A Case of Subclinical Malaria in the Salento Area (Apulia Region-Southern Italy) Identified by DASIT’s Sysmex XN 2000 Blood Cell Counter
L Biscozzo1, A Paiano2, M Serra2, M Cazzato2, M Laneve1, L Tagliaferro1*
1UOC Patologia clinica, Italy
2UOC Gastroenterologia - Ospedale “Veris Delli Ponti”, Scorrano (Le), Italy
Submission:March 09, 2024; Published: March 20, 2024
*Corresponding author: L Tagliaferro, Clinical Pathology Unit, Veris Delli Ponti hospital - Scorrano, Italy. Email: luigi.tagliaferro@asl.lecce.it
How to cite this article: L Biscozzo, A Paiano, M Serra, M Cazzato, M Laneve1, L Tagliaferro. A Case of Subclinical Malaria in the Salento Area (Apulia Region-Southern Italy) Identified by DASIT’s Sysmex XN 2000 Blood Cell Counter. Adv Biotech & Micro. 2024; 18(1): 555977. DOI:10.19080/AIBM.2024.17.555977
Abstract
Keywords: Plasmodium; Anopheles; Malaria; COVID-19; Giemsa; band shaped
Abbreviations: pRBC: Parasitized Red Blood Cells; ISS: Istituto Superiore di Sanità; WHO: World Health Organization
Introduction
Malaria is perhaps the most important human parasitic disease and among the main causes of morbidity and mortality, especially in tropical and subtropical areas, where it is endemic, without forgetting that even in countries that have always been free or where the disease has long been eradicated, thousands of cases of importation are recorded every year, due in particular to the exponential increase in international travels and the intensification of the migratory phenomenon [1].
Even in Italy, a country historically with a high malarial endemicity and declared malaria-free by the WHO in 1970, almost all of the notified cases are imported; the increase is due to the development of immigration flows from sub-Saharan African countries to Italy. Those who get sick are mainly immigrants who return to their country of origin to visit relatives or friends or Italians who for various reasons (work, tourism, volunteering) visited risk areas. In recent years there have also been very rare cases of malaria due to unusual modes of transmission, such as accidental bites with contaminated material and cases probably due to mosquitoes carried in luggage.
In accordance with the latest WHO World malaria report, 247 million cases of malaria were recorded in 2021, with an estimated number of deaths from malaria in the same year equal to 619,000. Between 2020 and 2021, in full COVID-19 pandemic, there have been 13 million more cases of malaria and 63,000 more deaths from malaria. According to the WHO, the African Region in 2021 recorded approximately 95% of all malaria cases and 96% of deaths and children under the age of five constituted over 80% of all malaria deaths in the Region [2].
The causal agent of malaria is Plasmodium, a protozoan transmitted by the bite of female mosquitoes of the Anopheles genus. There are five species of Plasmodium that cause malaria in humans: P. falciparum, P. vivax, P. malariae, P. ovale and P. knowlesi. The most lethal is P. falciparum, widespread mainly in the African continent, while P. vivax is the dominant malaria parasite in many countries outside the sub-Saharan area. The diagnosis of malaria should be made through a combination of medical history, clinical observation and diagnostic tests. Since the 1990s, new methods for diagnosing malaria have been introduced, such as molecular diagnosis and rapid diagnostic tests that detect malaria antigens, but the gold standard for confirming the presence and the identification of malaria parasites is microscopic examination of the peripheral blood smear [3].
It has been reported in several studies that the use of automated hematology analyzers, especially in non- endemic areas, supports the diagnosis of malaria, particularly in those cases where there is no clinical suspicion [4-6]. In this regard, we report a case of malaria diagnosed thanks to the automated counting of peripheral blood cells, without a specific request for the search for the malarial parasite. Although malaria is a low prevalence condition, the danger and rapid evolution of the infection make it necessary to prepare and maintain available professional skills and diagnostic methodologies that allow rapid recognition or exclusion of the pathology.
Materials and methods
A 31-year-old male patient originally from Burkina Faso, but in Italy for some time, arrived at the emergency room of the Veris Delli Ponti hospital (Scorrano-Italy) in the late morning of October 24th, due to general malaise, asthenia, and tremors. The clinical parameters were normal: heart rate 80/min, blood pressure 125/85 mm/Hg, O2 saturation 100%, body temperature 36.8°C. The physical examination by the doctor on call showed a treatable abdomen, not painful on palpation, no organ megalia; upon respiratory observation, diffuse pulmonary wheezes, upon cardiological observation, valid rhythmic tones. A blood gas analysis was performed, which revealed a state of respiratory alkalosis and several blood-chemical tests were urgently requested from the laboratory which revealed an anemic state (Hb 83 g/l); furthermore there was an increase in transaminases and gGT (GOT 237 IU/l, GPT 76 IU/l, GGT 1165 IU/l), hyperbilirubinemia (Tot 4.07mg/dl, direct 1.65mg/dl, indirect 2.42 mg/dl) and one state of dehydration (azotemia 9 mg/dl, creatinine 0.52 mg/dl, sodium 133 mEq/I, potassium not assessable due to haemolysed sampling). The emergency blood count was performed on Dasit's Sysmex XN 1000 analyzer.
The patient was sent to radiology for a chest X-ray which only revealed fractures on the right. In fact, the chest X-ray showed no alterations in the lung parenchyma; the pleural cavities were free from effusion, the cardio- mediastinal image was within limits. The diagnostic hypothesis was dehydration, poor nutrition, lung wheezing. The patient was admitted to the gastroenterology department of the same hospital. Here he reported alcohol abuse and in the last few days "dark" stools, therefore on October 25th an esophago-gastro-duodeno-scopy was performed from which only modest non-specific gastric inflammation emerged.
On the same day, further tests were requested from the laboratory, as per routine, including viral hepatitis A, B and C markers and some tumor markers (CEA, AFP, Ca-19.9), protein electrophoresis and urine analysis. Laboratory tests revealed a previous infection with hepatitis A and B viruses; CEA, AFP and Ca-19.9 were normal; while the urine analysis provided more interesting information for diagnostic purposes; in fact, the urine showed a light amber color, pH 8.5, bilirubin 1 mg/dl and urobilinogen > 12 mg/dl.
This time, as usually happens for non-urgent requests, the blood count test was performed in the hematology sector, by the Sysmex XN 2000 automatic analyzer from Dasit. The test confirmed the anemia already reported the previous day (Hb 70 g/l, RBC 2.18x1012/l, HCT 0.22) and provided the following values for white blood cells and platelets: WBC 4.44x109/l, PLT 181 x109/l; differential white blood cells count showed normal values of neutrophils 2.68x109/l, lymphocytes 1.06 x109/l, monocytes 0.68 x109/l, eosinophils 0.02 x109/l, and zero basophils; furthermore, the presence of erythroblasts equal to 5.6/100 WBC was noted. However, the instrument did not provide the alarms that usually appear in the presence of malaria infection such as "abnormal WDF scattergram" and "presence of parasitized red blood cells” (pRBC). But the area of the WDF graph between the cloud of neutrophils and that of eosinophils, where the parasitized red blood cells should be located, showed a certain number of events, although not numerous (figure 1).
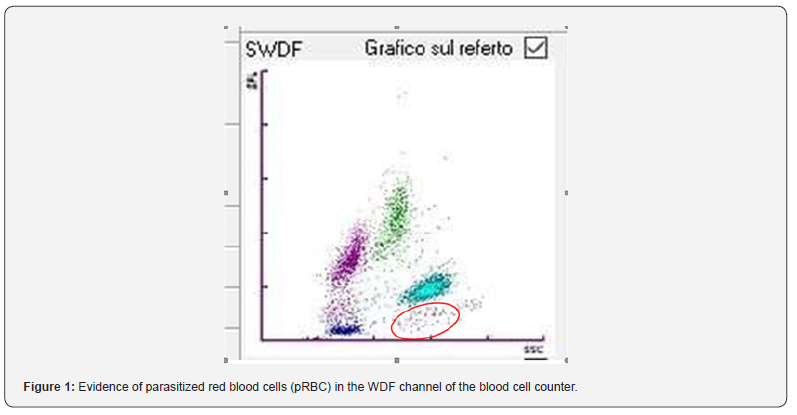
Therefore, considering the results of laboratory tests, which essentially pointed towards hemolytic anemia, it was decided to carry out the search for malarial plasmodium. In our laboratory the malarial parasite identification is conducted through microscopic observation of peripheral blood. We don’t have any reagents that allow us to carry out immunochromatographic research for malarial antigens. So, after carrying out the blood count, some slides were prepared for microscopic diagnosis, keeping in mind that a correct preparation of the smear allows even the determination of very low number of blood parasites. Uniform, thin slides were allowed to air dry for several minutes and stained with Wright-Giemsa using an automatic stainer (the WHO Standard Operating Manual of Procedure recommends the use of Giemsa stain). The microscopic examination confirmed the presence of trophozoites with ring form cytoplasm and some band shaped forms were also observed, with evident malarial pigment and some gametocytes; all this allowed the diagnosis of P. malariae malaria to be made (figures 2 and 3).
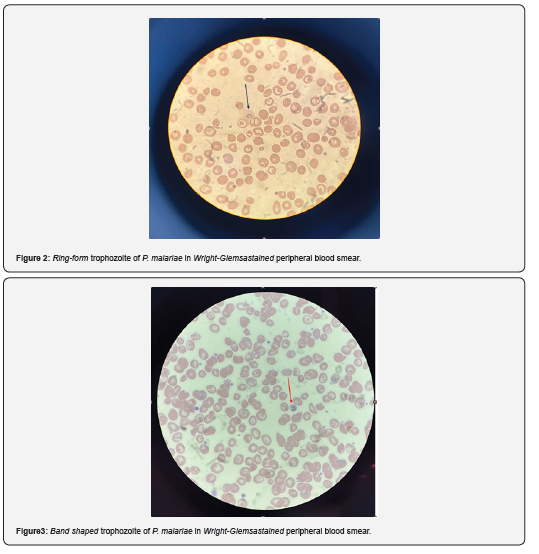
A rapid and accurate diagnosis represents the essential prerequisite for effective therapy in subjects suffering from malaria. Once the diagnosis of malaria was confirmed with the peripheral blood smear, the gastroenterology department was notified and the patient, on the same day, was sent to the Infectious Diseases department at a 2nd level hospital, where he was given therapy immediately (drug used Malarone, antimalarial containing two active ingredients, atovaquone and proguanil hydrochloride). Some smears were immediately sent to the laboratory of the Istituto Superiore di Sanità (ISS) in Rome, as per practice, for definitive confirmation.
Discussion
The search for the malarial parasite is necessary at the slightest anamnestic and/or clinical suspicion such as a patient returning from an endemic area with flu-like symptoms (fever, asthenia, gastrointestinal disorders, arthromyalgia, headache) and must be carried out even if the patient he has no fever and has undergone chemoprophylaxis. In our case, the diagnostic suspicion of malaria was placed by the analysis laboratory that performed the patient’s blood count, upon request from the gastroenterology department, but in the absence of a very specific clinical question.
The information provided by the laboratory tests which pointed towards a haemolytic anemia, together with the indications provided by the Sysmex XN 2000 blood cell counter (DASIT) with which the blood count was processed, led the laboratory to consider the possibility that the patient had malaria. In fact, although the “parasitized red blood cells” (pRBC) flag did not appear on the analyzer, nor an “abnormal WDF scattergram”, the careful analysis of the WDF graph further strengthened the diagnostic hypothesis.
There are mainly two reading channels in Sysmex instruments in which parasites present in the blood can be intercepted: one is the Reticulocyte channel (RET channel) and the other one is the White Blood Cell differential count channel (WDF channel). For both channels, specific fluorescent markers that bind to nucleic acids are used. The higher the intracellular content of nucleic acids (DNA and RNA), the higher the resulting fluorescent signal. The WDF channel differentiates leukocytes based on their nucleic acid content and their internal structure, not by size. This allows for an exact count of lymphocytes, monocytes, eosinophils, neutrophils, and immature granulocytes. Even if red blood cells infected by malaria parasites have a significant increase in nucleic acids compared to uninfected red blood cells, the resulting fluorescence signal is still too weak, compared to that of any nucleated cell, which contains DNA and will always provide a stronger fluorescence signal. Therefore, the red blood cells and reticulocytes, infected or not, will be placed in the ghost area of the WDF graph, the dark blue cloud that is normally positioned under that of the lymphocytes (figure 4).
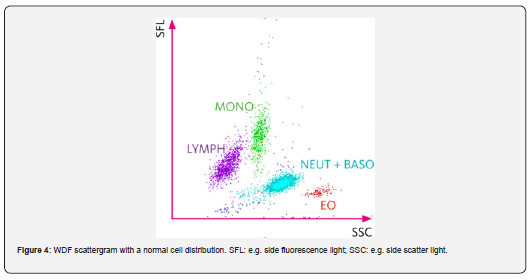
However, in the WDF channel, red blood cells are not completely lysed, so even a small number of lysis- resistant parasitized RBCs can result in increased fluorescence. Furthermore, when schizonts and gametocytes are present in the blood, which are not lysed like RBC, further events can be observed in the WDF graph in correspondence with the area of neutrophils and/or eosinophils, due to the increase in lateral refraction (SSC) and fluorescence intensity (SFL) [7].
Not all plasmodium species have a visible impact on the WDF graph. When this happens, such as when there is no clear separation between the “ghost area” and the “eosinophil area” on the WDF graph, the analyzer generally provides alarms. But even when a very specific alarm does not appear, as in the case in question, from a careful evaluation of the WDF graph it is most often possible to suspect the presence of malaria plasmodium. In fact, even if the sensitivity of Sysmex instruments is limited when compared to the sensitivity of conventional diagnostic methods, such as the peripheral blood smear stained with Giemsa, its specificity is high.
Conclusions
The ISS confirmed the diagnosis of P. malariae.
P. malariae, responsible for benign quartana (schizogonic blood cycle every 72 hours), is widespread in tropical and sub-tropical areas in a non-uniform manner and in any case always with a rather low frequency.
P. malariae can cause recrudescence even after decades. Forms of the parasite can remain in the bloodstream at a subclinical level, in order to evade the immune system and reactivate, causing a new malaria attack when the defenses are lowered for different reasons. According to the WHO, malaria is a curable disease if the diagnosis is timely and if the choice of therapy takes into account the area of origin.
The ability to intercept malaria plasmodium using automated hematology analyzers represents an advantage that helps in the diagnosis of malaria, particularly when clinical suspicion is low.
The peripheral blood smear remains the gold standard and must be performed during febrile attacks, when it is more likely to have trophozoites in circulation, but also in the absence of fever, given its urgency and, if negative, it must be repeated at the next febrile attack. From the blood smear it is possible to obtain the diagnosis of species, the calculation of parasitemia and recognize any other alterations in the red blood cells. It is a method that is easy to prepare and within the reach of all laboratories, even if observation under the microscope requires experienced personnel.
References
- World Malaria Report (2023) World Health Organization.
- (2021) European Centre for Disease Prevention and Control. Malaria – Annual epidemiological report for 2015. Stockholm
- Wongsrichanalai C, Barcus MJ, Muth S, Sutamihardja A, Wernsdorfer WH (2007) A review of malaria diagnostic tools: microscopy and rapid diagnostic test (RDT). Am J Trop Med Hyg 77(6 Suppl): 119-27.
- Campuzano-Zuluaga G, Alvarez-Sanchez G, Escobar-Gallo GE, Valencia-Zuluaga LM, Ríos-Orrego AM (2010) Design of malaria diagnostic criteria for the Susmex XE-2100 hematology Am J Trop Med Hyg 82(3): 402-11
- Huh HJ, Oh GY, Huh JW, Chae SL (2008) Malaria detection with the Sysmex XE-2100 hematology analyzer using pseudoeosinophilia and abnormal WBC Ann Hematol 87(9): 755-9
- Mohapatra S, Samantaray JC, Arulselvi S, Panda J, Munot K, et al. (2011) Automated detection of malaria with haematology analyzer Sismex XE-2100. Indian J Med Sci 65(1): 26-31
- (2015) Sysmex educational enhancement and “Malaria – the global burden”.






























