- Review Article
- Abstract
- Cryopreservation
- Main Text
- Types of Preservational Methods
- Comparison between Slow Freezing and Vitrification
- Types of Cryopreservation
- Challenges in Cryopreservation (Cryoinjuries)
- Innovations in Cryoprotectant’s Solution
- Types of Cryoprotectants
- Improving Viability and Post-thawing Recovery
- Applications of Cryopreservation Technique
- Limitations of the Cryopreservation Technique
- Future Strategies
- Ethical Considerations in the field of Cryopreservation
- Discussion
- Conclusions
- Author Contribution
A Brief Review of Current Strategies and Advances in Cryopreservation Techniques
Anisa Iftikhar1*, Irfan yaqoob2
1Department of Biology, Clarkson University, Potsdam, USA
2Department of Computer Science, Clarkson University, Potsdam, USA
Submission:February 26, 2024; Published: March 26, 2024
*Corresponding author: Anisa Iftikhar, Department of Biology, Clarkson University, Potsdam, USA.
Email: iftikha@clarkson.edu
How to cite this article: Anisa I, Irfan y. A Brief Review of Current Strategies and Advances in Cryopreservation Techniques. Adv Biotech & Micro. 2024; 18(1): 555978. DOI:10.19080/AIBM.2024.17.555978
- Review Article
- Abstract
- Cryopreservation
- Main Text
- Types of Preservational Methods
- Comparison between Slow Freezing and Vitrification
- Types of Cryopreservation
- Challenges in Cryopreservation (Cryoinjuries)
- Innovations in Cryoprotectant’s Solution
- Types of Cryoprotectants
- Improving Viability and Post-thawing Recovery
- Applications of Cryopreservation Technique
- Limitations of the Cryopreservation Technique
- Future Strategies
- Ethical Considerations in the field of Cryopreservation
- Discussion
- Conclusions
- Author Contribution
Abstract
Background
It is possible to store organs, cells, tissues, and other biological structures using a technique called cryopreservation.
Main Body
Theoretically interesting and practically relevant is how living cells react when ice forms. Because ice crystal formation, osmotic stress, and plasma membrane damage during freezing and thawing can kill cells, simple cooling or freezing is not an option for long-term storage of stem cells and other viable tissues with significant potential value in fundamental research and many medical applications. Cryoprotective agents and temperature control technology have led to an increase in the successful cryopreservation of cells and tissues during the past few years. Cryopreservation of cells or tissues and their clinical applications will only be successful if the physical and chemical changes that occur during the freezing and thawing processes are continuously understood.
Conclusion
This overview briefly discusses slow freezing, vitrification, and other representative cryopreservation processes and the available cryoprotective agents. In addition, some of the drawbacks of cryopreservation are discussed.
Keywords: Cryo-preservation; Cryodamage; Cryo-protective agents; Slow-freezing
Abbreviations: CPA: Cryoprotective agent; ROS: Reactive oxygen species; DMSO: dimethyl sulfoxide; EG: Ethylene glycol; MPD: 2-methyl-2,4-pentadiol; PEG: Polyethylene glycol; MP: Melting point; PVP: Polyvinylpyrrolidone; H-Bonging: Hydrogen Bonding; BHT: Butylated hydroxytoluene; BPSE: Beltsville poultry semen extender; TSE: Turkey semen extender; DMA: Dimethyl acetate; DMF: Dimethyl-formamide; HES: Hydroxyethyl starch; CRISPR-Cas9: Clustered regularly interspaced short palindromic repeats- associated protein 9.
- Review Article
- Abstract
- Cryopreservation
- Main Text
- Types of Preservational Methods
- Comparison between Slow Freezing and Vitrification
- Types of Cryopreservation
- Challenges in Cryopreservation (Cryoinjuries)
- Innovations in Cryoprotectant’s Solution
- Types of Cryoprotectants
- Improving Viability and Post-thawing Recovery
- Applications of Cryopreservation Technique
- Limitations of the Cryopreservation Technique
- Future Strategies
- Ethical Considerations in the field of Cryopreservation
- Discussion
- Conclusions
- Author Contribution
Cryopreservation
Cryopreservation is a method of preserving biological structures, such as organelles, cells, and tissues, by rapidly freezing samples at extremely low temperatures. There is both theoretical and practical value in studying how living cells react to the onset of ice. Because of the negative consequences of ice crystal formation, osmotic stress, and membrane damage during the freezing and thawing processes, the long-term preservation of stem cells and other useful tissues for various research and medicinal purposes is difficult. Simple cooling and freezing technologies are insufficient to keep these cells and tissues alive for long periods. Recently, there has been an increase in the successful cryopreservation of cells and tissues owing to the development of cryoprotective chemicals and temperature-control technology [1]. The ability of low temperatures to prolong the preservation of biological materials has been known since ancient times. Indeed, according to archaeological evidence, icehouses were utilized all over Mesopotamia as far back as 2000 BC [2]. Boyle and other early 17th century experimentalists were interested in the preservation effect of cold, and they made repeated attempts to freeze and resuscitate live animals, discovering species of frogs and fish that could withstand encasement in ice [3].
It is described as the use of low temperatures to secure cell tissues, organelles, or other biological tissues by cooling them for a long time. At these temperatures, the chemical and biological activities in living cells and tissues become dramatically low or cease temporarily; thus, structurally, all living cells and tissues can be preserved for a long time. It holds biological samples in pensile activity for any reasonable period at cryogenic temperatures; therefore, it can be used to secure the fine structures of living cells and tissues [4]. According to Onofre et al. [5], the success rate of cryopreservation techniques has steadily improved in recent years. This advancement is due to the use of appropriate cryoprotective chemicals and sophisticated temperature-control equipment. The application of these optimal parameters resulted in improved preservation outcomes and increased the vitality of the conserved materials.
The success of this technique depends on several aspects, such as improvement in cooling and warming rates, osmotic conditions, choice, suitable amount of cryoprotective agents, and equilibration time of cryoprotectants. All of these conditions could increase the persistence rate of cryopreserved cells and tissues. The main purpose of this process is to design a dehydrating atmosphere that preserves cells and tissues. Karlsson & Toner [6] concluded that cryopreservation of bulky cells and tissues is difficult to achieve as compared to single cell suspension because bulky cells and tissues have different mass and heat transmission effects during the procedures, which eventually make it tougher to attain quick chilling and thawing rates and the equal allocation of cryoprotective agents.
- Review Article
- Abstract
- Cryopreservation
- Main Text
- Types of Preservational Methods
- Comparison between Slow Freezing and Vitrification
- Types of Cryopreservation
- Challenges in Cryopreservation (Cryoinjuries)
- Innovations in Cryoprotectant’s Solution
- Types of Cryoprotectants
- Improving Viability and Post-thawing Recovery
- Applications of Cryopreservation Technique
- Limitations of the Cryopreservation Technique
- Future Strategies
- Ethical Considerations in the field of Cryopreservation
- Discussion
- Conclusions
- Author Contribution
Main Text
Methodology of Cryopreservation
Cryopreservation is a technique used to maintain the delicate cellular structure of biological samples by suspending them at extremely low temperatures for extended periods. This method involves freezing samples at temperatures below -150ᵒC to block biological processes and avoid degradation. Cryopreservation effectively maintains samples in this dormant state, allowing for long-term storage and a variety of applications in research, medicine, and other sectors [7,4].
Goa et al. (2016) concluded that the general cryopreservation process included different steps. First, fusion of cryoprotectants with living cells and tissues occurs before the cooling process begins. The melting point of water decreases because of the proper mixing of water with appropriate cryoprotectants. These cells and tissues are then cooled to very low temperatures and stored in liquid nitrogen. Thawing of these cells and tissues is achieved by heating them, and the last step is the removal of cryoprotectants from these cells and tissues. When cryoprotectants have been removed from the cell, the cell may swell first and then shrink again. Bhattacharya [8] concluded that excessive cell expansion may cause damage. When cryoprotectants have not been removed stepwise, they can cause over-swelling of the cell. If the thawing process occurs at a slower rate, small ice crystals may continuously grow and destroy cells [9]. In addition, recrystallization may occur, resulting in the restoration of extracellular ice fractions (Bank, 1973). For better results, the cryoprotectant concentration must be maintained between 5% and 15%.
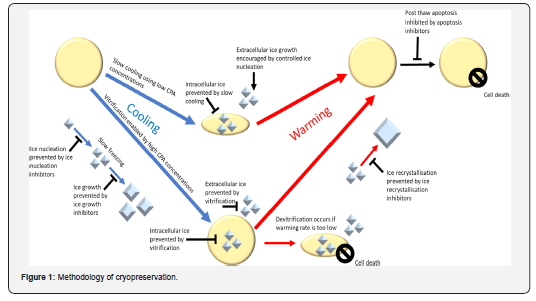
Outline of how Cryopreservation and CPAs work
Cryopreservation requires a high freezing rate or high concentration. CPA is used to prevent ice formation in the sample, or moderate cooling is used to allow water to flow out of the cell and prevent intracellular ice development. Inhibitors of ice nucleation, ice growth, and ice recrystallization are additional means by which ice expansion can be stifled. When a vitrified sample is gently heated, ice forms, a process known as devitrification. Cryopreservation can lead to stress-induced apoptosis, which can be prevented by using apoptosis inhibitors, as summarized in (Figure 1) [10].
- Review Article
- Abstract
- Cryopreservation
- Main Text
- Types of Preservational Methods
- Comparison between Slow Freezing and Vitrification
- Types of Cryopreservation
- Challenges in Cryopreservation (Cryoinjuries)
- Innovations in Cryoprotectant’s Solution
- Types of Cryoprotectants
- Improving Viability and Post-thawing Recovery
- Applications of Cryopreservation Technique
- Limitations of the Cryopreservation Technique
- Future Strategies
- Ethical Considerations in the field of Cryopreservation
- Discussion
- Conclusions
- Author Contribution
Types of Preservational Methods
Cryopreservation methodologies fall into different categories. These included:
(1) Slow freezing [11,12]
(2) Vitrification (in this process the liquid ambiance of the living cell and tissue solidifies into a non-crystalline glassy state also known as glass forming [13], without the threat of forming intra- and extracellular ice crystals during cooling or warming process beyond the rates of these processes
(3) Subzero non-freezing storage (preserving tissues and cells below -130ᵒC to -196ᵒC temperature known as subzero temperature by using different substances) and
(4) Preservation in a dry state (this is not possible because of many difficulties during the introduction of amino acids and disaccharide trehalose into the intracellular region [14]. In this method, small divisions of sperm samples (100-250µl) that were diluted with a suitable cryoprotectant were directly placed on dry ice and allowed to cool for 4-5 minutes. They are then transferred into cryovials and stored in LN2 [15].
Gao and Critser [16] concluded that there is a huge diversity in cryobiological responses, and cryo-survival of cells and tissues during freezing and thawing cycles depends on the types of cells and availability of given cells.
Slow Freezing and Vitrification
Gao and Critser [16] concluded that these processes are relatively dissimilar from each other. However, both are related to the same physicochemical properties to some extent. The main difference between the two is the use of various concentrations of cryoprotectants and cooling rates. In slow freezing, more than 3 hours are required for work, and it works only in closed systems. This is a much more expensive protocol owing to the requirement of freezing machines such as benchtop portable and sophisticated freezing containers. Manipulation is easy using this method. In this process, the required sample volume must be between 100-250µl. This requires a low concentration of cryoprotectant. These cryopreserved cells are at a high risk of cryoinjuries, such as the establishment of ice crystals, but the risk of cryoprotectant toxicity is low. The viability or viability of the cells after thawing was higher in this protocol. The benefit of this method is that cells have a low chance of contamination due to pathogenic agents during the procedure. (Figure 2)
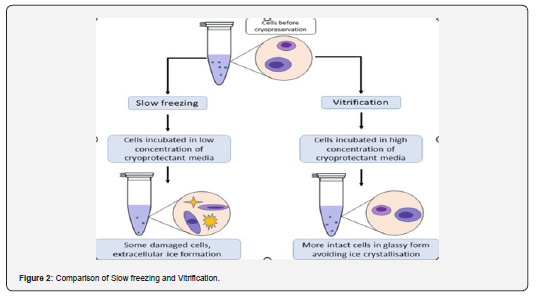
Gao and Critser [16] concluded that cells are chilled less than their chilling point during the process of slow cooling. At a slow freezing rate, when osmotic strength increases, cells can release intracellular water at a faster rate to avoid supercooling. Eventually, this lowers the chances of intracellular ice crystal formation. However, at a certain point below their freezing point, there are chances of the formation of spherical crystals that may damage the chronobiology of the cell [8], and the cell is at a higher risk of dehydration, which is not desired.
These ice crystals are also known as unfrozen masses or fractions. If these ice crystals continue to grow, they can freeze all the solutes in which cells are placed, because cells face very high concentrations of solutes during unfrozen fractions. If the amounts of cryoprotectants, sugars, and salts increase, ice crystals can be reduced. If the intracellular water is released at a faster rate, it can cause a 50% reduction in cell volume compared to its original volume. This eventually causes structural injury to cells.
Bhattacharya [8] concluded that in the process of slow cooling, almost 10% of cryoprotectants were initially added in each step. When cryoprotectants are added to the solution, they cause a classic shrink-swell response in cryobiology. In this process, cryoprotectants enter the cell membrane and substitute for water within the cytoplasm. As a result, cells first shrink because of osmosis and then swell again when cryoprotectants penetrate the cell, thus reducing cell damage and maintaining the cooling rate following the permeability of the cell membrane. When the amount of cryoprotectant becomes equal to the intra- and extracellular fluids of cells, swelling is prevented. Cells or tissues are ready for the freezing process after adjusting the volume and concentration within 10 min. However, cells are at high risk of extracellular ice crystal formation due to slow freezing. This protocol has a typical cooling rate of approximately 1°C/min in the presence of cryoprotectants less than 1.0M with the help of costly controlled-rate programmable freezers [11,12].
- Review Article
- Abstract
- Cryopreservation
- Main Text
- Types of Preservational Methods
- Comparison between Slow Freezing and Vitrification
- Types of Cryopreservation
- Challenges in Cryopreservation (Cryoinjuries)
- Innovations in Cryoprotectant’s Solution
- Types of Cryoprotectants
- Improving Viability and Post-thawing Recovery
- Applications of Cryopreservation Technique
- Limitations of the Cryopreservation Technique
- Future Strategies
- Ethical Considerations in the field of Cryopreservation
- Discussion
- Conclusions
- Author Contribution
Comparison between Slow Freezing and Vitrification
Cryopreservation is a widely used approach for preserving large quantities of sperm, oocytes, ovarian tissue, and fetal tissue, allowing for long-term cell and tissue storage [17]. Two basic cryopreservation procedures are freezing, in which the liquid phase transforms into a solid crystalline phase, and vitrification, which involves solidification into a glass-like state without the creation of ice [18]. Both progressive freezing and vitrification are possible possibilities, with changes in cryoprotective agent (CPA) concentrations and cooling speeds (Table 1).
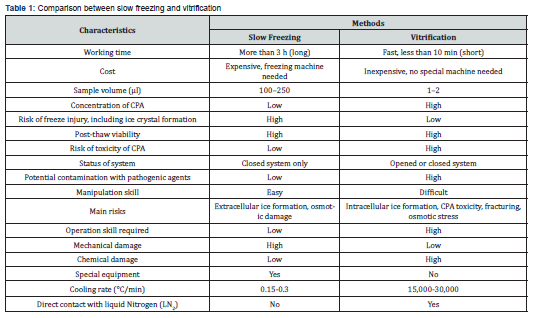
The cooling rate is important because slow cooling allows cells to progressively efflux intracellular water, limiting intracellular ice formation, and decreasing cell damage [14]. Slow freezing involves replacing water within the cytoplasm with CPAs using a controlled-rate freezer or a portable freezing container, resulting in a cooling rate of around 1ᵒC/min with less than 1.0M CPA concentration [11,12]. Slow freezing has several advantages, including ease of use, minimal handling, and a low risk of contamination during the procedure. However, it poses a significant risk of freeze injury owing to the generation of extracellular ice (Table 1).
Vitrification is an alternative to gradual freezing in which cell suspensions are directly exposed to liquid nitrogen, resulting in a transition from an aqueous phase to a glass state (Rall & Fahy, 1985) To avoid ice nucleation, cells or tissues are rapidly reduced to deep cryogenic temperatures while being exposed to cryoprotective agents (CPAs) at concentrations ranging from 40-60% (w/v) [19]. The rate of chilling and warming, sample volume, and sample viscosity are all important variables in the vitrification process. For good vitrification, it is critical to achieve a stable balance between these components. There are two methods of vitrification: equilibrium and non-equilibrium. Equilibrium vitrification involves creating multimolar CPA mixes and injecting them into cell suspensions. In contrast, non-equilibrium vitrification employs extremely high freezing rates and low CPA mixture concentrations. Nonequilibrium approaches are further subdivided into carrier-based and carrier-free systems. Vitrification has the advantage of a low risk of freeze injury, which allows the retention of a sufficient number of cells. However, specialist manipulation skills are required, and there is a substantial risk of infection with harmful microorganisms.
- Review Article
- Abstract
- Cryopreservation
- Main Text
- Types of Preservational Methods
- Comparison between Slow Freezing and Vitrification
- Types of Cryopreservation
- Challenges in Cryopreservation (Cryoinjuries)
- Innovations in Cryoprotectant’s Solution
- Types of Cryoprotectants
- Improving Viability and Post-thawing Recovery
- Applications of Cryopreservation Technique
- Limitations of the Cryopreservation Technique
- Future Strategies
- Ethical Considerations in the field of Cryopreservation
- Discussion
- Conclusions
- Author Contribution
Types of Cryopreservation
The different types of cryopreservation processes include (1) isochoric cryopreservation, (2) isobaric cryopreservation, and (3) hyperbaric cryopreservation [8]. Their descriptions are provided below.
Isochoric cryopreservation
Fowler et al. [20] concluded that isochoric cryopreservation involves a constant volume with a slow cooling rate within a system for the preservation of biological materials. This is another approach to the old-style method of cryopreservation because it is inexpensive, easy to control and perform, and requires fewer cryoprotective agents. This system is useful for the cryopreservation of materials, as well as in the vitrification process, as heterogeneous nucleation does not occur. Moreover, this system automatically maintains itself to minimize pressure at a given temperature, thereby reducing the toxic effects of pressure.
Isobaric cryopreservation
The isobaric process involved a constant pressure of 1 atm for cell preservation. Most cryopreservation depends on this protocol, where freezing occurs at a constant pressure of 1atm because this is much easier, both experimentally and in our natural environment. This protocol is generally used, but it has some limitations for cytocellular injuries that make it a highly ridiculous method, such as increased intracellular ionic concentration, which alters the cell’s chemical integrity during freezing and thus causes injuries to cells.
Hyperbaric cryopreservation
The hyperbaric preservation protocol allows the cells to sustain a suitable phase at a low temperature of 0ᵒC without freezing [21]. This process involves high pressure followed by a steady reduction in pressure at a low temperature of 0ᵒC which causes rapid freezing and reduces the ionic concentrations (Prehoda & Robert, 1969). This method is like the isochoric cryopreservation method. The hyperbaric pressure system is widely used in the preservation of food and cryomicroscopy fixation of tissue structures [22,7] and in the medical industry [22], regardless of the lethal effects of pressure maintenance. Moreover, by using this approach, many organs and tissues can be cryopreserved at low temperatures, such as the kidney [9] at 10,000atm, cells [21] at 200atm, or liver (Takahashi et al., 2000) at 700atm. In addition, RBCs could survive up to 200MPa.
- Review Article
- Abstract
- Cryopreservation
- Main Text
- Types of Preservational Methods
- Comparison between Slow Freezing and Vitrification
- Types of Cryopreservation
- Challenges in Cryopreservation (Cryoinjuries)
- Innovations in Cryoprotectant’s Solution
- Types of Cryoprotectants
- Improving Viability and Post-thawing Recovery
- Applications of Cryopreservation Technique
- Limitations of the Cryopreservation Technique
- Future Strategies
- Ethical Considerations in the field of Cryopreservation
- Discussion
- Conclusions
- Author Contribution
Challenges in Cryopreservation (Cryoinjuries)
During the process of cryopreservation, there are chances of damage to cryopreserved cells and tissues, such as cold shock at raid freezing temperatures and chilling injuries at slow freezing temperatures. Cryo-damage may occur before freezing and after thawing at between 0-40ᵒC temperature ranges [8]. These damages are linked to cryopreserved cells and tissues that face the phase change of water in both types of surroundings, that is, intra- and extracellular environments at very low temperatures. However, an accurate mechanism has not yet been clearly described [16].
Bakst et al. [23] concluded that the physiochemical and biophysical reactions of cells are mainly affected by cooling and thawing rates, which can alter the survival rate of cells and tissues. Moreover, these damages may mainly affect the plasma and cell membranes of the mitochondria. However, under the worst conditions, it can affect the nucleus. These injuries affect fertility and the duration of life. This is a crucial phenomenon in birds because semen remains in the uterovaginal glands of the female for a long period, generally from 1 week to several months, depending on the species.
Massip et al. [24] concluded that some acute points mainly affect the cell structures and metabolism throughout the process of cryo-preservation (1) interaction between the sperm cell and added cryoprotective agent; (2) temperature curve of freezing & thawing and (3) the type of semen packaging.
The mechanism of cryo-damage suggests that extra- and intracellular solutes and cellular ice formation are the main causes of osmotic damage in cells and tissues (McGann et al., 1998) [25]. These processes depend mainly on the cooling rate [14]. However, the freezing establishment of intra- and extra-cellular ice crystals eventually alters the chemical composition of the cells, which leads to cellular mechanical restraints and damage that is lethal to most living organisms [6]. Moreover, cells face the main obstacle in overcoming the water-to-ice phase transition at low temperatures [26,7].
Cellular damage caused by cryopreservation includes alterations in cell shape, features (e.g., adhesion and cell surface indicators), metabolic activity (e.g., proliferative ability and potency), function (e.g., immunomodulation), and even death. Some stress response pathways can be activated by a decrease in temperature, and thawing can trigger apoptotic and necrotic processes. Changes in the frozen sample that are stressful might cause significant cell harm, especially if ice crystals form [27].
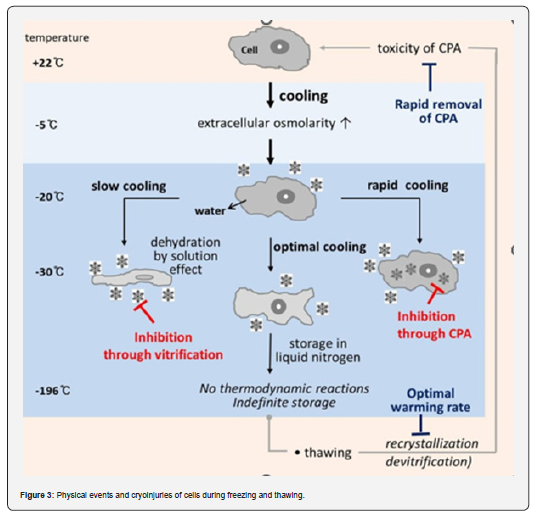
Effect of formation of both intra & extra-cellular ice crystals on cell
Dittrich et al. [28] concluded that intracellular ice crystal formation causes damage to the cryopreserved cells at a fast-cooling rate which produces lethal effects on the intracellular integration of the cells. As a result, rapid freezing of intracellular integration occurs in the cytosol, parenchyma, nucleus, and almost all cellular elements of the cell. Eventually, all the integral portion of the cells comes out, and thus, cells may die due to shrinkage [29,30]. Osmotic damage occurs because of disclosure to extremely concentrated intra-and extracellular solutes or because of the mechanical interplay between cells and extracellular ice crystals at slow cooling rates. In the presence of cryoprotective substances, the behavior of cells towards freezing could be altered because cryoprotectants affect nucleation, water transport, and ice crystal formation (Figure 3).
The chemical and biological responses of living cells slow significantly at low temperatures, which may allow long-term preservation. However, freezing is lethal to most forms of life because of the production of ice crystals both inside and outside of cells, which chemically alters the cellular environment and causes mechanical limitations and harm [6]. When the temperature drops, the water-to-ice phase shift presents cells with the greatest challenge [26,7]. When cells are subjected to rapid freezing, the formation of ice crystals within their internal structures can cause damage. On the other hand, cells may be damaged by osmotic changes caused by exposure to highly concentrated fluids inside and outside the cells or by mechanical contact between cells and extracellular ice if they are cooled slowly. Balancing these parameters is critical for creating appropriate cryopreservation techniques that reduce cell damage while ensuring successful preservation.
Effect of membrane lipids and proteins on the cell
The behavior and function of membrane lipids and proteins can be altered by variations in temperature. Membrane lipids that exist in the crystalline phase in normal conditions may become solidify at non-physiological temperatures thus their functions become altered that eventually initiates various processes that increase damage to membrane lipids such as distraction of lipid and proteins connections and various proteins including ion passages may translocated or lost their function [31], as well as the formation of ROS or destabilize the cellular membrane thereby loos its selective permeability which enhances the influx of ions, for example, Ca2+ and bicarbonates from extracellular space [32]. These changes are known as cryocapacitation or capacitation-like changes. To inhibit the generation of reactive oxygen species (ROS) molecules, Organisms have evolved a variety of defense mechanisms to inhibit ROS generation. The enzymatic defense system, which comprises enzymes such as superoxide dismutase, catalase, glutathione transferase, and glutathione peroxidase, is one such mechanism. These enzymes are critical for the neutralization and detoxification of reactive oxygen species (ROS). Antioxidants found in organisms include ascorbate (vitamin C), reduced glutathione, urate, vitamin E, and β-carotene. These antioxidants aid in the scavenging and neutralization of reactive oxygen species (ROS), thereby protecting cells and tissues from oxidative damage. Enzymatic defenses and antioxidants work together to maintain the redox balance inside organisms and protect against the detrimental effects of ROS.
Effect of reduced temperature on cell
Moreover, a reduction in temperature can cause irregularities in various cellular processes. The reason for this is that the rate of one procedure may be greatly affected compared with the other procedure. For example, variations in the dynamic equilibrium of the dissociation of tubulin filaments may cause fragmentation of metaphase spindles in oocytes.
Effect of Dehydration or very high solute concentration on cell
Dehydration or very high solute concentrations can cause loss of membrane stability and denaturation of proteins; thus, cells can die [33] (Crowe and Crowe, 1984; Hvidt and Westh, 1992). Very high solute concentrations can initiate a process known as solute landing, in which extracellular salts enter the cell (Daw et al., 1973; Griffiths et al., 1979). Moreover, if the cells are restrained into the narrow routes of the unfrozen fraction and pressurized between growing ice fractions, this can cause mechanical stress to the cells (Rapatz and Luyet, 1960). When cells or tissues face a hypertonic solution around them, they can also cause shrinkage or death of the cells or tissues. This mechanism also known as “solution effect injury” mainly occurs in the absence of cryoprotectants. Owing to this effect, the residual solution of cells may change the form and mechanical strength of the cells [8].
Effect of osmotic pressure, pH, and amount of cryoprotectants on cell
Bhattacharya [8] concluded that osmotic pressure, pH, and amount of cryoprotectants must be well managed during cryopreservation because any increase in any of these can cause cytoinjuries. For example, in some situations, the cell cytosol contains essential fatty acids, such as the plasma membrane of spermatozoa. The fatty acids present in the membrane of spermatozoa are of great significance. This is because they help retain the appropriate osmotic pressure and prevent shock throughout the process of freezing and thawing. Thus, fatty acids are useful for managing cell rehydration.
- Review Article
- Abstract
- Cryopreservation
- Main Text
- Types of Preservational Methods
- Comparison between Slow Freezing and Vitrification
- Types of Cryopreservation
- Challenges in Cryopreservation (Cryoinjuries)
- Innovations in Cryoprotectant’s Solution
- Types of Cryoprotectants
- Improving Viability and Post-thawing Recovery
- Applications of Cryopreservation Technique
- Limitations of the Cryopreservation Technique
- Future Strategies
- Ethical Considerations in the field of Cryopreservation
- Discussion
- Conclusions
- Author Contribution
Innovations in Cryoprotectant’s Solution
The creation of novel formulations that offer cells better protection during the freezing and thawing processes has been the focus of innovations in cryoprotectant solutions [34-36]. These are defined as chemicals that are used to hinder the injuries that can occur during the process of freezing cryopreserved cells and tissues. These molecules do not form ions in water and are not harmful to cells at room temperature. These solutions aim to reduce osmotic stress, preserve the integrity of the cell membrane, and stop the development of ice crystals [34,37,38].
Bhattacharya [8] concluded that these substances could enter preserved cells and tissues and moisturize them without producing any lethal effects on these cells and tissues, thereby increasing their survival. Cryoprotectants must be able to dissolve in the cryopreserving solution. After dilution with the preserving solution, cryoprotectants drop the melting point of the solution so that cells and tissues can freeze in this environment [39]. Cryoprotectants are called antifreeze such as DMSO, propylene glycol, and glycerol are mostly used in this type of cryoprotectant. The use of natural substances, such as trehalose and glycerol, which have been demonstrated to be successful in maintaining cell viability and functionality, is one of the advances in cryoprotectant solutions [34,1,40].
Wowk et al. [41] concluded that after the process of freezing and thawing, cells are preserved in suitable cryoprotectants with concentrations between 5 and 15% for the survival of preserving cells by producing large unfrozen pockets that help to inhibit the formation of intra- and extracellular ice crystals, which also prevent mechanical injury. In the absence of a suitable cryoprotectant, cells may be damaged at low temperatures as well, and cells may be injured in a transition phase at warming temperatures, resulting in the loss of cell numbness and hardness.
Pegg [26] concluded that they are liquids that lower cryoinjuries during the process. For this purpose, they must have the capacity to enter the membranes of cells and tissues, possess low toxicity, and should be biologically tolerable. Various types of cryoprotectants are available, including the cell banker series, dimethyl sulfoxide (DMSO), ethylene glycol (EG), glycerol, trehalose, proteins, polymers, 2-methyl-2,4-pentadiol (MPD), formamide, proline, glycerol 3-phosphate, sorbitol, diethyl glycol, propylene glycol, sucrose, polymers, polyethylene glycol (PEG), or triethylene glycol. The cryoprotectant used was determined by the cell type, desired preservation outcome, and precise freezing and thawing conditions used in the cryopreservation technique. Cryoprotectants differ in their ability to prevent ice crystal formation and protect biological structures during preservation.
Yavin and Arav [19] concluded that the ideal optimal survival rate of cells and tissues can be achieved by setting a suitable sample volume, optimized freezing and heating rates, and the appropriate amount of cryoprotective agents depending on the various cell types and contexts of tissues. Sambu [4] concluded that, along with conventional small molecules, biomaterials such as alginates, polyvinyl alcohol, and chitosan may also be utilized to block the formation of unfrozen masses. Moreover, using cryoprotectants, together with a few electrolytes and sugars, enhances the osmotic integrity of the solution, thus causing a positive outflow of moisture to the cell. During this process, ice crystal production becomes insignificant [8]. Cell death can also occur due to processes such as apoptosis during the cooling and thawing processes and the direct delay in the formation of ice crystals, both of which can be overcome with the use of different kinds of compounds and antioxidants [42].
Ploge et al. [43] concluded that in some cases cryoprotectants can cause toxicity to the cells because they are replaced with the water of the cryopreserved cells and tissues during the practice of cryopreservation. For example, at warm temperatures, every cryoprotectant behaves differently, as propylene glycol is non-toxic, but ethylene glycol can metabolize toxic substances. Moreover, recent research has shown that some cryoprotectants, known as penetrating cryoprotectants, can enter the cells very deeply owing to the lipophilic property of cryoprotectants, which could cause destabilization of cells.
Bhattacharya & Sankha [44] concluded that the electrical features of cryoprotectant solutions also cause membrane toxicity, with DMSO being highly toxic. Strong H-bonding can also cause membrane toxicity by disturbing the hydration layer present around the biomolecules. Fatty acids present in the membranes of spermatozoa are of great significance. They help sustain an appropriate osmotic pressure and prevent shock through the process of chilling and thawing. Thus, fatty acids are useful for managing cell rehydration.
Bhattacharya [8] concluded that a cryoprotectant solution might be a freezing solution or sometimes it acts as a carrier solution that supports life. The main objective of this solution is to maintain the continuity of life in cells that are close to the chilling environment. This solution is also called the “base perfusate”. It sustains stability and iso-osmolarity within cells thus it is capable of the cells to protect its form. For instance, the M22 cryoprotectant is used with a special carrier solution known as LM5. They provide sufficient support to cells at chilling temperatures. It contains various ingredients, such as nutritional salts, elements, some significant buffers, osmogens, and apoptosis inhibitors; therefore, it behaves as a buffering substance for preserving cells and tissues. When cells are placed in a carrier solution, they help to establish isotonic concentration; thus, cells can neither shrink nor swell. The concentration remains constant throughout the cooling process.
Bhattacharya and Sankha [44] concluded that the concentration of the carrier solution cannot be varied when changing the concentration of cryoprotectants. This should be constant for a given type of cryopreserved cell. Cryoprotectants also fall into various categories, such as penetrating and non-penetrating cryoprotectants. They also contain various ingredients in their solutions, which enhance their functions. The cryoprotectant solution also contains ice blockers that prevent the formation of ice crystals, such as low-molar weight polyglycerol and polyvinyl alcohol, known as x-1000 and z-1000. These ice blockers may bind ice inculcators to sunder-ice development at low temperatures and high pressures. Some of these are used only in the vitrification protocol.
Mode of action of cryoprotectant
These compounds, known as "extenders," are added to a solution to lower the freezing point and prevent the formation of ice crystals. They dissolve into the solution and reduce its freezing point, thereby allowing the solution to remain in a liquid state at lower temperatures. In the context of cell preservation, these extenders can help moisturize and preserve cells by invading them and preventing their dehydration. However, it is important to note that these extenders cannot change the inherent properties of cells, which could lead to harmful effects.
Cryoprotectant toxicity
Cryoprotectant toxicity occurs when the concentration of cryoprotectants exceeds an acceptable limit, leading to cell and tissue damage. This process can be lethal to the preserved cells. During the freezing process, water is replaced with cryoprotectants such as ethylene glycol, which can become toxic at freezing temperatures [8]. Additionally, the strong hydrogen bonding and electrical properties of cryoprotectants can produce toxicity. Of all cryoprotectants, dimethyl sulfoxide (DMSO) is the most toxic.
Proper concentrations
A study conducted by Rakha et al. [45] found that all cryopreservatives function effectively at appropriate concentrations. Polyvinylpyrrolidone (PVP) decreases the formation of crystals when used in low amounts, and it was discovered that between 1-5%, it has an optimal concentration. It was observed that 6% PVP produced a significant number of fertile eggs that hatched. Additionally, it was noted that PVP had the best results in terms of semen quality after thawing at a concentration of 6%. Regarding DMSO, it was observed that it performs well within the range of 1-10% [46]. Similarly, DMSO displays optimal results at low concentrations, with an optimal range of 1-3% [47]. In contrast, glycerol works best at a concentration of 20% [46]. Butylated hydroxytoluene (BHT) was found to function most effectively at concentrations of 1.5% and 2%.
Eradication of cryoprotectant
The removal of cryoprotectants from a cell can cause it to rapidly revert to its original state by absorbing a large amount of water, resulting in significant swelling. This phenomenon, known as osmoticity, makes cells more susceptible to swelling than to shrinking. Consequently, the addition of cryoprotectants is typically easier than their removal (Pegg, n.d.).
Extenders or Diluents
An extender is a solution that is free of cryoprotectants and consists of both organic and inorganic molecules. Its pH is like that of seminal plasma, and it is also known as a buffered salt solution or diluent. These solutions are primarily used to extend or dilute semen samples, protect them from physical and chemical impurities, and create a favorable environment for reproduction [48]. Examples of extenders and diluents include Beltsville poultry semen extender (BPSE) [49], EK extender [50], lake diluent [51], tselutin extender [52], chicken semen extender [53], and turkey semen extender (TSE).
Extenders help to maintain the viability of sperm cells by making them immotile and increasing the number of inseminated hens (sperm-to-egg ratio for fertilization). The motility and metabolic rate of sperm cells are largely dependent on the pH of the extender (Donoghue & Wishart, 1959). The ability of extenders or diluents to maintain semen integrity is highly species-specific [54].
Rakha et al. [45] concluded that the features of an ideal extender or diluent to endure viability and cell functions are:
Ø Supplies enough energy source for sperm cell metabolism.
Ø Regularity of pH and osmolarity
- Review Article
- Abstract
- Cryopreservation
- Main Text
- Types of Preservational Methods
- Comparison between Slow Freezing and Vitrification
- Types of Cryopreservation
- Challenges in Cryopreservation (Cryoinjuries)
- Innovations in Cryoprotectant’s Solution
- Types of Cryoprotectants
- Improving Viability and Post-thawing Recovery
- Applications of Cryopreservation Technique
- Limitations of the Cryopreservation Technique
- Future Strategies
- Ethical Considerations in the field of Cryopreservation
- Discussion
- Conclusions
- Author Contribution
Types of Cryoprotectants
There are two different types of cryoprotectants: (1) membrane-permeable and (2) non-membrane-permeable. These polymers reduce the number of ice crystals formed during this process [4]. Many reagents have been used as cryoprotectants. The choice between permeable and non-permeable cryoprotectants may be influenced by a variety of factors, such as the type of cells or tissues being cryopreserved, the desired outcome, and the specific cryopreservation methodology used. To obtain the required results, researchers may use a combination of permeable and non-permeable cryoprotectants.
Membrane-Permeable Cryoprotectants
Membrane-permeable cryoprotectants are also called penetrating cryoprotectants because they can enter the cell and facilitate its frozen survival like DMSO, dimethyl acetate (DMA), Dimethyl-formamide (DMF), propylene glycol, cell banker series, ethylene glycol, trehalose, methanol, dimethylacetamide and glycerol [55] Permeable cryoprotectants are often used in cryopreservation because they may protect both intracellular and extracellular tissues during freezing [56]. The defensive outcomes of these cryoprotectants are presumably because of their colligative properties. They can form H-bonds with polar head groups of cellular lipids, thus maintaining membrane lipids and providing protection to cells and tissues, which are essential in extremely dehydrated conditions (Crowe & Crowe, 1984) [57]. These CPAs can affect the cytoplasm viscosity, alter the diffusion rate, and change the cell membrane properties by penetrating a lipid bilayer [58].
Moreover, they can disturb the mechanical characteristics of an unfrozen segment, such as its viscosity and glass-producing features. The main function of penetrating cryoprotectants is to protect the cells against dehydration by maintaining proper moisture during the freeze-drying process. They can also reduce the establishment of intra- and extracellular ice crystals. They also lowered the lethal shrinkage of cells during slow cooling at any given temperature. They have a molar mass of <100 Da [8]. They are an important component of vitrification solutions. These properties are widely used in the pharmaceutical industry [8].
Non-Membrane-Permeable Cryoprotectants
Non-membrane-permeable cryoprotectants are also called non-penetrating cryoprotectants, such as 2-methyl-2,4-pentanediol and polymers, including PVP, hydroxyethyl starch, and various sugars [6], PEG [8], egg yolk, hydroxyethyl starch (HES), dextrin, glucose, and sucrose. Nonpermeable cryoprotectants, such as red blood cells, can be effective in maintaining tissues or structures where intracellular protection is not required [56]. They also inhibit the production of ice crystals. However, in some cases, they behave as toxic chemicals compared with membrane-permeable cryoprotectants at the same concentrations. Moreover, they can boost the tonicity of the vitrification solution, thus protecting cells from freezing damage [8]. These cryoprotectants are proteins that may be found in milk, egg yolk, and sugars as well as in compounds with high molecular weights.
- Review Article
- Abstract
- Cryopreservation
- Main Text
- Types of Preservational Methods
- Comparison between Slow Freezing and Vitrification
- Types of Cryopreservation
- Challenges in Cryopreservation (Cryoinjuries)
- Innovations in Cryoprotectant’s Solution
- Types of Cryoprotectants
- Improving Viability and Post-thawing Recovery
- Applications of Cryopreservation Technique
- Limitations of the Cryopreservation Technique
- Future Strategies
- Ethical Considerations in the field of Cryopreservation
- Discussion
- Conclusions
- Author Contribution
Improving Viability and Post-thawing Recovery
Enhancing cell viability and recovery during thawing is another area of research interest. Optimizing post-thawing procedures, such as removing cryoprotectants gradually and step-by-step, to avoid osmotic stress and reduce cellular damage [34,59,38]. Furthermore, improvements in medium formulations and cell culture methodologies have been made to augment the functionality and post-thaw recovery of cryopreserved cells [60,34,58]. Through the combination of these techniques, scientists hope to maintain the functional and structural integrity of cells during cryopreservation, enabling their successful application in a range of settings, including biobanking, cell therapy, and regenerative medicine [60, 61,34].
- Review Article
- Abstract
- Cryopreservation
- Main Text
- Types of Preservational Methods
- Comparison between Slow Freezing and Vitrification
- Types of Cryopreservation
- Challenges in Cryopreservation (Cryoinjuries)
- Innovations in Cryoprotectant’s Solution
- Types of Cryoprotectants
- Improving Viability and Post-thawing Recovery
- Applications of Cryopreservation Technique
- Limitations of the Cryopreservation Technique
- Future Strategies
- Ethical Considerations in the field of Cryopreservation
- Discussion
- Conclusions
- Author Contribution
Applications of Cryopreservation Technique
Cryopreservation has become an apex technology and has various applications in different areas such as cryopreservation of cells and organs [16], cryosurgery, molecular biology and biochemistry, environmental biology and plant physiology, food sciences, and various medical applications such as blood transfusion, bone marrow transplantation, artificial insemination, and in vitro reproduction [4,7,16] of cartilages, lymphocytes, or proteins [8].
Other benefits of this technique include the potential banking of cells for human leukocyte antigen typing for organ transplantation, the maintenance of a feasible time for transportation of cells and tissues between various chemical centers, and the availability of research sources to diagnose unknown transmissible diseases or pathogens [16,1] concluded that this technique is also helpful for the cryopreservation of numerous cells & tissues such as red blood cells, spermatozoa, ovarian tissue, oocytes, testicular cells or tissues, teeth and dental pulp, amniotic fluid and umbilical cord, adipocyte tissue, hepatocytes, platelet, microorganisms, embryo, and stem cells. Cryopreserved materials have numerous applications in regenerative medicine, organ transplantation, drug discovery, assisted reproductive technologies, and fertility preservation [60,62,34].
Polge et al. [39] concluded that mammalian spermatozoa were the 1st cells that cryo-preserved effectively. This discovery occurred because Polge et al. were busy working on the cryoprotective effect of glycerol as a cryoprotective agent. Similarly, 1st incident of embryo conservation for fecundity conservation was held in 1996, when the embryo of a woman with breast cancer was cryopreserved by an in vitro fertilization process before the initiation of chemotherapy. This method had no lethal side effects on parturition. This method can also be helpful for the conservation of sperm and semen, thus preserving the fertility of men facing different kinds of treatments that threaten their fertility, such as chemotherapy, surgery, and radiotherapy. Germ cell reduction can occur at any stage because of chemical and physical toxicity, diseases, or genetic predisposition [5].
Therefore, this technique acts as the first-line treatment for these patients by preserving fertility. Eventually, sperm and semen can be utilized endlessly after appropriate cryopreservation. Stem cells can be preserved using this technique without altering their cellular and genetic content, as they are used in various applications, such as tissue engineering, gene therapy, regenerative medicine, and transplantation of cells. Adult stem cells can be acquired from different sites, such as fat tissues, periosteum, amniotic fluid, and umbilical cord blood other than bone marrow cells, and they can differentiate into various types of specific cells [12]. They can differentiate into embryonic, mesenchymal stromal [63,64], and hematopoietic stem cells.
Fuller et al. [65] concluded that over the past 40 years, important applications of isolated hepatocytes have been seen in science and medicine in various fields, such as physiological studies, experimental and clinical transplantation, examination of liver metabolism, organ conservation, and drug decontamination. Currently, there is growing enthusiasm for various applications of liver progenitor cells in many scientific areas, such as regenerative medicine and biotechnology. Cryobanking is required for these purposes. Cryopreservation is a crucial enabling technology in regenerative medicine because it allows for stable and secure extended cell storage [62]. Cryopreservation enables the establishment and preservation of biobanks, which are vital resources for studies, clinical trials, and personalized treatment strategies [66,62,34].
- Review Article
- Abstract
- Cryopreservation
- Main Text
- Types of Preservational Methods
- Comparison between Slow Freezing and Vitrification
- Types of Cryopreservation
- Challenges in Cryopreservation (Cryoinjuries)
- Innovations in Cryoprotectant’s Solution
- Types of Cryoprotectants
- Improving Viability and Post-thawing Recovery
- Applications of Cryopreservation Technique
- Limitations of the Cryopreservation Technique
- Future Strategies
- Ethical Considerations in the field of Cryopreservation
- Discussion
- Conclusions
- Author Contribution
Limitations of the Cryopreservation Technique
Cryopreservation has some limitations in both basic and clinical research. Storage in liquid nitrogen (LN2) at -196°C may end the metabolizing process, causing potential side effects. Furthermore, in some cases, biological cells that are attached to lipids and proteins may have slow genetic variations that could alter cellular integrity and impair cellular structure and activity. Moreover, cryoprotectants can cause injuries to cellular chromosomal stability. For example, DMSO can cause tumors at high concentrations. Likewise, in some cases, cryoprotective agents make cells more susceptible to various types of contagious infections and diseases [8].
Cryopreservation is an important approach for storing biological materials for long periods; however, it has various limitations that must be considered. Cryoinjuries are a significant limitation of cryopreservation, reducing the usability of cryopreserved materials for research or clinical purposes, resulting from both freezing and thawing processes. Ice crystals formed during the freezing process might harm the cellular structure of the samples, resulting in diminished viability and function. Furthermore, cryopreservation may not be appropriate for all biological materials, and variances in methods can result in differences in cell survival and function after thawing [67].
Furthermore, it is difficult to store and transport cryopreserved materials. In addition, there are several ethical and regulatory concerns regarding cryopreservation. The use of cryopreserved human embryos and gametes for assisted reproductive technologies, for example, raises ethical considerations regarding the possibility of future offspring and the long-term effects of cryo-preservation on cells and tissues. Regulatory frameworks for their storage and use must be devised and updated to ensure the safety and ethical use of cryopreserved cells, tissues, and organs. To address the limitations of classical cryopreservation, continued research and development of new cryopreservation protocols and alternate storage technologies, such as vitrification, are required.
- Review Article
- Abstract
- Cryopreservation
- Main Text
- Types of Preservational Methods
- Comparison between Slow Freezing and Vitrification
- Types of Cryopreservation
- Challenges in Cryopreservation (Cryoinjuries)
- Innovations in Cryoprotectant’s Solution
- Types of Cryoprotectants
- Improving Viability and Post-thawing Recovery
- Applications of Cryopreservation Technique
- Limitations of the Cryopreservation Technique
- Future Strategies
- Ethical Considerations in the field of Cryopreservation
- Discussion
- Conclusions
- Author Contribution
Future Strategies
Cryopreservation has gone a long way, but considerable work is still required to improve its efficiency, safety, and usefulness. In terms of future strategies, there may be several possible methods. The creation of new cryoprotective agents (CPAs) that are more effective, less toxic, and more compatible with diverse cell and tissue types is an important prospect for cryopreservation research. Although several CPAs have been produced over the years, their efficacy varies depending on the type of cell or tissue being cryopreserved and their toxicity limits their use in clinical applications. Computer-aided molecular design, which can uncover novel compounds with specific cryoprotective characteristics, has been investigated to build more efficient and safer CPAs (Suzuki et al., 2019). Other options include the use of natural or biomimetic chemicals that can better mimic the properties of biological tissues and cells [68]. Creating more efficient and safer CPAs will allow for more successful cryo-preservation of different cell, tissue, and organ types and broaden the use of cryopreservation in research and clinical applications.
Another difficulty with cryopreservation is the limited storage capacity of traditional cryogenic facilities, which require significant amounts of liquid nitrogen and specialized equipment. This restricts the availability of cryopreserved cells and tissues, particularly in resource-constrained situations. Researchers are investigating new cryopreservation storage technologies such as dry storage, which utilizes desiccants to remove water from cells and tissues, and solid-state storage, which uses a solid medium to preserve cells at low temperatures (Karlsson et al., 2015). These techniques have the potential to make the cryopreservation of cells and tissues more affordable and accessible, which could have substantial consequences for research and clinical applications. The difficulty in retaining the functionality and viability of cells and tissues after thawing is a significant obstacle in cryopreservation, particularly for complex systems such as organs and tissues with intricate architectures. Tissue engineering and bioprinting advancements have opened new opportunities for generating sophisticated tissue constructions; however, cryopreservation of these structures remains a barrier. Researchers are exploring new ways to cryopreserve complex tissue constructs, such as vitrification-based technologies that use high concentrations of CPAs to reduce ice formation while preserving cell and tissue structure and function [69].
Another approach is to create new thawing technologies that reduce mechanical stress and cell damage during the thawing process, such as employing magnetic nanoparticles or acoustic waves to control ice formation and reduce cell damage. These methods offer potential solutions for the cryopreservation of complex tissue constructs, which could have far-reaching consequences in regenerative medicine and tissue engineering. Cryopreservation remains an important tool in modern medicine and research; however, it faces various obstacles that must be overcome before it can be widely used. New cryoprotective chemicals, storage technologies, and preservation methods for complex tissue constructs offer intriguing solutions to these difficulties; however, further studies are needed to maximize their effectiveness and safety.
- Review Article
- Abstract
- Cryopreservation
- Main Text
- Types of Preservational Methods
- Comparison between Slow Freezing and Vitrification
- Types of Cryopreservation
- Challenges in Cryopreservation (Cryoinjuries)
- Innovations in Cryoprotectant’s Solution
- Types of Cryoprotectants
- Improving Viability and Post-thawing Recovery
- Applications of Cryopreservation Technique
- Limitations of the Cryopreservation Technique
- Future Strategies
- Ethical Considerations in the field of Cryopreservation
- Discussion
- Conclusions
- Author Contribution
Ethical Considerations in the field of Cryopreservation
Consent, autonomy, and status of frozen materials are the main ethical issues in the field of cryopreservation [21,34]. These factors include individuals' agreement to have their tissues or cells cryopreserved, as well as matters concerning the ownership and management of preserved materials. Furthermore, any potential effects on the rights and welfare of people whose cells or organs are cryopreserved must be considered [70,71].
- Review Article
- Abstract
- Cryopreservation
- Main Text
- Types of Preservational Methods
- Comparison between Slow Freezing and Vitrification
- Types of Cryopreservation
- Challenges in Cryopreservation (Cryoinjuries)
- Innovations in Cryoprotectant’s Solution
- Types of Cryoprotectants
- Improving Viability and Post-thawing Recovery
- Applications of Cryopreservation Technique
- Limitations of the Cryopreservation Technique
- Future Strategies
- Ethical Considerations in the field of Cryopreservation
- Discussion
- Conclusions
- Author Contribution
Discussion
Cryopreservation is the process of freezing biological tissues or cells at extremely low temperatures for long-term storage. With little deterioration or damage, cryopreservation enables the long-term preservation of biological components, such as sperm, eggs, embryos, tissues, and organs. Cryopreservation techniques have significantly improved over time, improving the storage and preservation of biological resources. In this study, we examined the present cryopreservation techniques and their potential in the future.
The slow-freezing approach, which gradually lowers the temperature of biological materials to -80°C or below using a cryoprotectant solution, is the most widely used cryopreservation technique. Sperm, eggs, embryos, and various tissues can be preserved using this technique. However, it has drawbacks such as cellular structures being harmed and reduced viability of the preserved cells. Another method of cryopreservation that has recently grown in favor is vitrification. Using high concentrations of cryoprotectant solutions, this method entails rapid cooling of biological samples to extremely low temperatures, often below -196°C. Oocytes, embryos, and some types of tissues were successfully preserved using vitrification. For successful execution, a high level of skill and cutting-edge tools is required.
New technologies that promise to change the industry have been developed owing to improvements in cryopreservation methods. Cryopreservation employs one such technology: nanotechnology. It has been demonstrated that nanoparticles improve the penetration of cryoprotectants into cells and tissues, thereby increasing their effectiveness. This may result in substantial advancements in the preservation of biological materials. The application of gene editing in cryopreservation is an intriguing advance. Before cryopreservation, cells and tissues can be altered using gene editing technologies such as CRISPR-Cas9 to increase their resistance to damage from freezing and thawing. The viability and functionality of the preserved cells and tissues could be significantly improved. These developments could completely change the field of cryopreservation, improve the storage of biological materials, and pave the way for important developments in biomedical research, clinical applications, and conservation initiatives. In addition, current cryopreservation approaches struggle to keep these systems structurally intact during the freezing and thawing processes. To overcome this issue, researchers are investigating innovative techniques for increasing the structural integrity of these systems. One option is to use 3D printing technology to generate sophisticated tissue structures that might be cryopreserved and used for transplantation and other purposes.
- Review Article
- Abstract
- Cryopreservation
- Main Text
- Types of Preservational Methods
- Comparison between Slow Freezing and Vitrification
- Types of Cryopreservation
- Challenges in Cryopreservation (Cryoinjuries)
- Innovations in Cryoprotectant’s Solution
- Types of Cryoprotectants
- Improving Viability and Post-thawing Recovery
- Applications of Cryopreservation Technique
- Limitations of the Cryopreservation Technique
- Future Strategies
- Ethical Considerations in the field of Cryopreservation
- Discussion
- Conclusions
- Author Contribution
Conclusions
The fields of medicine, biology, bioinformatics, chemistry, and physics work together during cryopreservation. Scaling up existing approaches to greater volumes and more complex tissues represents the primary hurdle that must still be overcome. It takes more time to chill and warm an organ or tissue volume that needs to be vitrified because of its large size. The viscosity of cryoprotective agents (CPAs) in perfusion systems and their thermal conductivities are critical for cryopreservation. However, when dealing with larger human organs, additional considerations arise when dealing with cell lines or smaller tissues, such as sperm, eggs, or corneas. This is primarily because of the requirement for high concentrations of CPAs to suppress ice formation for long periods between the melting and glass transition temperatures. Certain solutions can be used to address these difficulties in small tissues. One strategy is to use CPAs with weak water interactions, which reduces the disturbance of the hydration layers surrounding the biomolecules. The use of CPAs with reciprocal toxicity-neutralizing effects is advantageous. Another method is to incorporate nonpenetrating CPAs and ice blockers to reduce the concentration of penetrating CPAs. These techniques assist in overcoming restrictions related to the high CPA concentrations required for larger organs. However, it is crucial to highlight that information regarding the mechanisms underlying these systems is limited. More studies are needed to gain a better understanding of the complexities involved in cryopreserving larger organs and to find more effective solutions to these obstacles.
Cryobiology has some obstacles but is not insurmountable. Innovative cryoprotectants with large cryoprotective effects depend on their concentrations. In silico molecular modeling, an improved understanding of the processes that occur during cryopreservation will be used in future research to avoid ice formation and reduce cryoprotectant toxicity. It is possible to imagine a universal cryoprotectant agent that can be used on a wide variety of tissues, and technological advances could allow for rapid cooling and warming or the manipulation of ice formation to allow for the vitrification or freezing of large volumes of material. Although the ideas have been around for quite some time, the science of cryobiology as a distinct discipline emerged approximately 70 years ago. It has come a long way from vitrifying spermatozoa in glycerol to vitrifying tissues and even tiny organs in complex multicomponent solutions. Given that cryopreservation is still a relatively obscure field of study, with little attention from schools or undergraduate courses and only a fraction of the cash dedicated to other causes, these advancements are truly astounding. Thus, there are still many possibilities ahead, ranging from near-term advancements in transplantation biology to far-fetched goals, such as the establishment of organ banks or prolonged suspended animation.
Cryopreservation is a rapidly emerging technology with numerous potential opportunities and concerns. Cryopreservation techniques will become more efficient and effective with the development of new cryoprotective chemicals, preservation procedures, and integration with other technologies. To ensure the safe and ethical use of cryopreserved cells, tissues, and organs in research and clinical applications, the ethical and regulatory aspects related to cryopreservation must also be properly addressed.
- Review Article
- Abstract
- Cryopreservation
- Main Text
- Types of Preservational Methods
- Comparison between Slow Freezing and Vitrification
- Types of Cryopreservation
- Challenges in Cryopreservation (Cryoinjuries)
- Innovations in Cryoprotectant’s Solution
- Types of Cryoprotectants
- Improving Viability and Post-thawing Recovery
- Applications of Cryopreservation Technique
- Limitations of the Cryopreservation Technique
- Future Strategies
- Ethical Considerations in the field of Cryopreservation
- Discussion
- Conclusions
- Author Contribution
Author Contribution
AI performed the research and wrote the manuscript, and IY helped with the writing and formatting of the manuscript. All authors have read and approved the manuscript.
- Review Article
- Abstract
- Cryopreservation
- Main Text
- Types of Preservational Methods
- Comparison between Slow Freezing and Vitrification
- Types of Cryopreservation
- Challenges in Cryopreservation (Cryoinjuries)
- Innovations in Cryoprotectant’s Solution
- Types of Cryoprotectants
- Improving Viability and Post-thawing Recovery
- Applications of Cryopreservation Technique
- Limitations of the Cryopreservation Technique
- Future Strategies
- Ethical Considerations in the field of Cryopreservation
- Discussion
- Conclusions
- Author Contribution
References
- Jang TH, Park SC, Yang JH, Kim JY, Seok JH, et al. (2017) Cryopreservation and its clinical applications. Integrative medicine research 6(1): 12-18.
- Love R (2009) Chillin'at the Symposium with Plato: Refrigeration in the Ancient World. Ashrae Transactions 115(1).
- Boyle R, Merret C, Hob (1973) New Experiments and Observations Touching Cold, or An Experimental History of Cold Begun: To which are Added an Examen of Antiperistasis and an Examen of Mr. Hobs's Doctrine about Cold. Richard Davis.
- Sambu S (2015) A Bayesian approach to optimizing cryopreservation protocols. PeerJ 3: e1039.
- Onofre J, Baert Y, Faes K, Goossens E (2016) Cryopreservation of testicular tissue or testicular cell suspensions: a pivotal step in fertility preservation. Human reproduction update 22(6): 744-761.
- Karlsson JO, Toner M (1996) Long-term storage of tissues by cryopreservation: critical issues. Biomaterials 17(3): 243-256.
- Mazur P (1970) Cryobiology: The Freezing of Biological Systems: The responses of living cells to ice formation are of theoretical interest and practical concern. Science 168(3934): 939-949.
- Bhattacharya S (2018) Cryoprotectants and Their Usage in Cryopreservation Process. Biotechnology In Biomedical and Biological Sciences 7.
- Rall WF, Fahy GM (1984) Ice-free cryopreservation of mouse embryos at -196 degrees C by vitrification. Nature 313(6003): 573-575.
- Bojic S, Murray A, Bentley BL, Spindler R, Pawlik, et al. (2021) Winter is coming: the future of cryopreservation. BMC biology 19(1): 1-20.
- Mandawala AA, Harvey SC, Roy TK, Fowler KE (2016) Cryopreservation of animal oocytes and embryos: Current progress and future prospects. Theriogenology 86(7): 1637-1644.
- Yong KW, Wan Safwani WKZ, Xu F, Wan Abas WAB, Choi JR, et al. (2015) Cryopreservation of human mesenchymal stem cells for clinical applications: current methods and challenges. Biopreservation and biobanking 13(4): 231-239.
- Zeron Y, Pearl M, Borochov A, Arav A (1999) Kinetic and temporal factors influence chilling injury to germinal vesicle and mature bovine oocytes. Cryobiology 38(1): 35-42.
- Crowe JH, Crowe LM (2000) Preservation of mammalian cells-learning nature's tricks. Nature biotechnology 18(2): 145-146.
- Kopeika E, Kopeika J, Zhang T (2007) Cryopreservation of fish sperm. In Cryopreservation and freeze-drying protocols. Humana Press Pp: 203-217.
- Gao D, Critser JK (2000) Mechanisms of cryoinjury in living cells. ILAR journal 41(4): 187-196.
- Taylor MJ, Weegman BP, Baicu SC, Giwa SE (2019) New Approaches to Cryopreservation of Cells, Tissues, and Organs. Transfusion medicine and hemotherapy: offizielles Organ der Deutschen Gesellschaft fur Transfusionsmedizin und Immunhamatologie 46(3): 197-215.
- Bojic S, Murray A, Bentley BL, Spindler R, Pawlik, et al. (2021) Winter is coming: the future of cryopreservation. BMC biology 19(1): 1-20.
- Yavin S, Arav A (2007) Measurement of essential physical properties of vitrification solutions. Theriogenology 67(1): 81-89.
- Fowler A, Toner M (2006) Cryo‐injury and biopreservation. Annals of the New York Academy of Sciences 1066(1): 119-135.
- Suppes GJ, Egan S, Casillan AJ, Chan KW, Seckar B (2003) Impact of high pressure freezing on DH5α Escherichia coli and red blood cells. Cryobiology 47(2): 93-101.
- Kalichevsky MT, Knorr D, Lillford PJ (1995) Potential food applications of high-pressure effects on ice-water transitions. Trends in Food Science & Technology 6(8): 253-259.
- Bakst MR, Wishart G, Brillard JP (1994) Oviducal sperm selection, transport, and storage in poultry. Poult Sci Rev 5: 117-143.
- Massip A, Leibo SP, Blesbois E (2004) Cryobiology of gametes and the breeding of domestic animals. In Life in the frozen state CRC Press Pp: 397-418.
- Toner M, Cravalho EG, Stachecki J, Fitzgerald T, Tompkins RG, et al. (1993) Nonequilibrium freezing of one-cell mouse embryos. Membrane integrity and developmental potential. Biophysical journal 64(6): 1908-1924.
- Pegg DE (2007) Principles of cryopreservation. In Cryopreservation and freeze-drying protocols. Humana Press Pp: 39-57.
- Meneghel J, Kilbride P, Morris GJ (2020) Cryopreservation as a Key Element in the Successful Delivery of Cell-Based Therapies-A Review. Front Med (Lausanne) 7:592242.
- Dittrich R, Maltaris T, Mueller A, Dimmler A, Hoffmann I, et al. (2006) Successful uterus cryopreservation in an animal model. Hormone and metabolic research 38(03): 141-145.
- Wang X, Chen H, Yin H, Kim SS, Tan SL, et al. (2002) Fertility after intact ovary transplantation. Nature 415(6870): 385-385.
- Arav A, Revel A, Nathan Y, Bor A, Gacitua H, et al (2005) Oocyte recovery, embryo development and ovarian function after cryopreservation and transplantation of whole sheep ovary. Human Reproduction 20(12): 3554-3559.
- De Leeuw FE, Colenbrander B, Verkleij AJ (1991) The role membrane damage plays in cold shock and freezing injury. Reproduction in Domestic Animals 1: 95-104.
- Watson PF (2000) The causes of reduced fertility with cryopreserved semen. Animal reproduction science 60: 481-492.
- Tanford C (1980) The Hydrophobic Effect 62(2).
- Yang J, Gao L, Liu M, Sui X, Zhu Y, et al. (2019) Advanced Biotechnology for Cell Cryopreservation. Transactions of Tianjin University 26(6): 409-423.
- Elliott GD, Wang S, Fuller B (2017) Cryoprotectants: A review of the actions and applications of cryoprotective solutes that modulate cell recovery from ultra-low temperatures. Cryobiology 76: 74-91.
- Chen J, Liu X, Hu Y, Chen X, Tan S (2023) Cryopreservation of tissues and organs: present, bottlenecks, and future. Frontiers in Veterinary Science 10.
- Liu X, Pan Y, Liu F, He Y, Zhu Q, et al. (2021) A Review of the Material Characteristics, Antifreeze Mechanisms, and Applications of Cryoprotectants (CPAs). Journal of Nanomaterials P: 1-14.
- Wang X, Wang E, Zhao G (2023) Advanced cryopreservation engineering strategies: the critical step to utilize stem cell products. Cell Regeneration 12(1).
- Polge C, Smith AU, Parkes AS (1949) Revival of spermatozoa after vitrification and dehydration at low temperatures. Nature 164(4172): 666-666.
- Baust JM, Vogel MJ, Buskirk RV, Baust JG (2001) A Molecular Basis of Cryopreservation Failure and its Modulation to Improve Cell Survival. Cell Transplantation 10(7): 561-571.
- Wowk B, Leitl E, Rasch CM, Mesbah-Karimi N, Harris SB, et al. (2000) Vitrification enhancement by synthetic ice blocking agents. Cryobiology 40(3): 228-236.
- Safa S, Moghaddam G, Jozani RJ, Kia HD, Janmohammadi H (2016) Effect of vitamin E and selenium nanoparticles on post-thaw variables and oxidative status of rooster semen. Animal reproduction science 174: 100-106.
- Polge C, Smith AU, Parkes AS (1949) Revival of spermatozoa after vitrification and dehydration at low temperatures. Nature 164(4172): 666-666.
- Bhattacharya MS (2016) A review on cryoprotectant and its modern implication in cryonics. Asian Journal of Pharmaceutics (AJP): Free full text articles from Asian J Pharm10(3).
- Rakha BA, Ansari MS, Akhter S, Zafar Z, Hussain I, et al. (2017) Cryopreservation of Indian red jungle fowl (Gallus gallus murghi) semen with polyvinylpyrrolidone. Cryobiology 78: 27-33.
- Rakha BA, Ansari MS, Akhter S, Zafar Z, Naseer A, et al. (2018) Use of dimethylsulfoxide for semen cryopreservation in Indian red jungle fowl (Gallus gallus murghi). Theriogenology 122: 61-67.
- Wowk B, Leitl E, Rasch C M, Mesbah-Karimi N, Harris SB, et al. (2000) Vitrification enhancement by synthetic ice blocking agents. Cryobiology 40(3): 228-236.
- Park C, Chapman FA (2005) An Extender Solution for the Short-Term Storage of Sturgeon Semen. North American Journal of Aquaculture 67(1): 52-57.
- Sexton TJ, Fewlass TA (1978) A new poultry semen extender: 2. Effect of the diluent components on the fertilizing capacity of chicken semen stored at 5°C. Poultry Science 57(1): 277-284.
- Lukaszewicz E (2002) An effective method for freezing White Italian gander semen. Theriogenology 58(1): 19-27.
- Lake PE (1960) Studies on the dilution and storage of fowl semen. Reproduction 1(1): 30-35.
- Tselutin K, Narubina L, Mavrodina T, Tur B (1995) Cryopreservation of poultry semen. British Poultry Science 36(5): 805-811.
- Blanco JM, Gee G, Wildt DE, Donoghue AM (2000) Species variation in osmotic, cryoprotectant, and cooling rate tolerance in poultry, eagle, and peregrine falcon spermatozoa. Biology of reproduction 63(4): 1164-1171.
- Blesbois E, Grasseau I, Seigneurin F (2005) Membrane fluidity and the ability of domestic bird spermatozoa to survive cryopreservation. Reproduction 129(3): 371-378.
- Pegg DE (2002) The history and principles of cryopreservation. In Seminars in reproductive medicine 20(1): 5-13.
- Fahy GM (2010) Cryoprotectant toxicity neutralization. Cryobiology 60(3): S45-S53.
- Crowe LM, Crowe JH, Rudolph A, Womersley C, Appel L (1985) Preservation of freeze-dried liposomes by trehalose. Archives of biochemistry and biophysics 242(1): 240-247.
- Holt WV (2000) Basic aspects of frozen storage of semen. Animal reproduction science 62(1-3): 3-22.
- Tie C, Zhao G (2021) Ice Inhibition for Cryopreservation: Materials, Strategies, and Challenges. Advanced Science 8(6).
- Gurruchaga H, Burgo LSD, Hernández RM, Orive G, Selden C, et al. (2018) Advances in the slow freezing cryopreservation of microencapsulated cells. Journal of Controlled Release 281: 119-138.
- Whaley D, Damyar K, Witek RP, Mendoza AJFN, Alexander M P, et al. (2021) Cryopreservation: An Overview of Principles and Cell-Specific Considerations. Cell Transplantation 30.
- Meneghel J, Kilbride P, Morris G (2020) Cryopreservation as a Key Element in the Successful Delivery of Cell-Based Therapies-A Review. Frontiers in Medicine 7: 592242.
- Miyagi-Shiohira C, Kurima K, Kobayashi N, Saitoh I, Watanabe M, et al. (2015) Cryopreservation of adipose-derived mesenchymal stem cells. Cell medicine 8(1-2): 3-7.
- Marquez-Curtis LA, Janowska-Wieczorek A, McGann LE, Elliott JA (2015) Mesenchymal stromal cells derived from various tissues: biological, clinical and cryopreservation aspects. Cryobiology 71(2): 181-197.
- Fuller BJ, Petrenko AY, Rodriguez JV, Somov AY, Balaban CL, et al. (2016) Biopreservation of hepatocytes: current concepts on hypothermic preservation, cryopreservation, and vitrification. CryoLetters 37(4): 432-452.
- Demirci U, Assal RE (2015) Bio-inspired cryoprotectants and microfluidics for cryopreservation. Cryobiology 71(1): 167-167.
- Fuller BJ (2004) Cryoprotectants: the essential antifreezes to protect life in the frozen state. Cryo Letters 25(6): 375-388.
- Fahy GM, Wowk B, Wu J, Phan J, Rasch C, et al. (2004) Cryopreservation of organs by vitrification: perspectives and recent advances. Cryobiology 48(2): 157-178.
- Chen H, Tian M, Chen L, Cui X, Meng J, et al. (2019) Optimization of composite cryoprotectant for freeze-drying Bifidobacterium bifidum BB01 by response surface methodology. Artificial Cells Nanomedicine and Biotechnology 47(1): 1559-1569.
- Acker JP (2005) Bio preservation of Cells and Engineered Tissues. Advances in Biochemical Engineering / Biotechnology PP: 157-187.
- Duarte JMB, Ramalho MFPDT, Lima VFHD, Jorge W (1999) A leukocyte cryopreservation technique for cytogenetic studies. Genetics and Molecular Biology 22(3): 399-400.






























