Preparation of an Autologous Fibrin Sealant and Its Functionalization with Antimicrobial Agents
Lucena MLC1,2, Lucena SCA2, Dias VVM1, G Brondani GL1, Lucena AES3, Leite ACL1*
1Departament of Pharmaceutical Science, Federal University of Pernambuco, Recife, Pernambuco, Brazil
1Fundação de Hematologia e Hemoterapia de Pernambuco (HEMOPE), Recife, Pernambuco, Brazil
1Empresa Brasileira de Hemoderivados e Biotecnologia (HEMOBRAS), Recife, Pernambuco, Brazil
Submission:September 19, 2025; Published: October 06, 2025
*Corresponding author:Leite ACL, Departament of Pharmaceutical Science, Federal University of Pernambuco, Recife, Pernambuco, Brazil
How to cite this article:Lucena MLC, Lucena SCA, Dias VVM, G Brondani GL, Lucena AES, Leite ACL. Preparation of an Autologous Fibrin Sealant and Its Functionalization with Antimicrobial Agents. Adv Biotech & Micro. 2025; 19(2): 556008.DOI:10.19080/AIBM.2025.19.556008
Abstract
This study aimed to develop a simple methodology for producing autologous fibrin sealant with the incorporation of an antimicrobial agent. To achieve this, fibrinogen (cryoprecipitate) and prothrombin (plasma euglobulin) were isolated from individual plasma units with negative serology using physicochemical protein precipitation techniques. Calcium salts at various concentrations were used to prothrombin-thrombin conversion. In the Clot Formation Test, immediate coagulation was observed upon mixing activated thrombin when it was prepared with 0.2 M calcium carbonate. The descriptive statistical analysis of thrombin concentration measurements reveals the mean and modal concentrations at 388.66 and 482.32 IU/mL, respectively. The incorporation of antimicrobial agents was designed to enable potential protection against pathogens associated with surgical site infections (SSIs). The viability of these components was evaluated qualitatively, and microbiological assays were performed to assess the antimicrobial activity of the antibiotic-enriched fibrin sealant against Staphylococcus aureus, one of the primary pathogens responsible for surgical site infections (SSIs). The fibrin matrix was able to inhibit local bacterial growth for up to 72 hours. The methodology developed proved effective for producing fibrin sealant on a blood center scale, with promising potential for SSI prevention. Developing a simplified and cost-effective approach for generating a fibrin matrix is significant not only for clinical and surgical applications but also for broader research and therapeutic innovation, given the versatility of this biological material and its wide-ranging potential in pharmaceutical technology and regenerative medicine.
Keywords: Fibrin Sealant; Fibrin Matrix; Fibrinogen; Prothrombin; Antimicrobial Agents
Abbreviations: SSIs: Surgical Site Infections; HEMOPE: Hematology and Hemotherapy Foundation of Pernambuco; CAAE: Certification of Ethical Approval; RDC: Resolution of the Collegiate Board; APTT: Activated Partial Thromboplastin Time; SEM: Scanning Electron Microscopy
Introduction
Surgical procedures often involve significant bleeding, which can complicate the intervention and increase the risk of infection, delayed healing, or even mortality. Fibrin sealants offer a targeted hemostatic solution, especially in cases where traditional methods such as sutures or cauterization are inadequate or difficult to apply [1]. Additionally, they enhance surgical outcomes by reducing operative time and the need for transfusions [2].
Beyond hemostasis, fibrin sealants play an important role in promoting tissue regeneration and delivering therapeutic agents. Recent studies have explored their use as carriers for antibiotics, growth factors, and stem cells, demonstrating their potential as drug delivery platforms and scaffolds in regenerative medicine [3,4]. This makes them particularly valuable in managing chronic wounds, reconstructive surgeries, and applications requiring localized, controlled release of bioactive molecules [5,6]. These sealants consist primarily of fibrinogen and thrombin, which, when combined, form a stable fibrin clot that promotes hemostasis, tissue adhesion, and wound healing [7,8]. The growing demand for fibrin sealants is driven by their versatility and effectiveness in a wide range of medical procedures, including cardiovascular surgery, neurosurgery, orthopedic interventions, and tissue engineering [9]. Fibrin sealants, also known as fibrin glues, have become an increasingly valuable tool in surgical and biomedical applications due to their biocompatibility, biodegradability, and ability to mimic the final stages of the coagulation cascade [10].
Despite their benefits, commercially available fibrin sealants are often derived from pooled human plasma or animal sources, raising concerns about immunogenicity, disease transmission, and cost-particularly in low-resource settings. These limitations have sparked interest in the development of autologous alternatives, which use the patient’s own plasma to mitigate safety risks and reduce costs [11]. Autologous fibrin sealants can also be customized to incorporate antimicrobial agents, enhancing their functionality in preventing surgical site infections [12,13]. In light of these advantages and evolving applications, the continued development of accessible, safe, and effective fibrin sealants remains a priority in both clinical practice and biomedical research.
Commercially available fibrin sealants are typically composed of concentrated, lyophilized human fibrinogen and thrombin, and may also contain stabilizing agents such as coagulation factor XIII and antifibrinolytic substances [14]. Manufactured by the plasmaderived biopharmaceutical industry, their production requires a sophisticated technological infrastructure, including advanced techniques such as affinity chromatography and ion-exchange processes-factors that contribute to their high production cost [15].
In this context, here we present a low-cost methodology for producing autologous fibrin sealants from a single plasma donation, designed for use in blood centers. The fibrin clot formation process was achieved using fibrinogen from cryoprecipitate and prothrombin was from cryo-free plasma after acid precipitation, both isolated from individual plasma units. Calcium salts at various concentrations were used to prothrombin-thrombin conversion. The clot formation was obtained by mix thrombin and fibrinogen. The incorporation of antimicrobial agents was designed to enable potential protection against pathogens associated with surgical site infections (SSIs) (Figure 1). The fibrin sealant/ antimicrobial formulation may offer a viable and accessible alternative for the use of this hemostatic product.

Materials and Methods
Ethical statement
The plasma bags used in this study were provided by the Hematology and Hemotherapy Foundation of Pernambuco (HEMOPE). The study protocol was approved by the Research Ethics Committee of the Brazil Platform, with the Certification of Ethical Approval (CAAE) number: 57637516.7.0000.5208. The patients were not identified, and the study data were analyzed anonymously. The plasma bags provided by the blood center were negative for HIV I/II, hepatitis B and C, syphilis, Chagas disease, and HTLV I/II (according to Resolution of the Collegiate Board (RDC) No. 34, dated June 11, 2014, of the Brazilian Health Regulatory Agency (ANVISA)) [14].
Obtaining plasma-derived fibrinogen and thrombin concentrates for fibrin sealant formulation
Cryoprecipitate preparation: A total of forty plasma bags, initially frozen at -80°C and stored at -30°C, were slowly thawed at -4°Cover a 12-hour period. Following thawing, the plasma bags were centrifuged at 3,500 rpm for 10 minutes at 4°C. The cryoprecipitate, containing fibrinogen, was collected by isolating the pellet. The resulting supernatant was reserved for subsequent steps involving plasma euglobulin precipitation.
Fibrinogen quantification in cryoprecipitate: Fibrinogen concentration in the cryoprecipitate was measured using automated coagulometric assays on an ACL TOP® analyzer (Instrumentation Laboratory, Kirchheim, Germany). Reagents (HemosIL®, Instrumentation Laboratory) from the same manufacturer were used, with all tests performed using single lots to ensure consistency. Calibration was carried out with standardized calibration plasma (HemosIL®). Plasma samples with known protein concentrations served as validated controls. Each assay was performed in duplicate, and descriptive statistical analysis of the results was conducted using Microsoft Excel®.
Plasma euglobulin precipitation: The cryoprecipitate supernatant (Cryo-Free Plasma, CFP) was collected, and conditions for euglobulin precipitation were adjusted accordingly. In a transfer plastic bag, 100 mL of CFP was mixed with 400 mL of 5% (w/v) glucose solution and 15 mL of 1 M HCl to adjust the pH to 5.3, which corresponds to the isoelectric point of euglobulins. The mixture was centrifuged at 3,500 rpm for 10 minutes at 4°C. The resulting euglobulin precipitate was collected, and the supernatant was discarded.
Standardization of calcium concentration for prothrombin-to-thrombin conversion: This step aimed to standardize the calcium ion source used to convert prothrombin into thrombin. Solutions of different calcium salts at various concentrations were prepared, including calcium chloride (0.02 M, 0.2 M, and 2 M) and calcium carbonate (0.02 M and 0.2 M). These solutions were used to resuspend the euglobulin precipitate obtained in Section 2.2.3. The mixtures of precipitate and calcium solution were allowed to rest for approximately 60 minutes at room temperature to facilitate the reaction. The effectiveness of each calcium formulation was subsequently evaluated (Section 2.2.6) by analyzing the coagulation capacity of the product resulting from the interaction between calcium ions and prothrombin.
Fibrin clot formation test: To evaluate fibrin clot formation, equal volumes (1:1) of the solution obtained in Section 2.2.4 and cryoprecipitate from Section 2.2.1 were combined in a test tube. The mixture was gently homogenized, and the time required for visible clot formation was recorded using a stopwatch.
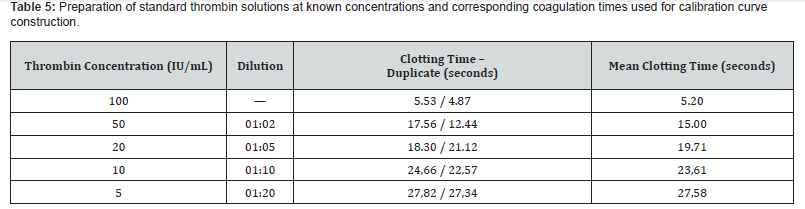
Determination of human thrombin concentration: The concentration of purified thrombin was determined using the inverted Clauss method [16]. In this approach, a known concentration of fibrinogen (from plasma) is added to the thrombin sample, and the clotting time is measured. A calibration curve was first established by preparing serial dilutions of standard thrombin (100 IU/mL) in distilled water at the following ratios: 1, 1:2, 1:5, 1:10, and 1:20. For the assay, standard plasma was diluted 1:10 by mixing 0.1 mL of plasma with 0.9 mL of 50 mmol/L imidazole buffer containing 0.05% sodium azide. The diluted plasma was incubated at 37°C for 5 minutes. Then, 0.1 mL of each thrombin dilution was added to 0.1 mL of the incubated plasma, and a stopwatch was started simultaneously to record the coagulation time. The calibration curve was plotted in Microsoft Excel®, with thrombin concentrations on the X-axis and clotting times on the Y-axis. To determine thrombin concentration in the purified samples, a 1:5 dilution was prepared, and the concentration was interpolated from the calibration curve. All measurements were performed in duplicate, and descriptive statistical analysis was carried out using Excel®.
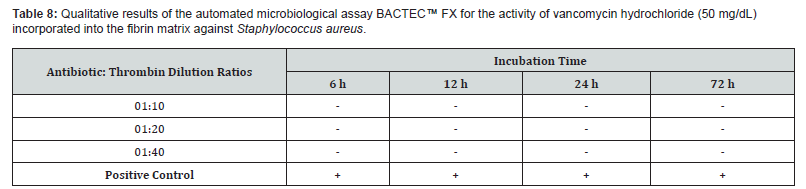
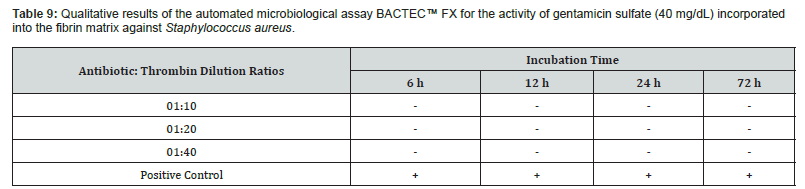
Incorporation of antimicrobial agents into the thrombin solution and clot formation test: Vancomycin hydrochloride (50 mg/mL) and gentamicin sulfate (40 mg/mL) were selected as the antimicrobial agents for this assay. Each was added to the thrombin solution in sterile test tubes at volume ratios of 1:10, 1:20, and 1:40, as shown in Table 1. After incorporation, the clotting ability of each thrombin-antimicrobial mixture was evaluated according to the procedure described in Section 2.2.6, in order to confirm that the addition of the antimicrobial agents did not interfere with the clot formation process or compromise the functional integrity of the fibrin sealant.
The antibiotic-enriched thrombin solutions, along with a control sample (thrombin without antibiotics), were each mixed with 5 mL of cryoprecipitate at a 1:1 volume ratio for a new clot formation test. Coagulation time was recorded using a stopwatch, and the resulting fibrin clots were evaluated for macroscopic characteristics such as firmness, homogeneity, and structural integrity. All experiments were performed in triplicate to ensure reproducibility.
Evaluation of the efficacy of antimicrobial agents incorporated into the fibrin matrix against staphylococcus aureus
Inoculum preparation: A stock culture of Staphylococcus aureus (ATCC 6538) was used for inoculum preparation. An aliquot of the microorganism was inoculated into Brain Heart Infusion (BHI) broth and incubated at 35°C for 24 hours. Following incubation, the culture was centrifuged, and the supernatant was discarded. The resulting bacterial pellet was resuspended in sterile saline solution to achieve a final concentration of 1 × 10⁸ CFU/ mL. The concentration was standardized using a turbidimetric method based on the McFarland scale.
Microbiological assay using automated blood culture system: The microbiological assay was performed using the BACTEC™ FX automated blood culture system. Each test bottle was prepared with 5 mL of thrombin solution enriched with an antimicrobial agent, 50 μL of the Staphylococcus aureus (ATCC 6538) suspension, and 5 mL of cryoprecipitate. A positive control bottle containing 5 mL of thrombin solution without antibiotics, along with the same volumes of bacterial suspension and cryoprecipitate, was prepared under identical conditions. Following fibrin clot formation, the bottles were incubated in the BACTEC™ FX system for up to 7 days. Bacterial growth was monitored at multiple time points-6, 12, 24, 48, and 72 hours-to assess the antimicrobial efficacy of each fibrin matrix formulation.
Results and Discussion
Figure 1 presented an overview of the methodology developed in this study for the extraction of fibrinogen and thrombin, designed to enable the formulation of an autologous fibrin sealant within the operational framework of blood centers.
Preparation of plasma-derived fibrinogen and thrombin concentrates for fibrin sealant formulation
Fibrinogen and thrombin concentrates were prepared from cryoprecipitate and supernatant fractions obtained by thawing 40 plasma units, followed by sequential centrifugation steps. Quantification of fibrinogen and thrombin levels was then performed, as illustrated in (Figure 2). The cryoprecipitate was promptly resuspended in 15 mL of plasma, following the guidelines set forth by Consolidated Ordinance No. 7 of the Brazilian Ministry of Health [17].

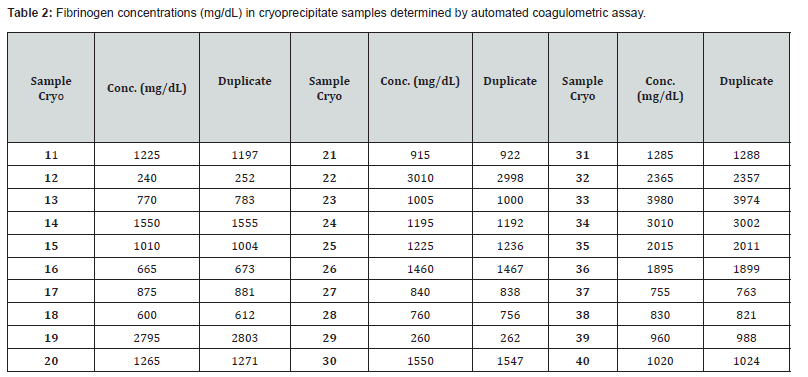
Fibrinogen measurement in cryoprecipitate: The results of fibrinogen measurements in cryoprecipitate samples are presented in (Tables 2&3). As shown in Table 2, all the tested samples exhibited fibrinogen concentrations above 150 mg/ dL, the recommended threshold. This value corresponds to the reference value established by Brazilian legislation as a quality parameter for cryoprecipitate, considered a therapeutic blood product [17].
The descriptive statistical analysis of the fibrinogen measurements in cryoprecipitate (CRIO), as presented in Table 3, revealed considerable variation between the minimum and maximum fibrinogen concentrations in the analyzed samples. This variability can be attributed to physiological differences among the donors, as the plasma fibrinogen levels naturally fluctuate between 3 and 30 g/L [18]. In industrial settings, fibrinogen concentration standardization in blood-derived products is typically achieved through technologies such as ultrafiltration, which allows for the concentration of fibrinogen in CRIO to reach approximately 80 g/L [15]. This standardization is crucial to ensure the uniformity of fibrin sealants produced. However, the lack of accessible laboratory methods for adjusting fibrinogen concentration in CRIO poses a significant challenge for the production of autologous fibrin sealants in blood centers. Different methodologies can be used to isolate fibrinogen from blood plasma, including cryoprecipitation, cold ethanol precipitation, ammonium sulfate, or polyethylene glycol [15,19]. According to Noori et al. [20], the main advantage of cryoprecipitation over other techniques is that no potentially cytotoxic chemicals are required for the fibrinogen extraction process, making it safer. However, the author highlights that cryoprecipitation is a more time-consuming process compared to the others, and the yield of fibrinogen is relatively low [20].
Qualitative evaluation of prothrombin activity using the activated partial thromboplastin time (aPTT) assay: In all analyzed samples, the formation of clots was observed, indicating the presence of thrombin concentrate within the precipitated mass. Upon the addition of calcium chloride, this precursor was successfully converted into functional thrombin capable of inducing coagulation. (Figure 3) shows some of the clots observed during the experiment in samples identified as PROT No. 1, PROT No. 15, PROT No. 24, and PROT No. 33.

The Activated Partial Thromboplastin Time (aPTT) assay was employed as a qualitative method to assess the presence and activity of prothrombin, as it is a laboratory test based on measuring the clotting time of citrated plasma. The assay is performed by adding reagents containing cephalin, ellagic acid, and phospholipids-which act as platelet substitutes-along with calcium chloride. This combination promotes the activation of the coagulation cascade via the intrinsic pathway [21,22]. The results obtained in this phase of the study indicate that prothrombin activity is preserved following plasma dilution in a glucose solution and pH adjustment to 5.3 using 1M HCl.
Standardization of calcium concentration for prothrombin-to-thrombin conversion and clot formation test: To the standardization, the precipitates A1-30, B1-30, C1-30 and D1-30 were treated at 10mL of different sources of calcium (concentrations 0.02 M, 0.2 M, and 2 M). (Table 4) shows that CaCl2 solutions were not effective in inducing fibrin clot formation. The tested concentrations may have been insufficient to convert precipitated prothrombin into active thrombin. When CaCO3 was used, it was observed that calcium carbonate solution at 0.02 M was also ineffective. In the Clot Formation Test, immediate coagulation was observed upon mixing activated thrombin (prepared with 0.2 M calcium carbonate) with fibrinogen-rich cryoprecipitate. Clots formed uniformly throughout the contents of the test tube, adhering firmly to the inner walls, as shown in Figure 4.
Calcium chloride is commonly used as the clot activator in the preparation of fibrin sealants¹⁸ and in laboratory coagulation assays [23]. However, other calcium-containing salts, such as calcium carbonate, can also promote the conversion of prothrombin into thrombin [15]. It is possible that the calcium concentration required for thrombin activation, within the methodology developed in this study, is directly related to the amount of prothrombin present in the resuspended euglobulin precipitate. In this context, measuring thrombin concentration in the subsequent phase of the study was crucial for clarifying why calcium carbonate concentrations below 0.2 M were insufficient to activate thrombin and, consequently, to promote clot formation.
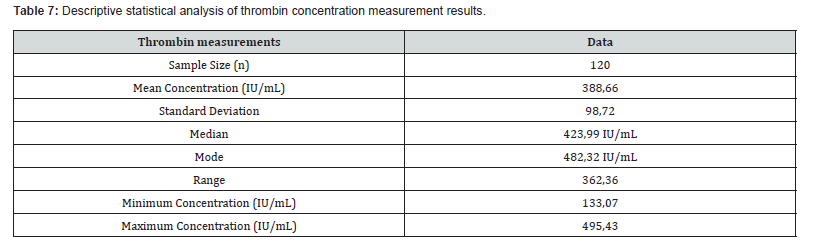
Determination of the concentration of the human thrombin solution: The calibration curve for thrombin quantification using the inverse Clauss method is shown in (Figure 5) and (Table 5). The resulting linear equation was Y = -0.2156X + 26.198, with a correlation coefficient of R² = 0.9616. Table 6 presents the thrombin concentration values of the samples, calculated based on the linear equation obtained from the calibration curve and by multiplying the resulting value by the sample dilution factor, while Table 7 provides a descriptive statistical analysis of the results. All thrombin solutions obtained by resuspending the euglobulin precipitate with a calcium source for prothrombin conversion had a final volume of 10 mL.

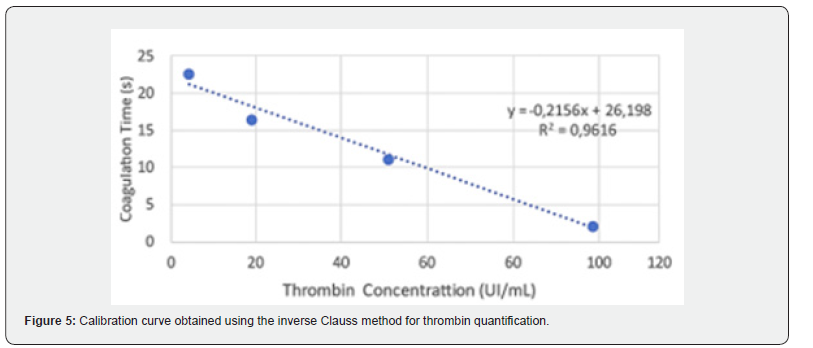
The descriptive statistical analysis of thrombin concentration measurements (Table 7) reveals a wide range between the minimum and maximum values expressed in IU/mL. This variability is inherent to the physiological differences among individual plasma donors. The final thrombin concentrations observed in this study were higher than those reported in other publications describing methods for the production of autologous fibrin sealants [24,25]. Furthermore, the mean and modal concentrations (388.66 and 482.32 IU/mL, respectively) exceeded the value reported by Aizawa et al. (238 IU/mL), who employed large-scale industrial techniques for thrombin production and purification [26].

* Either vancomycin hydrochloride or gentamicin sulfate


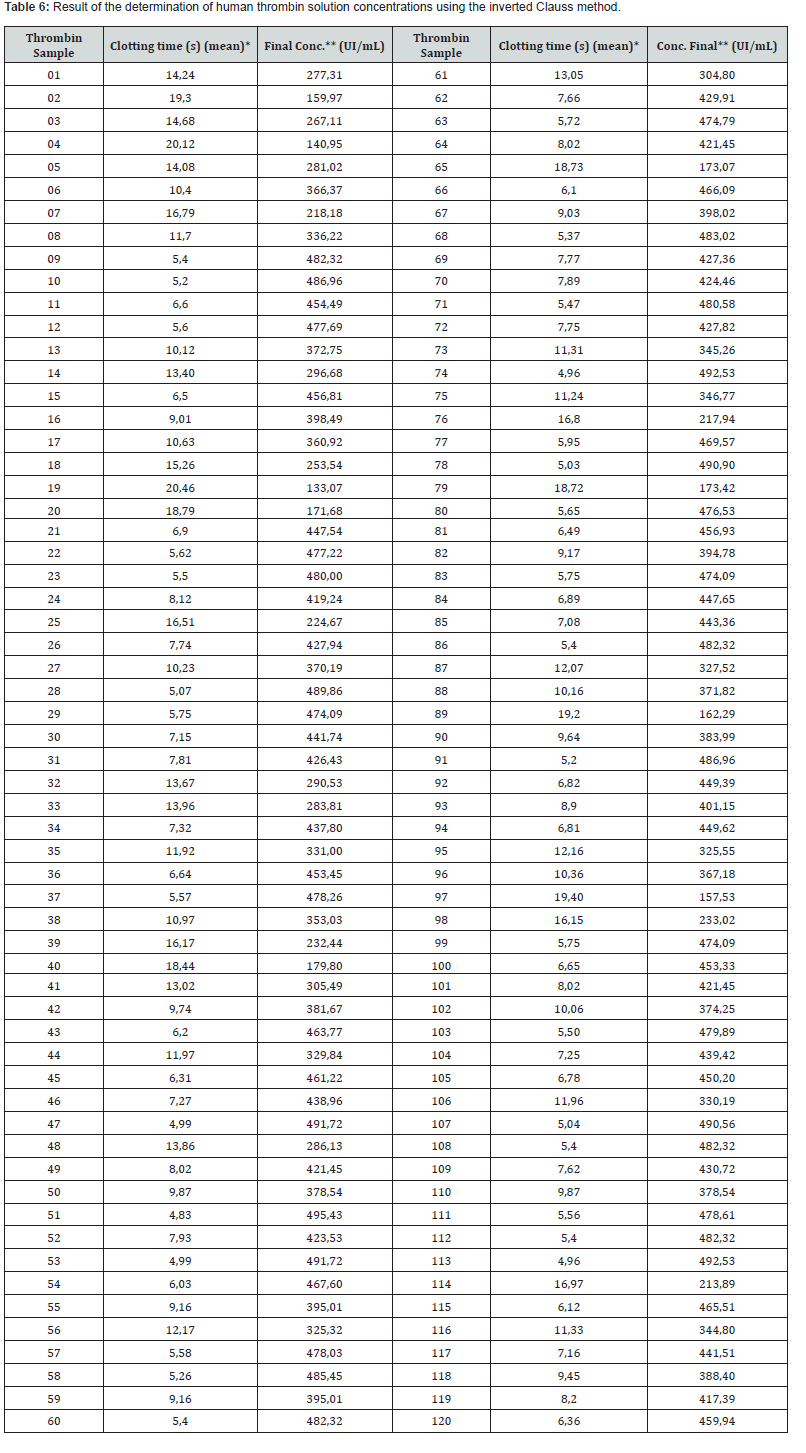
* Mean of the duplicate; ** Value multiplied by the sample dilution factor (dilution factor = 5).
(+): Positive result for bacterial presence / (−): Negative result for bacterial presence
(+): Positive result for bacterial presence / (−): Negative result for bacterial presence.


Incorporation of antimicrobial agents into the thrombin solution and clot formation test
All fibrin clots were formed immediately, regardless of the type or volume of antibiotic added to the thrombin solution. However, clots containing gentamicin sulfate or vancomycin hydrochloride showed reduced consistency and weaker adhesion to the walls of the test tube compared to the control group. This effect was proportional to the volume of antibiotic added, becoming more pronounced at higher concentrations (Figure 6). No significant macroscopic differences were observed between clots containing vancomycin and those with gentamicin (Figure 7). The observed changes in the physical properties of antibiotic-enriched fibrin clots may be attributed to molecular interactions between the drugs and the fibrin matrix. These interactions can influence structural characteristics of the clot, such as thickness, porosity, fibrin mesh diameter, permeability, and consequently, the clot’s ability to retain and release substances [23]. In this context, further analysis using scanning electron microscopy (SEM) could significantly enhance the detailed morphological characterization of the modified fibrin matrix.
Evaluation of the effectiveness of antimicrobial agents incorporated into the fibrin matrix against staphylococcus aureus
Both antimicrobial agents added to the fibrin matrix were able to inhibit staphylococcus aureus contamination for up to 72 hours, across all tested antibiotic: thrombin ratios (Table 8 & 9). These results suggest that the fibrin matrix was effective in releasing the drugs to perform their local action, indicating that the use of this fibrin sealant, produced at a blood center level, may be effective in preventing infections caused by Staphylococcus aureus, one of the main species responsible for surgical site infections (SSIs).
Conclusion
Based on the results obtained, it can be concluded that the methodology developed in this study was effective for producing an autologous fibrin sealant using a single plasma unit as the source of fibrinogen (cryoprecipitate) and prothrombin, the latter converted into thrombin using 0.2 M calcium carbonate. This novel technique offers a promising and cost-effective alternative for laboratory-scale production of autologous fibrin sealants within Brazilian blood centers. However, the challenge of standardizing the concentrations of protein components remains a limiting factor in the formulation process. The descriptive statistical analysis of thrombin concentration measurements reveals the mean and modal concentrations at 388.66 and 482.32 IU/mL, respectively. The fibrin matrix was able to inhibit local bacterial growth for up to 72 hours. The incorporation of vancomycin hydrochloride and gentamicin sulfate into the fibrin sealant, followed by the observed inhibition of Staphylococcus aureus growth, suggests that this blood-derived product may contribute to the prevention of surgical site infections (SSIs). Nevertheless, further studies are required to assess the effectiveness of the incorporated antimicrobial agents against other SSI-causing pathogens. The development of a simple, low-cost method for producing a fibrin matrix is highly relevant not only to clinical and surgical applications but also to broader areas of research and therapeutic innovation. Given the versatility of this biological material, there is considerable potential for its use in pharmaceutical technology and regenerative medicine, particularly as a platform for drug delivery systems and human cell incorporation.
- WD Spotnitz (2014) Fibrin Sealant: The Only Approved Hemostat, Sealant, and Adhesive-a Laboratory and Clinical Perspective. ISRN Surg p. 1-28.
- MR Jackson (2001) Fibrin sealants in surgical practice: An overview. Am J Surg 182(2): S1-S7.
- B Bujoli, J-C Scimeca, E Verron (2019) Fibrin as a Multipurpose Physiological Platform for Bone Tissue Engineering and Targeted Delivery of Bioactive Compounds, Pharmaceutics 11(2019): 556.
- R Squires (2023) Fibrin sealant as a delivery vehicle for cells, antibiotics, growth factors, and painkillers. J Appl Biotechnol Bioeng 10(2): 56-64.
- WD Spotnitz, S Burks (2008) Hemostats, sealants, and adhesives: components of the surgical toolbox. Transfusion 48(7): 1502-1516.
- P Heher, S Mühleder, R Mittermayr, H Redl, P Slezak (2018) Fibrin-based delivery strategies for acute and chronic wound healing. Adv Drug Deliv Rev 129: 134-147.
- T Burnouf, C‐Y Su, M Radosevich, H Goubran, M El‐Ekiaby (2009) Blood‐derived biomaterials: fibrin sealant, platelet gel and platelet fibrin glue. ISBT Sci Ser 4(1): 136-
- JW Weisel (2005) Fibrinogen and Fibrin. Advances in Protein Chemistry 70: 247-299.
- P Goczyńska, J Lasocka, E Lachert (2021) Fibrin glues - the current state of knowledge. J Transfus Med 14: 214-224.
- M Beudert, M Gutmann, T Lühmann, L Meinel (2022) Fibrin Sealants: Challenges and Solutions. ACS Biomater Sci Eng 8: 2220-2231.
- E Anitua, A Pino, R Prado, F Muruzabal, MH Alkhraisat (2024) Biochemical and biomechanical characterization of an autologous protein-based fibrin sealant for regenerative medicine. J Mater Sci Mater Med 35: 15.
- K Tofuku, H Koga, M Yanase, S Komiya (2012) The use of antibiotic-impregnated fibrin sealant for the prevention of surgical site infection associated with spinal instrumentation. Eur Spine J 21: 2027-2033.
- JD Cashman, JK Jackson, C Mugabe, S Gilchrist, HM Burt, et al. (2013) The use of tissue sealants to deliver antibiotics to an orthopaedic surgical site with a titanium implant. J Orthop Sci 18(1): 165-174.
- (2024) Agência Nacional de Vigilância Sanitária (Brasil) Farmacopeia Brasileira 7a edição: volume II - Hemocomponentes e hemoderivados - versão RDC no 940/2024 – VIGENTE 62.
- T Burnouf (2018) An overview of plasma fractionation. Ann Blood 3: 33-33.
- I Mackie, A Casini, M Pieters, R Pruthi, C Reilly‐Stitt, et al. (2024) International council for standardisation in haematology recommendations on fibrinogen assays, thrombin clotting time and related tests in the investigation of bleeding disorders. Int J Lab Hematol 46(1): 20-
- Ministério da Saúde, Portaria De Consolidação No 5, De 28 De Setembro De 2017. Consolidação das normas sobre as ações e os serviços de saúde do Sistema Único de Saúde pp. 856.
- J Callum, ME Farkouh, DC Scales, NM Heddle, M Crowther, et al. (2019) Effect of Fibrinogen Concentrate vs Cryoprecipitate on Blood Component Transfusion After Cardiac Surgery. JAMA 322(20): 1966-1976.
- M Dietrich, J Heselhaus, J Wozniak, S Weinandy, P Mela, et al. (2013) Fibrin-Based Tissue Engineering: Comparison of Different Methods of Autologous Fibrinogen Isolation. Tissue Eng Part C Methods 19(3): 216-226.
- A Noori, SJ Ashrafi, R Vaez-Ghaemi, A Hatamian-Zaremi, TJ Webster (2017) A review of fibrin and fibrin composites for bone tissue engineering. Int J Nanomedicine 12: 4937-4961.
- YT Pinheiro, ECL Da Silva, MA Maciel, ET de Sousa (2017) Hemofilias e Doença de von Willebrand: uma revisão de literatura. Arch Heal Investig 6(5).
- Chronolab Systems SL (2020) APTT Ellagic acid: Quantitative determination of Activated Partial Thromboplastin test. Sant Climent de Llobregat Spain.
- J Gessmann, D Seybold, F Ayami, E Peter, H Baecker, et al. (2018) Peripheral Blood Plasma Clot as a Local Antimicrobial Drug Delivery Matrix. Tissue Eng Part A 24: 809-818.
- E Semple, T Madsen, JW Semple (2005) Quality of thrombin produced from the patient’s own plasma using the TPD, a new Thrombin-processing Device. J Extra Corpor Technol 37(2): 196-200.
- V Kumar, JR Chapman (2007) Whole blood thrombin: development of a process for intra-operative production of human thrombin. J Extra Corpor Technol 39(1): 18-23.
- P Aizawa, S Winge, G Karlsson (2008) Large-scale preparation of thrombin from human plasma. Thromb Res 122(4): 560-567.






























