Fertility. A handbook of Methods, (2nd edn), CAD International, Wallingford U.K, pp. 221.
- Thomas GW (1982) Exchangeable Cations. In: Page AL, Miller RH, Keeny DR (Eds.), Methods of Soil Analysis part 2. (2nd edn), Agronomy and Monograph No.9 ASA, and SSA, Madison, WI, USA, pp: 159-165.
- Blakemore LC, Searlesp PL, Daly BK (1987) Methods for Chemical analysis of Soil. New Zealand soil bureau scientific report. New Zealand, pp. 103.
- Agboola AA, Omueti JAI (1982) Soil fertility Problem and its Management in Tropical Africa: International Conference on Land clearing and Development. Proceedings IITA 16: 234-240.
- Hakim N, Agustain S, Soepardi G (1989) Effects of Lime, Fertilizers and Crop Residues on Yield and Nutrient Uptake of Uplant Rice, Soybean and Maize in intercropping Systems. In: JVD Heide (Ed.), Nutrient Management for Food Crop Production in Tropical Farming System. Haren. The Netherlands, pp. 349-360.
- Parks GA, de Bruyn PL (1962) The Zero Point of Charge of Oxides. J Phys Chem 66(6): 967-973.
- Buni A (2015) Effects of Liming Acidic Soils on Improving Soil Properties and Yield of Haricot Bean. J Bioremed Biodeg 6: 278-285.
- Ritchie GSP (1989) The Chemical Behaviour of Aluminium, Hydrogen and Manganese in Acid Soils. In: Robson AD (Ed.), Soil Acidity and Plant Growth. Academic Press, Marrickville, Australia, pp. 1-49.
- Omueti JA (2015) A Rexamination of the Criterion for Liming Tropical Acid Soils of Nigeria (in Press).
- Caries EF, Barth G, Garbuio FJ (2006) Lime application in the establishment of a no-till system for grain crop production in Southern Brazil. Soil and Tillage Research 89(1): 3-12.
- Unagwu BO, Asadu CLA, Ezeaku PI (2013) Residual Effects of Organic and NPK Fertilizers on Maize Performance at Different Soil pH Levels. IOSR J of Agric and Vet Sci 5(5): 47-53.
- Amarasiri SL, Olsen SR (1973) Liming as related to solubility of phosphorus in plant growth in an acid tropical soil. Soil Sci Soc Amr J 37(5): 716-721.
- Sanchez PA (1976) In Properties of Management of Soils in the Tropics. John Wiley and Sons, New York, USA, 70: 393-410.
- International Institute of Tropical Agriculture (IITA) (1981) Research Highlights.
- Parnes R (1990) Fertile Soil.A grower’s guilde to Organic and Inorganic Fertilizer. Los Angeles University of California Press, USA, pp. 80.
- Udo EJ, Ogunwale JA (1986) Laboratory manual for analysis of Soil, Plant and Water Samples. (2nd edn.), Imperial Binding Services, Ibadan, Oyo State, Nigeria, pp.174.






























