Efficacy of Bridelia ferruginea Stem Bark Aqueous Extract as Anaesthesia on Clarias gariepinus (Burchell 1822) Juvenile
Nwakpa J N1, Awoke J S2,3*, Agim M U4, Nkwuda P J2 and Ogueji E O2
1Department of Fisheries and Aquaculture, Ebonyi State University, Abakaliki, Nigeria
1Department of Fisheries and Aquaculture, Alex Ekwueme Federal University Ndufu Alike, Nigeria
1Department of Biology Education, Ebonyi State College of Education, Ikwo, Abakaliki, Nigeria
1Department of Agricultural Education, Federal College of Education Obudu, Cross River State, Nigeria
Submission:September 19, 2025; Published: September 26, 2025
*Corresponding author:Awoke J S, Department of Fisheries and Aquaculture, Alex Ekwueme Federal University Ndufu Alike, Nigeria
How to cite this article:Nwakpa J N, Awoke J S, Agim M U, Nkwuda P J, Ogueji E O. Efficacy of Bridelia ferruginea Stem Bark Aqueous Extract as Anaesthesia on Clarias gariepinus (Burchell 1822) Juvenile. Adv Biotech & Micro. 2025; 19(2): 556007.DOI:10.19080/AIBM.2025.19.556007
Abstract
This study investigated the efficacy of Bridelia ferruginea stem bark aqueous extract as an anaesthetic agent and its effect on the haematology of Clarias gariepinus. 150 healthy C. gariepinus juveniles were exposed to different concentrations (20, 25, 30, 35 mg/l) of B. ferruginea stem bark aqueous extract. Fish behaviour was observed and time taken for complete immobilization and recovery from anaesthesia was recorded at the different stages and concentrations with a digital stopwatch. Blood samples were collected from the test fish before and after the exposure to check for any alterations in the blood parameters. Results from the test showed that the induction time decreased significantly with increase in concentration of B. ferruginea stem bark aqueous extract with the shortest induction time of 137±28 s and recovery time of 224±32 s. Furthermore, there were no significant changes on the haematological profiles of the exposed fish. All the haematological parameters assessed in this study were within the recommended physiological ranges for C. gariepinus. The findings show that B. ferruginea stem bark aqueous extract is a good anaesthetic agent. Hence, concentration range of 20-35 mg/l aqueous extract of B. ferruginea stem bark is recommended for use as an anaesthetic for C. gariepinus.
Keywords: Anaesthesia, Bridelia ferruginea, Bark aqueous extract, Induction, Recovery
Abbreviations: MS: Methanesulphonate; CRD: Completely Randomized Experimental Design; EDTA: Ethylene Diamine-Tetra Acetic Acid; PCV: Packed Cell Volume; RBC: Red Blood Cell; WBC: White Blood Cell; PLT: Platelet; LYMP: Lymphocyte; MCH: Mean Cell Haemoglobin; MCV: Mean Cell Volume; MCHC: Mean Cell Haemoglobin Concentration; EBSU: Ebonyi State University; NESREA: National Environmental Standard Regulations Enforcement Agency
Introduction
Bridelia ferruginea is a member of the family Euphorbiaceae and a native medicinal species in Africa, Asia and Australia [1]. The plant is a shrub of about 15m high with curved stem and up to 1.8m in girth. It is characterized by branches that are long and thin with short spines [2]. It is commonly found in the savannah, though often present in the forest vegetation. It bears fruits that are drupe-shaped with green or blue pericarp when mature. The stem bark of B. ferruginea is dark grey, cracked, rough and even markedly scaly [3]. B. ferruginea plant (roots, bark, fruits and leaves) is composed of chemical compounds. It is reported that phytochemicals found in extracts obtained in different parts of B. ferruginea contain phenolics, phytosterol, glycosides, tannins, flavonoids, alkaloids, saponins and triterpenes [4-7]. [8,9] particularly reported that phytochemicals such as quinones, gallic acid, sterols, alkaloids, polyterpenes, polyphenols, saponisides, reducing sugars, flavonoids and catechic tannins were found in aqueous stem bark extract of B. ferruginea. In traditional African medicine, B. ferruginea decoctions are often used to manage medical conditions like diabetes, arthritis, dysentery, constipation, diarrhea, thrush, contusion, intestinal disorder and as antidote for arrow poison in West Africa [10,1]. However, these phytochemicals found in the aqueous extracts of B. ferruginea are also known to exhibit anaesthetic properties [7,11].
Anaesthesia is a biological state in which there is partial or complete loss of sensation or loss of voluntary neuromotor control induced by chemical or nonchemical means [12,13,14]. Anaesthetic agents have become indispensable in present-day aquaculture [15]. This is because aquaculture operations such as transportation, sorting, tagging, grading, vaccination and artificial fish propagation procedures often expose fish to handling. These practices subject fish to stressful conditions which negatively impact their health and general wellbeing. Anaesthetic agents are often used to reduce pain in fish and bring a calming effect followed by lack of equilibrium, kinesis and consciousness during such operations [16,17]. They can as well be employed to restrain fish in order to grip them more effortlessly for the period of harvesting, sampling, vaccination and spawning procedures [18]. In aquacultural research, requiring surgical operations and physiological investigations where the fish must be held motionless for extensive periods, anaesthetic agents are often used to mitigate stress, reduce mortality and diminish stressinduced problems such as decrease in food intake and immunity [19,20,21].
[22] reported three criteria that an anaesthetic applied in aquaculture must accomplish. The anaesthetic must be effective, safe and inexpensive. The criteria for efficacy includes that, the fish must be sedated within 3 minutes and after 15 minutes of exposure to anaesthetic agent, fish must regain normal swimming in 10 minutes. More so, all anaesthetized fish must survive. The effect of an anaesthetic on fish depends on a number of factors such as concentration of an anesthetic agent, water temperature, fish size and species [23]. The anaesthetic agent must not have toxic side effects on either the fish or the handler. It should be biodegradable and have properties which allow the body to clear it from the tissues following exposure. It should have no persisting physiological, immunological or behavioural effects which could reduce the likelihood of survival of the fish or interfere with later measurements [24]. It is imperative that preliminary tests be performed with small numbers of the fish to determine the optimal dosage and exposure. Due care should be taken to control the level of anaesthesia desired, through the application of appropriate concentration, and to maintain constant observation of fish as they go through the various stages of anaesthesia [25- 27].
According to [24] two categories of anesthetic agents are used in aquaculture. They are synthetic and natural anaesthetics. Synthetic or chemical agents are proscribed because of safety issues and residues, while natural agents are more developed and projected to have greater potential prospects [21,24]. Several synthetic agents frequently used to anaesthetize fish include: tricaine methanesulphonate (MS 222), quinaldine sulphate, benzocaine, phenoxyethanol, Isoflourane, propoxate, metomidate and etomidate [28]. Naturally occurring anaesthetic agents commonly derived from plant extract include clove oil (Eugenia aromatica) [29-32], seeds from the fish poison tree (Baringtonia asiatica) [33], rubber seeds (Hevea brasiliensis) [34] and tuba roots (Derris elliptica) [35,36]. Furthermore, Derris (rotenone), tephrosia (tephrosin), Erythrophleum (alkaloid and tanin), Pyrus (tannin and saponin) and Tobacco (nicotine) have also been reported to have anaesthetic effects on fish [37,38,39]. Natural anaesthetic agents have been shown to be better than synthetic anaesthetics in several ways [40]. They have higher activity, leave behind no residues after use, have relatively fewer environmental impacts, more effective at lower concentrations, relatively cheaper, readily available and do not impact on the taste/flesh of fish [24].
The African Catfish, Clarias gariepinus belongs to the family Clariidae (Air breathing catfishes), order Siluriformes (catfish). There are over hundred species in this family occurring naturally throughout most of Africa and the Southern half of Asia to Java and the Philippines [41]. The experimental fish, C. gariepinus was used for this study because it is the most sought-after species among fish farmers and consumers in Nigeria. Many of the species are of great economic importance in both fisheries and fish culture. Its culture is becoming more fashionable among fish farmers in Nigeria because they can be mass produced through artificial propagation procedures in the hatchery. Thereafter, these hatcheries transport the fry, fingerlings and juveniles of C. gariepinus to different farms scattered in far and remote parts of the country for rearing to table sizes. Anaesthesia is often used to calm the fish, reduce mortalities and ensure a safe and successful transportation to the fish farms. Due to the inherent advantages of natural anaesthetic agents over synthetic anaesthesic, there is need to investigate other cheaper, easily available and local botanical sources of anaesthesic such as B. ferruginea bark aqueous extract as alternative anaesthetic agent for use in aquacultural operations.
In ecotoxicological investigations, haematological parameters are often considered as the most reliable biomarkers in toxicity testing of organisms. This is because blood parameters adjust very quickly with changes in activities around the fish [42]. So, they are considered pathophysiological indicators of the whole body and are essential in the diagnosis of morphological and health condition of fish exposed to toxicants [43]. B. ferruginea stem bark aqueous extract contains phytochemicals of plant origin. Information about its sedative capabilities is still very scanty, though it is used locally in traditional medicine to treat several ailments in humans. However, [44] and [45] had investigated its toxicity effect on the blood parameters of rodents but no research has been carried out on its toxicity on aquatic organisms like fish, especially now that it is being considered as a novel anaesthetic for fish culture. To this end, it is imperative to investigate if B. ferruginea stem bark aqueous extract has any adverse effect on the haematological characteristics of C. gariepinus.
Furthermore, there has been a trend towards the use of non-synthetic or botanical anaesthetic agents in aquaculture management [26,46]. This has stimulated a renewed interest in anaesthetic research and the search to develop ‘green anaesthetic’ with little ecological and health risks [40]. Research into the enhancement of anesthesia through the use of agents such as clove oil, and other plant extracts are veritable alternatives to chemical anesthesia [11]. Therefore, this study is aimed at determining the effectiveness of B. ferruginea bark aqueous extract as an anaesthetic agent and any possible toxicological effect on the haematological profiles of C. gariepinus juvenile when exposed to different concentrations of the anaesthetic agent.
Materials and Methods
Procurement of experimental fish
A total of 180 C. gariepinus juvenile were used for the study. The experimental fish were procured from Regina Pacis Fish Farm in Abakaliki and transported to the Wet Laboratory Unit of the Department of Fisheries and Aquaculture, Ebonyi State University. The experimental fish were acclimated in plastic tanks for seven days, before the commencement of the experiment. Water was changed every two days, and the experimental fish were fed with commercial feed at 5 % body weight twice daily by 8:00 am and 6:00 pm each day. No mortality was recorded during the period of acclimation and feeding was discontinued 12 hours before the commencement of the experiment.
Plant collection and Extract Preparation
The fresh stem bark of B. ferruginea plant (Figure 1) was collected from Ntsuruakpa bush in Ezzamgbo, Ohaukwu Local Government Area Ebonyi State, Nigeria. Botanical identification and authentication were done by a plant scientist in the Department of Crop Science and Landscape Management, Faculty of Agriculture and Natural Resource Management, Ebonyi State University, Abakaliki. A voucher specimen was later deposited in the herbarium of Ebonyi State University, Abakaliki. Fresh B. ferruginea stem bark were cut into pieces and dried at room temperature of 25°C for two weeks then pulverized into powder using grinding machine and sieved with 100-micron net to obtain a fine powder. Fifty grams of ground stem bark of B. ferruginea were weighed into a conical flask and 100mls of distilled water, mixed and shaken prior to filtration by means of a dried Whatman No.1 filter paper into a graduated 1 litre measuring cylinder to obtain cold water extract. The extract was decanted 24 hour later and the filtrate was concentrated and evaporated to dryness at 60°C, using rotary evaporator (Stuart Barloworld, Model RE 300). Thereafter, the extract which has a dark brown color was stored at 20°C in an air-tight container pending further tests.

Experimental design
A completely randomized experimental design (CRD) was adopted for this research. At the end of the acclimation period, 10 juvenile fish were randomly distributed into the 30L plastic aquaria tanks in three replicates containing varying concentrations of aqueous extract of B. ferruginea stem bark and a control containing water without aqueous extract of B. ferruginea. The treatment groups consisted of 0.00 mg/l (control), 20 mg/l, 25 mg/l, 30 mg/l and 35 mg/l,. Each treatment had a total of 30 juveniles in the five treatments while the total fish used was 150 juveniles.
Water quality parameters
For the duration of the study, the following water quality parameters were monitored in the experimental and control tanks: temperature, pH, dissolved oxygen, alkalinity, conductivity and total hardness. The water temperature was measured using the mercury in glass thermometer. Conductivity was assessed with conductivity meter (PACM 35 Model) and pH was measured with pH meter (Model 3015 Jenway). Dissolved oxygen was measured with a digital oxygen meter. Alkalinity was determined by standard methods [47]. Total hardness was determined by ethylene diamine-tetra acetic acid (EDTA) titration method.
Exploratory test
Preliminary screening was carried out to determine the appropriate and optimal concentration range for the test chemical. The concentrations of the aqueous extract of B. Ferruginea were prepared and tested on fish for range finding. Four concentrations were used to determine induction time (anaesthesia) of aqueous extract of B. Ferruginea following the method of [26]. The concentrations were 1 mg/l, 10 mg/l, 50 mg/l, 100 mg/l. Four plastic bowls (30L) were cleaned and randomly labeled and each filled with 20L of borehole water for induction test and 5L of water for recovery. The different concentrations were prepared by serial dilution of the stock solution. The mixture was stirred with a glass rod for homogeneous mixing. Each treatment was stocked with 10 fingerlings and monitored for the onset of induction (anaesthesia) for 30 minutes as a period greater than this was considered impractical for routine fish handling procedures [27,48]. At the end of the exploratory test, mortality was recorded from the concentration of 50 mg/l and above. This shows that the concentration from 50 mg/l and above were higher concentration that should not be used for anaesthesia on C. gariepinus. Test fish in the concentration less than 50 mg/l were induced within 30 minutes and recovered within 3 minutes. As a result of this, the following concentrations 20 mg/l, 25 mg/l, 30 ml/l, 35 mg/l were selected to induce the fish.
Determination of induction and recovery times of experimental fish
After the range finding test, lower concentrations within the range of 20 mg/l, 25 mg/l, 30 mg/l, 35 mg/l were used for the definitive test. Plastic aquaria tanks (30L) were cleaned and randomly labeled and each filled with 10L of borehole water for induction test and 5L of water for recovery. The different concentrations were prepared by serial dilution of the stock solution. The mixture was stirred with a glass rod for homogeneous mixing. Each treatment was stocked with 10 fingerlings in triplicates and monitored for the onset of induction (anaesthesia). The test fish were observed following the various stages of induction and recovery time using a digital stopwatch. Any fish that lost balance and ceased respiratory movement of the opercula i.e. stage IV (deep anaesthesia) was removed at once and transferred to 5L of B. Ferruginea extract free water. Alterations in the physiological condition and behavioural responses of the anaesthetized fish were assessed in four successive stages for induction and three stages for recovery as described by [27] (Table 1). The time taken to reach each pre-determined stage of anaesthesia was recorded. None of the revived fish was re-used for further experimentation but were kept in another bowl to determine the fastest and most effective concentration. The time for the fish to enter the desirable anaesthesia level (induction) and that which is required for an anaesthetized fish to regain equilibrium and begin active swimming (recovery time) were recorded for each concentration. The efficacy of B. ferruginea stem bark aqueous extract was decided on the time taken to reach stage IV (deep anaesthesia) and the recovery time. The induction time is defined as the time taken from the moment the fish is exposed to the anaesthetic to the moment respiratory movement of the opercula stopped. Recovery time is defined as the time taken from the moment the fish was considered anaesthetized until the moment regular regular respiratory movements were resumed. [25] recommended desirable induction and recovery times of anesthesia of fish as 180 and 300 seconds respectively.

Haematological analysis
Prior to commencing of the experiment, baseline blood samples were collected from five fish randomly selected from all the fish for haematological analysis. This served as a control. At the end of the experiment, 2ml of blood samples were collected from each treatment (concentration) for haematological analysis. Blood was drawn from the caudal peduncle of the fish using a disposable needle and syringe into an EDTA sample bottle to avoid clotting. Packed cell volume (PCV) was analyzed with microhaematocrit by means of heparinized 25mm capillary tubes. Red blood cell (RBC), white blood cell (WBC) platelet (PLT), lymphocyte (LYMP) counts were analyzed as described by [49].
While haemoglobin concentration (Hb) was estimated using the method outlined by [50]. Other haematological indices like mean cell haemoglobin (MCH), mean cell volume (MCV) and mean cell haemoglobin concentration (MCHC) were determined using the formular put forward by [51] thus:
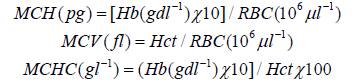
Statistical analysis
Data obtained from the various stages of induction and recovery times as well as haematological indices of the experimental fish were subjected to statistical analysis. Differences among time for dosage to achieve various stages of anesthesia and recovery time were subjected to one way analysis variance (ANOVA). The significance of difference between means was determined by Duncan’s Multiple Range Test (p<0.05) using SPSS for windows (version 21). Values were expressed as means ± SE.
Results
Water quality parameters of test water
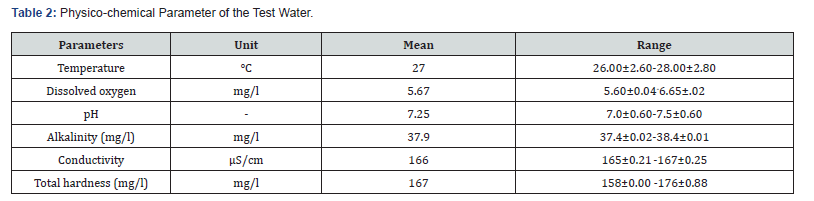
Result of water quality parameters recorded during the experiment is presented in (Table 2). There was no significant difference (p>0.05) in the mean values from those of the control parameters. Mean water parameters obtained during anaesthetic exposure of juveniles of C. gariepinus gave values of various ranges of water quality parameters. Temperature ranged from 26.00±2.60-28.00±2.80°C, pH ranged from 7.0±0.60-7.5±0.60, Dissolved oxygen (DO) ranged from 5.60±0.04-6.65±.02 mg/l, Alkalinity ranged from 37.4±0.02-38.4±0.01, mg/l, Conductivity ranged from 165±0.21-167±0.25 μS/cm and Total Hardness ranged from 158±0.00-176±0.88 mg/l throughout the experimental period.
Induction and recovery times of c. gariepinus juveniles anaesthetized with b. ferruginea stem bark aqueous extract
Result of the various time (s) required for Induction and recovery of C. gariepinus juveniles anaesthetized with B. ferruginea stem Bark Aqueous Extract are shown in (Table 3). Fish that were exposed to less than 30ml/l of aqueous extract of B. ferruginea were completely immobilized in less than 246±86 s. While fish exposed at 30ml/l and 35ml/l were immobilized faster at less than 150±29 s. Immediately after transferring those into the recovery tanks normal locomotive activity were observed within 194±12 s to 224±32 s. The increase in the concentration of aqueous extract of B. ferruginea proportionally decreased the time required for sedation (induction). This result shows that, there was significant difference (P>0.05) in the time required for the stages of induction of the test fish in the different concentrations of aqueous extract of B. ferruginea.
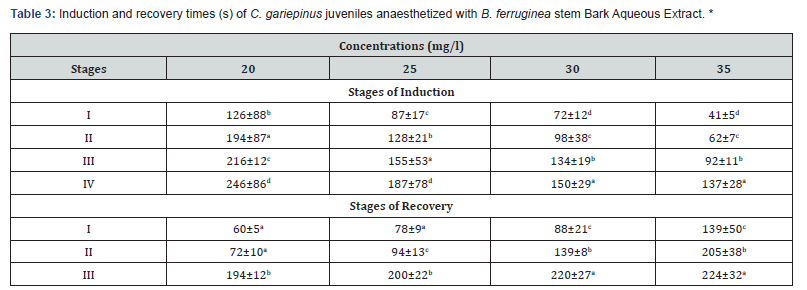
The time of recovery were significantly fast following the exposure from the lowest concentration (20-35 ml/l). There was no uniform sedation of the catfish (C. gariepinus) fingerlings exposed to different concentration of aqueous extract of B. ferruginea. There was also a variation in time of recovery by the fish exposed to the aqueous extract as the concentration increased, the recovery time equally increased from (194±12- 224±32 s). This finding has also shown that, the time required for the different stages of recovery of the test fish induced in different concentrations of aqueous extract of B. ferruginea was significantly different (P>0.05). So, induction and recovery times were dose dependent. No mortality were recorded from the lowest concentration to the highest concentration, there was 100% survival rate. The sequential progression through the various concentration of the anesthesia with increasing dosage, time of induction and the recovery of anesthetized fish, all followed the pattern of typical fish anaesthetic according to [25].
Haematological responses of c. gariepinus juveniles anaesthetized with b. ferruginea stem bark aqueous extract
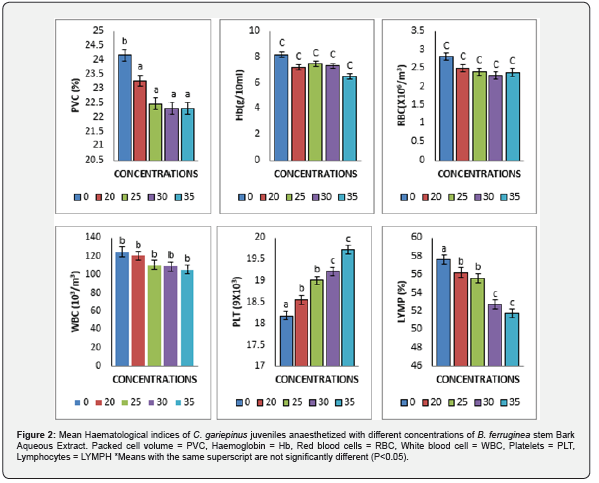
The result of mean values of haematological parameters of C. gariepinus juveniles exposed to different concentrations of B. ferruginea stem bark aqueous extract are shown in (Figure 2). The values were compared with the corresponding values in the control. Mean values of PVC, PLT and LYMP showed a significant (P<0.05) reduction from the control as the concentration of B. ferruginea stem bark aqueous extract increased. Mean values of packed cell volume (PCV) decreased from 24.17±2.81-21.25±1.22, platelets (PLT) reduced from 124.50±9.16-105.47±7.68, while lymphocytes (LYMP) decreased from 57.63±4.13-51.73±2.63. There was no significant (P>0.05) change in mean values obtained for Hb, RBC. WBC. The mean values recorded for haemoglobin (Hb) reduced from 8.18±0.87-6.48±0.40, red blood cell (RBC) decreased from 2.80±0.53-2.38±0.42 and mean values obtained for white blood cells (WBC) increased from 18.18±1.84-19.72±1.64.
Discussion
The use of anti-stress chemicals is unavoidable in present day fish culture. However, [52] posited that certain natural factors enhance or diminish the efficacy of an anaesthetic agent on the test organism. Such factors are age, size, weight, species, phase of life cycle, fat content as well as disease status of the organism. Others are ecological factors such as temperature and pH [53,13]. These factors impinge on the metabolic rate of the organism and indirectly the pharmacokinethics of the anaesthetic chemical [17].
In the present study, induction time recorded reduced significantly (P>0.05) as the dosage of the aqueous extract of B. ferruginea stem bark increased. While the recovery time of the fish increased notably with the increase in the concentration of the aqueous extract of B. ferruginea bark Table 3. Furthermore, there was significant difference (P>0.05) in the induction time and recovery time among the different concentrations of B. ferruginea stem bark aqueous extract. This study has revealed that stem bark aqueous extract of B. ferruginea stem bark acted as an anaesthetic agent in sedating juveniles of C. gariepinus. [25] posit that an anaesthetic agent is regarded as being effectual if it gives a total induction within 180 s and recovery within 300 s for fish. Therefore the concentration range of 20-35 mg/l of B. ferruginea stem bark aqueous extract has achieved the recommended efficacy criteria of complete anaesthesia induction within 180 s and recovery within 300 s. In the present study, exposure of fish to 20-35 mg/l of aqueous extract of B. ferruginea stem bark resulted in rapid induction, total immobilization and quick recovery in C. gariepinus juveniles. The results are in conformity with previous studies on bony fishes [54-57,17].
The present finding had a high correlation between anaesthetics dosage and recovery time. Smaller doses of the aqueous extract of B. ferruginea stem bark could be utilized in sedating fish for a longer period. This trend followed the typical patterns of fish anaesthetic as reported by [25,13,38,21]. This study is also in agreement with the observation of [46,42] who reported that fish exposed to anaesthetics usually exhibit decreased opercula movements, erratic swimming movements, loss of equilibrium and lethargies. The qualities required of an anaesthetic agent to sedate fish varies, depending on nature, mode of application and fish species. Most importantly, it should have a quick induction and recovery time which allows for maximum manipulation of fish in aquaculture. It should be cheap, safe, easy to handle, readily available and accessible to the fish farmers in different parts of the country. In the light of present finding, stem bark aqueous extract of B. ferruginea has a rapid induction and recovery time. It is cheap, safe, readily available and accessible to the fish farmers.
Haematological indices are useful in monitoring toxicity especially with plant or herbal constituents that affect blood formation. They are veritable parameters that reveal the response of the animal to dynamics of its external environment. In this study, all the haematological parameters assessed were within the recommended physiological ranges documented for C. gariepinus. Though the mean values of packed cell volume (PVC), platelets (PLT) and lymphocytes (LYMP) were significantly (P<0.05) reduced from the control, but the values are within acceptable optimal limits for the rearing C. gariepinus. Therefore these did not constitute an impediment to the test fish health. The mean values recorded for Hb, RBC, and WBC in this study were not significantly different from the control. This is an indication that the aqueous extract of B. ferruginea stem bark did not adversely affect the haematological parameters of the test fish. This finding agrees with the earlier work by [45] who reported that administration of aqueous extract of B. ferruginea stem bark showed a non-significant increase in red blood cell, hemoglobin, platelet and the packed cell volume of the treated rodents except at higher dose above (1000 mg/kg), where there was a slight decrease when compared with control group. According to [45], absence of significant changes on RBC, Hb, PCV, WBC, PLT, LYMP of treated rats might be an indication that administration of the extract did not lead to damage of mature red blood cells. The result of this study equally aligns with the report of [44] that low concentrations of aqueous extracts of B. ferruginea stem bark did not significantly alter the haematology profile of albino rats.
Fish used for this study were closely monitored for five days after the end of the experiment. No mortality was recorded during this period and fish showed normal swimming activity. This is an indication that the aqueous stem bark extract of B. ferruginea had no undesirable toxic effects on the fish. This aligns with the findings of [58] that aqueous stem bark extracts of B. ferruginea was relatively safe when administered on rodents. However, the pharmacokinetics and pharmacodynamics of the phytochemicals in aqueous stem bark extract of B. ferruginea is not understood and need to be elucidated. Therefore, further researches are required to discover the active ingredient in B. ferruginea that exerts this sedative property and any possible side effects on physiology of fish.
Conclusion
The use of aqueous extract of B. ferruginea bark as an anaesthetic agent in sedating juveniles of C. gariepinus is efficacious. It is a veritable alternative to other conventional or synthetic fish anaesthetics. Moreover, it is cheap, environmentally safe, easy to handle, readily available and accessible to the fish farmers even in remote parts of the country. Furthermore, the recovery time is shorter than other conventional anaesthetics. Therefore, aqueous stem bark extract of B. ferruginea is highly effective as an anaesthetic and its effect was found to be reversible within the concentration of 20-35 ml/l. From the findings of this study, there is strong recommendation of the use of aqueous extract of B. ferruginea bark as an anaesthetic agent for fish culture by local fish farmers.
Ethical Approval
All experimental trials were approved by the Ebonyi State University (EBSU) Abakaliki ethics clearance committee. This was done in accordance with the university’s animal welfare act and the National Environmental Standard Regulations Enforcement Agency (NESREA) Act of Nigeria on the protection of animals against cruelty.
- Yeboah GN, Owusu FWA, Archer M, Kyene MO, Kumardoh D, et al. (2022) Bridelia Ferruginea Benth; An ethnomedicinal, phytochemical, pharmacological and toxicological review. Heliyon 8(8): e10366.
- Afolayan M, Srivedavyasasria RO, Asekunb OT, Familonib OB, Rossa SA (2019) Chemical and biological studies on Bridelia ferruginea grown in Nigeria. Nature Productivity Research 33(2): 287-291.
- Nene-Bi SA, Ramachandran V, Vengal RP, Gopalakrishnan R, Dhanabal SP, et al. (2016) Subchronic toxicity studies of the aqeous stem bark extract of Bridelia ferruginea in Wister rats. Bulletin of Environmental Pharmacology and Life Science 5(10): 14-21.
- Oliver-Bever B, (1986) Medicinal Plants in Tropical West Africa, Cambridge University Press 2. London, UK. 75: 99-106.
- Cimanga K, DeBruyne T, Apers S, Dieters L, Totte J, et al. (1999) Complement-inhibiting constituents of Bridelia ferruginea stem bark. Plant Medicine 65(3): 213-217.
- Rashid MA, Gustafson KR, Cardellina JH, Boyd MR (2000) A New Podophyllotoxin Derivative from Bridelia, Academic Press, Inc. 3: 139-152.
- Okwu DE, Ukanwa N (2010) Isolation and characterization of flavonoids chalcones and anthocynidines from Bridelia ferruginea Der Chemica Sinica 1: 21-28.
- Néné-Bi SA, Zahoui OS, Soro TY, Traoré FL (2012) Diuretic and Natriuretic Activity of an Aqueous Extract of Bridelia Ferruginea (Euphorbiaceae) in Normal Rats. Journal of Physiology and Pharmacology Advances 2(10): 349-353.
- Owoseni AA, Ayanbamiji TA, Ajayi YO, Ewegbenro IB (2010) Antimicrobial and phytochemical analysis of leaves and bark extracts from Bridelia ferruginea. African Journal of Biotechnology 9(7): 1031-1036.
- Olajide OA, Aderogba MA, Okorji PU, Fiebich BL (2012) Bridelia ferruginea Produces Antineuroinflammatory Activity through Inhibition of Nuclear Factor-kappa B and p38 MAPK Signalling. Hindawi Publishing Corporation Evidence-Based Complementary and Alternative Medicine pp. 546873.
- Tsuchiya H (2017) Anesthetic Agents of Plant Origin: A Review of Phytochemicals with Anesthetic Activity. Molecules 22(8): 1369.
- Iwama GK, Pickering AD, Sumpter JP Schreck CB (1997) Fish stress and health in aquaculture. Cambridge University Press UK pp. 278.
- Ross LG, Ross B (1999) Anaesthetic and Sedative Techniques for aquatic animals. Oxford: Blackwell Science Fish. University of Sterling, Scotland: Institute of Aquaculture p. 35-52.
- Kanani G, Mirzargar SS, Soltani M, Ahmadi M, Abrishamifar A, et al. (2011) Anesthetic effect of tricane methanesulfanate, clove oil and electroanesthetic on lysozyme activity of Oncorhynchus myksis. Iranian Journal of fisheries Science 10: 393- 402.
- Pawar HB, Sanaye SV, Sreepada RA, Harish V, Suryavanshi U, et al. (2011) Comparative efficacy of four anaesthetic agents in the yellow seahorse, Hippocampus kuda (Bleeker, 1852). Aquaculture 311(1-4): 155-161.
- Summerfelt RC, Smith LS (1990) Anaesthesia, Surgery and related techniques. In: C.B. Scherelk and P.B. Moyle (eds) Methods for fish biology. American Fisheries Society Bethesda Maryland pp. 213-272.
- Ogretmen F, Gokcek K (2013) Comparative Efficacy of Three Anesthetic Agents on Juvenile African Catfish, Clarias gariepinus (Burchell, 1822). Turkish Journal of Fisheries and Aquatic Sciences 13: 51-56.
- Coyle SD, Durborow RM, Tidwell JH (2004) Anesthetics in Aquaculture Kentucky State University Aquaculture Research Center. Southern Regional Aquaculture Center (SRAC) Publication No. 3900.
- Opiyo MA, Ogello EO, Charo Karisa H (2013) Effectiveness of Sodium Bicarbonate as an Anesthetic for different size of Nile tilapia, Oreochromis niloticus Juveniles. International Journal of Aquatic Science 4(1): 14-22.
- Husen MA, Sharma S (2015) Anaesthetics Efficacy of MS-222, Benzoak® vet, AQUI-S® and Clove oil on Common Carp (Cyprinus carpio) Fry. International Journal of Research in Fisheries and Aquaculture 5(3): 104-114.
- Okey IB, Keremah RI, Gabriel UU (2018) The efficacy of clove (Eugenia caryophyllata) powder as anaesthesia on African catfishes (Clarias gariepinus and Heterobranchus bidorsalis) fingerlings. Journal of Aquaculture and Marine Biology 7(4): 182-
- Gilderhus PA, Marking LL (1987) Comparative efficacy of 16 anesthetic chemical in rainbow trout. North American Journal of Fisheries management 7(2): 288-292.
- Velisek J, Wlasow T, Gomulka P, Svobodova Z, Novotny L (2007) Effects of 2-phenoxyethanol anaesthesia on sheatfish (Silurus glanis L). Veterinarni Medicina 52(3): 103-110.
- Purbosaria N, Warsikic E, Syamsuc K, Santosod J (2019) Natural versus synthetic anesthetic for transport of live fish: A review. Aquaculture and Fisheries 4(4): 129-133.
- Marking LL, Meyer FP (1985) Are better anesthetics needed in fisheries. Fisheries 10(6): 2-5
- Bell G (1987) An outline of anesthetics and anesthesia for salmonids, a guide for fish culturists in British Columbia can Tech rep fish aquat sci. Biotechnology 8: 650-660.
- Iwama GK, McGeer JC, Pawluk MP (1989) The effects of five fish anaesthetics on acid-base balance, hematocrit, cortisol and adrenaline in rainbow trout. Canadian Journal of Zoology 67(8): 2065-2073.
- Sajevan S (2019) Anaesthesia and anaesthetics for the controlled management of freshwater Fishes Aquafind. Aquatic fish data base.
- Abdolazizi S, Ghaderi E, Naghdi N, Kamangar BB (2011) Effects of clove oil as an anesthetic on some hematological parameters of Carassius auratus. Journal of Aquaculture Research & Development 2(1): 1-3.
- Sindhu MC, Ramachandran A (2013) Acute toxicity and optimal dose of clove oil as anaesthetic for blue hill trout Barilius bakeri (Day). Fishery Technology 50: 280-283.
- Sutili FJ, Gressler LT, Baldisserotto B (2014) Clove oil, eugenol effective anesthetics for silver catfish, other Brazilian species. Global Aquaculture Advocate p. 71-
- Aydın I, Akbulut B, Kucuk E, Kumlu M (2015) Effects of temperature, fish size and dosage of clove oil on anaesthesia in turbot (Psetta maxima Linnaeus). Journal of Fisheries and Aquatic Science 15(4): 899-904.
- Septiarusli IE, Haetami K, Mulyani Y, Dono D (2012) Potensi senyawa metabolit sekunder Dari ekstrak biji buah keben (Barringtonia asiata) Dalma proses anestesi ikan kerapu macan (Ephinephelus fuscoguttatus). Jurnal Perikanan dan Kelautan 3(3): 295-299.
- Hasan H Farida, Ertiyasa G (2016) Different concentration of rubber seed extract (Hevea brasiliensi) for anesthetic to the survival of prospective brood stock milk fish by using closed transportation. Jurnal Ruaya 4(2): 55-
- Irawan O, Efendi E, Ali M (2014) Efek pelarut yang berbeda terhadap toksisitas ekstrak akar tuba (Derris elliptica). E-Jurnal Rekayasa dan Teknologi Budidaya Perairan 2(2): 259-265.
- Prariska D, Tanbiyaskur, Azha MH (2017) Toxicity test root extract Tuba (Derris elliptica) in Red Tilapia (Oreochromis sp). Jurnal Ilmu-ilmu Perikanan dan Budidaya Perairan 12(1): 41-48.
- Mgbenka BO, Ejiofor EN (1998) Effect of extract of dried leaves of Erythrophleum suaveolens as anaesthetics on Clariid Catfish. Journal of Applied Aquaculture 8(4): 73-80.
- Agokei OE, Adebisi AA (2010) Tobacco as an anaesthetic for fish handling procedures. Journal of Medicinal Plants Research 4(14): 1396-1399.
- Adebayo TO, Fasakin EA, Popoola OM (2010) Use of Aqueous Extracts of Avocado Pear, Pyrus Communis, Leaf as Anaesthetic in Gonadectomy of African Catfish, Clarias gariepinus. Journal of Applied Aquaculture 22(2): 117-122.
- Ramanayaka JC, Atapattu NSBM (2006) Fish Anaesthetic Properties of Some Local Plant Material Tropical. Agricultural Research & Extension p. 1-6.
- Teugels G, Sudarto G, Pouyaud L (2001) Description of a New Clarias Species from Southeast Asia Based on Morphological and Genetical Evidence (Siluriformes, Clariidae) Zoological Studies 25(1): 81-92.
- Olatunji AE, Sunday IK (2023) Effect of Aqueous Extract of Ichthyotoxic Plant Bridelia micrantha (Hochst) (Baill) on Haematology, Histopathology and Carcass Characteristics of African Catfish (Clarias gariepinus) (BURCHELL, 1822) Juvenile. Journal of Marine Science Research and Oceanography 6(3): 106-122.
- Salim AM, Yusuf MA, Bichi AH (2021) Haematological Responses of African Catfish (Clarias gariepinus, Burchell, 1822) Juveniles Exposed to Acute Concentrations of Butachlor (Herbicide). FUDMA Journal of Agriculture and Agricultural Technology 7(2): 110-115.
- Olarewaju OI, Oloyede OI, Ojo OA, Onikanni SA (2013) Effects of aqueous extract of Bridelia ferruginea stem bark on some haematological parameters of albino rats. Innovations in Pharmaceuticals and Pharmacotherapy 1(2): 70-75.
- Awodele O, Amagon KI, Agbo J, Prasad MNV (2015) Toxicological evaluation of the aqueous stem bark extract of Bridelia ferruginea (Euphorbiaceae) in rodents, Interdisciplinary Toxicology 8(2): 89-98.
- Abu OMG, Ayaobu-Cookey IK, Okoye Oti, CO (2023) Efficacy of Nutmeg (Myristica fragrans) as Anaesthetics in Three Life Stages of African Catfish (Clarias gariepinus). International Journal of Oceanography and Aquaculture.
- APHA, AWWA, WPCF (2005) Standard methods for the examination of water and waste water, 21st 401 ed. American Public Health Association, Washington, DC.
- King W, Hooper B, Hillsgrove S (2005) The use of Clove oil, metomidate, tricaine methanesulphonate and 2-phenoxyethanol for inducing anaesthesia and their effect on the cortisol stress response in black sea bass (Centropristis striata L.). Aquaculture Resources 36(14): 1442-1449.
- Blaxhall PC, Daisley KW (1973) Routine haematological methods for use with fish blood. Journal of Fish Biology 5: 771-781.
- Wedemeyer GT, Yasutake WT (1977) Clinical methods for assessment of the effects of environment stress on fish health. Technical Report of United States Fish and Wildlife Service p. 17.
- Dacie JV, Lewis SM (2001) Practical Haematology 9th ed. Churchill Livingstone. London 633.
- Iversen M, Finstad B, Mac Kinley RS, Eliiassen RS (2003) The efficacy of metomidate, clove oil, Aqui-S and Benzoak as anaesthetics in Atlantic salmon (Salmo salar L.) smolts and their potential stress-reducing capacity. Aquaculture 221: 549-566.
- Burka JF, Hammell KL, Horsberg TF, Johnson GR, Rainnie DJ, et al. (1997) Drugs in salmonid aquaculture. Journal of Veterinary Pharmacology and Therapeutics 20: 333-349.
- Mylonas CC, Cardinaletti G, Sigelaki I, Polzonetti- Magni A (2005) Comparative efficacy of clove oil and 2-phenoxyethanol as anaesthetics in aquaculture of European sea bass (Dicentrarchus labrax) and gilthead sea bream (Sparus auratus) at different temperatures. Aquaculture 246: 467-481.
- Gullian M, Villanueva J (2009) Efficacy of tricaine methanesulphonate and clove oil as anaesthetics for juvenile cobia Rachycentron canadum. Aquaculture Research 40: 852-860.
- Weber RA, Peleterio JB, Garcia Martin LO, Aldegunde M (2009) The efficacy of 2- phenoxyethanol, metomidate, clove oil and MS-222 as anaesthetic agents in the Senegalese sole (Solea senegalensis Kaup, 1858). Aquaculture 288: 147-150.
- Heo GJ, Shin G (2010) Efficacy of benzocaine as an anaesthetic for Crucian carp (Carassius carassius). Veterinary Anaesthetics and Analgesics 37: 132-135.
- Galalain AM, Aliyu BS (2021) Evaluation of the Toxicity Profile of Bridelia ferruginea Methanol Stem Bark Extract. Dutse Journal of Pure and Applied Sciences (DUJOPAS) 7(4b): 1-8.






























